Abstract
A novel methodology for the synthesis and purification of drug-like compound libraries has been developed through the use of a microwave reactor with an integrated high-performance liquid chromatography–mass spectrometry (HPLC-MS) system. The strategy uses a fully automated synthesizer with a microwave as energy source and robotic components for weighing and dispensing of solid reagents, handling liquid reagents, capper/crimper of microwave reaction tube assemblies, and transportation. Crude reaction products were filtered through solid-phase extraction cartridges and injected directly onto a reverse-phase chromatography column via an injection valve. For multistep synthesis, crude products were passed through scavenger resins and reintroduced for subsequent reactions. All synthetic and purification steps were conducted under full automation with no handling or isolation of intermediates, to afford the desired purified products. This approach opens the way to highly efficient generation of drug-like compounds as part of a lead discovery strategy or within a lead optimization program.
Introduction
Pharmaceutical drug discovery research is under increased pressure to improve efficiency in the generation of drug candidates in a cost-effective and time-efficient manner. Previously, we have described the successful integration of mass-directed preparative high-performance liquid chromatography (HPLC) to a segmented flow reactor. 1 The integrated system combined synthesis, purification, and compound management in a single continuous operation with full automation. Cycle time for generating small focused libraries of compounds (48–96 members) ready for bioassay and compound registration was reduced to 3 days. 2 However, an inherent limitation for carrying out chemical transformations in flow mode is the obvious requirement of homogeneous reaction conditions. This prerequisite has restricted the integrated platforms to performing only a subset of chemical transformation where no solid reagents/catalysts are involved. This limitation consequently hampered the utilization and application of the integrated platform for a wide variety of chemistries requested from medicinal chemists within AbbVie. A non-flow-based system is therefore needed to complement the flow-based systems.
The use of a microwave for heating to carry out organic chemical transformation has been widely studied3,4 and applied in synthetic laboratories. 5 The advantages of this enabling technology have proven particularly valuable for drug discovery projects where speed is often a critical factor. 6 About 50% of the compounds made by the AbbVie High-Throughput Chemistry (HTC) group have used microwave as the heating source. Single-mode microwave heating equipment that carries out reactions one at a time has been the standard equipment of choice. 7 As part of our efforts to expand the scope of chemistries to non-flow-amenable transformations (such as reactions that require heterogeneous catalysts or reagents with poor solubility), we have developed a microwave and hotplate-based platform that is capable of carrying out microwave/hotplate reaction, filtration, and preparative HPLC in a fully automated fashion. Together with the already developed segmented flow chemistry system, this microwave/hotplate platform provides chemists in AbbVie the necessary tools to generate the majority of libraries within HTC, from start to purified products, and further reduces the cycle time to within 24 h.
To demonstrate the effectiveness of the integrated system, we have selected two reactions that are widely used by medicinal chemists—namely, the Sonogashira coupling 8 for C-C bond formation and Buchwald-Hartwig amination9,10 for C-N bond formation ( Fig. 1A ). For multistep synthesis, we have designed a three-step synthetic sequence involving a sulfonylation reaction followed by a nucleophilic substitution and a Suzuki-Miyaura coupling reaction to yield the final product with good yield ( Fig. 1B ).
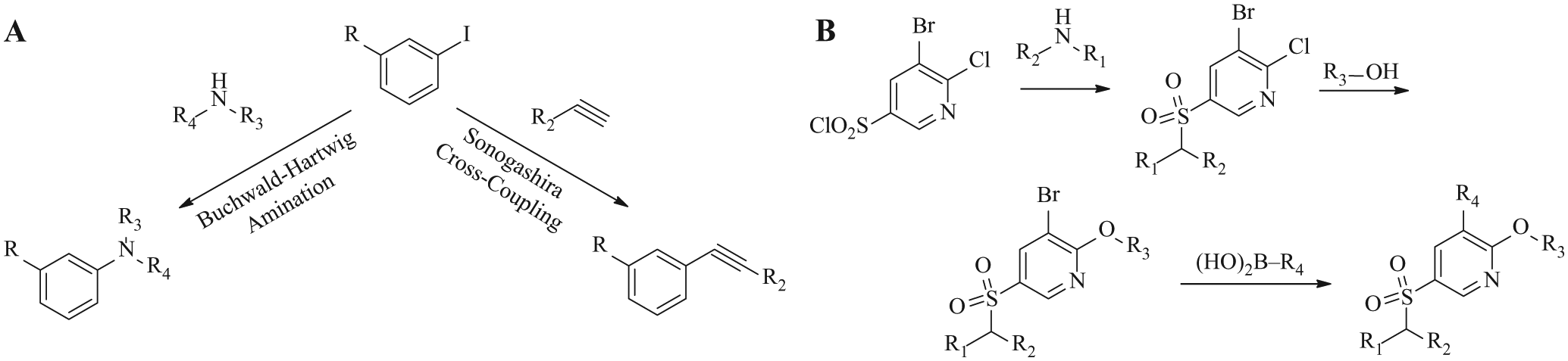
Selected reactions for library demonstrations. (
Materials and Methods
General Library Setup Procedure
Core, reagents, and monomers (in either 4-mL or 40-mL vials) were dissolved while resin/solid reagents were placed inside the solid dispensing bottle. Experimental design was created using the AutoSuite software (Chemspeed Technologies, Augst, Switzerland), which controlled various components in assembling the reaction mixture according to a predetermined amount of each reagent. Microwave heating conditions (temperature, time of heating, and solvent absorption characteristics) were transmitted to the microwave oven via integrated software. In the case of hotplate library synthesis, the user placed fully assembled reaction mixtures in 4-mL vials on the heating plate and set heating temperature by adjusting the temperature controller. Heating time was controlled by a countdown timer switch (Traceable Countdown Controller, part number 5095; Control Company, Friendswood, TX). A corresponding wait time was programmed into AutoSuite to wait for the completion of the hotplate heating step before subsequent solid-phase extraction (SPE) workup/HPLC injection.
Sample lists for the HPLC-UV/mass spectrometry (MS) were created with targeted molecular weight (MW) data imported from external Laboratory Information Management System (LIMS) system. These MW data and various UV/MS threshold settings were used to customize the purification and collection method for each compound output from the SWAVE synthesizer.
Pure fractions collected from HPLC-UV/MS were than labeled using a custom labeling and reracking robot. A 5% (v:v) sample of the HPLC fraction was transferred for nuclear magnetic resonance (NMR) analysis using a custom liquid handler with liquid-sensing capability. The remaining HPLC fractions were dried down under vacuum.
Specific Library Setup Procedure
Buchwald-Hartwig amination library ( Fig. 2A ).
