Abstract
Since the adoption of Labcyte Echo Acoustic Droplet Ejection (ADE) technology by AstraZeneca in 2005, ADE has become the preferred method for compound dosing into both biochemical and cell-based assays across AstraZeneca research and development globally. The initial implementation of Echos and the direct dosing workflow provided AstraZeneca with a unique set of challenges. In this article, we outline how direct Echo dosing has evolved over the past decade in AstraZeneca. We describe the practical challenges of applying ADE technology to 96-well, 384-well, and 1536-well assays and how AstraZeneca developed and applied software and robotic solutions to generate fully automated and effective cell-based assay workflows.
Introduction
Acoustic droplet ejection (ADE) technology uses sound energy to transfer small-volume liquid droplets between reservoirs and/or microtiter plates, and a detailed description of the technology is provided in this journal edition by Ellson et al. 1 ADE has been used in life sciences applications for over a decade, 2 and the benefits of using ADE technology to dose compounds into biological assays (particularly compared with using aqueous serial dilutions) have been described extensively elsewhere. 3 The small-volume transfers enabled by ADE mitigate the impact of the use of DMSO on biological assays. DMSO is the routine solvent for pharmaceutical small-molecule compound collections; however, DMSO has been shown to induce a range of effects on cells, including differentiation, growth arrest, and reduced cell viability,4–6 and DMSO tolerance in cell-based assays is typically 0.1% to 0.5% v/v. Miniaturization of assays into 384-well and 1536-well formats is also enabled. In addition to small-volume capability, ADE provides significant advantages in the form of noncontact dispensing. This avoids problems observed with both manual handling and conventional automated liquid handling, including compound adsorption into plastic or metal pipette tips, and compound carryover during serial dilution. The elimination of compound carryover/loss and the high level of accuracy and precision provided by ADE have been shown to offer an improvement in assay IC50 data quality, including a reduction in false negatives and loss of potency.7,8 The most significant improvement in data quality is observed when comparing ADE dosing to performing manual serial dilutions in buffer or assay media.8,9 When serial dilutions are automated and performed in 100% DMSO and then transferred into an assay plate, errors associated with compound carryover/loss are reduced 10 but potentially not eliminated. 9
In 2004, emerging screening data from AstraZeneca Oncology in Alderley Park (AP) indicated that low data consistency was observed in projects working with compound series with high lipophilicity. Further investigation led to the identification of a number of liquid handling factors during compound solubilization, storage, and dilutions during the screening workflow, which were exacerbated by increasing lipophilicity of test compounds. As a result, a number of projects were closed due to the lack of confidence of the screening data and failure to establish structure-activity relationships (SARs) in the project medicinal chemistry effort. The AP Oncology group deployed extensive effort to search for a standard compound handling workflow to resolve all critical issues relating to compound storage and dilution/dosing workflows to prevent similar impact to future projects. A large number of liquid handling platforms were evaluated, but none were found to eliminate the cross-contamination and compound precipitation issues observed when DMSO-solubilized compounds were diluted into aqueous reagents, such as cell culture medium or other diluents. Incidentally, in 2004, Labcyte (Sunnyvale, CA) introduced its novel ADE platform, the Echo 550, targeting the compound management market, and the AP Oncology group initiated a collaboration with Labcyte to explore the application of the Echo 550 to dispense DMSO-solubilized compounds directly into inverted cell and culture media–containing microplates. As a result of the successful collaboration, AstraZeneca invested in Echo 550 systems for its research and development (R&D) sites at AP and Wilmington, and the AP Oncology group adopted the Echo 550 as its standard compound handling workhorse, replacing the entire fleet of automated XYZ liquid handlers throughout the group. In addition to the scientific rationale described, the financial benefit of moving to acoustic dispensing was also considerable. A single acoustic dispenser replaced four traditional aqueous liquid handling systems, reducing servicing costs, footprint, and overhead within the laboratory. In addition, there was a reduction in DMSO utilization, reducing revenue spend by $160K per year and significantly reducing the volume of both solvent and plastic waste within screening. Despite the relatively high capital cost, the return on investment for a single acoustic dispenser was shown to be less than 24 months.
Echos were first implemented in the Oncology cell biology group in AP for cell-based IC50 determination assays 11 and in Wilmington for the creation of predosed plates for both biochemical and cellular assays. 12 Prior to this in AP, most cell assays were performed using manual compound dilution and dosing and resourced in a decentralized, project-centric model, with around 2000 compound IC50 results generated annually across the AP Oncology portfolio. Biochemical IC50 determination assays were already performed in a centralized team within AP Oncology using automation and automated compound plate dilution processes. These processes originally employed contact dilution and dispensing systems, but all biochemical assays in AP moved to Echo-dosed plates by 2007. 8
The Echo acoustically transfers 2.5-nL droplets from any well in a source compound plate to any well in a destination assay plate, transferring multiple droplets to achieve larger final volumes/concentrations (therefore, all Echo dispense volumes must be a multiple of 2.5 nL). The maximum volume to be transferred into the assay plate is limited to the DMSO sensitivity of the assay, with typical ranges of 1% to 10% in biochemical assays and 0.1% to 0.5% in cell-based assays. Within this working volume range of 2.5 to 500 nL in a 100-µL cell-based assay, a 5-point half-log dilution series can be achieved from a single stock concentration (e.g., dispensing 250, 75, 25, 7.5, and 2.5 nL from a single source well). Unfortunately, this 100-fold concentration range is inadequate for most IC50 determination studies, so intermediate dilutions needed to be generated. A number of AP Oncology assays support multiple drug discovery projects by testing different chemical series in parallel with potencies ranging from µM to low nM; therefore, a 12-point half-log concentration range was preferred. The Echo is capable of creating these intermediate plates prior to generating the final assay plate, thereby maintaining a fully contactless compound dispense workflow, 7 but this workflow requires three separate Echo transfer steps and two different source/intermediate plates to create the 12-point concentration range. This workflow was considered too complex to be practical and consequently not cost-effective; therefore, AstraZeneca implemented the pragmatic hybrid two-step method described by groups in AP11,13 and Wilmington. 12 In the AP method shown in Figure 1 , a 96-head Agilent VPrep liquid handler (Agilent Technologies, Santa Clara, CA) is used for the first step to create a four-point DMSO serially diluted “Echo-ready” source plate, typically with 1:100 compound dilutions from a 10-mM stock to give 10-mM, 100-µM, 1-µM, and 10-nM compound wells. In the second step, the Echo uses all four serial dilutions on this source plate to create the final concentration range in the assay plates, dispensing both the compounds and any DMSO backfill required to ensure a consistent DMSO concentration in the final assay. For biochemical assays, the Echo doses compounds into empty plates, termed assay-ready plates (ARPs),12,13 which are then sealed and can be stored before having assay components dispensed directly onto the compounds.
Description of the two-step hybrid method for Echo dosing. (
For cell-based assays, Echo compound dosing can be split into two different workflows: indirect and direct:
Indirect dosing uses the Echo to generate ARPs. Across the AstraZeneca R&D sites in AP, Mölndal, and Gatehouse Park (GHP), the use of ARPs for cell-based assays offers an efficiency benefit because they are created within centralized compound management groups, which also generate ARPs for biochemical assays. ARPs are suitable for assays where cells can be dispensed onto the compounds for an immediate compound incubation start, whether for a short-term homogeneous assay or a long-term proliferation/differentiation assay. 14 In addition, assay media or buffers can be added to ARPs, to be used as intermediate plates for assay technologies that incorporate a compound addition step within the detection system (e.g., FDSS, FLIPR, Epic, and automated electrophysiology platforms).
