Abstract
Endotoxin, present in the outer membrane of all gram-negative bacteria, can pose serious risks to human health, from irreversible shock to death. Therefore, it is essential to develop sensitive, accurate, and rapid methods for its detection. The rabbit pyrogen test is the first standard technique for endotoxin detection and, nowadays, has been replaced by the Limulus Amoebocyte Lysate test, which is the most popular detection technique for endotoxin. With in-depth understanding of endotoxin, biosensors based on endotoxin-sensing components are promising alternatives to pursue in developing low-cost, easy-operation, and fast-response endotoxin detection techniques. This article summarizes the recent advances of endotoxin detection methods with a particular emphasis on optical and electrochemical biosensors based on various sensing elements ranging from nature biomolecules to artificial materials. As the research and technological revolution continues, the highly integrated and miniaturized commercial devices for sensitively and reliably detecting endotoxin will provide a wide range of applications in people’s daily life.
Introduction
Endotoxin was introduced in the 19th century to describe the component of gram-negative bacteria responsible for the pathophysiological phenomena associated with gram-negative infections. 1 In the late 20th century, the chemical structures and biological properties of endotoxin were gradually revealed as the result of improvements in analytical techniques and apparatuses.
Biochemical Structure of Endotoxin
Endotoxin (whose more proper biochemical description is lipopolysaccharide [LPS]), is referred to an inherent fraction present in the outer membrane of all gram-negative bacteria ( Fig. 1 ). Endotoxin is supposed to represent a nonconcrete category of biomolecules that are released after the death of bacterial cells and cause toxin effects, such as fever, septic shock, multiorgan failure, and even death. In fact, these immune responses are in charge of Lipid A, which is the active moiety of the LPS biochemical structure. 2 Lipid A is the membrane anchor of LPS molecules, composed of a hydrophilic, negatively charged bisphosphorylated diglucosamine backbone and a hydrophobic domain of six (Escherichia coli) or seven (Salmonella) acyl chains in amide and ester linkages. 3 Because of its unique structure, Lipid A is responsible for LPS molecules’ biological function, specificity, and affinity to the relative proteins.
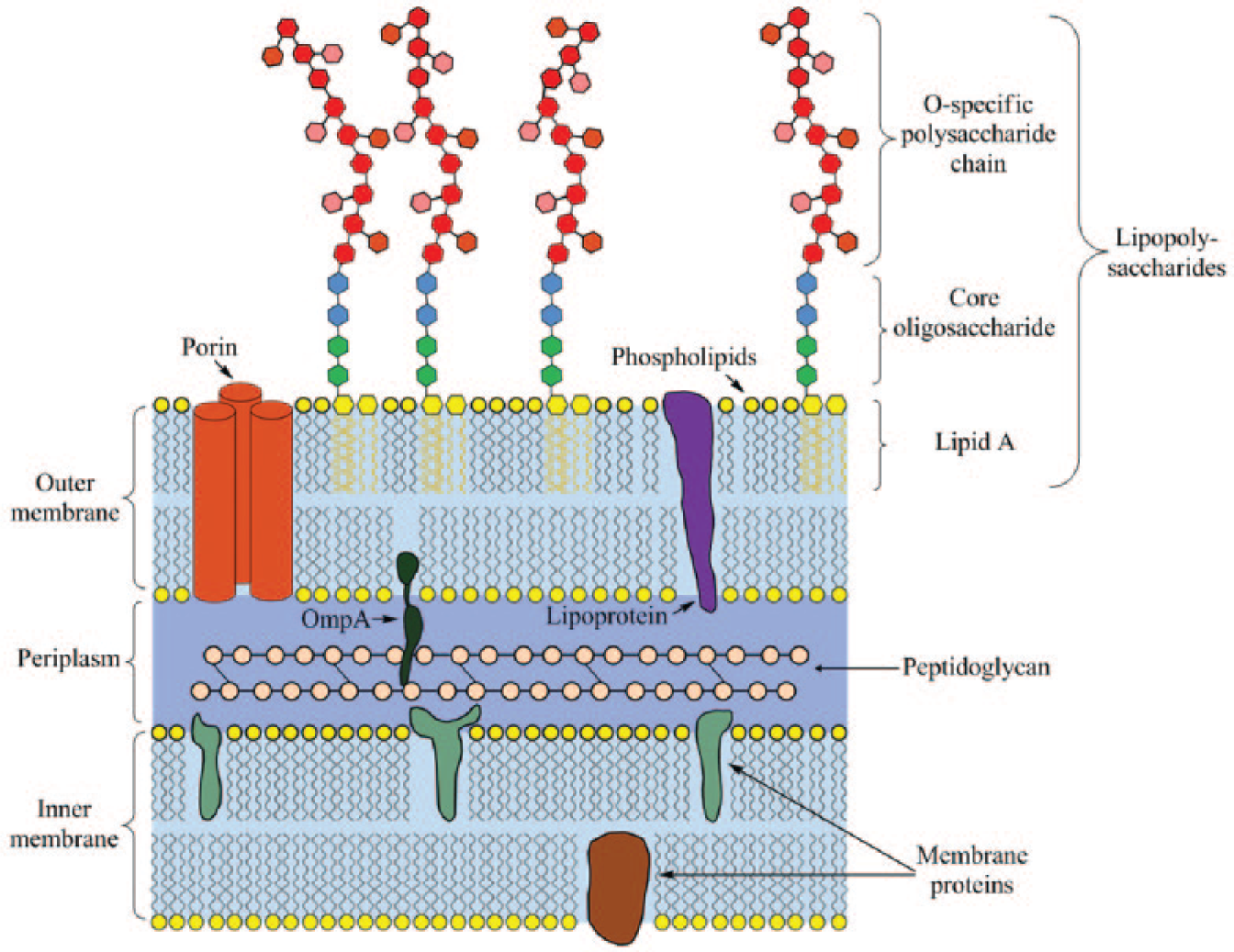
Illustration of the lipopolysaccharide biochemical structure existing in the outer membrane of gram-negative bacteria.
