Abstract
The aim of this study was to establish a dual-label time-resolved fluorescence immunoassay (TRFIA) for the simultaneous determination of cardiac troponin T (cTnT) and myoglobin (MYO) for the early diagnosis of acute myocardial infarction. The sandwich immunoassay was used to detect the concentration of cTnT and MYO in serum. cTnT and MYO in serum were captured by anti-cTnT and anti-MYO antibodies immobilized on microtiter wells and then banded together with another anti-cTnT and anti-MYO labeled with europium(III) Sm3+ and samarium(III) Eu3+ chelates, followed by fluorescence measurement using time-resolved fluorometry. The performance of this TRFIA was evaluated using the clinical serum and compared with the commercial assays. The linear correlation coefficients (R2) of the cTnT and MYO standard curves were 0.9993 and 0.9995, respectively. The sensitivity for cTnT detection was 2.21 pg/mL (linear dynamic range was 3.24–963.71 pg/mL), and the average recovery was 100.57%. The sensitivity for MYO detection was 3.24 ng/mL (linear dynamic range was 4.95–976.85 ng/mL), and the average recovery was 99.79%. High correlation coefficients (R2) were obtained between the commercial assays and dual-label TRFIA (R2 = 0.999). The present dual-label TRFIA has high sensitivity, specificity, and accuracy in clinical sample analysis. It is a good alternative to the single-label diagnostic methods.
Keywords
Introduction
Acute myocardial infarction (AMI) is a major health problem caused by coronary atherosclerosis that leads to high rates of mortality and morbidity. Coronary atherosclerosis is a chronic heart disease characterized by stable and unstable periods. During unstable periods with activated inflammation in the vascular wall, patients may develop a myocardial infarction. 1 Annually, over 800,000 people in the United States experience an AMI, of which 27% result in mortality.2,3
Electrocardiogram (ECG) is the current method used to measure and diagnose abnormal rhythms of the heart and helps to diagnose heart disease. Unfortunately, ECG alone is often insufficient to diagnose early AMI despite its importance. 4 The main limitation of EGG is that significant ECG changes are absent in numerous AMI patients. In addition, ST segment deviation, which could signal an AMI, may also be observed in many other cardiac and noncardiac conditions, 5 and the sensitivity of ECG is only 20%. Therefore, ECG is not a reliable method for establishing early AMI. To overcome these shortcomings, cardiac biomarkers of AMI become good strategies.
In recent years, serum tests for biomarkers of AMI have emerged as a simple and attractive method to distinguish AMI. 6 There are many serum markers such as troponins T and I, creatine kinase (CK) and its isoenzyme MB isoforms (CK-MB), and myoglobin.7,8 Cardiac troponin T (cTnT) and cardiac troponin I (cTnI) are regarded as more sensitive and specific than other cardiac biomarkers for AMI detection. cTnT is a structural protein of the heart and has tissue specificity as well as high clinical sensitivity, thereby reflecting even microscopic zones of myocardial necrosis. Therefore, it is a highly sensitive biomarker.9,10 The high level of sensitivity, accuracy, and specificity makes cTnT a better candidate for early diagnosis of myocardial damage, which is fatal for cardiovascular disease patient therapy. 11
Myoglobin is a small (17.8-kD) cytoplasmic heme protein found in all muscles. Serum levels of myoglobin increase within 1 to 3 h in myocardial necrosis, usually peaks within 6 to 9 h, and comes back to normal within 24 h, 12 whereas troponins and creatine-kinase MB (CK-MB) are released after 3 and 6 h, respectively. 13 In addition, serum myoglobin levels of AMI increase sharply from 90 pg/mL to 250 ng/mL within 90 min. 14 Moreover, myoglobin possesses higher specificity and sensitivity and therefore is a better detector for AMI.15,16
Time-resolved fluoroimmunoassay is a sensitive, simple, and inexpensive detection method that requires relatively small sample volumes.17,18 In this study, a highly sensitive and specific dual-label time-resolved fluorescence immunoassay (TRFIA), using lanthanide europium(III) Sm3+ and samarium(III) Eu3+, was developed for the simultaneous determination of cTnT and myoglobin (MYO). The present dual-label TRFIA has high sensitivity, specificity, and accuracy in clinical sample analysis. It is another alternative to the commonly used single-labeled diagnostic methods.