Direct dosing uses the Echo to dispense compounds directly into the cell media, and this is necessary in assays that require the cells to incubate in the assay plate prior to compound dosing (e.g., to allow adherent cell lines to form a cell monolayer or to allow stem cells to differentiate). Although intermediate plates can be used (i.e., ARPs with added buffer as described above), this requires an additional, unnecessary compound handling step to transfer the diluted compounds into the final cell plate. Direct Echo compound dosing into cell-based assays has been reported by AstraZeneca11,15–17 as well as a number of other groups18–21 and offers the advantage of a simplified workflow without any additional steps or tip/plate consumables, reducing costs and overall assay time. Direct dosing also removes any concerns related to the long-term storage of ARPs (e.g., evaporation issues that can lead to variations in assay data quality).10,22
Direct Echo dosing is now the preferred model for a large number of cell-based assays run across the global AstraZeneca R&D sites; however, the initial implementation of Echos and the direct dosing workflow into the Oncology cell biology group in AP provided us with a unique set of challenges. In this article, we outline how direct Echo dosing has developed and evolved over the past decade in AstraZeneca. We describe the practical challenges of applying ADE technology to 96-well, 384-well, and 1536-well assays and how we have developed and evolved software and robotic solutions to generate fully automated and effective cell-based assay workflows.
Materials and Methods
Experimental Protocol: Cell Monolayer Disruption and Compound Mixing
Cell Culture
The MDA-MB-468 human breast adenocarcinoma cell line was obtained from ATCC (HTB-132; ATCC, Manassas, VA) and cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% heat-inactivated fetal calf serum and 2 mM L-glutamine.
Cell Monolayer Damage in 96-Well Plates
As previously described, 11 MDA-MB-468 cells were seeded in 96-well Costar tissue culture–treated plates (Corning, Corning, NY) at 4000 cells/well in a total media volume of 100 µL. Cell plates were incubated prior to use for 24 h in a rotating incubator at 37 °C, 5% CO2, and 95% relative humidity. Prior to Echo dosing of each plate, 70 µL of the culture media was aspirated from each well and held by a 96-head FluidX XPP-721 liquid handler (FluidX, Nether Alderley, UK). The cell plate with remaining 30 µL media was loaded into an Echo 555, and 300 nL of either phosphate-buffered saline (PBS) or 100% DMSO was dispensed into the center of each well. After dosing, the 70 µL of culture media was returned to the corresponding well. Following either a 0- or 2-h incubation at 37 °C, cells were fixed by the addition of 20 µL of 20% formaldehyde (3.3% final concentration) and room temperature incubation for 40 min. Fixed cells were then washed twice with PBS solution, and 50 µL PBS was added to all wells. Following this, a further 20 µL PBS containing 5 µg/mL Hoechst 33342 was added to stain the cell nuclei and a black-colored light blocking seal applied to each plate. The resultant stained cell monolayer was imaged on an Acumen Explorer plate reader (TTP Labtech, Royston, UK). Following excitation at 405 nm, cell object fluorescence was detected at the emission wavelength range of 420 to 500 nm.
Compound Mixing in 384-Well Plates
MDA-MB-468 cells were seeded in 384-well Costar tissue culture–treated plates at 1500 cells/well in a total media volume of 40 µL. Cell plates were incubated prior to use for 24 h in a rotating incubator at 37 °C, 5% CO2, and 95% relative humidity. The Echo 555 was used to dispense volumes from 5 to 125 nL of 2 mg/mL Hoechst 33342 in DMSO, and plates were then immediately placed in the Acumen Explorer plate reader. Cell object fluorescence was detected as previously described at time points after Echo dispensing.
Experimental Protocol: Hydration of DMSO Source Plates with Different Lid Options
A 384-well Labcyte-certified polypropylene source plate was uniformly filled with 40 µL/well 100% DMSO. Plates were positioned within the room temperature, humidity-uncontrolled environment of Integrated Echo System I (21 °C, 35% relative humidity) and were either unlidded or lidded with a Greiner 6-mm low-profile lid (Greiner Bio-One GmbH, Frickenhausen, Germany), Costar 9-mm high-profile lid, or Labcyte MicroClime lid prefilled with 6 mL 100% DMSO. Hydration of wells in the unlidded plates was analyzed at 1.5, 4.5, 7, 24, and 48 h, and hydration in the lidded plates was determined at 24 h. Analysis of water content in each well was performed by the Echo 550 DMSO audit function by measuring fluid acoustic impedance. 23
Experimental Protocol: Use of the TipCharger to Prevent Cell Carryover
The effectiveness of the TipCharger cold plasma cleaning system (Ionfield Systems, Moorestown, NJ) at preventing carryover of cells between different plates was tested on a VPrep liquid handler equipped with disposable plastic tips. Hela-GFP cells were seeded into 96-well plates at 58,000 cells/well. Immediately following cell seeding, the VPrep was used to aspirate and dispense 70 µL media three times in the cell plate and then empty the tips completely, before moving to a plate containing only fresh media and aspirating and dispensing 80 µL media three times. The media-only plates were then incubated for 4 days in a rotating incubator at 37 °C, 5% CO2, and 95% relative humidity to allow any viable cells to proliferate. Following incubation, wells were examined using a microscope, and 50 µL Cell-Titer Glo reagent (Promega Corporation, Madison, WI) was added to each well to determine a value for the cell carryover. The number of cells carried over was compared between plates where no tip wash was conducted and where a standard TipCharger cleaning cycle had been used between plates.
Automation Systems
Integrated Echo System I
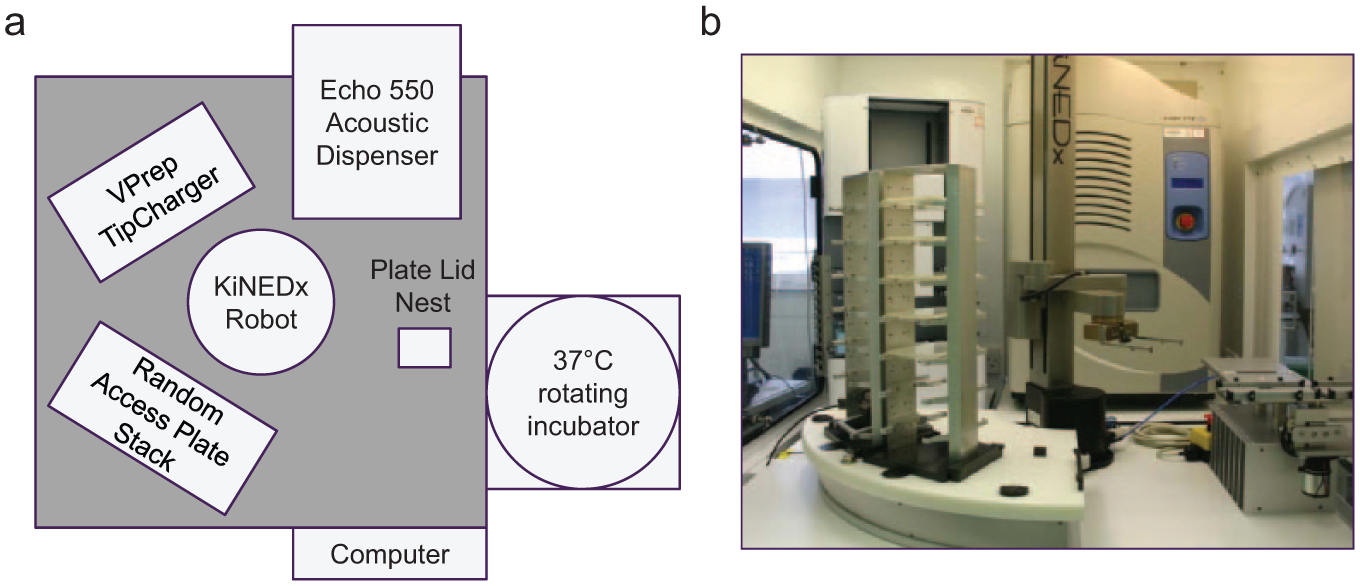
The first custom-built work cell from PAA (Peak Analysis and Automation, Farnborough, UK) was equipped with an Echo 550 and a PAA KiNEDx robotic arm for plate handling and lidding/delidding of source and destination microplates. Storage for up to 11 Echo source plates was supplied by a room temperature KiNEDx random-access plate hotel, and a 37 °C LiCONic STX220 rotating incubator (LiCONic UK, Macclesfield, UK) provided cell plate incubation with up to a 220-plate capacity. A mobile barcode scanner, accessible by the robotic arm, was used to track source plate barcodes. A VPrep was used for all 96-well pre- and post-Echo dosing liquid handling steps. The work cell was fitted with an overhead CAS BioMat laminar flow module (Contained Air Solutions, Manchester, UK) to provide a sterile environment to maintain sterility of the cell plate during the operation.