The other two parts of LPS molecules are O-specific polysaccharide chain and core oligosaccharide. The core oligosaccharide 4 of LPS can be divided into an inner subdomain, which is linked directly to Lipid A, and an outer subdomain, which provides an attachment site for the O-specific polysaccharide chain. The covalent structures of outer cores are more variable than those of inner cores. Other than providing linkage, the function of the core oligosaccharide is unknown.
The O-specific polysaccharide chain 5 is attached to the terminal of the outer core and is exposed to the external environment of bacterial cells. As a kind of immunogenic substance, the O-specific polysaccharide chain may help bacteria evade the immune system. Compared with core oligosaccharide and lipid A, the O-specific polysaccharide chain has highly variable structures. There are more than 1000 distinct immunochemical variants of the O-specific polysaccharide chain in Salmonella, whereas O-specific polysaccharide chains may absent from some kinds of LPS molecule structures.
Toxicity of Endotoxin
LPS molecules of high chemical stability are released to the environment with the death of gram-negative bacteria cells. Injured intestinal mucosa allows access of LPS molecules to human blood, which can introduce endotoxemia, 6 symptoms affecting the structure and function of organs and cells, changing metabolic functions, raised body temperature, modification of hemodynamics, and septic shock. When LPS molecules access the human system by circulating through the liver, various inflammatory cytokines, 7 such as tumor necrosis factor, interleukin-6, platelet activating factor, and so on, are overexpressed by the activation of the innate immune system and lead to systemic inflammatory response syndrome, which has been reported as the cause of death related to severe acute respiratory syndromes, cancers, large-area burns, and acute peritonitis.8–12 The high mortality rate associated with endotoxin-induced shock remains a major clinical problem, especially in debilitated and immunosuppressed patients.
Furthermore, endotoxin is not eliminated when the organism is killed by the sterilization process; instead, the release of LPS takes place upon the death of cells. Although the products may have been sterilized, the endotoxin of the organism still remains if gram-negative organisms were present prior to sterilization. Testing for this LPS in the finished products is an important part of ensuring the safety of the sterilized products, especially in the fields of biological products, medical devices, parenteral drugs, food and water security, and so on.
In this article, LPS detection methods are introduced and the development of new detection techniques are described. The biosensors taking LPS affinity components as sensing elements are summarized because of their inherent properties of fast response, easy operation, low cost, high sensitivity, and great specificity, which satisfy the development requirements of endotoxin detection. With the improvement of microfabrication and nanomaterials, integrated and miniaturized endotoxin detection devices of high sensitivity and great stability will be commercially available in the future.
Endotoxin Detection Methods
Rabbit Pyrogen Test
The rabbit pyrogen test 13 was the first method approved by US Food and Drug Administration for LPS detection. It was developed in the 1920s on the basis of the rise in temperature of rabbits after an intravenous injection of a test solution. The rabbit pyrogen test has obvious defects related to cost and time period, so it has been gradually replaced by the Limulus Amoebocyte Lysate (LAL) test in practical use. Nowadays, the rabbit pyrogen test is applied only as supplement of the LAL test to analyze pyrogenic materials in the earlier development phase of parenteral devices.
LAL Test
Limuluspolyphemus, existing in the blood of horseshoe crabs, can form a clot when exposed to LPS. 14 Based on this unique property, the most popular LPS detection technique, the LAL test, has been developed. The protease cascade employed in the LAL test is initiated by the combination of LPS and zymogen Factor C. The activated Factor C stimulates Factor B, which converts the proclotting enzyme to the clotting enzyme. Finally, two peptide bonds in coagulogen are catalytically cleaved to form coagulin gel. 15
The simplest form of LAL test is the LAL gel clot assay, in which a piece of LAL gel will form a clot with a certain amount of LPS. 16 Because the critical LPS amounts clotting the LAL gel pieces are known, when a dilution of sample containing LPS is applied to a given piece of gel, a clot will form in the amount equivalent to the LPS sensitivity of the given piece of gel. The LPS concentration is approximated by continuing to apply the sample to an LAL gel of less sensitivity (higher critical LPS amount) until a negative reaction (no observable clot) is obtained, a process that can take up to several hours.