Materials and Methods
Reagents and Chemicals
Two clones of monoclonal antibodies against different epitopes of cTnT (capture antibody 1C3 and detection antibody 4F1) or MYO (capture antibody 2A5 and detection antibody 3B2) were obtained from Youdi Biotechnology (Guangzhou, China). The cTnT and MYO standard were purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). Europium(III) (1244-302) and samarium(III) (1244-303) labeling kits were obtained from PerkinElmer (Waltham, MA). Bovine serum albumin (BSA) was obtained from Merck (Darmstadt, Germany). Centrifugal filters with a molecular weight cutoff of 10 KDa and 50 KDa were purchased from Millipore (Bedford, MA). A Sephadex G50 column was obtained from GE Healthcare (Uppsala, Sweden). The 96-well microplates were purchased from Corning (3585; Corning, New York, NY). The cTnT Test Kit and MYO Test Kit were purchased from Radiometer (AQT90 FLEX; Copenhagen, Denmark). One hundred clinical serum samples came from Shenzhen Chinese Traditional Medical Hospital and were aliquoted and freshly frozen in liquid nitrogen and stored at −80 °C. This study was approved by the Institutional Review Board of Shenzhen Chinese Traditional Medical Hospital, and all participants gave written informed consent.
Coating of Microplates
The 96-well microplates were coated with 200 µL/well of coating buffer (50 mmol/L carbonate, pH 9.6) containing 2.5 µg/mL and 2 µg/mL, respectively, of capture antibodies (monoclonal antibody for cTnT 1C3 and MYO 2A5) overnight at 4 °C. After coating, plates were washed three times with phosphate buffer containing 0.05% Tween-20 (v/v) followed by addition of blocking buffer 250 µL/well (50 mmol/L Tris-HCl containing 5% BSA, m/v, pH 7.4) for 2 h at 37 °C. The blocking buffer was then removed and the coated plates were dried in vacuum and stored at −20 °C.
Labeling Antibody
Labeling was performed using europium(III) Sm3+ and samarium(III) Eu3+ kits according to the manufacturer’s instructions. Briefly, 1 mg of detection antibody cTnT 4F1 was washed four times with labeling buffer (50 mmol/L Na2CO3, pH 9.0), suspended in 200 µL of labeling buffer, and mixed gently with 1000 µg of Sm3+ chelates. The mixture was incubated overnight at room temperature and purified using Tris-HCl (50 mmol/L, pH 7.8) buffered Sephadex G50 column. The conjugated detection antibody was preserved in Tris-HCl buffer (50 mmol/L, 0.1% BSA, m/v, pH 7.8) at 4 °C. MYO detection antibodies 3B2 were labeled with Eu3+ using the same procedure.
cTnT and MYO Standards
Pure reference cTnT and MYO standards were mixed in equivalent volume in the phosphate-buffered saline (PBS, 50 mmol/L Tris-HCl, pH 7.2, with 1.5% BSA and 0.15% NaN3) at final concentrations of 0, 1, 10, 100, 500, and 1000 pg/mL of cTnT standards and 0, 1, 10, 100, 500, and 1000 ng/mL of MYO standards.
Assay Procedure
Twenty-five microliters of standards, controls, or samples were added. Then, 200 µL of Sm3+- and Eu3+-labeled cTnT and MYO antibodies (1 µg/mL) was added, and the plate was incubated for another 1 h with continuous horizontal shaking at room temperature. After washing six times in PBS supplemented with 0.05% Tween 20, 200 µL/well of enhancement solution was added, and the plate was shaken gently for 5 min followed by the fluorescence readings (Auto DELFIA 1235; PerkinElmer). Sm3+ fluorescence was measured at an excitation wavelength of 340 nm and an emission wavelength of 600 nm. Eu3+ fluorescence was measured at an excitation wavelength of 337 nm and an emission wavelength of 615 nm.
Standard Curves and the Sensitivity Assays
The standard curves of the assay were evaluated using serial standard dilutions of cTnT (0, 1, 10, 100, 500, 1000 pg/mL) and MYO (0, 1, 10, 100, 500, 1000 ng/mL). We used the mean concentration plus 3 standard deviations (SD) of the blank assayed in duplicate in 20 independent measurements as the definition of limit of blank (LOB). The sensitivity was determined by using both the measured LOB and test replicates of a sample known to contain a low concentration of cTnT and MYO (limit of detection [LOD]: LOD = LOB + 1.645 [SDlow concentration sample]).
