Abstract
MicroRNAs (miRNAs) are globally dysregulated in human carcinomas. However, the specific miRNAs that mediate gastric cancer metastasis have not been identified. We identified 100 miRNAs that are dysregulated in gastric cancer and used a self-assembled cell microarray method to systematically evaluate their capacity to regulate cell migration. MiR-451, which is down-regulated in human gastric cancer samples, potently modulated multiple metastatic phenotypes including cell migration, invasion, proliferation, and epithelial-mesenchymal transition. These effects were achieved via down-regulation of the miR-451 target gene, ERK2. These findings provide new insight into the physiological effects of and potential therapeutic uses for miRNAs in gastric cancer.
Introduction
Gastric cancer (GC) is the second leading cause of cancer-related mortality worldwide, 1 particularly in eastern Asia. 2 Over the past decade, treatment strategies for this disease have improved. 3 However, the mortality rate of patients with advanced GC remains very high because of cancer metastasis. 4 The concept of metastasis as a multistep process is an active area of study, 5 and some famous metastasis-promoting and -suppressing genes include c-Myc 6 and TP53. 7 However, the molecular mechanisms that underlie metastatic processes remain poorly understood. Therefore, further study of the signaling pathways and molecular mechanisms that underlie GC metastasis is imperative for the improvement of GC therapies.
A new class of signaling molecules known as microRNAs (miRNAs) negatively regulates gene expression, typically by binding to the 3′UTR of target mRNAs. 8 A small number of miRNAs have been demonstrated to actively participate in the regulation of tumor development.9–13 In addition, recent studies have also identified specific roles for miRNAs in cancer metastasis, 14 and there are new hopes that we can interpret the molecular pathways underlying each step through miRNAs. Moreover, miRNAs have also been found to be involved in cancer pathogenesis, 15 indicating that miRNAs may play a central regulatory role in the initiation and progression of most cancers analyzed thus far. 13 Strikingly, miRNAs were found to be globally dysregulated in human cancer samples 16 and may be used as potential biomarkers for GC metastasis.17,18 However, the specific miRNAs that mediate GC metastasis have not been identified. To address this question, we identified 100 miRNAs known to be dysregulated in GC19,20 and used a high-throughput screen to identify which of these factors altered cell migration as a functional surrogate endpoint for cancer metastasis. 21
Although high-throughput, cell-based RNA interference screens are routinely performed in 96- or 384-well plate formats, these screens are not suitable for the analysis of cell migration because of the high cost and sophisticated techniques required for these assays. Recently, a novel approach called Self-Assembly Cell Microarray (SAMcell) has been successfully implemented to screen for miRNAs or siRNAs that influence cell migration.22–24 In the present study, we identified 100 miRNAs known to be dysregulated in GC and then used the SAMcell method to determine which of these miRNAs influenced cell migration in the SGC-7901 human GC cell line. To this end, we identified miR-451 as a potent regulator of cell migration.
Although miR-451 was previously identified as a novel prognostic marker in human GC, 25 its target gene remained unknown. In the present study, we found that miR-451 mediates various steps in the metastatic process including cell migration, invasion, and epithelial-mesenchymal transition (EMT) by repressing the target gene ERK2, which is a component of the MAPK signaling pathway and is known to promote cancer metastasis.26–28
Materials and Methods
Cell Culture and Transfection
The SGC-7901 cell line was obtained from the American Type Culture Collection (ATCC; Manassas, VA) and was cultured in Dulbecco’s Modified Eagle’s Medium containing 10% fetal bovine serum, 100 U mL−1 penicillin and 0.1 mg mL−1 streptomycin under humidified conditions of 95% air and 5% CO2 at 37 °C. Transfection protocols were performed with the lipofectamine RNAiMAX (Invitrogen, Carlsbad, CA) transfection reagent according to the manufacturer’s instructions.
Fabrication of SAMcell
The SAMcell fabrication protocol was performed as previously described. 23 Briefly, the slides were covered with poly(N-isopropylacrylamide) and etched via a shadow mask by oxygen plasma. After ultraviolet (UV) sterilization, reverse transfection reagent containing miRNA mimics was printed on each chip using a nanodispenser (Phoenix, Art Robbins Instruments, Sunnyvale, CA). Next, the slides were fixed in a six-well plate with melted wax. Approximately 3 mL of 37 °C culture medium containing 5 × 105 cells was transferred to each well. Approximately 20 h later, the dishes were moved to room temperature for 5 min and were washed with phosphate-buffered saline (PBS) three times to ensure total removal of the polymer.