With time, some advanced techniques (e.g., chromogenic, 17 turbidimetric, 18 or viscosity 19 ) are introduced to improve the traditional LAL test. In particular, various commercial LPS detection kits have been developed, 20 such as the endpoint chromogenic LAL assay (Thermo Scientific; Sigma; GenScript), kinetic turbidimetric LAL assay (Lonza BioScience), and kinetic chromogenic LAL assay (Lonza BioScience), providing faster and more economical LPS detection methods of quantity or quality.
However, LAL is based on an enzymatic reaction. When applied to actual cell lysate, the LAL results are susceptible to the effect of protease. Moreover, it has been reported that LAL has shown reactivity to some polymeric forms of glucose. 21 β-(1,3)-D-glucan, existing in the cells of fungi, algae, and yeasts, is one of them. Although β-(1,3)-D-glucan is not pyrogenic, it can activate Factor G, which is able to trigger the coagulation cascade, interfering with the LAL response to LPS. 22
Biosensors Based on Endotoxin-Affinity Components
In addition to the rabbit pyrogen test and LAL test, biosensors based on LPS affinity components are promising alternates to pursue in developing a rapid and accurate endotoxin detection technique. Biosensors23–26 are compact analytical devices that detect the presence of a target by using a biological recognition element and then monitoring the optical, electronic, mass, or magnetic signal variation that is induced by the interaction of the biological recognition elements and the analyte of interest. Usually, the biological recognition elements that affect the selectivity of biosensors should possess a number of essential features: high specificity to the targets, invariability under storage and detection conditions, and good stability over a large number of assays. More importantly, the reactions of recognition elements and targets should be accurate, rapid, reliable, atoxic, and controllable from environmental factors (e.g., pH, ionic strength, temperature, and so on).27–30
Protein-Based Biosensors
LPS molecules are structurally able to interact with suitably modified surfaces via electrostatic and hydrophobic interactions, coordination through the phosphate groups, or, more selectively, binding to recognized proteins or aptamers. Scientists have explored and described several LPS-affinity components in plasma, 31 some of which have been used as biological recognition elements to construct endotoxin detection biosensors.
Cluster of differentiation 14 (CD14) accompanies lipopolysaccharide-binding protein (LBP) 32 forming a receptor complex. LBP is a lipid-transfer protein described to accelerate the transfer of monomeric LPS molecules from LPS aggregates to the hydrophobic binding pocket of CD14. Interaction between LBP and CD14 is important for this transfer process. CD14 transfers the monomeric LPS to a complex of Toll-like receptor 4 (TLR4) and myeloid differentiation protein-2 (MD-2), 33 which causes the rearrangement of TLR4 and promotes the secretion of proinflammatory cytokines after the combination of LPS and the TLR4/MD-2 complex ( Fig. 2 ). Another protein from the LBP family is bactericidal/permeability-increasing protein (BPI), 34 which can potently inhibit LPS activation at certain concentrations. Similar to BPI, serum amyloid P component 35 and cationic protein 18 (CAP 18) 36 are demonstrated to have combination and detoxification capacities to LPS at local sites of inflammation. The last three proteins are also known as endotoxin-neutralizing proteins (ENPs), which are related to a category of proteins capable of binding and detoxing to LPS. ENPs were firstly isolated from amebocytes of the horseshoe crab 37 (i.e., Limulus anti-LPS factor), slowly expanding to other alike functional proteins, such as the above-mentioned proteins from human plasma and the lectins from plants.