The following reagent preparation procedures were carried out in a dry box under nitrogen atmosphere. Bromide
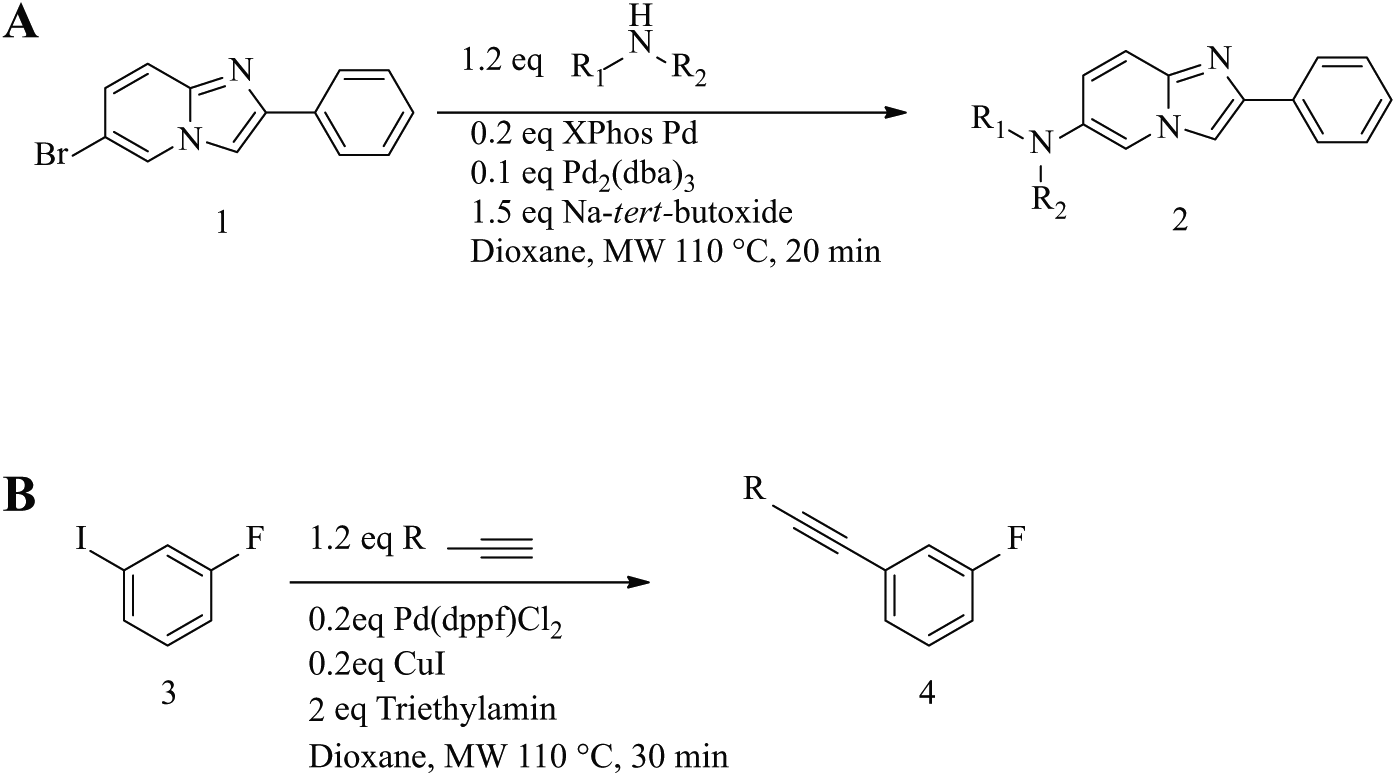
(
Sonogashira library ( Fig. 2B )
The following stock reagents were prepared and placed on the deck of the synthesizer.
1-Fluoro-3-iodobenzene
Alkyne monomers (0.6 mmol; Aldrich Market Select) in dioxane (1 mL; Sigma-Aldrich; CAS: 123-91-1)
Triethylamine (10 mL; Sigma-Aldrich; CAS: 121-44-8)
Pd(dppf)Cl2 (200 mg powder; Sigma-Aldrich; CAS: 72287-26-4)
CuI (200 mg powder; Sigma-Aldrich; CAS: 7681-65-4)
An application was created using AutoSuite where 1-fluoro-3-iodobenzene
Three-step matrix library ( Fig. 3A )
The following stock solutions, reagents, and reaction microwave reaction vessels were prepared and placed on the deck of SWAVE:
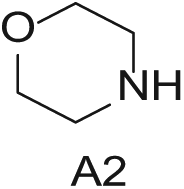
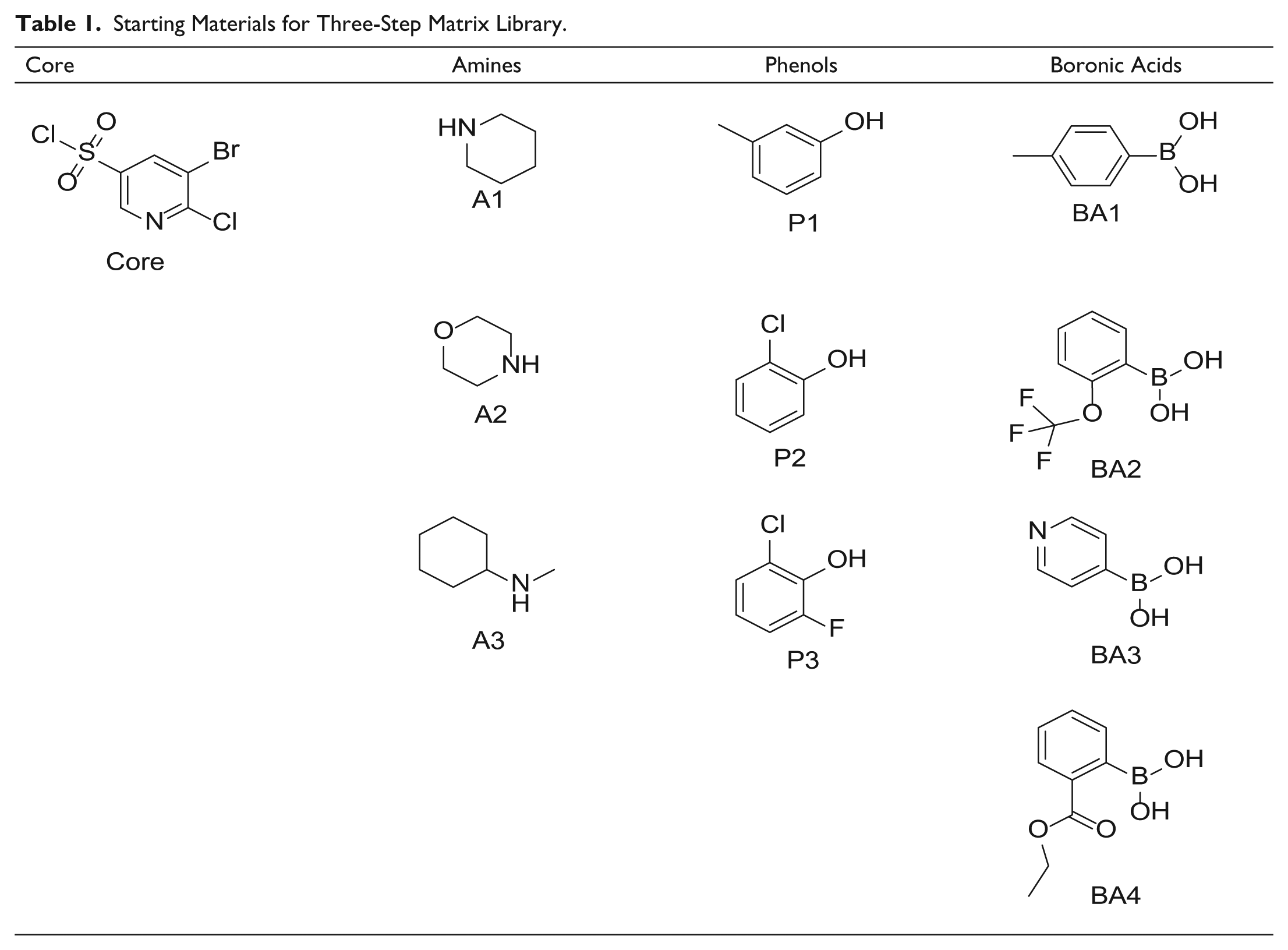
Three 4-mL vials with a stir bar each containing core
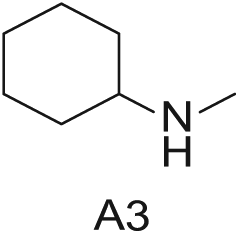
Three 4-mL vials, each containing one of the three amines (200 mg in DMA; Aldrich Market Select)
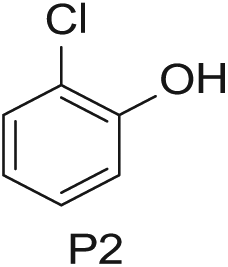
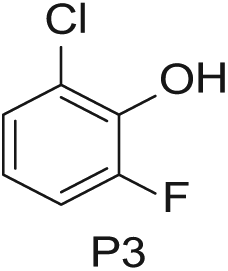
Three 4-mL vials, each containing one of the three phenols (200 mg in DMSO; Aldrich Market Select)
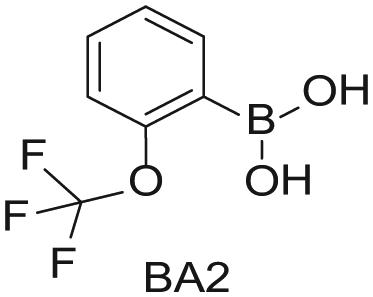
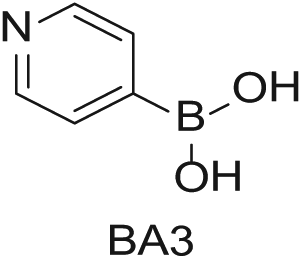
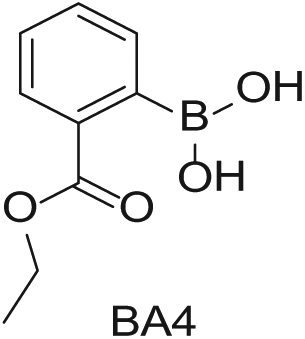
Four 4-mL vials, each containing one of the four boronic acids (100 mg in EtOH; Aldrich Market Select)
Diisopropylamine in a 4-mL vial (2.0 mL; Sigma-Aldrich; CAS: 108-18-9)
Isocyanate resin (2.0 g; Sigma-Aldrich; CAS: 59990-69-1, 1.5 mmol/g loading) in solid dispensing bottle 1
Cesium carbonate (2.0 g; Sigma-Aldrich; CAS: 534-17-8) in solid dispensing bottle 2
Nine empty microwave tubes (set 1) for step 2 synthesis and 36 empty microwave tubes (set 2) for step 3 synthesis
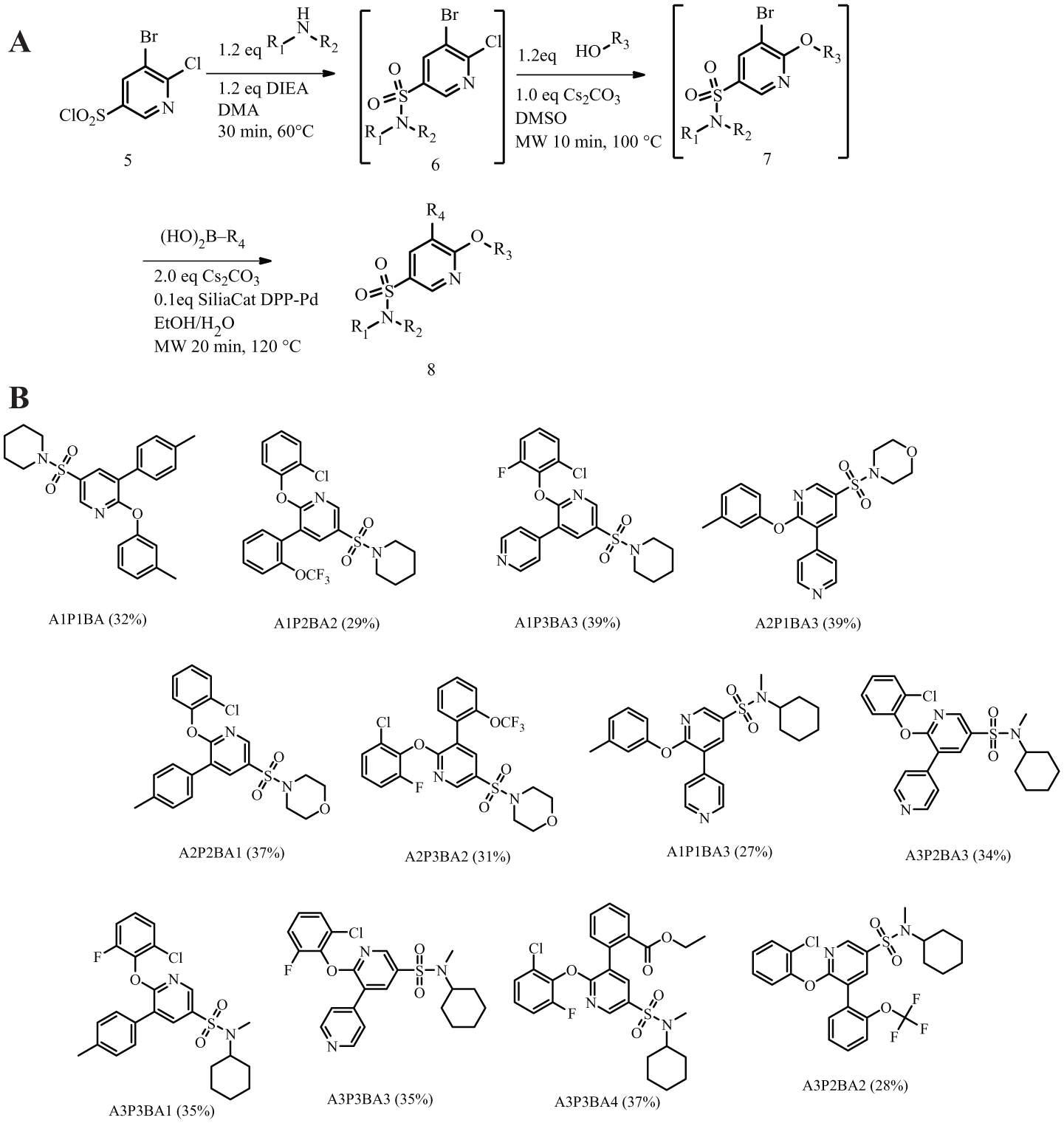
(