Transwell Migration and Invasion Assay
For migration assays, SGC-7901 cells were seeded into the upper chamber of a transwell insert (pore size, 8 µm; Costar, Corning, NY) in 100 µL of serum-free medium per well. In total, 600 µL of medium containing 10% serum was placed into the lower chamber to act as a chemoattractant. At the conclusion of the assay, nonmigratory cells were removed from the upper chamber using a cotton swab. The cells remaining on the lower surface of the insert were then fixed with 4% formaldehyde (Sigma-Aldrich, St. Louis, MO) and stained with DAPI (Roche, Basle, Switzerland). For invasion assays, cells were seeded in a Matrigel (BioRad, Hercules, CA) -coated chamber and were incubated at 37 °C.
Cell Proliferation Assay
SGC-7901 cells were seeded in a 96-well plate at a density of 3 × 103 cells per well. After 1 day, cells were transfected with the indicated miRNAs or siRNAs. Two days later, cells were incubated with 100 µL of fresh medium containing 10% CCK-8 reagent (DoJinDo Laboratories, Tokyo, Japan) for 1 h at 37 °C. Then, the absorbance values of the wells at 450 nm were detected using an automatic spectrometer (Multimode Reader, Enspire). This procedure was repeated 4, 5, 6, and 7 days after cell seeding. The normalized absorbance values of different samples at each time point were plotted in one figure.
Immunofluorescence
Cells were seeded onto sterile cover slides and allowed to attach overnight. The cells were then fixed with 4% formaldehyde, permeabilized with 0.1% Triton X-100, and blocked with 2% bovine serum albumin for 1 h at room temperature. The expression of E-cadherin and vimentin was examined using primary antibodies (anti-E-cadherin: ab76055; abcam, Cambridge, MA) (anti-vimentin: ab8978; abcam) and visualized using an anti-rabbit IgG (H + L) F(ab′)2 fragment (Alexa Fluor 488 conjugate; Cell Signaling Technologies, Danvers, MA). All antibodies were diluted 1:200. Cell nuclei were stained with DAPI. Immunofluorescence was examined using a fluorescence microscope (EcliPSE Ti-U, Nikon, Tokyo, Japan).
Luciferase Assay
For the luciferase reporter assay, 4.0 × 104 SGC-7901 cells were co-transfected with 200 ng of miRNA mimics, together with 200 ng of the indicated pGL3 firefly luciferase construct and 20 ng of a normalization control pGL3 Renilla luciferase construct. The culture medium was changed 6 h posttransfection, and luciferase activity was measured after 48 h using the dual luciferase reporter assay system (Promega, Madison, WI).
qRT-PCR Assay
The total RNA was extracted from cell lines and tissue samples using TRNzol (Tiangen, Beijing, China) according to the manufacturer’s protocol. The concentration of the total RNA was quantified by measuring the absorbance at 260 nm with a NanoDrop Spectrophotometer (ND-1000, ThermoFisher, Waltham, MA). The reverse transcriptase reaction for the formation of cDNA from total RNA was performed in a 20 µL volume and contained 2 µg of total RNA, 2.5 µM primers (both random primers for ERK2 and a specific primer for miR-451; RiboBio, Guangzhou, China), 200 U PrimeScript reverse transcriptase (Takara, Dalian, China), 0.5 mM dNTPs, and reaction buffer (pH 8.3, 50 mM Tris-HCl, 75 mM KCl, and 3 mM MgCl2). The reaction was incubated at 42 °C for 60 min, followed by an incubation at 95 °C for 5 min to inactivate the enzyme. Real-time PCR was performed on an ABI 7300 (Life Technologies, Carlsbad, CA). The dsRNA PCR templates, 10 µL of Trans-StartTM SYBR Green qPCR Supermix (TransGen Biotech, Beijing, China), and 0.4 µL each of 10 µM forward and reverse primers were mixed, and water was added to each reaction to a final volume of 20 µL. The reactions were incubated in a 96-well plate at 95 °C for 2 min, followed by 40 cycles at 94 °C for 15 s, 60 °C for 31 s, and 72 °C for 15 s. All reactions were run in triplicate. The threshold cycle (CT) was defined as the cycle number at which the fluorescence exceeded the fixed threshold.
