Abstract

Traditionally, the processing of laboratory data occurs on three levels (Figure 1):
On-board the analytical instruments
Within the laboratory information system (LIS)
In the hospital information system (HIS)
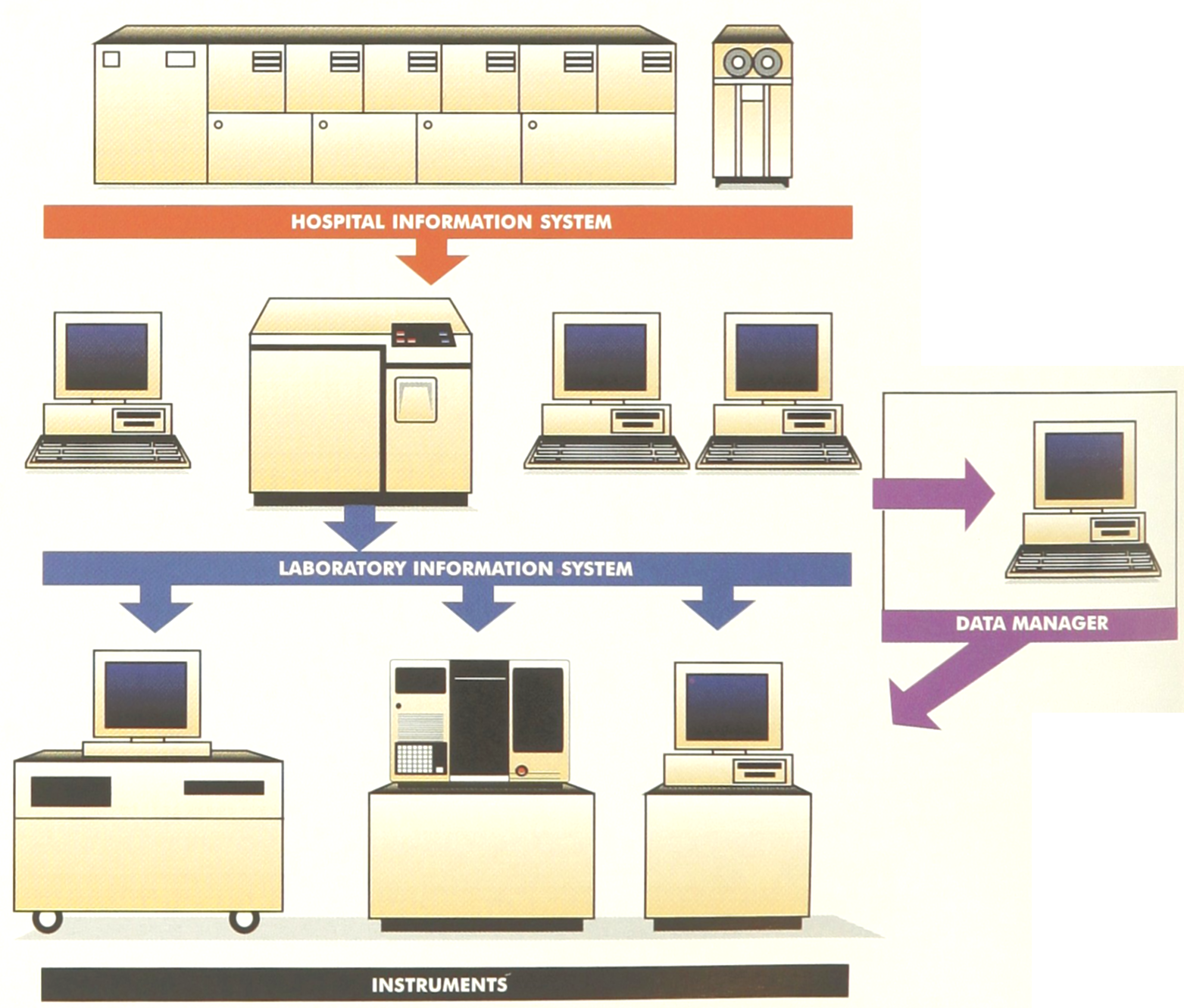
Laboratory data processing as it currently exists, or with the addition of a data manager (inset).
In many hospitals, especially in Europe, the HIS provides patient demographics to the LIS in an unidirectional manner, whereas in some cases, especially in the United States, there is a bi-directional communication exchange between the HIS and the LIS.: Orders can be down-loaded to the lab and results are reported back, thus allowing for an increasingly paperless process.
Manufacturer-Specific Data Managers
More recently some diagnostic manufacturers have begun to offer specialized data management systems to support their own line of analytical instruments with selected added-value functions.
This results in an additional information processing layer between the analyzer and the LIS level which has certain advantages both for the lab manager and the manufacturer:
Consolidation between instruments reduces interfacing with the LIS thereby cutting costs of instrument-host connections.
Those software functions that are common to all connected analyzers can be moved to the data manager resulting in a reduction in the complexity of the analyzer software and training times for technologists.
Added-value functions such as QC or statistics can be provided by the manufacturer without modifying analyzer software.
The data managers provide a common user interface for information handling, even in cases where the manufacturer's analyzer software looks completely different from instrument to instrument.
On the other hand manufacturer-specific data management systems have certain disadvantages:
Additional computer hardware is needed.
Some data managers offer only a limited spectrum of functionalities which often duplicate functions found in commercial LIS software.
There are overlaps with LIS functions that may create logistic problems and data inconsistency in cases where the data manager and a LIS are run in parallel.