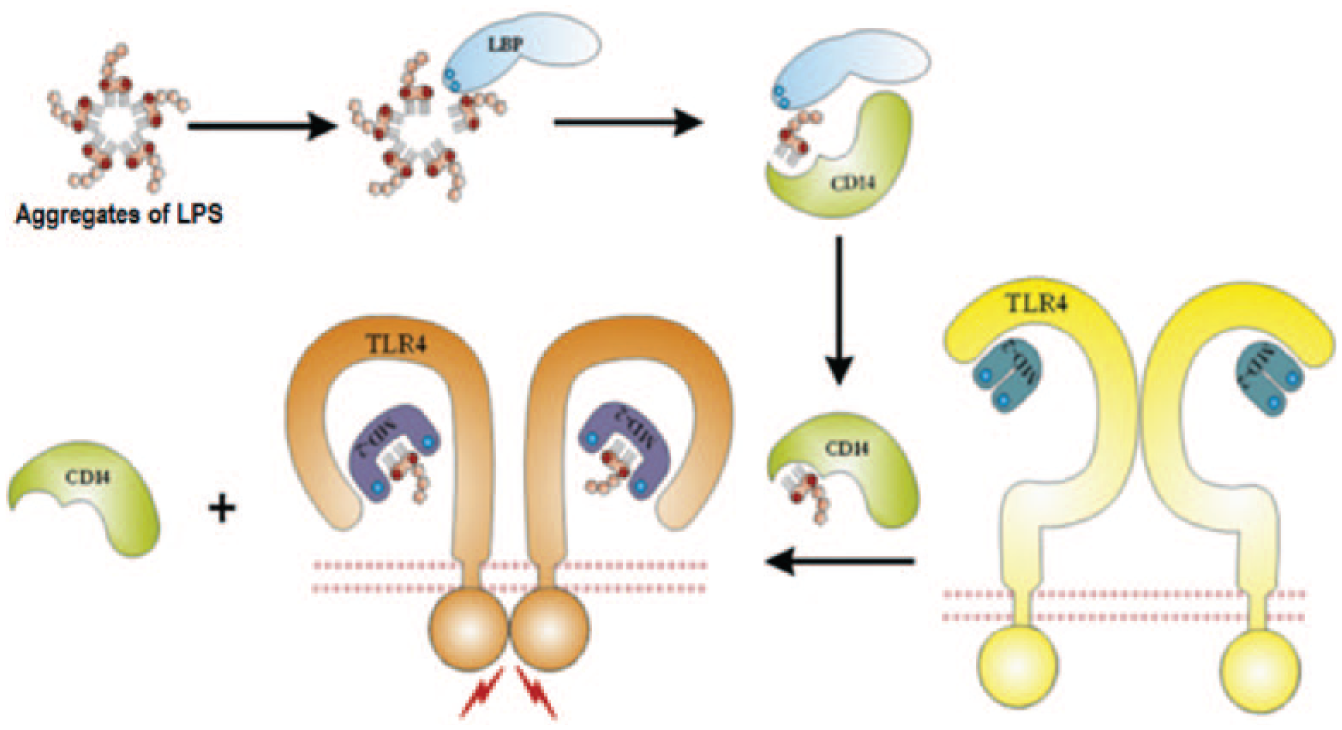
Schematic representation of the recognition and transfer process of lipopolysaccharide by lipopolysaccharide-binding protein, cluster of differentiation 14, myeloid differentiation protein-2, and Toll-like receptor 4. Reproduced from reference 33 with permission from Elsevier.
As the first described receptor for LPS, CD14 was purified by Burkhardt et al. and was used to develop a novel electrochemiluminescent assay to detect bioactive LPS. 38 Yeo et al. used the complex of human recombinant TLR4 and MD-2 as the recognizing component to develop an electrochemical sensor that can detect LPS in the range of 0.0005 to 5 EU/mL and avoided the interference of dipalmitoyl phosphatidylcholine, an agent structurally similar to LPS. 39 Concanavalin A (ConA), as a lectin, is also considered an LPS recognition element. Hreniak et al. 40 applied ConA to develop a luminescence assay that can detect LPS from 0.2 to 20 µg/mL, whereas da Silva et al. 41 had ConA covalently bonded on polyaniline-covered steel electrodes to develop an impedimetric sensor for LPS detection. CramoLL lectin was adsorbed on the surface of L-cysteine–modified gold nanoparticles to develop an electrochemical impedance spectroscopy (EIS) biosensor that can detect LPS from 25 to 200 µg/mL. 42 A capacitive biosensor based on lectins, directly self-assembling on the gold electrode surface, was also fabricated with a detection limit of 1.0 × 10−13 M and linearity ranging from 1.0 × 10−13 to 1.0 × 10−10 M. 43 When applied to endotoxin samples purified from an E. coli cultivation, the capacitive biosensor achieved a good agreement to the LAL test. Priano et al. developed an amperometric competitive assay44–46 by taking ENP (a recombinant version of the Limulus anti-LPS factor) as the recognition element to detect LPS from Salmonella minnesota. Competing with horseradish peroxidase–labeled LPS, the free LPS in samples can be detected as low as 100 pg/mL.
As the research developed, many kinds of proteins and synthetic substrates with affinity to LPS were acquired, promoting the detection of LPS by electrochemical or optical biosensors. Limulus factor C is the key protein in the LAL test. Because of the finite resource of horseshoe crabs, recombinant Factor C (rFC) 47 has been developed from the clone of their DNA. Inoue et al. 48 developed an rFC-based biosensor that can detect LPS concentration as high as 1000 ng/L. rFC was first activated by endotoxin and then hydrolyzed a synthetic substrate, Boc-Val-Pro-Arg-p-nitroanilnide, which generated an electrochemical active compound, p-nitroaniline to give rise to a detecting signal at −0.75 V by differential pulse voltammetry (DPV). Later, they applied the synthetic substrate to the last step of the LAL test to realize the electrochemical detection of endotoxin with the LAL test. The electrochemical signal was enhanced along with the increased concentration of endotoxin in the range of 0.5 to 1000 EU/L. 49 Finally, they constructed the endotoxin biosensor on a screen-printed electrode overlaid by a hydrophilic cover and a fluidic channel to produce a disposable and practical chip sensor. 50 Other electrochemical LAL test reports have been reviewed in reference 51.
Nevertheless, these nonspecific LPS adsorbents usually cross-bind to other molecules and give rise to the significant loss of target molecules and the large margin of errors during the detection process. For example, CD14 has a lack of specificity to LPS and can also recognize other pathogen-associated molecular patterns. 52 There are still a lot of challenges in the development of protein-based LPS detection biosensors with optimal platform and suitable strategies.