A script was created in AutoSuite to carry out the entire three-step matrix library synthesis. The hotplate was preheated to 60 °C, and the amines were added separately to the three vials containing core with the liquid handler; a programmed pause of 30 min in the script allowed the reaction of step 1 to take place. After the 30 min, the content from each 4-mL vial was transferred to the SPE station and passed through cartridges containing isocyanate resin, and the filtrates were collected. The filtrate of each reaction was aspirated and dispensed into three empty microwave vessels in equal portion to create nine reaction vessels for step 2 synthesis (set 1). Solid cesium carbonate was added to each of the nine reaction vessels using the solid dispensing unit, followed by the addition of phenols (phenol 1 to MW tubes 1, 4, and 7; phenol 2 to MW tubes 2, 5, and 8; phenol 3 to MW tubes 3, 6, and 9). All MW tubes were capped and crimped by the capper/crimper tool. The assembled MW tubes were transported to the microwave oven for heating (100 °C, 10 min). After the heating was done, the content of each MW tube from step 2 (set 1) synthesis was dispensed into four empty MW tubes (set 2) in equal portions to create 36 tubes for step 3 synthesis. Four boronic acids were added to the 36 tubes created for step 3 synthesis (boronic acid 1 to MW tubes 1, 5, 9, 13, 17, 21, 25, 29, and 33; boronic acid 2 to MW tubes 2, 6, 10, 14, 18, 22, 26, 30, and 34; boronic 3 to MW tubes 3, 7, 11, 15, 19, 23, 27, 31, and 35; boronic acid 4 to MW tubes 4, 8, 12, 16, 20, 24, 28, 32, and 36). All microwave tubes were capped and crimped by the capper/crimper tool. The assembled MW tubes were transported to the microwave oven for heating (120 °C, 20 min). Simultaneous synthesis-purification was used here: upon finishing a microwave heating step, the system placed the next tube inside the microwave oven and started the microwave heating step (20 min), and during this wait time, the system aspirated and dispensed the crude reaction material from the previous microwave tube through the SPE cartridge and injected the filtrate into the HPLC injector and purified.