System I was equipped with a customized assay configuration and graphic user interface, known as Batch Manager, and OVERLORD event-driven scheduling software, both from PAA.
Integrated Echo System II
The second custom-built work cell from PAA was equipped with an Echo 555, as well as a Stäubli TX60 6-axis robotic arm (Stäubli Limited, Telford, UK) and HighRes LidValet vacuum delidding hotel (HighRes Biosolutions, Manchester, UK) for plate handling and lidding/delidding. Room temperature storage for up to 12 source plates was provided by a LiCONic LPX44 plate hotel with integrated barcode scanner, and the two plate stacks were modified to allow rear-loading of source plates without needing to handle the stacks. A 37 °C STX220 provided cell plate incubation. A XPP-721 was used for all 96-well pre- and post-Echo dosing liquid handling steps. XPP-721 tip decontamination and sterilization between cell plates was performed by the TipCharger, and the work cell was fitted with a CAS BioMat laminar flow module as described earlier. Online cell fixation capability was integrated with this system, provided by a Multidrop Combi liquid dispenser (Thermo Fisher Scientific, Waltham, MA) fed by an addressable Hamilton eight-way valve (Hamilton, Bonaduz AG, Switzerland), LiCONic STX44 enclosed plate hotel for room temperature incubation of plates containing fixative, and a BioTek ELx405 plate washer (BioTek, Winooski, VT), with waste disposal controlled by a Brandel autodrain/waste unit (Alpha Biotech Limited, London, UK). A 4 °C STX220 was used to provide storage for fixed cell plates to maintain integrity of cellular structure after fixation. Fixative aerosols and fumes were removed using directed extract vents positioned at critical positions around the STX44, ELx405, and Multidrop Combi instruments.
System II was equipped with an updated customized front-end interface program, known as Batch Manager 3, and OVERLORD2 event-driven scheduling software, both from PAA.
Results and Discussion
Practical Considerations for Direct Echo Dosing into Cell-Based Assays
Considerations for Direct Echo Dosing into 96-Well Assays
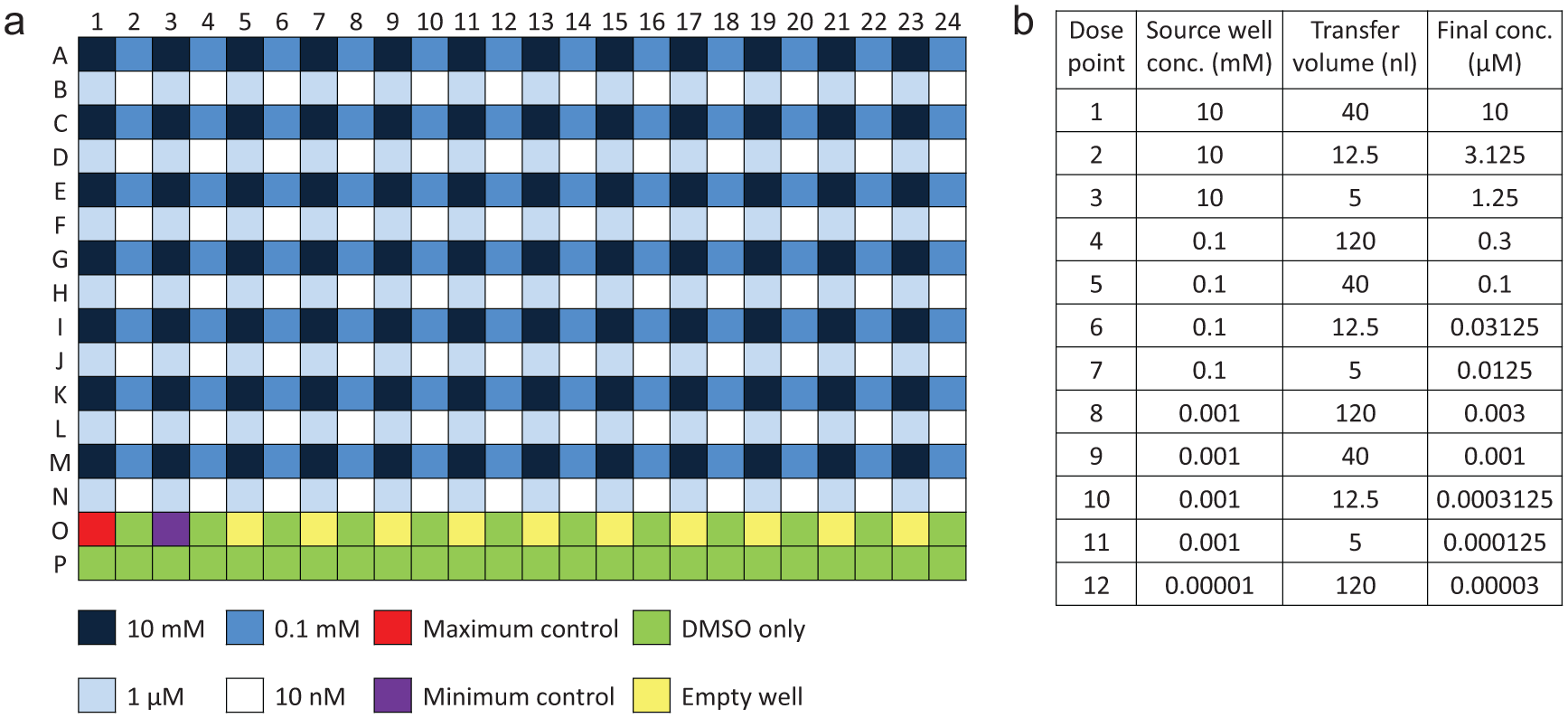
The ability of liquid to remain inside inverted plate wells has previously been demonstrated with 100 µL of various assay fluids (including DMEM cell culture media) in 384-well plates. 2 When Echos were introduced into the AP Oncology cell biology group in 2006, all routine cell-based assays were run using adherent cell lines seeded in 100 µL media, incubated at 37 °C overnight to allow the formation of a uniform cell monolayer prior to any compound treatment. However, these were all 96-well assays, rather than 384-well, and it was found that 100 µL/well media would drain out of a 96-well plate upon inversion. Consequently, some of the media needed to be removed before the plate was inverted inside the Echo, and in-house experiments showed that the optimum volume to leave inside each well was 30 to 35 µL. 3 A disposable tip-based liquid handler was used to aspirate 70 µL media from each well, hold the media while the plate was inverted and dosed inside the Echo, and then immediately return the media to the original well. Aspirate and dispense speed and position settings were optimized to ensure that the cell monolayer was maintained and undamaged.