Peptide-Based Biosensors
Besides these LPS-binding proteins from human plasma or horseshoe crabs, polymyxin B (PmB), sushi peptide, and many other designing peptides derived from the above LPS-binding proteins53–55 were involved not only in the detection but also the detoxin or neutralizing of LPS.
PmB is related to a family of polypeptides that are positively charged and can bind to the negatively charged LPS molecules, leading to the death of gram-negative bacteria by their outer membrane destabilization. Ding et al. 56 used EIS to detect the interaction of LPS and PmB molecules, which were immobilized on the 4,4-dithiodibutyric acid-coated gold electrode surface. The biosensor based on this principle was sensitive to the presence of LPS in the range of 0.2 to 0.8 ng/mL. A planar interdigital sensor based on PmB was developed to detect LPS at a high concentration level. Carboxyl-functional polymer (3-aminopropyltriethoxysilane) and thionine were chosen to cover the sensor surface and immobilize PmB. Taking EIS as a sensing signal, the planar interdigital sensors can detect LPS ranging from 0.1 to 1000 µg/mL. 57 Thompson et al. 58 published preliminary research on a real-time and label-free detection of LPS in human whole-blood plasma using ultra-high-frequency acoustic wave sensing in PmB-modified piezoelectric quartz discs. Kato et al. 59 used ferrocene-labeled PmB as a signal probe, which was captured by a poly-epsilon-lysine-modified ultraflat nanocarbon film electrode. The detection limit of LPS was achieved to 2 ng/mL. 59 Ferrocene-labeled PmB was also employed to construct an enzymatically amplified electrochemical biosensor to detect LPS as low as 50 ng/mL within 5 min. 60
Two peptide variants derived from CD14 were fluorescently labeled and applied to detect and discriminate LPS down to the submicromolar concentration range. 61 Suzuki et al. 62 reported an instance of quantitative detection of free LPS in plasma using a designated peptide Li5-025(K′YSSSISSIRAC′) as pretreatment agent. This LPS-affinity peptide was bonded to silica beads and was used to capture free LPS in plasma. The treated LPS in plasma can be measured by LAL test at a concentration of 1 pg/mL. 62 Recently, Lim et al. 63 designed a fluorescent labeled LPS-binding peptide that was self-assembled with graphene oxide in buffer solution. When selectively binding to LPS, the specific peptide released from GO, leading to fluorescence recovery and a limit of detection of 130 pM. 63
However, protein-based sensors would cross-bind to other components because the specificity of the proteins to LPS was not as high as expected. The biosensors based on designer peptides were high cost and had a complex detection process.64–67 Thus, ligands with high specificity and sensitivity to LPS are highly required.
Antibody-Based Biosensors
Antigen-antibody binding is essential for immunology and is basically able to detect all microbes and various subjects including endotoxin, virus, spore, and bacteria. 68 Because of the high specificity and excellent affinity between antigens and antibodies, detection of endotoxin with its antibody has been achieved by a chemiluminescence technique in a concentration ranging from 0.01 to 10 ng/mL. 69 Hochel et al. 70 applied polyclonal antibodies to detect Campylobacter and L. monocytogenes. Wang et al. 71 tested several kinds of polyclonal and monoclonal antibodies to detect Salmonella captured by PmB-coated polyester cloth, setting up a polymyxin-cloth enzyme immunoassay. A colony blot-radioimmunoassay based on murine monoclonal antibodies was set up and revealed to successfully detect endotoxin from purified Haemophilus ducreyi at a level of 25 pg/mL. 72 Nieradka et al. 73 developed a microcantilever array biosensor with monoclonal antibodies to detect gram-negative bacteria LPS. Luk and Lindberg 74 coupled monoclonal antibodies specific for Salmonella LPS to magnetic beads and developed a rapid immunomagnetic assay with a sensitivity of 103 to 104 bacteria/mL. A test assay containing murine monoclonal IgM antibody against lipid A of LPS was built by Romaschin et al. 75 Through the measurement of an enhanced chemiluminescent response of the neutrophils in the presence of LPS-antibody complexes, it can be used to quantify LPS with a detection sensitivity at 20 pg/mL.
Although the specific recognitions of antibodies to antigens are much better than the protein-based reactions, antibody-based biosensors still suffer from being high cost, time consuming and, labor intensive.
Aptamer-Based Biosensors
Aptamer is a promising affinity ligand that forms a specific three-dimensional structure based on its primary sequence in order to recognize and bind to its target by molecular shape complementarities; stacking of aromatic rings; electrostatic, Van der Waals interactions; or hydrogen bondings. 76 During the past two decades, aptamers have been applied to various classes of targets.77,78 Aptamers with high specificity and affinity can, in principle, be selected in vitro for any given target, ranging from small molecules to large proteins and even cells, thus making it possible to develop a wide range of aptamer-based biosensors. Aptamers, once selected, can be synthesized with high reproducibility and purity from commercial sources. 79 Also, in contrast to protein-based antibodies or enzymes, DNA aptamers are usually highly chemically stable. 80 Therefore, the aptamer-based biosensors exhibit a lot of advantages compared with biosensors that use natural receptors such as antibodies, enzymes, and peptides.81,82
Several aptamers have been isolated against LPS for different purposes. Ding et al. 83 reported that their anti-LPS aptamers, isolated from the 96-well microplate-based selection, were able to bind and neutralize the toxicity of LPS. Bruno et al. 84 harnessed LPS-coated magnetic microbeads for screening of aptamers showing affinity to LPS and proved that the selected aptamers could inhibit the growth of E. coli, making them potential aptamer-based antibiotics. Wen et al. 85 also obtained LPS-binding aptamers through 12 rounds of nitrocellulose-based SELEX and proved that mice could be efficiently protected from LPS-induced sepsis in the presence of the screened aptamers to inhibit LPS-induced toxicity.