HPLC Purification Method
The HPLC system consists of the following components: Gilson (Middleton, WI) 305 and 306 HPLC pumps with 50-mL pump heads; Gilson 806 manometric module; Gilson 155 UV detector with preparative flow cell (0.05 mm path length, 0.7 µL volume); Gilson FC204 fraction collector with four code 22 racks for 18 × 150-mm test tubes, for a total capacity 176 tubes; and Gilson 506C interface box for digital input/output (I/O) communication with the SWAVE system and acquisition of analog UV signals from the UV detector. The system is plumbed in a two-pump “at-column dilution” configuration, 11 with the organic phase pump plumbed through the Valco (Houston, TX) injection valve (W type; 5-mL sample loop) and then the stream mixed with the flow from the aqueous phase pump via a mixing tee immediately prior to the HPLC guard column. Typical injection volumes range from 1 to 3.5 mL. The MS system consists of a Thermo Scientific (Waltham, MA) MSQ-Plus mass spectrometer operating in APCI mode: probe temperature, 550 °C; cone voltage, 30 V; scan time, 0.5 s; and mass range, 170 to 1300 amu. A 4000:1 split of the stream from the preparative HPLC flow is achieved using an Agilent (Santa Clara, CA) G1968D active splitter and delivered to the MS using a Gilson 305 HPLC pump with a 5-mL pump head, operating at 1 mL/min. A mixture of methanol/water (7:3) with 0.1% formic acid is used as the makeup solvent.
The column used is a Phenomenex (Torrance, CA) Luna C8(2), 5 µm, 100 Å, 50 × 30 mm, with a SecurityGuard 15 × 30-mm guard column. A typical HPLC method uses a gradient of acetonitrile (A) and 0.1% trifluoroacetic acid in water (B), at a flow rate of 40 mL/min (0–1.0 min 10% A, 1.0–9.0 min linear gradient 10%−100% A, 9.0–9.5 min 100% A, 9.5–10.0 min linear gradient 100%−10% A). Column equilibration time was dependent on reaction time but was a minimum of 2 min.
The HPLC-MS system is controlled through a combination of Thermo Xcalibur 2.0.7 software and a custom application written in-house using Microsoft Visual Basic 6.0 (Microsoft Corp., Redmond, WA). The Thermo Xcalibur software is used for MS method creation and MS data acquisition. The custom application uses the Thermo Xcalibur developers kit (XDK) to send sample sequences to Xcalibur, initiate data acquisition, and read MS data in real time. National Instruments (Austin, TX) Measurement Studio controls are also used for displaying chromatograms. All HPLC components are controlled by the custom application using Gilson serial I/O commands (GSIOC). Communication between HPLC components, the MS, and the SWAVE system is made through digital I/O signals from the Gilson 506C interface box.
The purification data browser application was similarly written in Visual Basic 6.0 and uses the Thermo XDK and National Instruments Measurement Studio controls for displaying chromatograms and mass spectra. All raw data files are archived and available over the network to multiple users.
Results and Discussion
The synthesis portion of the integrated system employed the commercially available Chemspeed SWAVE (Chemspeed Technologies, Augst, Switzerland), which is capable of assembling and delivering both solid and liquid reagents to a microwave tube ( Fig. 4A ). The preparative HPLC-MS purification system was developed in-house using components from several vendors ( Fig. 4B ).

Integrated robotic instrument for synthesis and purification. (
SWAVE Control Software
The control software for the SWAVE system (AutoSuite) is highly flexible and is capable of controlling various components and scheduling robotic movements in batch or sequential mode. Since the integrated Biotage (Uppsala, Sweden) microwave system is a set up for sequential heating, a linear synthesis-purification workflow could be lengthy as each individual microwave step could be over 30 min or more in some cases. To speed up the overall process, an AutoSuite application was developed whereby microwave heating and purification of the previous product can be carried out simultaneously ( Fig. 5A ). With a typical HPLC gradient method of 10 min, purification is usually completed before microwave synthesis, making the synthesis portion of the operation rate limiting for the overall process. However, a typical hotplate reaction protocol employed fully assembled reaction vessels from a standard liquid handling robot, placed on the SWAVE and heated for a specific time period and temperature. At the end of the heating step (typically between 1 and 4 h), all compounds are available for purification one at a time by the integrated HPLC system. Under this operation mode, purification time will become rate limiting for the overall process. To address this, we developed software capability to halt the HPLC purification after the compound of interest has been collected (see HPLC components section below for detail). This capability proved to be effective in reducing purification time and had the added benefit of savings in solvent use.
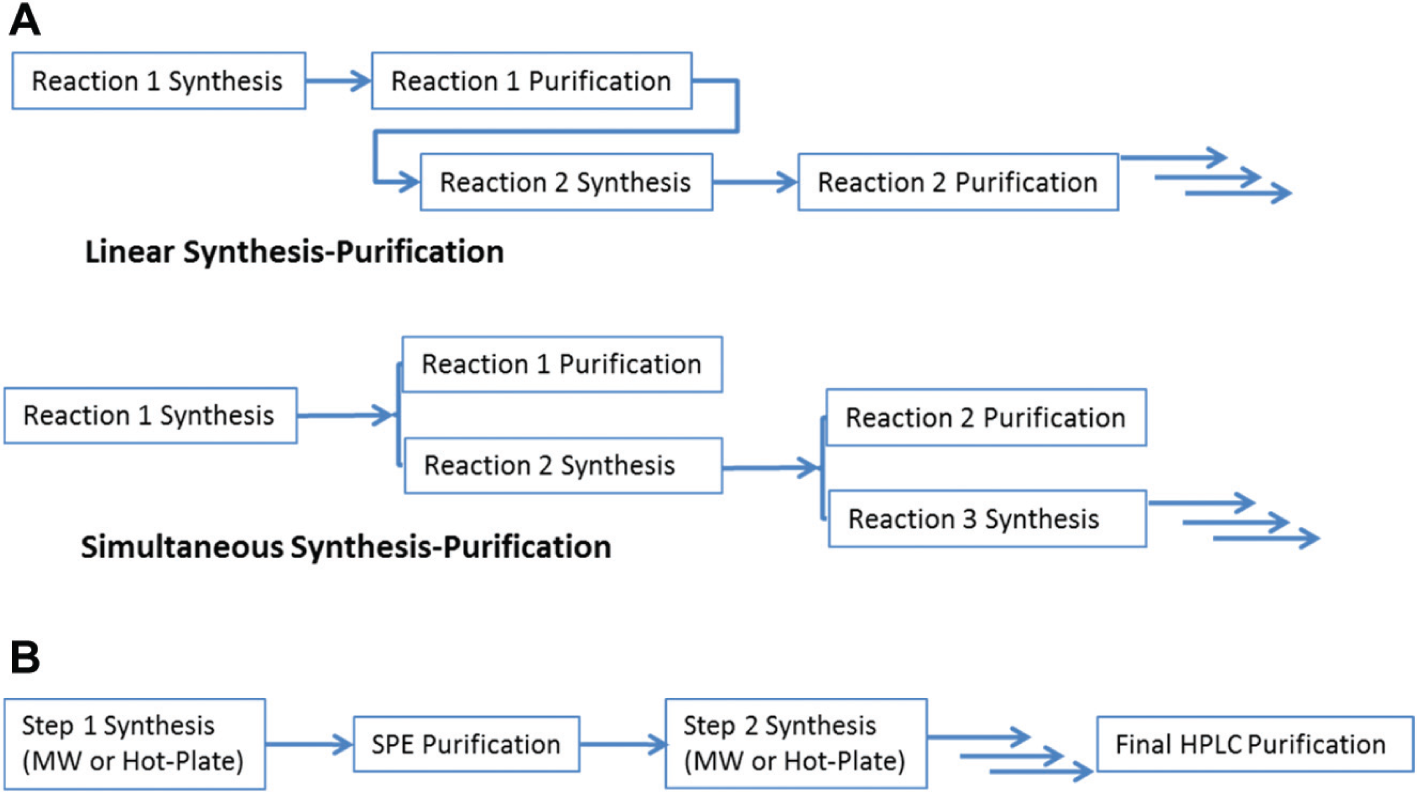
(
Another important advantage of the fully automated system versus traditional bench-top library assembly is the “just-in-time” delivery of reagents. Traditionally, chemists would mix all reagents for the entire library at the beginning of the library production process, and reaction mixtures were then sent to the microwave for heating one at a time. This translates to significant wait time for reactions late in the queue, and side reactions can take place while idling, affecting product yield and complicating purification. We addressed this by programming the mixing of reagents only when the microwave oven is available and by limiting wait time to less than 2 min from the addition of the last reagent to the start of microwave heating.