The data managers of different manufacturers are not compatible with each other.
New Laboratory Software Products
Observations at US and European trade fairs such as AACC or Medica indicate further evolution and fast expansion in the field of laboratory software products.
Current trends of new software development include automation and on-line control of pre-analytical procedures, scheduling and workflow optimization, decision support at the medical validation and interpretation level, data collection from remote testing sites and, especially in the United States, communication with hospital AI systems and managed care. Some examples are given in Figure 2.

Current trends in software development with AI systems and managed care include work-area-specific dataflow and management systems, state-of-the-art computer technology and systems for rapid response testing.
These do not represent product importance nor high market shares but have been selected from a vast variety of commercial offers for illustration purposes.
Open and Scalable Software Architecture (OSSA)
It is to be expected that the more the spectrum of laboratory-specific software products will grow, the less likely it will be that software complexity can be handled within the rigid framework of the traditional 3- or 4- level architecture.
Current analyzer and laboratory software structures are not advanced enough to cope with the requirements of the future. Hence we have proposed an open and scalable software architecture (3). A schematic view of this concept is given in Figure 3.
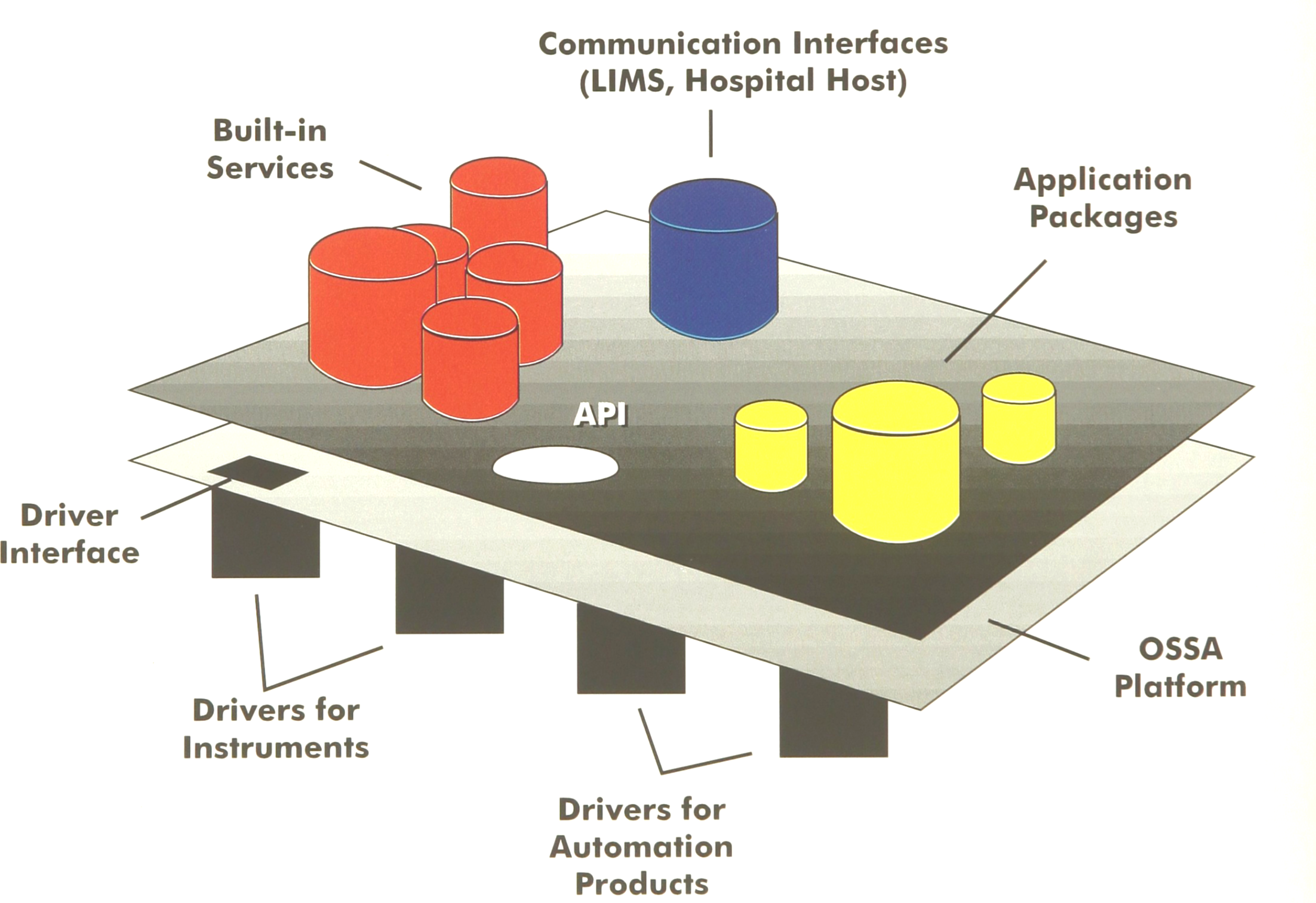
Open and scalable software architecture.
An open and scalable laboratory automation architecture provides the scalability necessary for a flexible automation system. For example, a client server relational database will adapt to the needs of the small laboratory as well as to the large, multi-hospital consortium laboratory.
quire interconnectivity between the electronic communication protocols in use on the many analytical systems used in most laboratories. Mechanical interfacing between instruments and distribution systems from multiple vendors will allow the customer to design a laboratory that will adapt to this uncertain environment.