Recently, Su et al. selected LPS-recognizing aptamers and applied one of them to develop a series of electrochemical sensors capable of sensitively and selectively detecting endotoxin from crude biological liquors.86–88 The single-stranded DNA aptamer with high affinity and excellent specificity to LPS was decorated with thiol groups at its 5′-end and was self-assembled on gold nanoparticles, which were electrochemically deposited and orderly arranged on a conducting polymer 89 -modified electrode surface. With the assistance of EIS as sensing signal, the detection limit of LPS was as low as 0.1 pg/mL. 90
Based on the sandwich structure consisting of two endotoxin-binding aptamers and LPS, a new flow cytometry–based magnetic aptasensor was developed with magnetic beads. The reproducible magnetic aptasensor was able to detect LPS rapidly (<1 min) and selectively (in the presence of bovine serum albumin, RNA, sucrose, and glucose) within a broad dynamic detection range of 10−8 to 1 mg/mL. 91
For sensitively detecting endotoxin, Bai et al. 92 developed an electrochemical aptasensor with signal amplification by three-way DNA junction-aided enzymatic recycling and graphene nanohybrid ( Fig. 3 ). There were three steps involved in the endotoxin detection process: first, a certain amount of LPS-binding aptamer association with the determinand LPS concentration was obtained and involved in the next enzymatic recycling step; in the presence of nicking endonuclease, the LPS-binding aptamers coming from the first step were repeatedly hybridized with assistant probes and capture probes located on a gold nanoparticle-modified glass carbon electrode, resulting in more and more cleaved capture probes remaining on the electrode surface. Finally, the cleaved capture probe was hybridized with a signal amplification nanocomposite fabricated from complementary DNA sequences, electroactive toluidine blue, graphene, and gold nanoparticles. Thus, a DPV electrochemical sensor with amplified signal by the enzymatic recycling and nanocomposite was obtained with the detection limit of LPS concentration down to 8.7 fg/mL and a linear range of 10 fg/mL to 50 ng/mL. 92
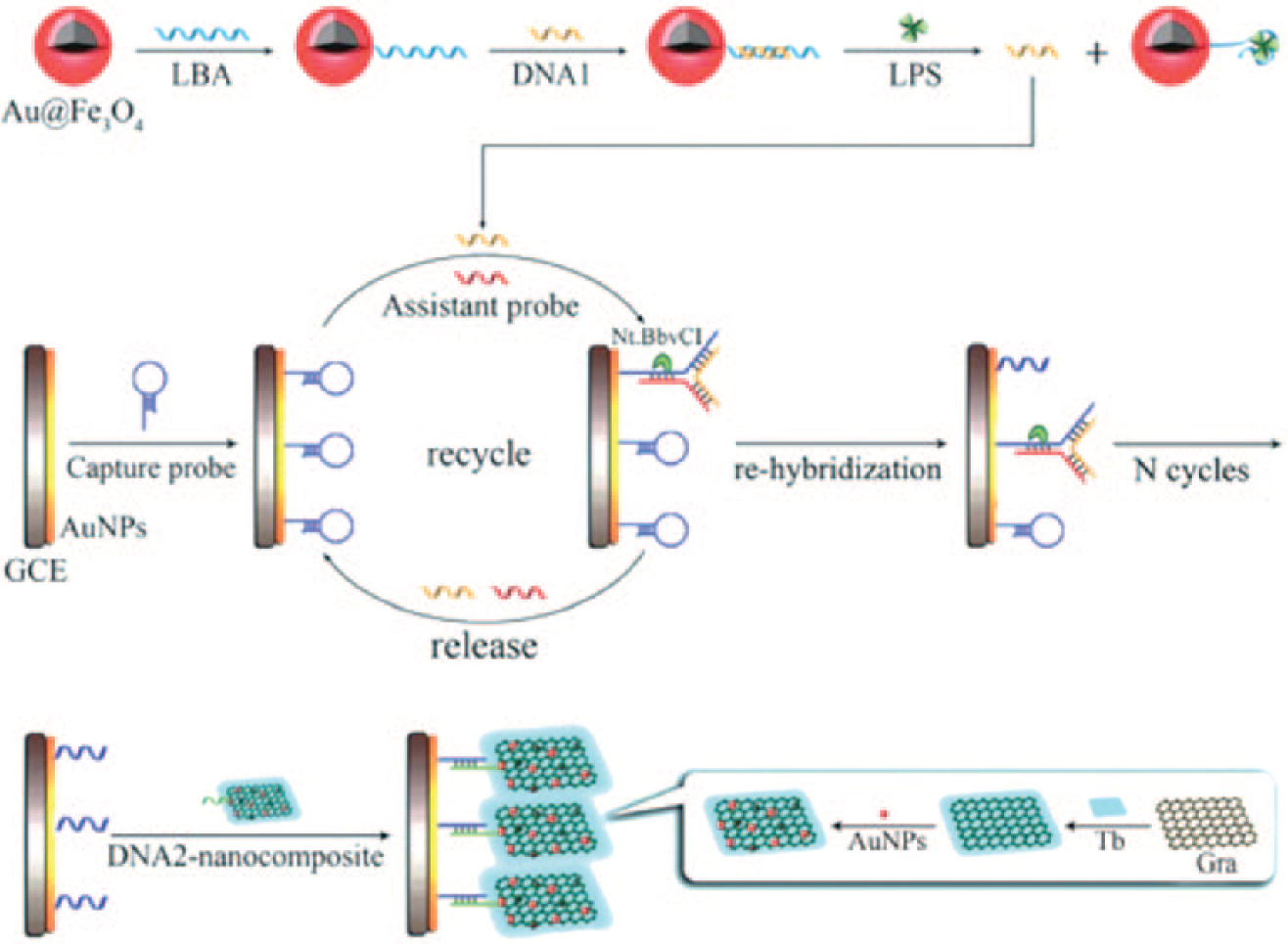
Schematic representation of the signal amplification strategy for LPS detection in reference 92. Adapted from reference 92 with permission from RSC.
Cell-Based Biosensors