The highly flexible control software also enables the setup of multistep synthesis within the same operation. Crude product from step 1 is aspirated from microwave or 4 mL vials and dispensed into a SPE cartridge containing solid sequestering agents, and the filtrate is then reintroduced for the next step, heating with the microwave oven or hotplate on deck. The final product would then be purified with the integrated HPLC to yield product in high purity ( Fig. 5B ).
Solid Dispensing Module
One of the main advantages of the SWAVE system is the ability to dispense solid materials accurately. The overhead gravimetric solid dispensing module incorporates an overhead balance and a solid dispensing bottle housing an extruder screw that rotates within the cap to dispense solid material to targeted container. During the solid dispensing step, the overhead balance monitors the weight of material dispensed and stops rotating the extruder screw once the targeted weight is achieved. 12
Liquid Handling/Custom Needle
The handling of liquids is performed by a module with four needles capable of 360-degree rotation for ease of access to vials on the deck. Custom coaxial needles (Angel Science Technologies, Edmonton, Canada) with beveled tips were used for aspirating and dispensing liquid between various septum-capped vials. The same needles were also used to transport crude material to the HPLC injection port.
Capper/Crimper
After various reagents (solid and/or liquid) were added to the microwave tube, the capper/crimper module was used to cap, crimp, and transport the microwave vials to and from the microwave oven. Caps are stored in a tube on the module and are automatically placed on each vial. The cap reservoir tube can be automatically refilled with caps during an application.
Microwave Unit
The integrated Biotage Initiator EXP microwave oven is controlled via API software within AutoSuite, allowing the user to specify microwave conditions for each reaction (irradiation time, temperature, and solvent dielectric property). Fully assembled microwave tubes were transported to the microwave cavity by the capper/crimper module.
SPE Module
Since crude material is injected into HPLC without further workup, a filtration step is necessary to avoid clogging of the HPLC injector port and sample loop by solid materials. This was achieved by passing crude material through the SPE block equipped with various cartridge types (single filter, double filter, single filter with celite) (MMHT.CHAN, Durrington, UK) prior to HPLC injection. To ensure minimum retention of material inside the cartridge, a rinse step was incorporated followed by positive air pressure push created by dispensing air with the needle through the air-tight cartridge cap. Scavenging materials can also be placed inside an empty cartridge for specific removal of the targeted compound. An example of such use was described in the three-step matrix library synthetic sequence ( Fig. 3A ).
Hotplate/Stirrer
Historically, about 45% of all compounds made from the AbbVie HTC group required simple heating on a hotplate. To include these type of libraries, we have added an in-house modified heater/stirrer (IKA Works, Wilmington, DE, RCT Basic, part number: 3810001) equipped with a temperature controller (IKA ETS-D5, part number 3378000) attached to the side of the heating rack. The hotplate was placed under the aluminum racks, and the position was adjusted by a lab-jack, as shown in Figure 1 . To focus the heating to the intended racks only, the lab-jack/hotplate combo was raised to push against the bottom of the aluminum rack and created a 1-mm air gap between the aluminum rack and the rail. This proved effective in isolating the heating to only the heating rack and not to the adjacent equipment. For hotplate reactions that required no SPE filtering, the material was aspirated directly from the 4-mL vials to the HPLC injection port. To maximize the transfer of material, custom racks built by AutoChem Pte Ltd (Singapore) with a 6-degree tilt were used to hold the reaction vials.
Inert Atmosphere
Some reagents are sensitive to moisture, oxygen, or carbon dioxide, 13 and many useful reactions routinely used by medicinal chemists use these sensitive reagents. An example would be the Buchwald-Hartwig amination, which requires inert atmosphere conditions that were normally done by adding reagents and assembling reaction vessels inside a glove box, which discouraged the practical use of automation due to limited space inside the glove box. The SWAVE system is constructed inside an airtight chamber with inert gas distribution tubing around the lower perimeter of the deck. When the front hatch is lowered and inert gas is pumped inside the chamber, an inert atmosphere is created to carry out sensitive reactions. Sensitive reagents can be prepared in various size vials with a septa cap to preserve their integrity and placed under inert atmosphere inside the SWAVE chamber.
HPLC Injection Valve
Injection of the crude reaction products onto the preparative HPLC system is made via an injection valve (Valco, W type) fitted with a 5-mL stainless steel sample loop and injection port (Gilson, part number 26035413). The injection port is accessible on the SWAVE bed, and the crude product is introduced via a needle on the four-needle module (1.5 mm OD; Angel Science Technologies, part number AST1877).
HPLC Components
The custom preparative HPLC-MS system ( Fig. 4B ) was developed in-house using components from several vendors and is similar to our previously described system 1 with software modifications to allow interfacing to the microwave synthesizer. A two-pump preparative HPLC system configured for “at-column dilution” 11 introduces crude reaction mixtures directly from the HPLC injection valve onto the preparative HPLC column (Phenomenex Luna C8 (2), 5 µm, 100 Å, 50 × 30 mm). An active flow splitter 14 and makeup pump are used to deliver a 4000:1 split of the column eluent to the mass spectrometer, operating in APCI mode, with the main stream being delivered via the UV detector flow cell to the fraction collector.
The HPLC-MS system is controlled through a custom software application, written in-house ( Fig. 6A ) to control all HPLC components and interface with the mass spectrometer. The purification system synchronizes with the SWAVE system through digital I/O signals for “Inject start” and “HPLC ready” conditions. The purification software allows the user to import run-list information from a structure-data file, provided by the LIMS software, and configure the purification run parameters. Full control of the LC gradient, UV wavelengths used for detection, and MS method parameters are provided. In addition, a variety of fraction collection options are available to allow collection of the desired product when the MS-extracted ion chromatogram of the target mass, the UV signal intensity, or both are above-specified thresholds. Fraction collection parameters can be used in combination with time collection windows. Further options are available to enforce collection of only one fraction per sample, stop the run if fractions are not collected for a specified number of samples, stop the run upon loss of system pressure or system overpressure, and allow the user to trigger fraction collection manually. Parameters can also be set for MS-directed fraction collection to collect unreacted starting material, or a common adduct or by-product, in addition to the desired product. To maximize throughput and reduce solvent consumption, the software allows the user to optionally select parameters to stop the HPLC gradient, as soon as the product of interest has been collected, and switches the HPLC pumps to 100% organic for a specified time to flush the column of remaining components. After each run is completed, a PDF report is generated and archived along with the raw data for viewing within the LIMS software.