Accuracy Assays
The accuracy of the assay was evaluated by analyzing the low, median, and high concentrations of clinical samples. The intra- and interassay variations were obtained from 10 independent experiments.
Specificity Assays
For the specificity test, different concentrations of cTnT, MYO, cTnI, cardiac troponin C (cTnC), aspartate transaminase (AST), and lactate dehydrogenase (LDH) were added into the same serum matrix, respectively, and then the dual-label TRFIA measured the concentration of cTnT and MYO. Data were obtained from 10 independent experiments.
Recovery Assays
The recovery was assessed by adding different concentrations of cTnT and MYO standards to the three clinical samples. Recovery was expressed as the percentage measured of the amount added. The equation was as follows: Recovery (%) = (measured concentration/spiked concentration) × 100.
Methods Comparison and Practical Application of TRFIA Assays
Parallel tests comparing the dual-label TRFIA method with cTnT and MYO signal-label TRFIA kits were performed using 50 clinical samples from Shenzhen Chinese Traditional Medical Hospital. The Radiometer kit assay was performed according to the manufacturer’s instructions.
Statistical Analysis
Quantitative data were presented as mean ± SD. Comparisons between quantitative data were performed using the paired-sample t test, whereas correlations between groups were calculated with Spearman’s rank correlation. SPSS 19.0 (SPSS, Inc., an IBM Company, Chicago, IL) was used in the statistical analysis; p < 0.05 was considered statistically significant.
Results
TRFIA Standard Curves
Standard curves plotted for cTnT and MYO are shown in Figure 1 . The linear concentration ranges were 3.24 to 963.71 pg/mL for cTnT and 4.95 to 976.85 ng/mL for MYO. The linear correlation coefficients (R2) of the cTnT and MYO standard curves were 0.9993 and 0.9995, respectively. Both calibration plots exhibited well-defined linear relationships between the concentration and fluorescent intensity. The sensitivity (or LOD) of the assay for cTnT was 2.21 pg/mL, and the sensitivity for MYO was 3.24 ng/mL.
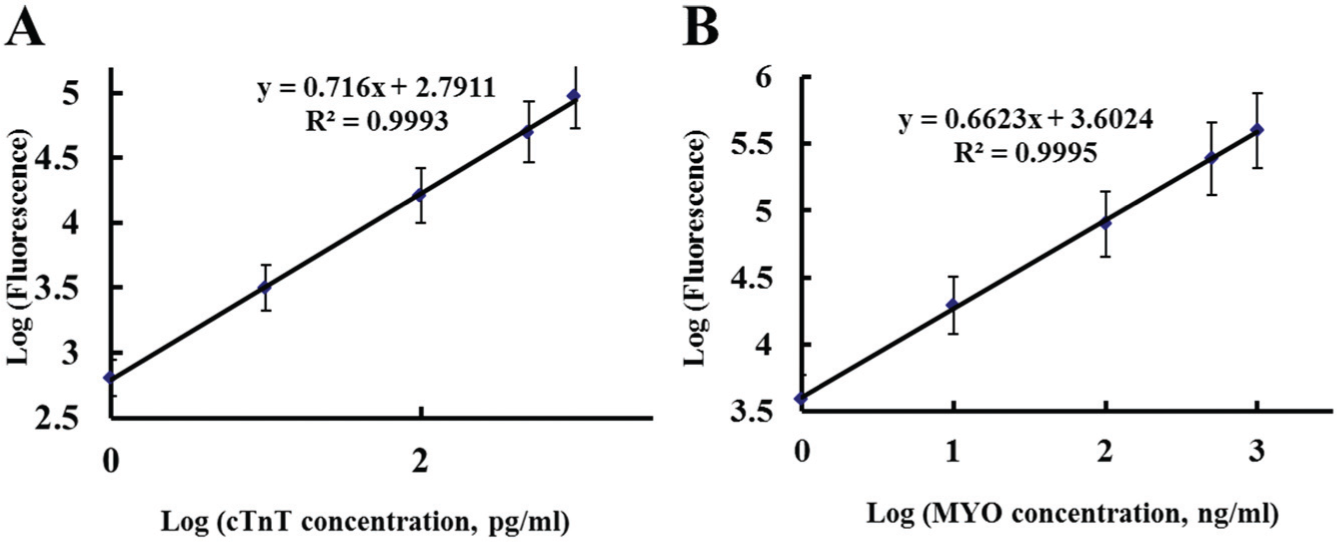
The standard curves of cardiac troponin T (cTnT) (
Accuracy
The accuracy was determined using three clinical samples with low, median, and high concentration of cTnT or MYO, and the results are shown in Table 1 . The intra-assay coefficients of variation (CVs) for cTnT were between 3.64% and 8.85%, and the interassay values ranged from 5.08% to 9.09%. The intra-assay CVs for MYO were between 3.90% and 8.14%, and the interassay CVs ranged from 6.07% to 8.18%. All coefficients of variation were less than 10%, indicating suitable performance of the assay.