Cells are the basic structural and functional units of living organisms. The bioactivities and biological functions of cells are performed with the subcellular components. As a recognition element applied to construct a cell-based biosensor, there would be a serial of complex biological reactions, which may multiply the difficulties in monitoring the sensing signal. Somehow, it is possible to amplify the sensing signal through certain cascades in cells.
Microarrays of single macrophage cell-based sensors were developed to detect LPS based on the significant and consistent shifts of the synchrotron Fourier transform infrared spectroscopy spectrum after exposure of the cell-based system to LPS. 93 Gold electrode microarrays were patterned on silica oxide substrate and coved by fibronectin to immobilize macrophage cells. After 21 h of exposure, 0.1 µg/mL LPS can be sensed by the developed cell-based sensor of microarray gold electrodes with an area of 100 µm2.
An amperometric cell-based sensor was fabricated with genetically engineered cells, which can secrete alkaline phosphatase (i.e., SEAP) in the presence of endotoxin. The electrochemical device was cultured in the medium containing p-aminophenyl phosphate and various concentrations of endotoxin for 24 h. The sensing signal produced by the oxidation of p-aminophenol, generated by SEAP-catalyzed hydrolysis, at +0.35 V was revealed to increase with the concentration of endotoxin in the range of 0.01 to 1 ng/ml. 94
When exposed to LPS, endothelial cells will rapidly induce the expression of the cell adhesion molecule E-selectin and keep its high expression for 4 h. A cell-based immunofluorescent assay was developed to detect LPS by means of evaluating the expression of E-selectin in endothelial cells exposed to various concentrations of LPS. A comparable sensitivity of the cell-based immunofluorescent assay to the LAL test was achieved after 4 h of exposure to LPS. 95 Interestingly, the release of LPS-introduced inflammatory mediators was used to value the platform for cell adherence. 96 Endothelial cells were immobilized on a fibronectin, a protein promoting the adhesion, and polystyrene deposited surface of a thiol functionalized gold electrode. Because LPS can increase vascular endothelial permeability, there was a dose-dependent decrease in EIS measurement. In another words, this cell-based EIS sensor was able to detect LPS with a detection range of 0.5 to 200 µg/mL.