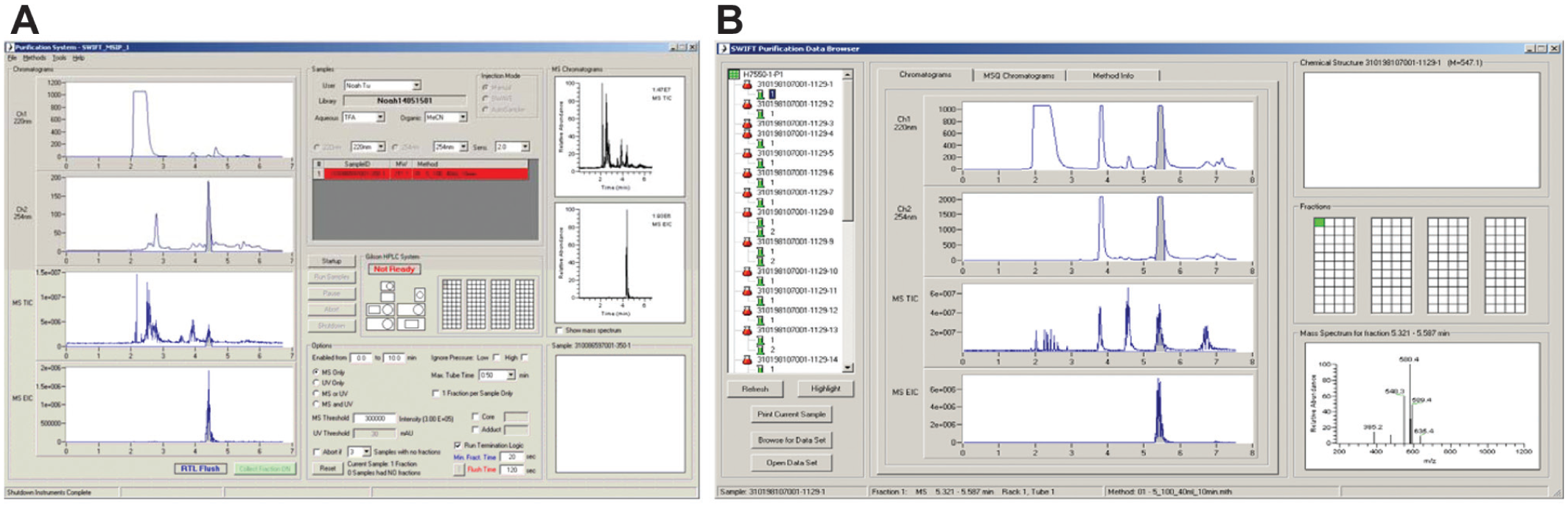
(
A second software application allows the user to monitor the library purification data throughout the run ( Fig. 6B ) so that modifications can be made to the HPLC method and fraction collection parameters, if required.
Example Libraries
Three-Step matrix library
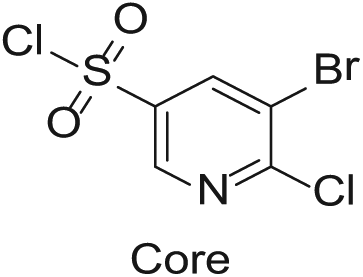
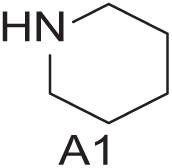
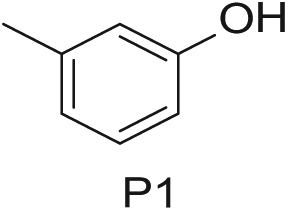
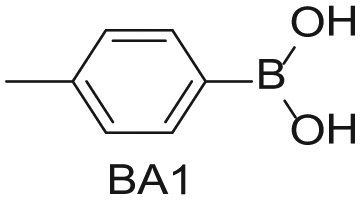
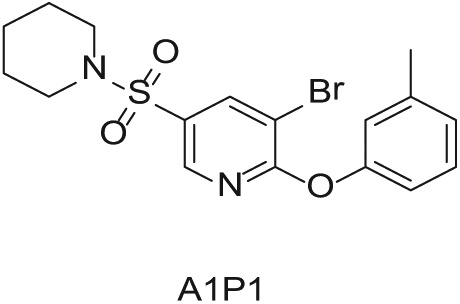
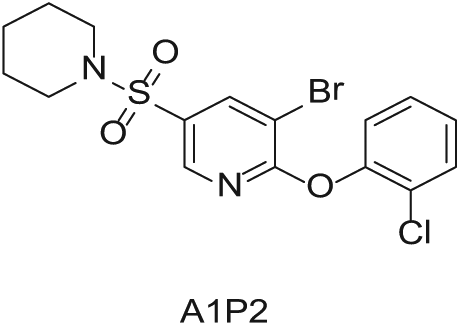
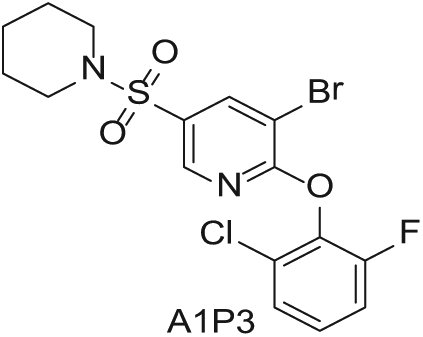
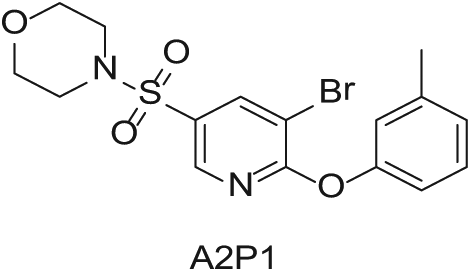
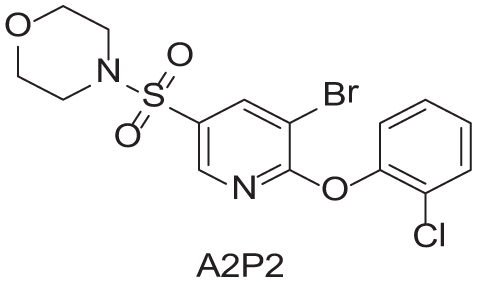
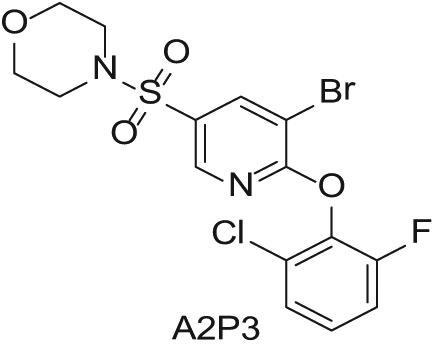
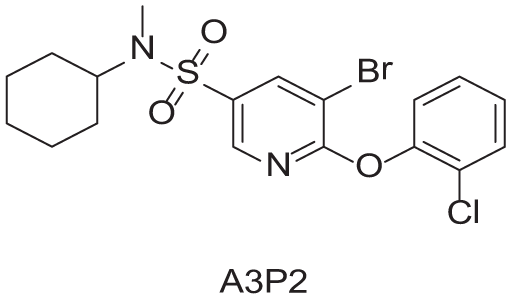
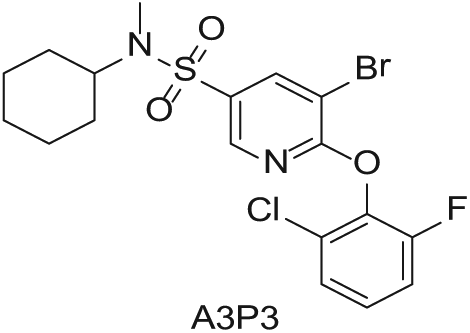