Accuracy of the Dual-Label Time-Resolved Fluorescence Immunoassay.
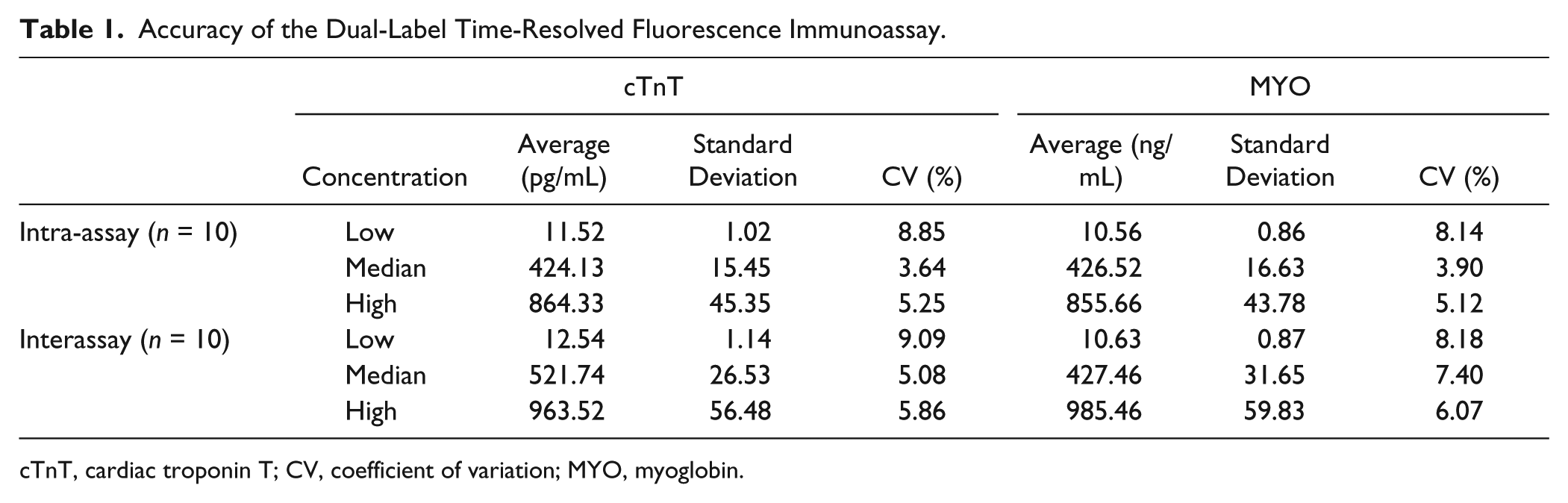
cTnT, cardiac troponin T; CV, coefficient of variation; MYO, myoglobin.
Specificity
Four proteins at various concentrations were investigated as potential interferents. As shown in Table 2 , the cross-reactivity was very low. The results showed that the present dual-labeled TRFIA had high affinity and specificity for cTnT and MYO.
Cross-Reactivities of the Dual-Label Time-Resolved Fluorescence Immunoassay (n = 10).