Immunoblotting
The lysates were resolved by electrophoresis, transferred to a poly-vinylidene difluoride membrane (Millipore Corporation, Billerica, MA), and probed with antibodies against ERK2 (ab32081; abcam) or GAPDH (ab9485; abcam). The antibodies were diluted 1:1000.
Human Gastric Samples
All human gastric samples were obtained from the First Affiliated Hospital of Soochow University. Before surgery, all of the patients provided written informed consent to donate any excess tissue to research. None of the patients received radiotherapy or chemotherapy before surgery. In total, 20 pairs of fresh tumor and paraneoplastic tissue samples were obtained, snap frozen, and stored in liquid nitrogen.
Statistical Analysis
All of the results are expressed as the means or totals from independent experiments. When comparing two groups, the two-tailed Student’s unpaired t test was used. For all of the tests, a p value <0.05 was considered significant. The Benjamini and Hochberg false discovery rate was used as a correction for multiple comparisons. A * indicates p < 0.05; ** indicates p < 0.01. Error bars represent the standard deviations of at least three independent experiments.
Results
High-Throughput Screening Identifies miR-451 as a Negative Regulator of GC Cell Migration
To identify miRNAs that mediate GC metastasis, we designed the study workflow depicted in Figure 1A . We conducted loss-of-function screening using the highly metastatic SGC-7901 GC cell line and selected 100 candidate miRNAs, most of which have been reported to be dysregulated in GC.19,20 We performed cell migration screening using the aforementioned SAMcell method. 23 Briefly, we first printed a reverse transfection mixture containing miRNA mimics or scrambled siRNAs onto each chip and seeded cells on top of this mixture. After release of the polymer, cell islands were recorded by microscopy (Image Xpress, Molecular Devices, Sunnyvale, CA) immediately and after 18 h. Each experiment was performed in quadruplicate, and the mean of each group was recorded. The areas of the cell islands increased as a linear function of time within a given period, such that the slopes of the areas represented the migratory activities of each island. 