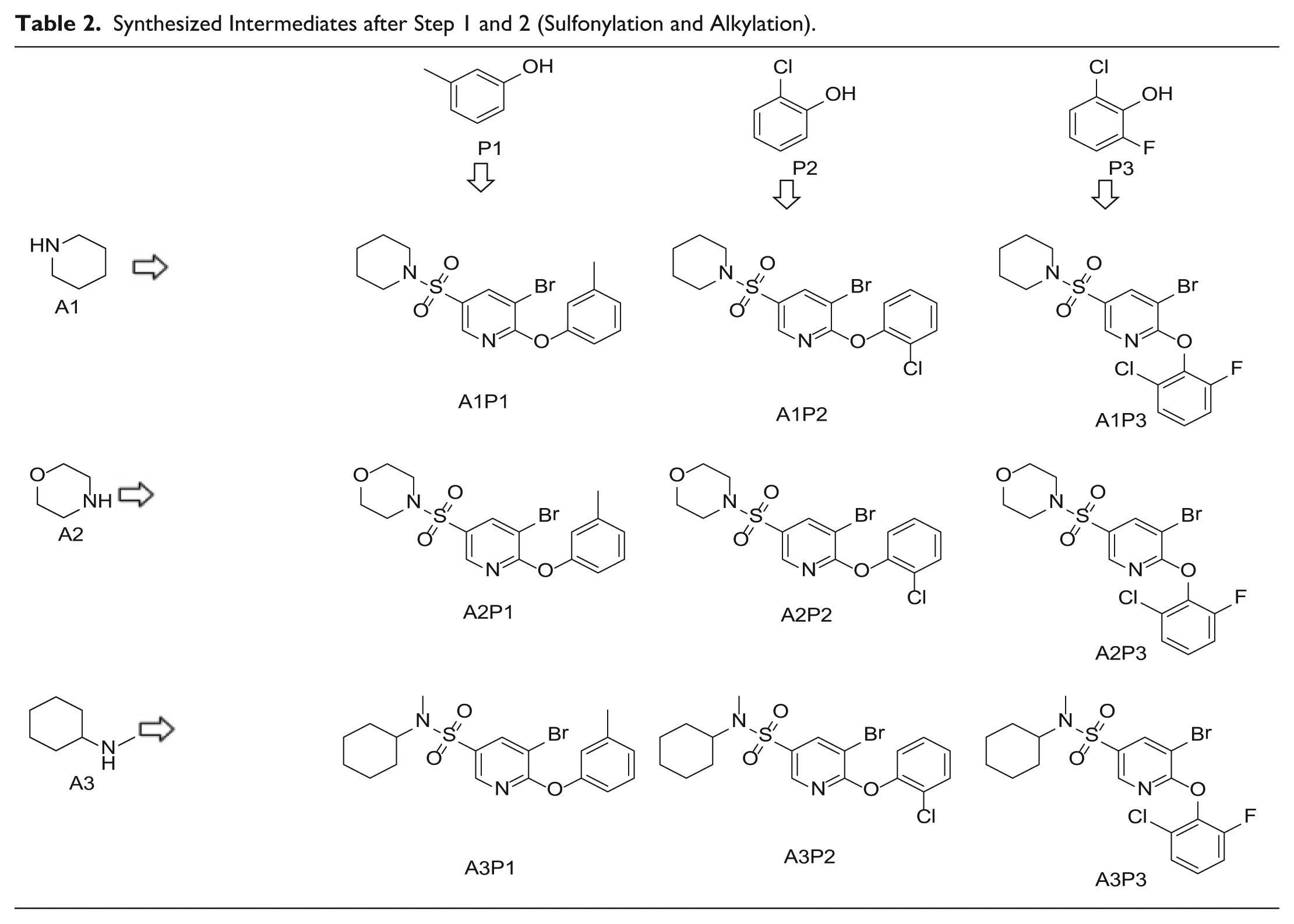
To demonstrate the flexibility and versatility of the system, a three-step matrix library of 3 × 3 × 4 using 5-bromo-6-chloropyridine-3-sulfonyl chloride
Starting Materials for Three-Step Matrix Library.
Synthesized Intermediates after Step 1 and 2 (Sulfonylation and Alkylation).
Buchwald-Hartwig cross-coupling reaction
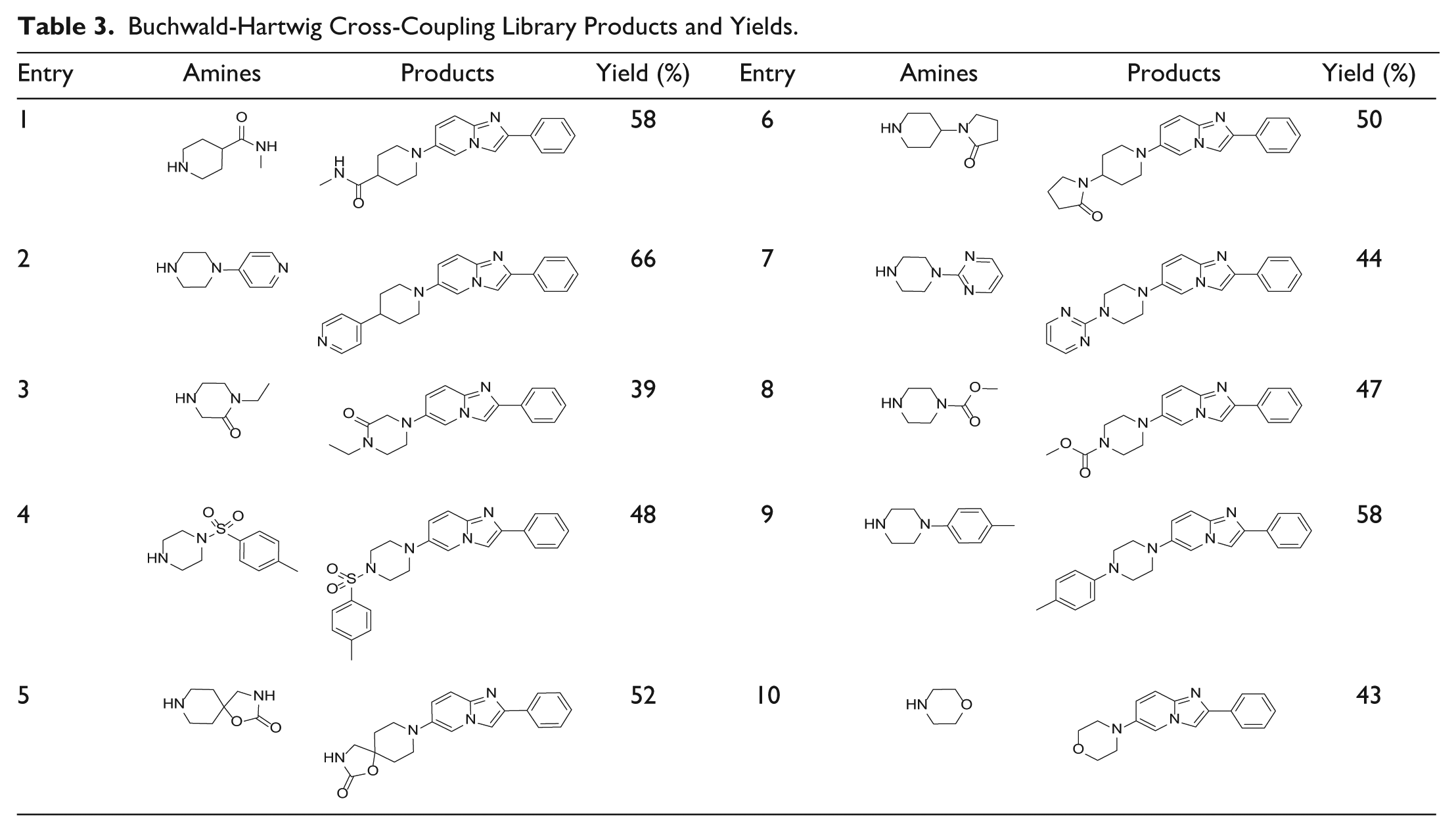
The palladium-catalyzed coupling of amines with aryl halides or aryl alcohols is widely used in natural product synthesis and drug discovery laboratories due to its versatility and reliability. However, automated Buchwald reactions have been a challenge in the past mainly because of its sensitivity to air and the need for a glove box for reagent preparations and reaction assembly.