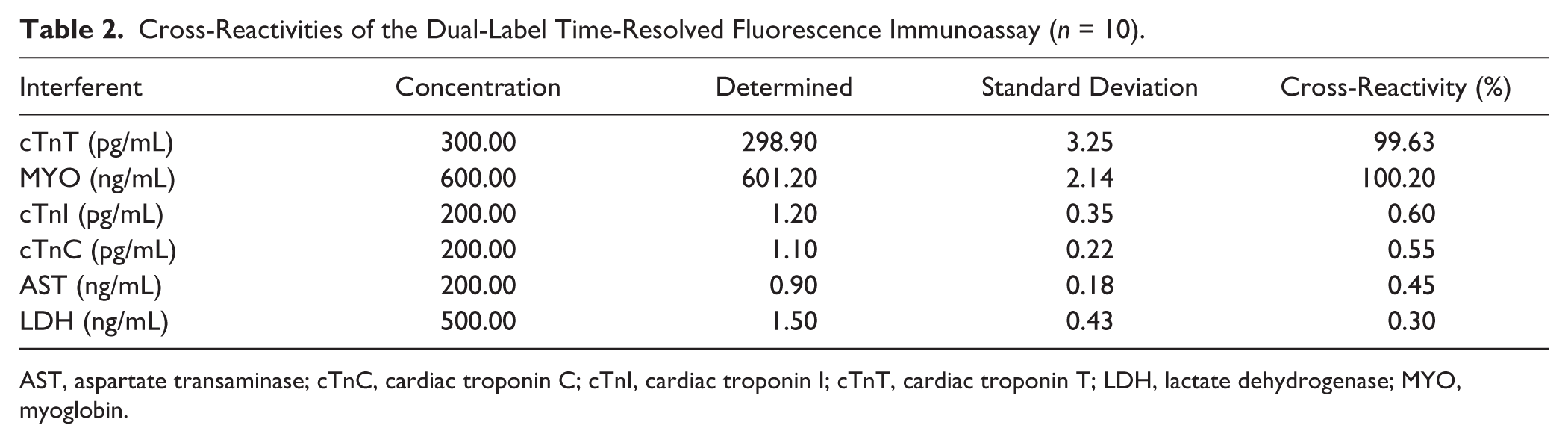
AST, aspartate transaminase; cTnC, cardiac troponin C; cTnI, cardiac troponin I; cTnT, cardiac troponin T; LDH, lactate dehydrogenase; MYO, myoglobin.
Recovery
Three clinical serum samples with known concentration of cTnT and MYO were used in this study. The initial concentrations of cTnT and MYO were 2.6 pg/mL, 252.4 pg/mL, 635.8 pg/mL and 7.5 ng/mL, 323.6 ng/mL, 665.4 ng/mL, respectively. After cTnT and MYO standards were added, the final concentrations of cTnT and MYO were 5.0 pg/mL, 500.0 pg/mL, 900.0 pg/mL and 10.0 ng/mL, 500.0 ng/mL, 1000.0 ng/mL, respectively. As shown in Table 3 , the average recoveries for cTnT were between 98.0% and 104.0%, while MYO values ranged from 97.0% to 103.0%. The results showed that this assay was free from interferences in serum.
Recoveries of the Dual-Label Time-Resolved Fluorescence Immunoassay.
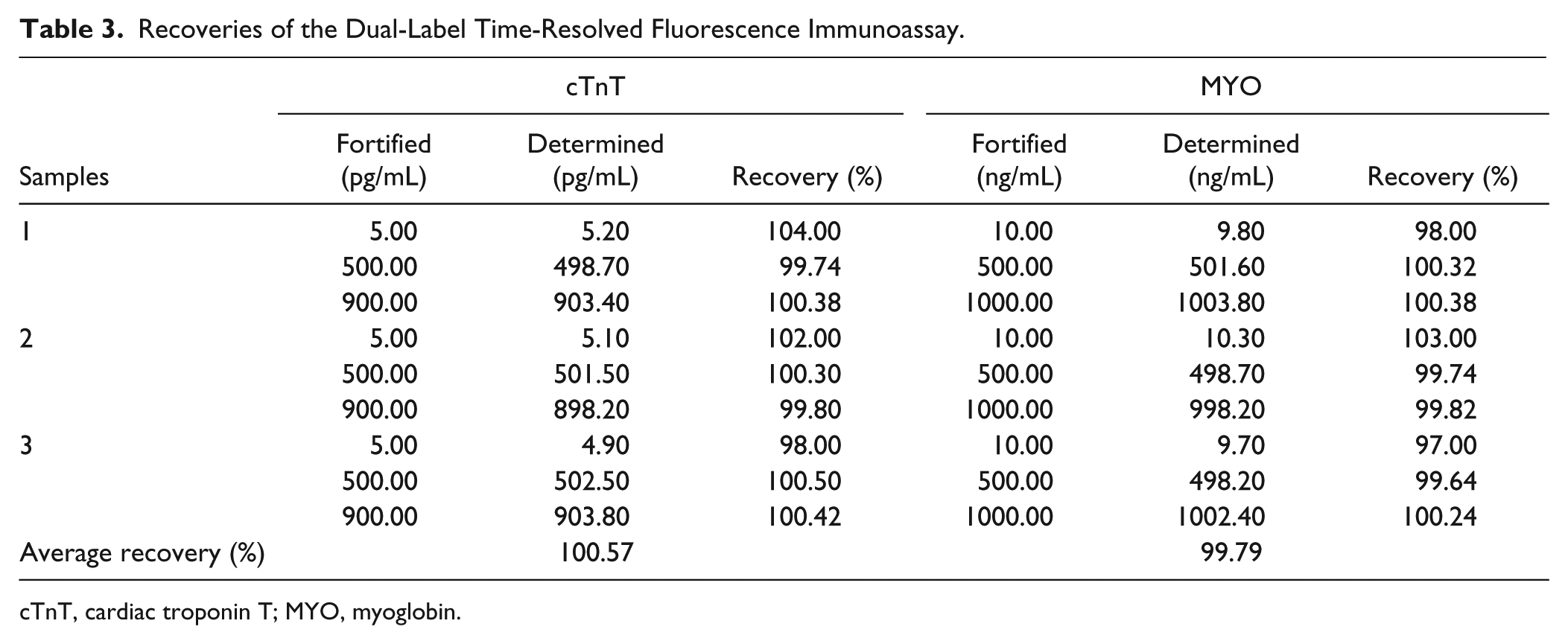
cTnT, cardiac troponin T; MYO, myoglobin.
Comparison with Commercial Kits
Fifty clinical samples were analyzed by the dual-labeled TRFIA and Radiometer kits, as shown in Figure 2 . High correlations between the methods were obtained. The calibration equation for cTnT was y = 0.983x + 8.611, with a correlation coefficient of 0.999 (p < 0.001). For MYO, the calibration equation was y = 0.999x − 0.093, with a correlation coefficient of 0.999 (p < 0.001). The results indicated that the dual-labeled TRFIA was a reliable method for the simultaneous determination of cTnT and MYO in clinical samples.
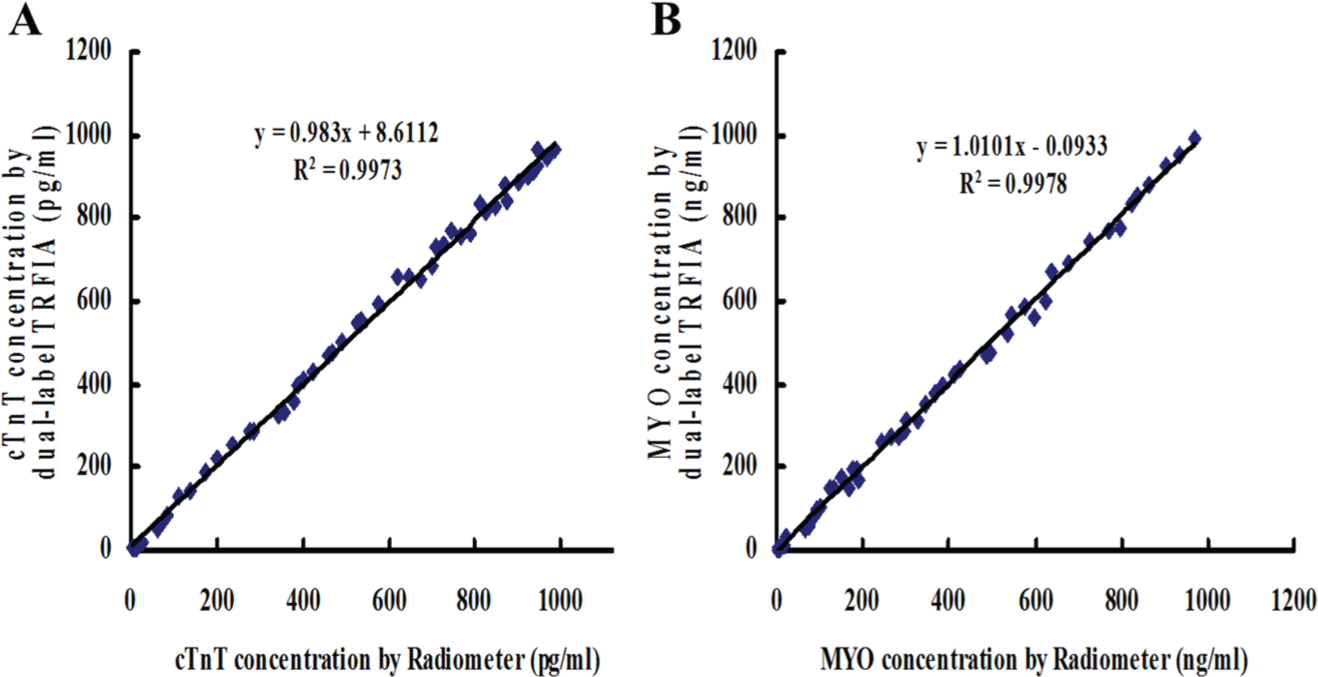
Correlation of cardiac troponin T (cTnT) (
Discussion