Although 30 to 35 µL/well media will remain in an inverted 96-well plate, most of the media pools in the corners and sides of the well. This results in only a very thin layer of media left over the cell monolayer, leading to cell detachment caused by the Echo dosing directly onto the cells.3,11 Figure 2 shows the results of a study to determine whether cell detachment is caused by the physical impact of dispensing DMSO directly onto the cell monolayer or by the toxic effect of localized high concentrations of DMSO formed directly above the cells (as previously suggested3,18). Following Echo dosing, cells were immediately fixed to limit the number of cells that would detach but then reattach elsewhere in the plate following typical 2-h assay incubations (as previously suggested 3 ). Cell detachment is clearly observed with 300 nL DMSO but not with the same volume of PBS buffer (experiments also included dosing up to 1000 nL PBS with no effect on the cell monolayer), suggesting that cell detachment is caused by DMSO toxicity.

Cell detachment and monolayer damage in 96-well format plates: (
In Echo protocols designed for direct dosing into 96-well plates, we implemented offset dispensing by reducing the destination plate A1 column offset value in the Labcyte software, so that the 2.5-nL droplets were fired into the shoulder of the meniscus rather than the center of the well ( Fig. 3a ), and this successfully prevented any damage to the cell monolayer.3,11 This offset position was less than 1 mm in from the well wall and had a very tight tolerance; too close to the wall, the 2.5-nL droplets would attach to the wall and not come into contact with the media, but too far from the wall, the droplets would cause cell detachment. This offset position provided a further benefit for the post-Echo media replacement step required for 96-well assays. Media was removed from the center of each well but was replaced high up the wall on the opposite side of the well to where the Echo compound dispense had been fired. No mixing step was added to this procedure, and so the disposable plastic tip on the liquid handler did not come into contact with the dispensed compound when returning media and, when tested in multiple assays, this was found to successfully avoid carryover of any compound into the next assay plate. As all plates in a single assay contained the same cell line and same media, we were able to avoid changing or washing the tips between cell plates in a single assay, reducing consumables costs and avoiding lengthy wash protocols.
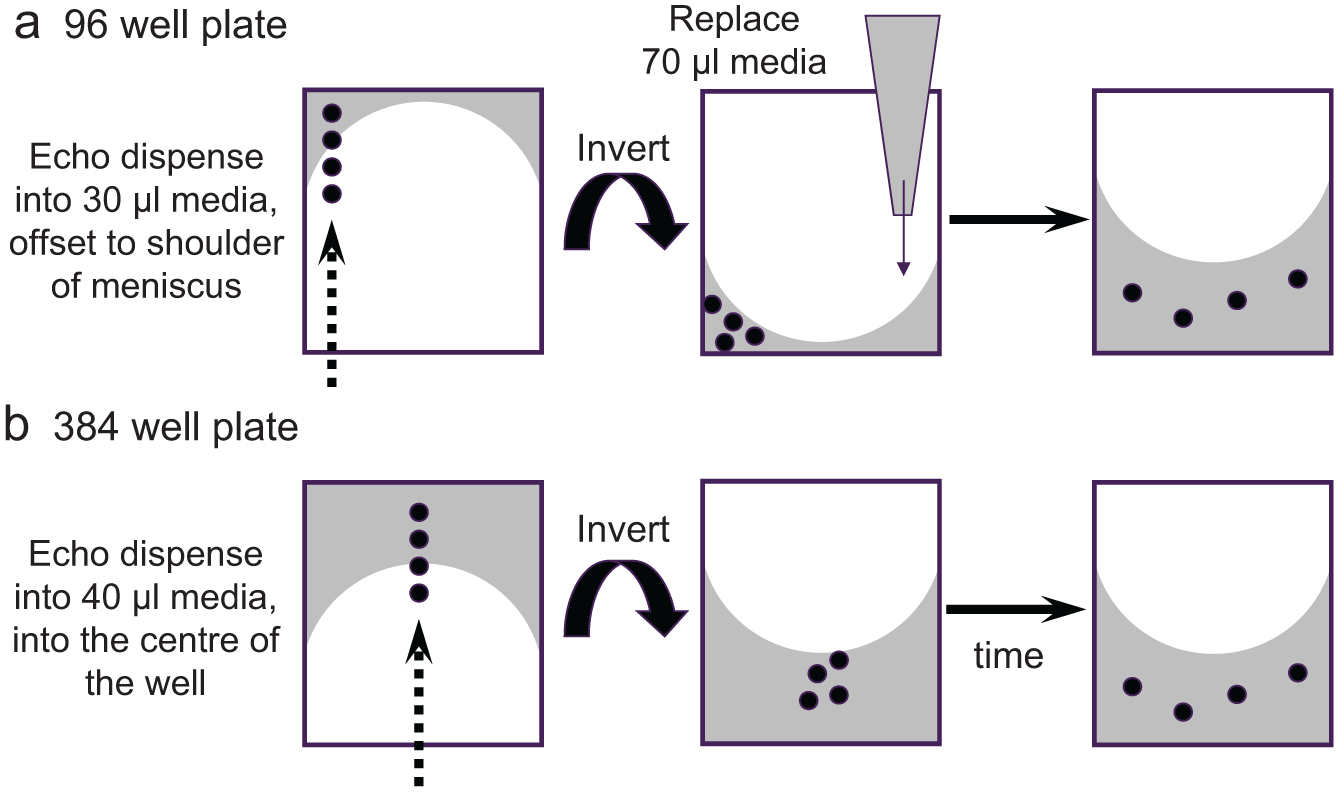
Diagram showing direct Echo dosing workflows in 96-well and 384-well format cell-based assays. (
The workflow for direct dosing into 96-well plates does not include any specific compound mixing steps. DMSO is a highly polar and water-miscible organic liquid, with a high affinity for mixing with water 24 and, at small scale, there is often very rapid mixing due to differences in the surface tensions of the two fluids. 25 The plate rotation inside the Echo system and post-Echo replacement of 70 µL media does, however, provide a quasi-mixing step.
Direct Echo dosing was the preferred method for compound dosing into 96-well cell-based assays in AP from 2007; however, it was not possible for every assay we developed. For example, the method described by Bradbury et al. 26 for an androgen-receptor down-regulator assay used an intermediate dosing workflow using ARPs with added media and a plate-to-plate compound transfer step. This more complicated dosing method was required because, during assay development, we observed that the LNCaP cell line was not able to withstand plate inversion and direct Echo dosing without significant cell disruption and loss (even when using offset dispensing). Subsequent validation of a 384-well version of this assay showed that direct dosing was possible in this format without significant disruption to the LNCaP cells (data not shown).
Considerations for Direct Echo Dosing into 384-Well Assays
The introduction of the Echo, as well as other 384-well compatible liquid handlers, plate washers, and plate readers, enabled the transition of cell-based assays from a 96-well to 384-well format. As Figure 3b shows, this significantly improved and simplified the direct Echo dosing workflow. The 40- to 50-µL/well media used to seed cells in 384-well plates remains in inverted wells in the majority of cases, removing the requirement for pre- and post-Echo liquid handling steps. In addition, offset dispensing is not necessary; cell monolayer damage has never been observed in 384-well assays in AstraZeneca, in agreement with previous reports. 18
The removal of the pre- and post-Echo liquid handling steps was a simplification and improvement in workflow, but the lack of this “quasi-mixing” step raised concerns for direct Echo dosing into 384-well assays ( Fig. 3 ). In the 384-well assay workflow, although there will be some movement of the media when the plate is inverted, there have been concerns that nanoliter droplets of compound will not adequately mix and disperse uniformly, leading to cells experiencing variable and inaccurate concentrations of compound. Although data variability may not be expected in long-term assays (e.g., 72-h incubation proliferation assays 18 ), a large number of the cell-based assays performed in AP have 2-h compound incubations.