22 To calculate the migratory activity, the migration fold change of cells transfected with miRNA mimics was normalized to that of scrambled siRNA-transfected controls. The p values were then adjusted using the Benjamini Hochberg false discovery rate method to determine statistical significance. The results from this screen are summarized in a volcano plot ( Fig. 1B ).
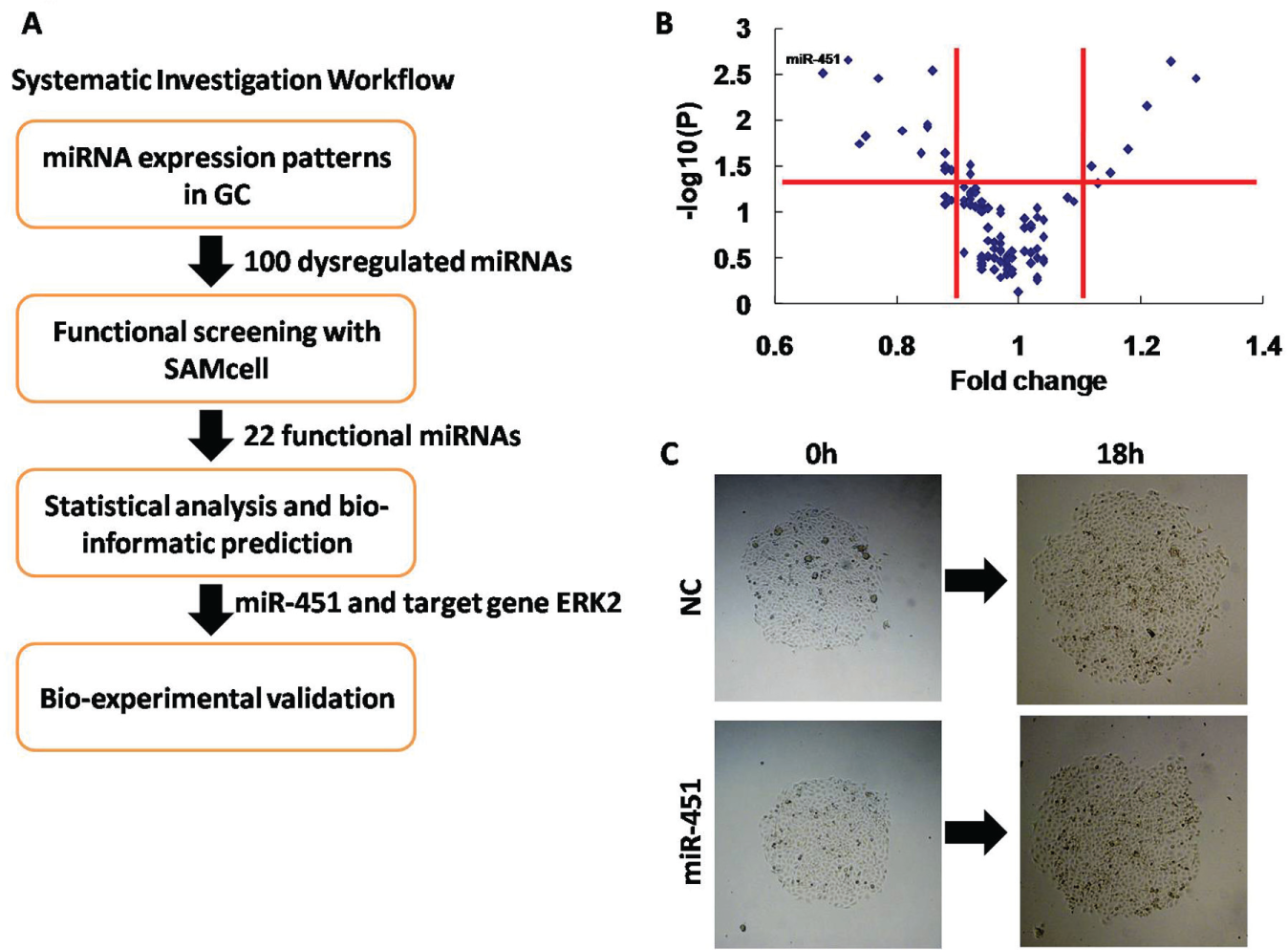
SAMcell functional screening in SGC-7901 cells identifies miR-451 as a negative regulator of gastric cancer metastasis. (
In total, 22 functional miRNAs were identified, including 15 miRNAs that suppressed migration and 7 miRNAs that promoted migration
MiR-451 Inhibits Metastatic Phenotypes in GC Cells
To investigate the role of miR-451 in GC metastasis, functional assays were performed in the highly metastatic SGC-7901 cell line. When miR-451 and negative control (NC) mimics were transfected into SGC-7901 cells individually, by transwell migration and invasion assay, the migratory and invasive capabilities of the cells with a high expression of miR-451 were almost 0.5 times lower than those of NC ( Fig. 2A ).
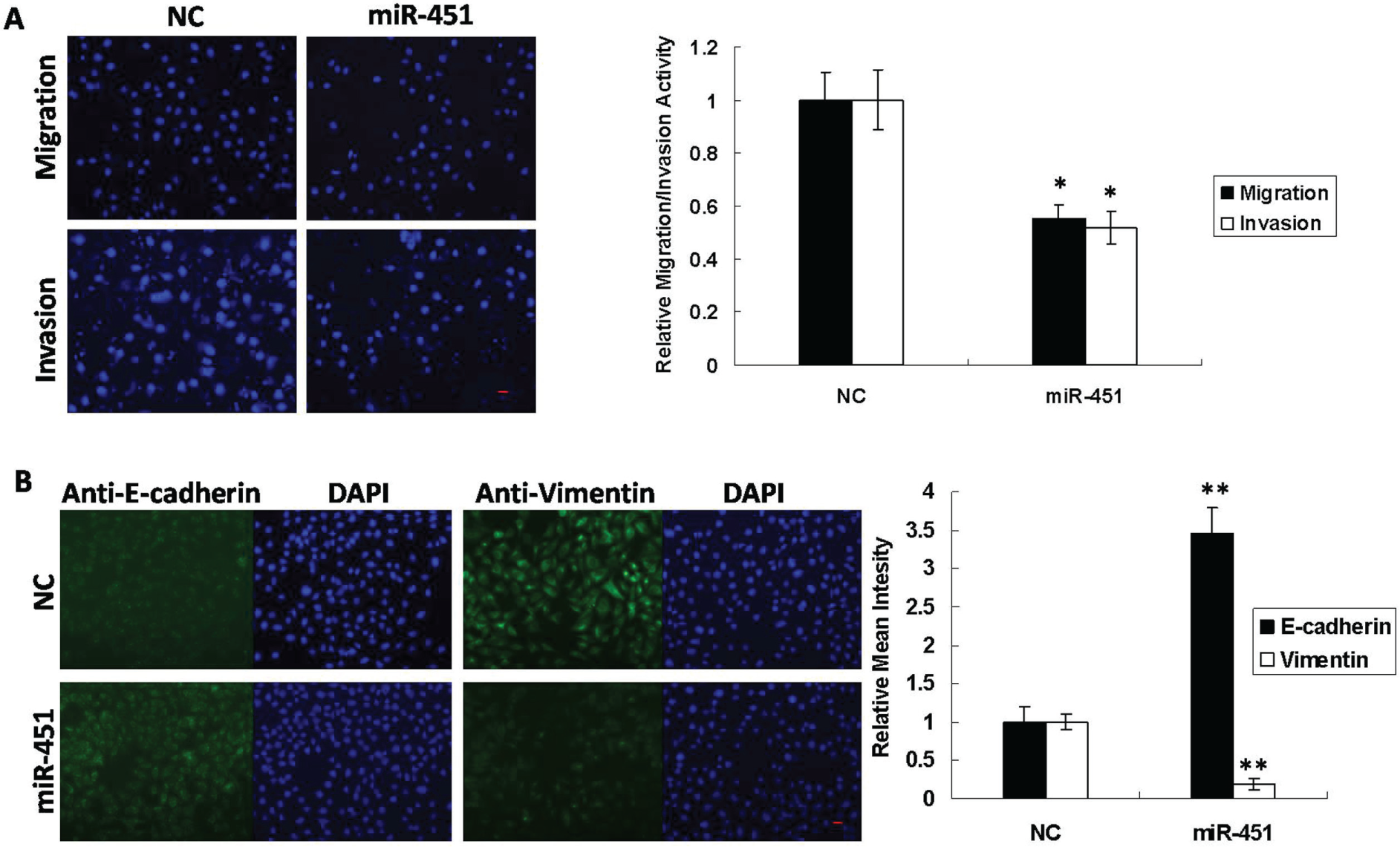
MiR-451 modulates metastatic phenotypes in gastric cancer cells. (