There are several causes of chest pain that can present as a myocardial infarction. Most notable include acute pericarditis, pulmonary embolism, acute aortic dissection, costochondritis, and gastroesophageal reflux disease. In addition, diagnosing early stage AMI patients with chest pain is urgently needed to initiate appropriate treatment. Hence, it is extremely necessary to develop strategies for rapidly and accurately detecting early AMI. In 1410 chest pain patients with suspected acute coronary syndromes, the quantitative measurement range of cTnT was 100 to 2000 pg/mL, with values below 50 pg/mL considered negative. For the MYO test, the quantitative measurement range was 30 to 700 ng/mL, with values below this range determined as low and values above as high. 19 To diagnose AMI, the sensitivities of all detection methods should reach levels of pg/mL for cTnT and ng/mL for MYO.
Common methods for detection of cTnT and MYO were nephelometric immunoassay, chemiluminescence, and immunochromatographic assay.20,21 These methods are time and labor consuming as well as high cost. In recent years, the research on cTnT detection using biosensor technology has been a popular study point. Surface plasmon resonance, dual quartz crystal microbalance-based biosensors, and conductometric and electrochemical-based biosensors have been successfully investigated for the detection of cTnT by using various nanomaterials and surface chemistries to develop a highly sensitive immunosensor. 22 However, the LOD for the immunosensor only reaches the ng/mL level and requires specialized technical personnel and equipment. 