Figure 4 shows the results of a study examining the dispersal of a cell-permeable DNA stain, Hoechst 33342, when fired into 40 µL media over a cell monolayer in a 384-well plate. For the successful staining of live animal cells with Hoechst, the manufacturers recommend a final concentration of 0.25 to 5 µg/mL incubated for 20 to 30 min. Using a 2-mg/mL stock solution of Hoechst, the Echo was used to dispense volumes from 5 nL (0.25 µg/mL final concentration) to 50 nL (2.5 µg/mL final concentration), and wells were imaged after 5, 15, 30, and 60 min. Figure 4a shows cell staining after 5 min (an incubation time significantly shorter than recommended), displaying how coverage of the entire cell monolayer increases with the dispense volume/final concentration and reaches full coverage between 25 and 50 nL. Figure 4b shows a time course of staining with a 5-nL dispense (giving the lowest recommended final concentration), achieving full coverage of the cell monolayer between 30 and 60 min. Consistent with these results, we have observed neither variable nor shifted IC50 data caused by short incubation times or low-volume compound dispenses in any of the cell-based assays we have run, and the simpler 384-well workflow has led to direct Echo dosing being implemented across the AstraZeneca R&D sites in AP, Mölndal, and GHP.
Mixing and dispersal of a cell-permeable DNA stain in a 384-well cell plate. (
Considerations for Direct Echo Dosing into 1536-Well Assays
Recently, the AZ Oncology group successfully developed and validated a 1536-well high-throughput cell-based assay, using direct Echo dosing, to evaluate the synthetic lethality of Oncology drug combinations using an imaging-based live/dead cell assay format. The development of the 1536-well format was critical to the application due to the exponential scaling of in vitro drug combination profiling across a panel of over 200 tumor cell lines. The main benefits of conducting such a profiling screen in 1536-well format included the reduction in cell supply requirements, more test conditions per assay plate, and the >75% reduction in plate handling. With the imaging quantification being performed by an automated Acumen Explorer plate reader, the imaging time was identical between the 384-well and 1536-well format, and so the miniaturized format represented an increase of over 200% in screening throughput for each assay slot.
During assay miniaturization from the 384-well to 1536-well format, key challenges were the choice of final assay volume, the design of the compound dilution and dosing regimen, and cell and reagent dispensing into 1536-well plates. The full assay development and validation of the 1536-well in vitro combination assay will be the subject of a manuscript currently under preparation. However, with respect to achieving direct Echo dosing in the 1536-well format, the final cell incubation volume was fixed at 10 µL, and a final DMSO concentration of 0.1% v/v was required to be within the DMSO tolerability of this assay. With these parameters and the 2.5-nL droplet size of the Echo system, a modification to the compound dilution workflow was required. To cover the broad 12-point compound dose range with half-log increments, the prediluted “Echo-ready” source plate was modified from the previous four-point 1:100 dilution series to a six-point 1:10 dilution series. The Echo was then used to dose multiple droplets of 2.5 nL from each 1:10 dilution into the cell plates, resulting in a final compound concentration range between 10 µM and 25 pM while limiting the final DMSO concentration at 0.1% v/v.
The downscaling of the cell line handling procedure proved to be less technically challenging than expected; cell numbers per well were reduced, and the duration of the overall assay incubation was also reduced to successfully maintain cell growth/metabolism in the smaller media volume. The standardization of our data analysis platform also ensured the seamless transition from 384-well to 1536-well.
Although direct Echo dosing into 1536-well cell-based assays has been performed in AstraZeneca, most 1536-well cell-based assays have been indirectly dosed using ARPs (use of ARPs for 1536-well assays has also been reported by other groups 27 ). In a high-throughput screening (HTS) setting where batch sizes typically reach 1300× 1536-well plates, this is due to the time required within the assay for direct Echo dosing (10 min per 1536-well plate for compound dosing and DMSO backfill) compared with adding cells to a predosed ARP (30 s per 1536-well plate for adding 5 µL cells to each well). When an HTS assay requires cells to incubate in the assay plate prior to compound dosing, speed considerations mean that an intermediate dosing workflow is preferred using ARPs with added media and a plate-to-plate compound transfer step. Direct compound dosing into 1536-well assays using a pin-tool stamping system has also been reported. 10
Considerations for Direct Echo Dosing into More Complex Assay Formats
A number of cell-based assays in AstraZeneca require incubation times of up to 14 days (e.g., cell differentiation and mitobiogenesis assays). In these assays, media is often replaced at intervals throughout the incubation, and cells are redosed with fresh compound. This can cause workflow issues for direct Echo dosing into 384-well plates, as Echo dosing must occur immediately following the media replacement step to maintain the correct compound concentration, but media dispense often wets the walls of the well and can lead to media loss when the plate is inverted. Therefore, in these cases, pre- and post-Echo liquid handling steps are employed similar to those required for 96-well assays. For example, in a 14-day adipose cell differentiation assay, cells are seeded in 45 µL media, and then 20 µL is removed prior to Echo dosing, with 20 µL fresh media added immediately afterward.
Many drug discovery programs across the industry have moved beyond small molecules, and as we move toward more varied drug molecules (e.g., small molecules, proteins, antibody therapies, RNA interference [RNAi]), a challenge for future screening is a system that can handle a mixture of dilution agents to maintain a uniform screening process regardless of the drug molecule type. With the introduction of the OMICS capability, enabling dispensing of any solvent/diluents, the Echo appears to be well suited to this task.
There has been increasing interest in the development of more physiologically relevant cell models for drug discovery, using primary cells, cocultures, and 3D cell assay systems. Assays using primary cells and cocultures can be directly Echo dosed with no additional workflow considerations; however, 3D systems, particularly cell spheroid assays, provide practical challenges. For example, although direct Echo dosing into a 3D coculture soft-agar colony formation assay has been described, 21 the soft-agar format is particularly amenable to plate inversion. In contrast, it may be difficult to design a practical workflow for direct dosing into the hanging-drop plates currently available from multiple manufacturers. These new assay formats may provide challenges, but they are also providing opportunities; ADE technology has already been employed to dispense single cells 28 and also to generate multiplexed cell cocultures. 29
Automation Solutions for Direct Echo Dosing into Cell-Based Assays
The standalone Echo systems introduced into AstraZeneca AP in 2005 were housed within custom-built cabinets providing a down-flow of clean filtered air. This was considered essential to prevent contamination of cell media when the assay plate was unlidded during compound dosing. Subsequent automated systems for compound dosing have also provided sterility, with filtered airflow hoods providing clean air over the entire work-cell deck where cell plates are delidded and transferred in and out of the Echo. Although AstraZeneca AP has continued to provide a sterile environment for direct Echo dosing, it may not be essential; Rodriguez et al. 18 described a proliferation assay study performed at MDS PharmaServices over a 13-week period using 297 distinct cell lines where direct Echo dosing occurred outside the protection of a hood (although all other plate handling was performed within the hood), and no contamination of the cell plates was observed despite a 72-h incubation and antibiotic-free media. The authors suggested that the inversion of the cell plate inside the Echo, coupled with contactless dispensing and rapid robotic plate handling, contributed to reducing potential sources of contamination. Recent experience in both AstraZeneca Mölndal and GHP also confirms this finding, where direct dosing into cell assays is now being successfully provided by the nonsterile automated Echo work-cells used by the compound management groups to generate ARPs.