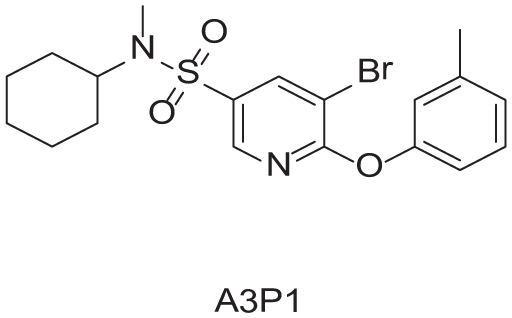
To demonstrate the glove box–like environment of the system, we have carried out a 10-membered library using bromide
Buchwald-Hartwig Cross-Coupling Library Products and Yields.
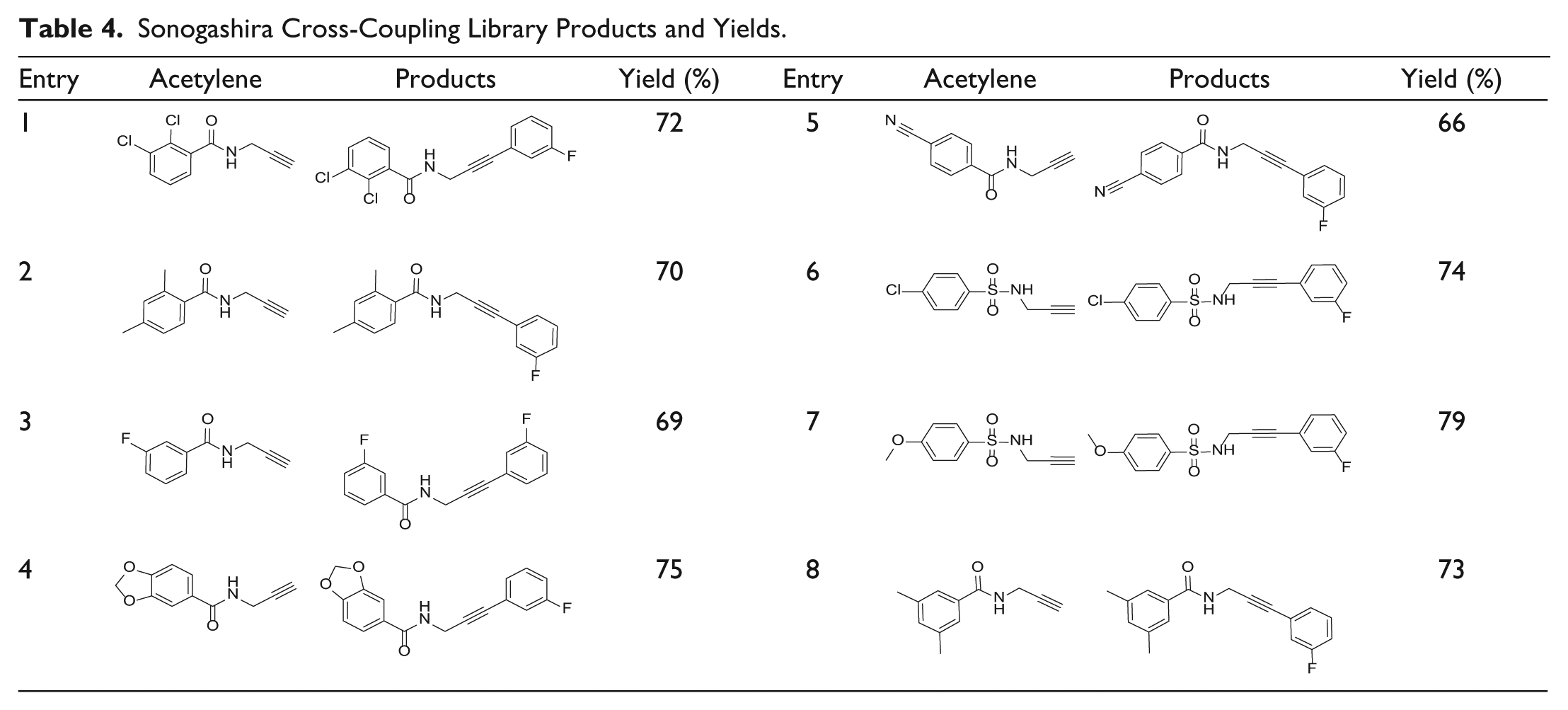
Sonogashira cross-coupling reaction
The Sonogashira cross-coupling reaction has been employed in a wide variety of areas, due to its usefulness in the formation of acetylenic compounds (
Fig. 2B
). An eight-membered library using 1-fluoro-3-iodobenzene
Sonogashira Cross-Coupling Library Products and Yields.
The integrated synthesis-purification system highlighted in this article has provided a versatile and efficient platform for the production of compound libraries. The average cycle time to synthesize and purify a 48-member library of 30-mg compounds was 24 h. Although the time needed for a compound to complete the heating step in a microwave oven can be 30 min or more, the ability for the system to simultaneously handle compounds at multiple production stages shortens the overall production time for the library. The in-house added hotplate modification expanded the system capability to include parallel synthesis and shorten the time needed for synthesis. The ability of the HPLC control software to automatically stop the purification gradient upon collection of the expected product further reduces cycle time and solvent usage. The combined microwave/hotplate platform inside a glove box–like environment is capable of handling most, if not all, chemical transformations for a medicinal chemist.
In summary, we have developed a novel, first-in-class, fully automated synthesis-purification system based on the SWAVE platform and in-house developed robotics. The microwave/hotplate-based synthesizer with integrated purification system complements the flow synthesis-purification platforms and fills in the gaps where library production using flow chemistry is not amenable. The system demonstrated exceptional cycle times as well as versatility for library production.
Footnotes
Acknowledgements
The authors thank Ana L. Aguirre for assistance with reaction testing. They also thank Mark Myers and Mark Levorse of Chemspeed for technical support. All contributors are AbbVie employees.
Author Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NPT, PAS, and KS are all employees of AbbVie. This research was sponsored by Abbvie, Inc. AbbVie, Inc. contributed to the study design, research, and interpretation of data; reviewing; and approving the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.