Next, given that the EMT is an essential early step in cancer metastasis,
31
we investigated whether miR-451 might mediate this process. Immunofluorescent staining revealed that overexpression of miR-451 led to an increase in E-cadherin expression as well as a decrease in vimentin expression (
Fig. 2B
). In addition, we also examined cell proliferation using the CCK-8 assay and found that the overexpression of miR-451 inhibited cell proliferation by 20% compared with control cells (
MiR-451 Down-Regulates Target Gene ERK2
To determine the molecular mechanisms by which miR-451 suppresses metastatic phenotypes, we identified ERK2 as a putative miR-451 target gene using the RNA22 algorithm
32
(
Fig. 3A
). To verify whether ERK2 was, indeed, an miR-451 target gene, the ERK2 3′-untranslated region (UTR) was cloned into a luciferase reporter construct (pGL3; Promega). Compared with cells transfected with an NC mimic, overexpression of miR-451 reduced luciferase activity by 65% (
Fig. 3C
). In addition, when the miR-451 binding site in ERK2 was mutated, the luciferase activity was restored (
Fig. 3B
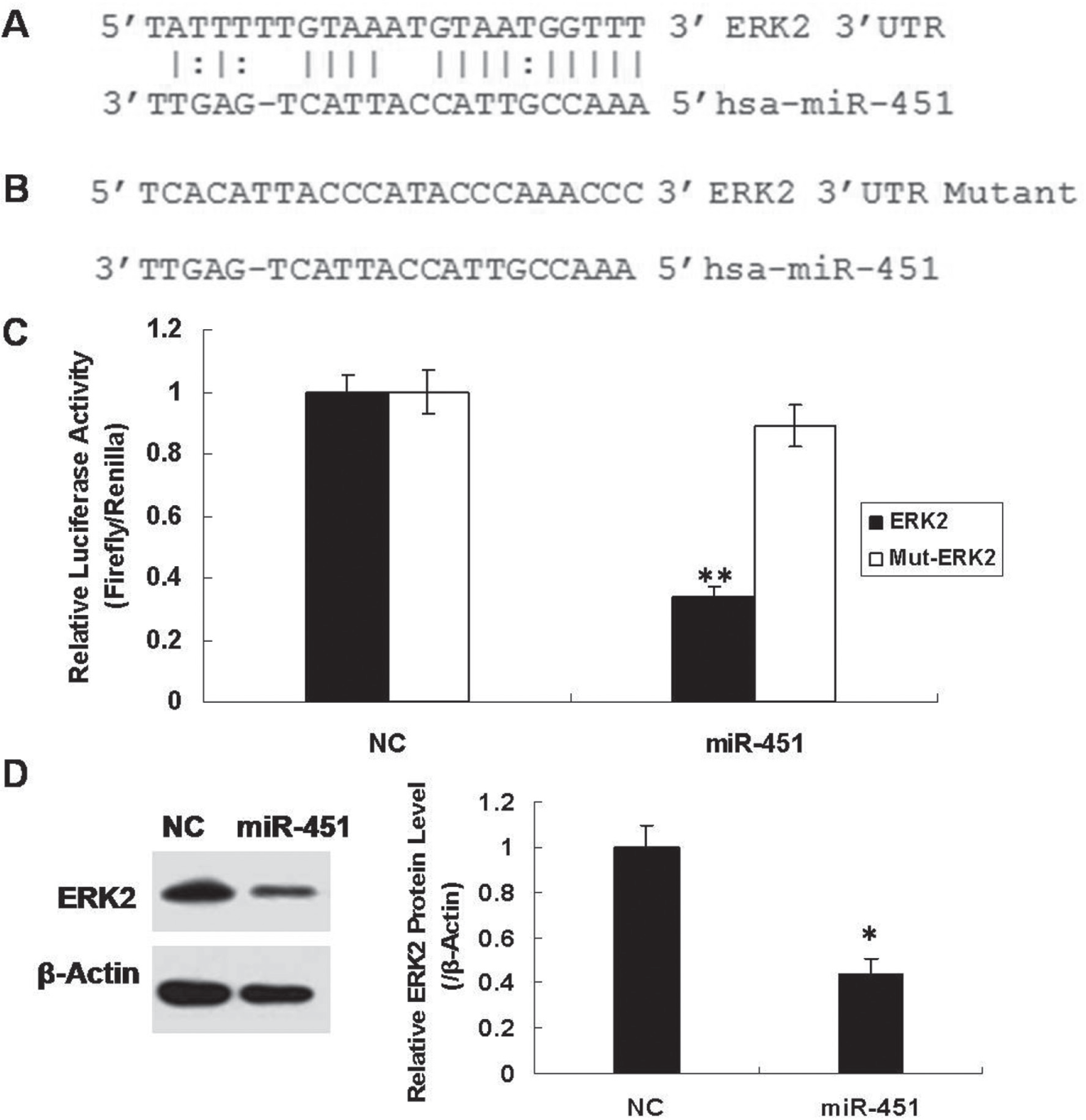
MiR-451 directly regulates ERK2. (
Silencing of ERK2 Inhibits Metastatic Phenotypes in GC Cells
To investigate the role of ERK2 in GC metastasis, we first synthesized three siRNAs against ERK2 and determined their silencing efficacy at both the mRNA and protein levels (
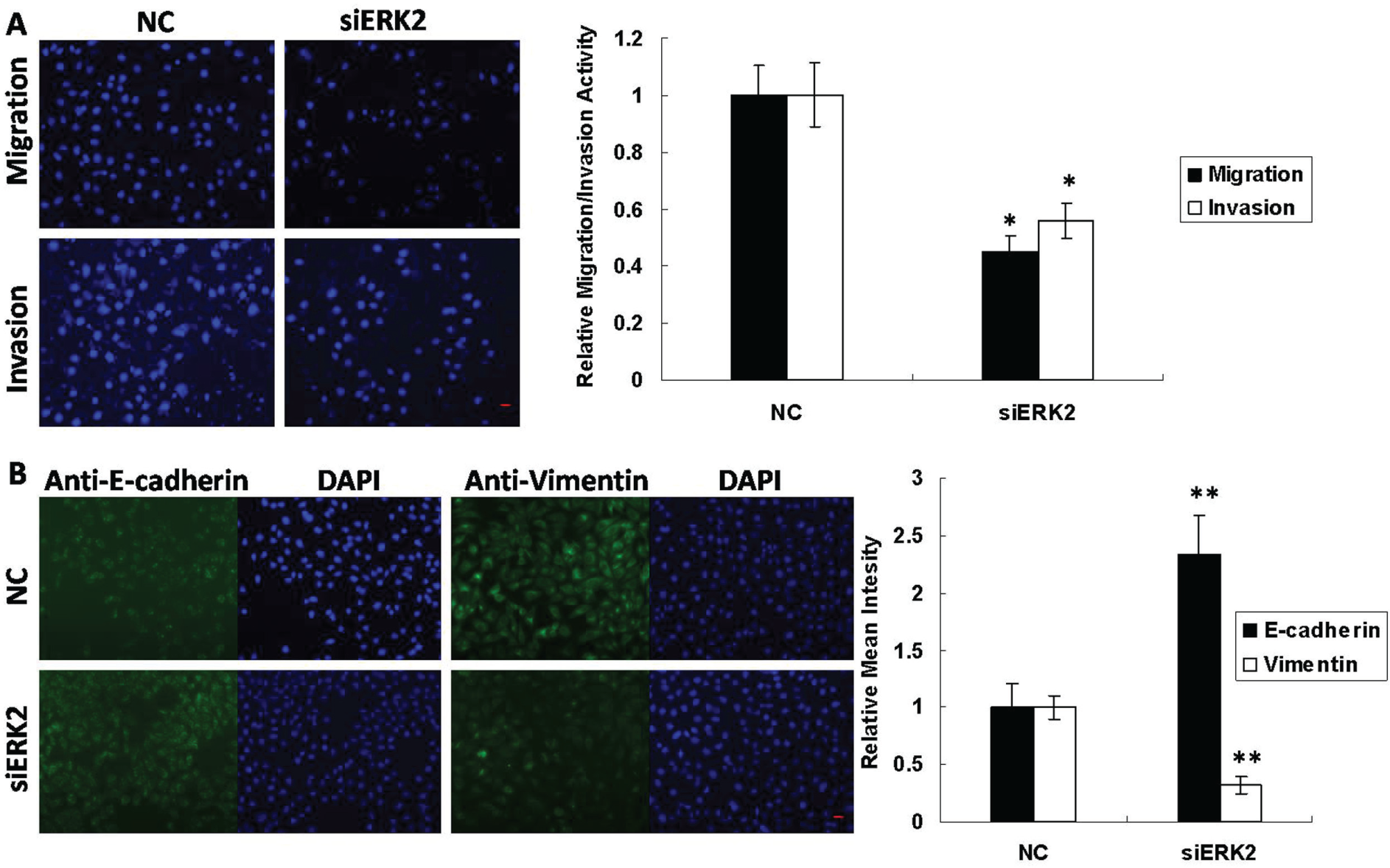
Silencing of ERK2 modulates metastatic phenotypes in gastric cancer cells. (
Expression Patterns of miR-451 and ERK2 in Human GC Samples
To evaluate the prognostic value of miR-451 in GC, we collected 20 pairs of human GC and cancer-adjacent normal tissue samples. Using qRT-PCR, we observed that the mean expression of miR-451 was reduced in GC samples relative to cancer-adjacent normal tissue (n = 20, p = 5.12 × 10−5; Fig. 5 ), suggesting a significant association between the expression levels of miR-451 and early stages of GC progression. Furthermore, we also detected protein expression of ERK2 in GC samples. Pairwise comparisons of ERK2 protein expression between tumor and cancer-adjacent normal tissue samples from six patients indicated that ERK2 expression was significantly lifted in GC.