Labcyte’s initial Echo control software offered limited compound dosing options, focused on single-volume transfers (same volume dispensed to every well) and simple plate reformatting (producing identical copies of the source plate or reformatting 384 to 1536 or vice versa). To create the concentration ranges required for IC50 determination, AstraZeneca developed in-house Echo control software that used prewritten comma-separated values (.csv) files. These files were created manually or with the help of simple Excel macros, with each file providing enough dispensing information for a single assay plate (source well, destination well, volume), and this workflow required serially diluted test compounds, reference controls, and 100% DMSO for backfill, all to be located on a single Echo source plate. Subsequent automated systems for creating ARPs use reference controls positioned on separate source plates and cherry-pick from multiple source plates to prepare a single assay plate. This offers compound savings when particular compounds are required for multiple assays, but this also increases the number of source plate movements in and out of the Echo for each assay plate and can increase total dosing time from 3 min (one source plate) to 15 min (three source plates) for a 384-well assay plate. This extended time is acceptable when creating ARPs, but it is not acceptable for cell-based assays for two reasons: (a) the amount of time a cell plate is held inverted within the Echo should be kept to a minimum, particularly in 96-well plates where the cell monolayer may be exposed, and (b) assay compound incubation starts as soon as the compound is dosed, so any time difference between the first and last well on a single plate should be kept to a minimum (particularly for short incubation assays of <2 h).
Integrated Echo System I—Original Design
A custom-built work-cell (layout as shown in Fig. 5 ) was purchased from PAA in 2006 and was in routine use by 2007. The design specification for Integrated Echo System I (System I) was originally scoped to provide fully automated single-concentration or IC50 compound dosing for 96-well and 384-well cell-based assays. Cell plates were stored in a 37 °C incubator, removed to be compound dosed in the Echo, and then returned to the incubator. For 96-well assays, the workflow included pre-Echo media removal and post-Echo media replacement steps.
(
OVERLORD1 software with event-driven scheduling was used by PAA to generate and operate the automated procedures, including all robotic plate handling operations and controlling the programs and actions of all third-party equipment. This included the homing of axes and focus calibration of the Echo at the beginning of each run. Any errors on the system during a run were reported to a group of system administrators via an automatically generated email.
Batch Manager 1 software was custom-designed by PAA for AstraZeneca to interface the end user with OVERLORD1 and was used to generate individual assay protocols. The most significant improvement over the standalone Echo workflow was the on-the-fly generation of Echo control .csv files. The initial in-house Echo control software required one .csv file per assay plate, so that a 10-plate assay would require 10 different .csv files. As a range of different plate layouts and 6-, 8-, and 12-point concentration ranges with different top concentrations were initially used in AP, a large library of .csv files gradually built up, and this led to a small number of mistakes when Echo users selected incorrect dosing files for their assays. Instead, Batch Manager 1 generated .csv files on-the-fly, using the following information: source plate barcodes and the positions of selected compounds within each source plate (files generated by an in-house LIMS and compound management database), source plate map (file showing source plate position of serially diluted compound wells, 100% DMSO for backfill, and reference control compounds), assay plate map (file showing assay plate position of control and concentration-response wells), and compound/DMSO volumes for each concentration point (volumes entered into Batch Manager 1 during protocol creation). Via the compound list files generated by the in-house LIMS, Batch Manager 1 software allowed full cherry-picking of compounds (i.e., selected compounds on selected source plates could be dosed into individual cell plates, rather than dosing all compounds from each source plate). This improved speed and efficiency by reducing the total number of cell plates required in an assay, and this functionality, coupled with the replacement of the manually written .csv files for routine work, led to Batch Manager 1 and OVERLORD1 being subsequently installed on the standalone Echo units as well as the automated work-cells.
Integrated Echo System I—Postimplementation Enhancements
Once System I was in routine use, ever-increasing demands on capacity and capability highlighted various system limitations. Issues focused on the requirement to run a higher number of different assays back-to-back, meaning that scientists needed to set up multiple assays at the same time, often many hours before an individual assay would start. Specific issues concerned overall speed and throughput, potential cross-contamination between 96-well assay plates caused by the VPrep pre- and post-Echo steps, progressive hydration of DMSO source plates, and out-of-hours use.
System I was originally equipped with an Echo 550, but this was later upgraded to an Echo 555 to improve the speed of compound dispensing (for typical cell assay protocols, 6 min per 384-well plate was reduced to 3 min per plate).
The VPrep used on System I used a single set of plastic 96-head tips for each assay, without DMSO/ethanol washing steps between each assay plate, as the VPrep protocol avoided any contact between the tips and compounds, and all cell plates in a single assay contained the same cell line. However, this limited System I’s use for cell panel assays (routinely up to 20 cell lines per assay), assays requiring multiple dosing steps (e.g., where the Echo dispensed ligands/stimulants in addition to compounds but with time delays between dispenses), and different assays run back-to-back without user intervention to change the VPrep tips between assays. Therefore, we evaluated different tip-washing protocols for VPrep tip decontamination and sterilization between cell plates, and this included evaluation of the TipCharger cold plasma cleaning system (Ionfield Systems, Moorestown, NJ). The benefit of the TipCharger compared with standard washing procedures is that no DMSO/ethanol is required, the washing protocol is faster, and the tips are completely dry before being used for the next assay plate. The effectiveness of the TipCharger at preventing carryover of cells between different plates was tested, using a highly concentrated suspension of fast-growing Hela-GFP cells (58,000 cells per well) and comparing cell carryover into a media-only plate between protocols with no tip washing or a standard TipCharger cleaning cycle. Cell plates were incubated for 4 days to allow any viable cells to proliferate but, in both protocols, no cell carryover was observed using the Cell Titer Glo reagents. However, examination of wells using a microscope showed a number of wells containing small cell colonies (<50 cells) in plates with no tip washing versus only apparent cell debris in wells where the TipCharger had been used. The success of the TipCharger to prevent cross-contamination in screening assays has also been demonstrated with low molecular weight compounds, nucleic acids, and bacterial liquid transfers. 30
DMSO is hygroscopic and rapidly absorbs water from the atmosphere, and studies have shown that water uptake in compound DMSO solutions significantly contributes to compound degradation, precipitation, and reduction in concentration.31,32 An open Echo source plate filled with 100% DMSO at under half capacity will equilibrate to a moisture content of over 30% within 24 h under standard laboratory conditions 23 ; therefore, storage conditions aimed at limiting hydration both prior to and during use are an important consideration. In AP, compound source plates for cell assays were created up to 1 week before the assay, foil sealed, and stored within a StoragePod nitrogen enclosure at 5% relative humidity (Roylan Developments Ltd., Leatherhead, Surrey, UK). Prior to loading into System I, plates were centrifuged, their seals were removed, and they were lidded with Greiner universal low-profile lids. In the majority of assay workflows, this occurred immediately before the start of compound dosing, so source plates were unsealed only for a short period of time. However, as users required the ability to load source plates longer in advance or the previous day for assays with start times before 8 a.m., this could extend exposure times up to 16 h. Therefore, we examined source plate hydration in the System I environment and compared hydration patterns using three different lid types. Figure 6a shows the average hydration over 48 h of an unlidded 384-well source plate containing 40 µL/well DMSO, reaching 25% hydration within 24 h. Figure 6b displays a hydration map of each well at 24 h using the Greiner universal low-profile lid, clearly showing the difference between inner and outer wells previously described by other studies,23,31 with the greatest hydration observed in the corner wells. In the System I environment, this spatial distribution of hydration could cause a significant difference in quality between compounds located in the outer and inner source plate wells. The hydration pattern was slightly improved with the use of a deeper Costar lid ( Fig. 6c ); however, a significant improvement was observed with the Labcyte MicroClime lid ( Fig. 6d ). The MicroClime lid incorporates a fluid-absorbing matrix that can be prefilled with a range of solutions to create a vapor barrier between the microplate and the environment. DMSO added to the lid prior to use creates a high-DMSO vapor layer with the intention of limiting hydration of the source plate wells. Although Figure 6d shows that hydration still occurs in the edge wells over the 24-h time period, the difference between the inner and outer wells is less than 1.5%. Based on these results, all subsequent use of System I incorporated this new lid type for Echo source plates.