23 In a commercially available MYO kit (immunoturbidimetric assay), the upper limit of the reference interval was 75 ng/mL, with a sensitivity cutoff value below 50 ng/mL. The detection sensitivity of cTnT and MYO needs to be improved. In addition, AMI detection may be enhanced by the use of two targets together instead of just one. Therefore, our dual-label TRFIA may be much more useful and important, and to save time for AMI patients, a simultaneous detection for cTnT and MYO is urgently needed.
TRFIA has been widely used in various fields, such as clinical research, molecular biology, and drug analysis. 24 The unique fluorescent properties of lanthanide chelates render TRFIA with many advantages, such as narrow emission peaks and high quantum yields. 25 Conventional fluorescent labels have broad spectra and backgrounds, which are not suitable for multianalyte assays. Accordingly, the combined usage of cTnT and MYO only within 1 to 2 h could get earlier therapeutic decisions, and the sensitivity is 2.21 pg/mL for cTnT and 3.24 ng/mL for MYO. There is no obvious cross-reaction with potential interferents. In comparison with commercial kits, the present dual-labeled TRFIA is a reliable method for the simultaneous determination of cTnT and MYO in clinical samples. Therefore, our research provides a convenient, highly sensitive, and fast method for the early diagnosis of AMI.
In conclusion, a simple, sensitive, and fast dual-label fluoroimmunoassay was developed for the determination of cTnT and MYO for AMI patients. This method has an advantage to be used for the early diagnosis of the AMI patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article is supported by the National Natural Science Foundation–funded project of 2015 (NO. 81573922), science and technology project of Guangdong Province of 2015 (NO. 2014A020221002), and Guangdong Medical Scientific Research Fund of 2016(NO.A2016231).