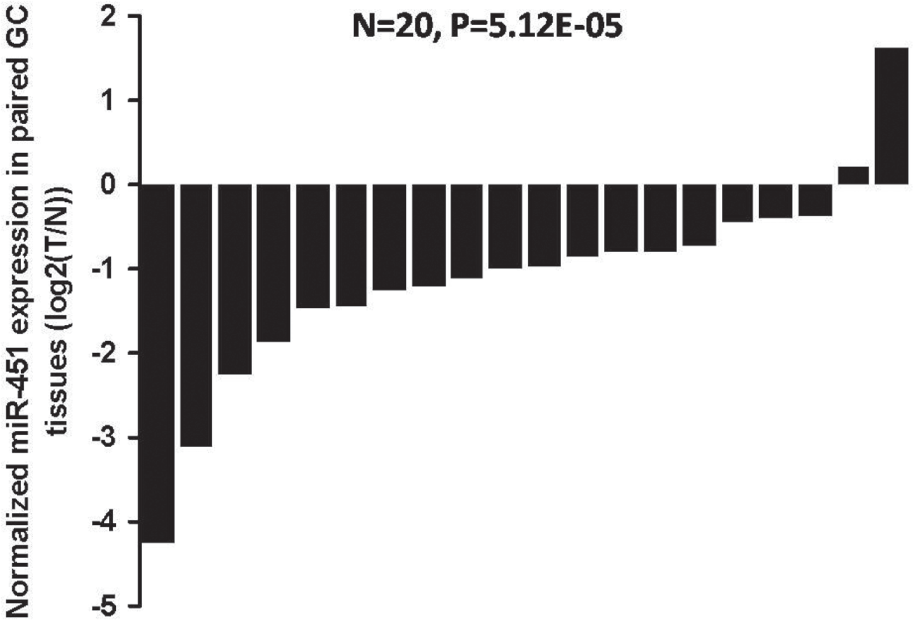
Expression patterns of miR-451 in human gastric cancer (GC) tissue samples. Pairwise comparisons of miR-451 expression between tumor (T) and cancer-adjacent normal (N) tissue samples indicated that miR-451 expression was significantly reduced in GC.
Discussion
It is well established that miRNAs participate in carcinogenesis. However, it is essential to identify miRNAs with potential therapeutic value. 33 In this study, we systematically investigated the effects of individual miRNAs on GC cell migration. High-throughput screening is a powerful tool for identifying the roles of large quantities of genes in a short period of time, and it would probably help us scratch the surface of the cancer metastatic process. Traditional screening methods for cell migration, such as the Transwell and wound-healing assays, are not suitable for high-throughput screening because of low throughput, poor efficiency, and sophisticated operations. However, the SAMcell method integrates microfabrication technology, reverse-transfection technology, and RNA interference technology and investigates cell behavior directly on a cell chip. To perform the migration screen on a large scale, we used the SAMcell method, which demonstrates certain important advantages, including higher throughput and accuracy, compared with the traditional Transwell assay. Functional analyses such as these improve on the conventional differential expression strategies.
From previous reports, we identified 100 miRNAs known to be dysregulated in GC and performed functional screening on these miRNAs. MiR-451 was identified as the most powerful regulator capable of reducing cell migration. We believe that this functional pattern provides an overview of miRNAs that regulate metastatic phenotypes in GC and will therefore contribute to the identification of physiologically important miRNAs with therapeutic relevance. Other miRNAs that have been previously shown to alter GC cell function were also represented in our screening results (
In addition, our functional and mechanistic analyses demonstrated that miR-451 negatively regulates GC cell migration, invasion, proliferation, and EMT processes by repressing ERK2 expression. In fact, the tumor-suppressive functions of miR-451 have been reported in recent decades. Specifically, Bandres et al. 36 initially found that miR-451 expression was decreased in both gastric and colorectal cancer compared with noncancerous tissues and that its down-regulation was associated with a worse prognosis. Furthermore, Su et al. 25 identified miR-451 as a novel prognostic marker and potential therapeutic target in human GC, which is consistent with the results of our study. However, in this previous work, the downstream molecular mechanism mediated by miR-451 was unclear.
In this study, we demonstrated that miR-451 suppressed metastatic phenotypes through the down-regulation of its target gene, ERK2. This 42 kDa protein was first identified by Boulton et al. 37 in the 1990s and is a member of the serine/threonine family of protein kinases that is activated by phosphorylation in response to insulin and NGF. Phosphorylated ERK2 is then translocated to the nucleus, where it regulates the activity of transcription factors such as AP1, NF-kappa B, Jun, c-Myc, Fos, Atf2, and Max. 26 In agreement with our findings, previous reports have also demonstrated that ERK2 mediates metastatic phenotypes in many tumor types, such as breast cancer, 38 oral cancer, 27 and melanoma. 39
In summary, the results of this study have important significance for our understanding of how dysregulated miRNAs influence human GC progression and promote metastasis. We believe that these findings provide new insight into the physiological and therapeutic importance of miRNAs in human gastric cancer.
Footnotes
Acknowledgements
We thank Suzhou Genepharma Biotech for the miRNA mimics and for assistance with siRNA design and synthesis.
Supplementary material for this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the National Natural Science Foundation of China (grant 31270919).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