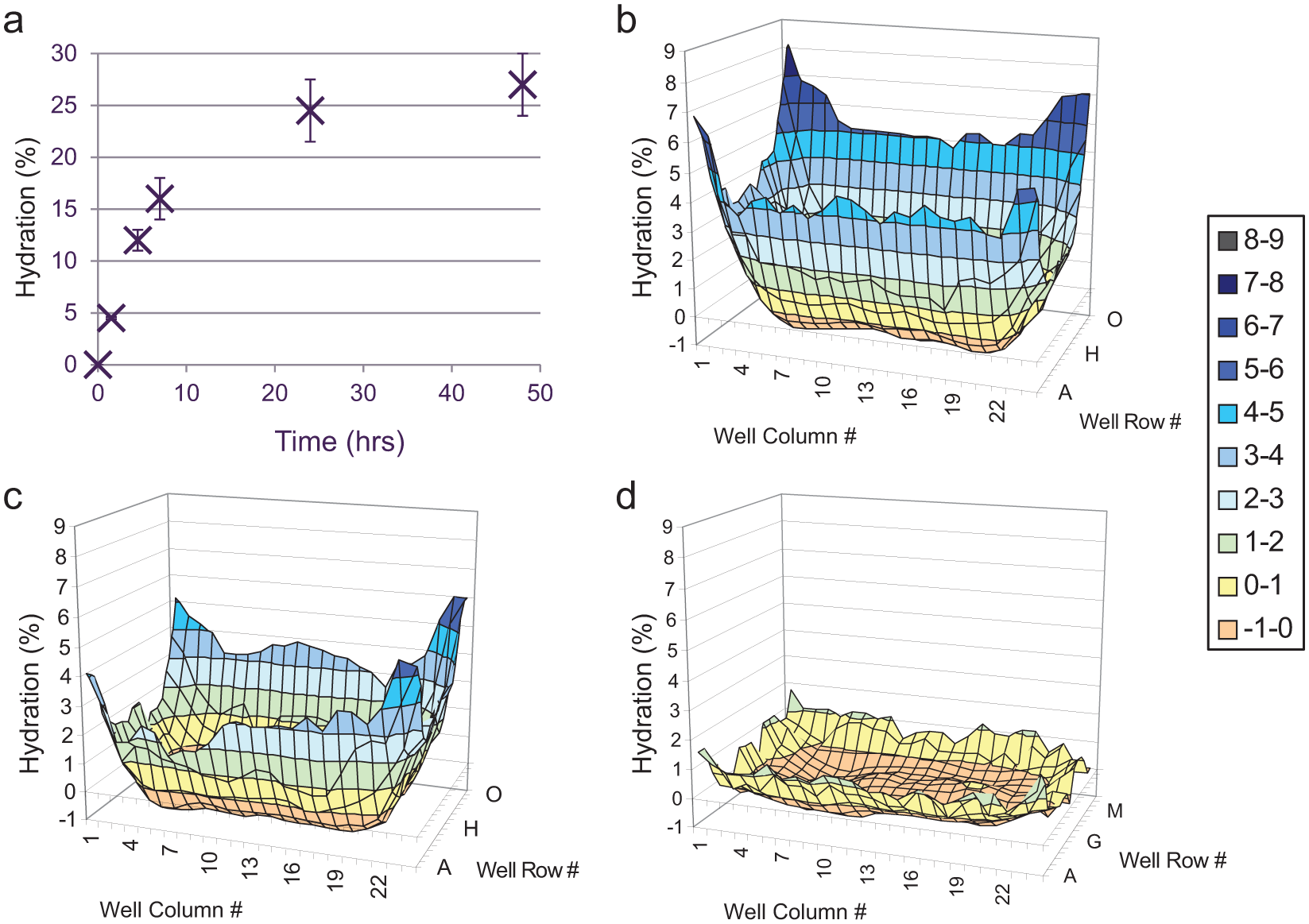
Data showing hydration of DMSO in 384-well source plates in the room temperature, humidity-uncontrolled environment of Integrated Echo System I. (
Despite the learnings and postimplementation enhancements described above, one major limitation of System I could not be addressed. Following Echo dosing on System I, the length of compound incubations was controlled by manual removal of cell plates from the 37 °C incubator and offline addition of fixative. This requirement to complete the incubation manually limited the work-cell’s use overnight and during weekends.
Integrated Echo System II
All plate-based cell assays run within AP Oncology had migrated from manual dosing to Echo dosing by 2008, and so a second Integrated Echo System (System II) was purchased from PAA in 2009 to meet the higher capacity requirement (system layout as shown in Fig. 7 ). Although both the hardware and software specifications were based on the original System I design, a number of new capabilities were added, not least the provision of cell fixation and long-term plate storage to allow and promote out-of-hours use.
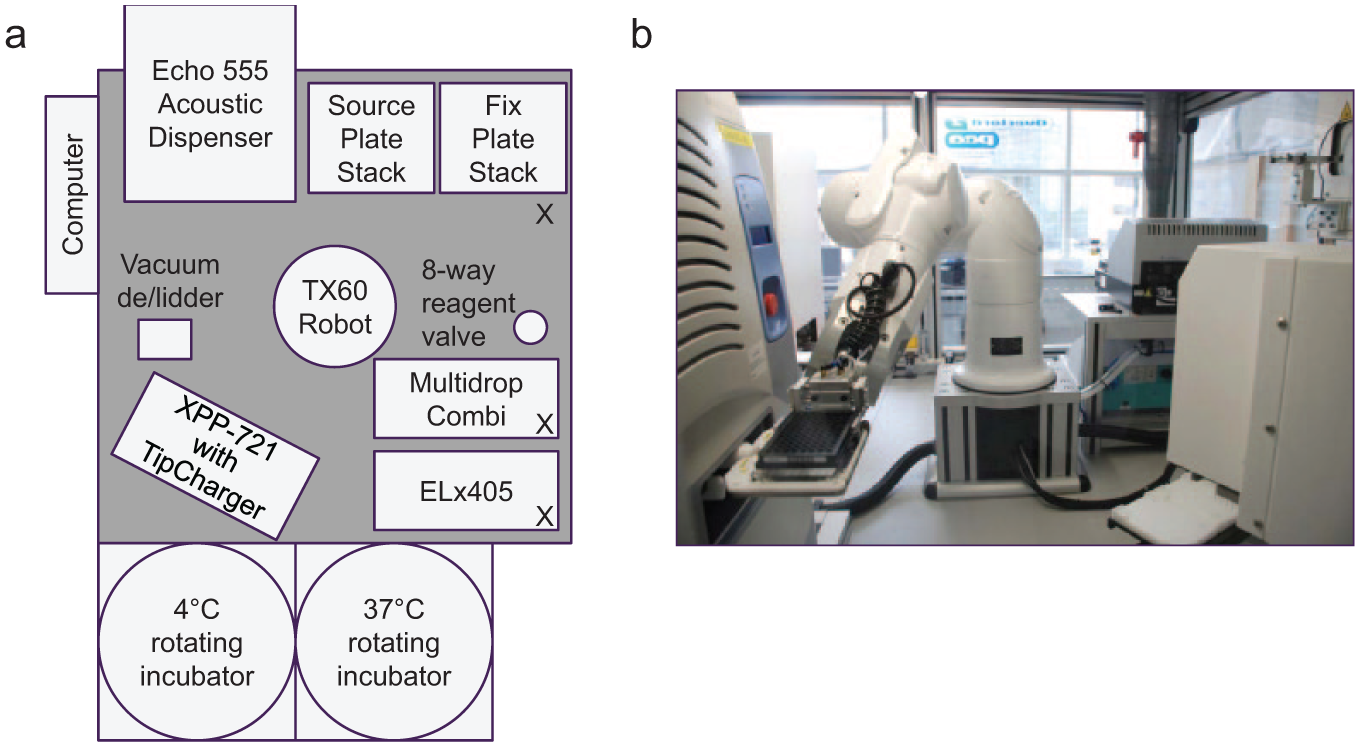
(
Compared with System I, the robustness and speed of the plate and lid handling in System II were improved with a Stäubli TX60 robotic arm. The use of a HighRes LidValet vacuum delidding hotel and barcode scanners integrated in each plate storage location reduced the number of procedures carried out by the robotic arm and, therefore, improved speed and parallel processing.
OVERLORD2 software with event-driven scheduling was used by PAA to generate and operate the automated procedures, including the control of all third-party equipment. This included homing of axes and focus calibration of the Echo at the beginning of each run, priming and washing of the Multidrop Combi and ELx405 platewasher, and emptying of the Brandel Waste Unit into a connected drain at the end of each run.
Batch Manager 3 software was custom-designed by PAA for System II to interface the end user with OVERLORD2. On-the-fly generation of Echo control files, full compound cherry-picking, and 96-well pre- and post-Echo liquid handling were controlled in the same manner as System I. However, Batch Manager 3 offered the following additional capabilities:
A fixation procedure (composed of a liquid addition, room temperature incubation, and wash) could be used in conjunction with an Echo dosing protocol or as a protocol in isolation. The former scenario was particularly relevant for short-term assays, as precise compound incubation timings from dose to fix could be maintained.
Batch Manager 1 stopped individual cell plates being dosed more than once, and this prevented accidental redosing on a system designed to support multiple users working in parallel on different assays. However, as experiments became more complex, this functionality proved limiting, and complicated workarounds were required to run these experiments. Batch Manager 3 still generated warnings when selected plates were already allocated to a dosing batch but allowed redosing to be requested and processed. This enabled complex time course experiments, repeat dosing on long-term assays, and simple combination dosing, as well as allowed the Echo to be used to add ligands and reagents pre- or postcompound dosing for stimulated cell assays. For example, direct stimulatory ligand dosing was enabled for functional nuclear hormone receptor antagonism assays, 17 and this was utilized extensively on the Integrated Echo II System, enabling the timing and order of addition of compound and ligand to be explored during assay development. The system was also amenable to codosing of compound plus other modulators that could be used to discern compound mechanism of action in a routine manner. 17
During the assay, the user was provided with continuously updated details of the running stage, incubation end time, and current location of each assay plate. At completion, all assay details and final timings were saved in an accessible database. Notification of the completion of a run, or any errors on the system during a run, was reported to the user via an automatically generated email.
Echo-generated output files for each individual assay plate (containing details of the success or failure of compound and DMSO dispensing into each destination well) were automatically combined and reformatted into a single results file at the end of each assay. A “Check Dispense” option in the interface displayed the number of successful Echo dispenses compared with the number attempted. If the assay contained any failures, the user could then open the results file to locate any failed well and adjust data analysis accordingly.
Impact of Acoustic Dosing and Future Developments
In combination with investment in automation enabling 384-well assay formats and the use of cryopreserved cells, the investment in Echo technology has had a major impact on both the quantity and quality of cell screening output in AP. The observed improvement in both the accuracy and precision of compound dosing concentrations led to a change in assay plate layouts; concentration ranges previously run in duplicate or triplicate on the same plate were abandoned in favor of a singlicate plate layout. This reduction in the number of plate wells required per assay enabled a change from 6- or 8-point half-log concentration ranges to a 12-point half-log range; the extended concentration range increased the likelihood that an accurate IC50 result would be obtained first time for compounds across a range of potencies, and this yielded the benefit of reducing the number of repeats required and decreasing the turnaround time for IC50 results. From 2005 through 2009, the number of cell-based assay IC50s generated in AP Oncology effectively doubled year on year from ~2k/annum to ~30k/annum across 25 assays supporting the majority of the Oncology portfolio. In terms of IC50 curve quality, by the end of 2010, on average 99% of IC50 values generated had a confidence interval ratio (CIR) of less than 4, with the fidelity of compound dosing a major contributor to this performance.
The on-the-fly generation of .csv files to control the Echo has been the preferred route for all routine screening work, but Batch Managers 1 and 3 both allow the use of manually written .csv files for assay development and bespoke studies, including simple combination dosing experiments. AstraZeneca has recently developed new software tools to enable more complex combination screening, and one of these tools, Apothecary, is described in this journal edition by Cross et al. 33
Since implementing automated Echo-dosed cell-based assays in AP, assay workflows have always been split between the Integrated Echo Systems (providing compound dosing, incubation, and cell fixation) and two AutoAssay Systems 34 providing all downstream assay-processing steps (i.e., addition and wash of assay components such as permeabilizing, blocking, antibody, and detection reagent solutions). As a number of different plate-based readers have been historically required for the wide range of cell-based assays we perform, readers have also been kept on a separate automated system. This separation of prefixation steps from downstream assay staining procedures and final end-point detection has both advantages and disadvantages. The impact of automation issues and breakdowns is more easily managed where each work-cell has only a narrow and discrete function within each assay; however, user intervention is required throughout a single assay to transfer plates between work-cells, and this model requires the use and maintenance of multiple automated work-cells, most of which have overlapping capabilities.
With AstraZeneca’s transfer of UK R&D activities from Alderley Park to Cambridge, including the transfer of cell assay capability during 2015, it has been necessary to reexamine the capacity and capability of replacement automation systems using a different perspective. In a future space-constrained laboratory environment, any large automated work cell should be in operation 24 h/day, 7 days/week. However, the reality of cell-based assays, particularly those that use cell lines that require manual cell culture before use, is that cells are often seeded and then dosed at the beginning of the week, downstream assay staining often occurs during the middle of the week, and end-point detection and plate reading often occur at the end. With this type of working pattern, separate work cells with specific and narrow capabilities may be in use only for limited time periods. Consequently, we have recently designed a new automated system in collaboration with HighRes BioSolutions, based on its six-sided MicroStar Hexapod base. This system contains all the equipment required to run an assay end to end (i.e., cell seeding through to plate reading), and the instrument docking capability on HighRes systems means that different equipment and readers can be swapped in or out of the system depending on individual assay requirements. Their dynamic scheduling software, Cellario, allows multiple protocols to be run in parallel. This system is currently being manufactured and is expected to be in routine use in Cambridge by the end of 2015. The design, validation, and use of the new system will be the subject of a future manuscript.
Footnotes
Acknowledgements
We thank Jon Orme for early evaluation of Echo dosing into 96-well and then 384-well cell assays, as well as the bioscientists past and present in AP Oncology Assay Sciences and then Discovery Sciences for designing and implementing new assays using direct Echo dosing. We thank our technical colleagues in Labcyte and PAA for working collaboratively with AstraZeneca over the past decade to develop and evolve software and hardware solutions for our cell assays, with particular thanks to Rob Harkness, Ian Pye, Andy Mash, and Jon Newman-Smith. We also thank Roger Clark and Jonathan Wingfield for helpful discussions around the issues of using direct Echo dosing in HTS and a historical perspective on the introduction of Echos into AstraZeneca.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