Biosensors Based on Other Materials
Gold nanoparticle (Au NP) is a promising candidate as a sensing substrate because of its excellent biocompatibility and unique optical/electronic properties. Sun et al. 97 applied cysteamine-modified gold nanoparticles to detect LPS by UV-Vis spectrum, and the detection limit was decreased to 3.3 × 10−10 mol/L. 97 More facilely, a colorimetric biosensor fabricated with gold nanorods was developed and can detect LPS in the concentration range of 0.01 to 0.6 µM. Nanorods of a high aspect ratio were also demonstrated to show superiority in sensing LPS ( Fig. 4 ). 98
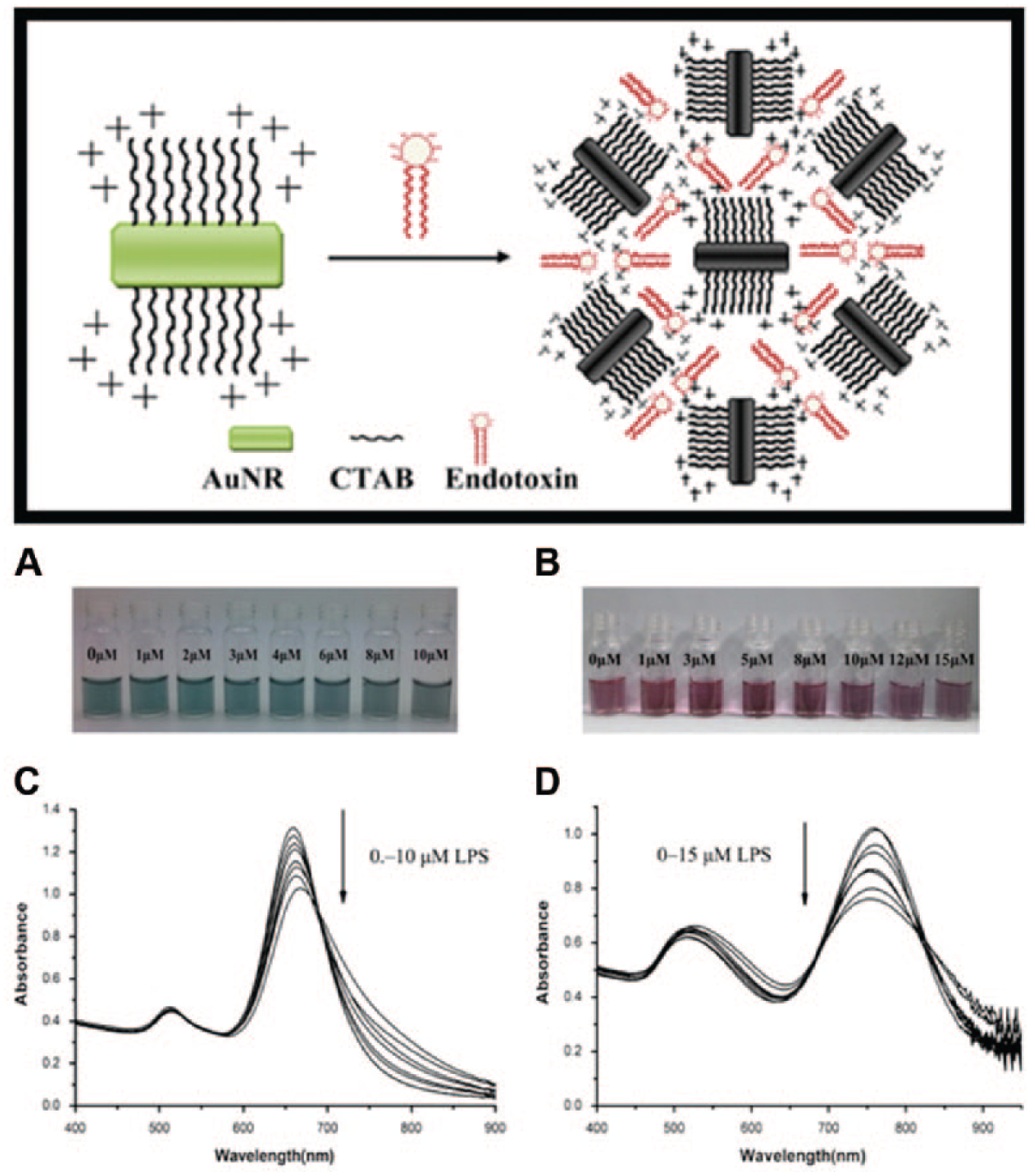
Schematic representations of the interaction between endotoxin and gold nanorods. Photos and UV-Vis spectra of green (short) and red (long) gold nanorods interacted with endotoxin at different concentrations. Reproduced from reference 98 with the permission of Elsevier.
The first synthetic sensor for LPS was also based on a colorimetric change to discriminate LPS from different species with a lipid-functionalized polydiacetylene liposome. 99 Using 3-phenylthiophene–based water-soluble copolymer as fluorescent substrate, LPS of 30 nM at most can be detected. 100 Pyrene derivatives were designed to fabricate biosensors and applied as a fluorescent probe for sensing bacterial endotoxin with a limit of detection estimated in the range of 10 to 100 ng/mL.101,102 By polyacrylamide gel electrophoresis, Eosin B can be used to detect LPS from 20 to 40 ng/mL. 103 With an assistance of the copper ion and nitrilotriacetic acid complex, which can specifically bind to the O-specific polysaccharide chain of E. coli 055:B5 (L4524), an EIS biosensor was performed in the LPS concentration range of 0.1 to 100 pg/mL and easily regenerated by EDTA solution, exhibiting a great potential in real-time detection of endotoxin. 104
Finally, an optical assay for LPS from E. coli (0111:B4) was developed based on the combination of molecular imprinted polymers (MIPs) and surface plasmon resonance (SPR) sensor. 105 MIPs are artificial receptors potentially tailored to any target with high affinity and selectivity, mimicking binding characteristics of antibodies with additional benefits of good stability, reasonable cost, easy preparation, and modification. As the recognition component of the developed optical sensor, the LPS-templated nanoMIPs with dissociation constants of 3.24 to 5.24 × 10−8 M were covalently immobilized on an 11-mercaptoundecanoic acid-modified SPR gold chip, leading to an LPS detection range of 15.6 to 500 ng/mL ( Fig. 5 ).
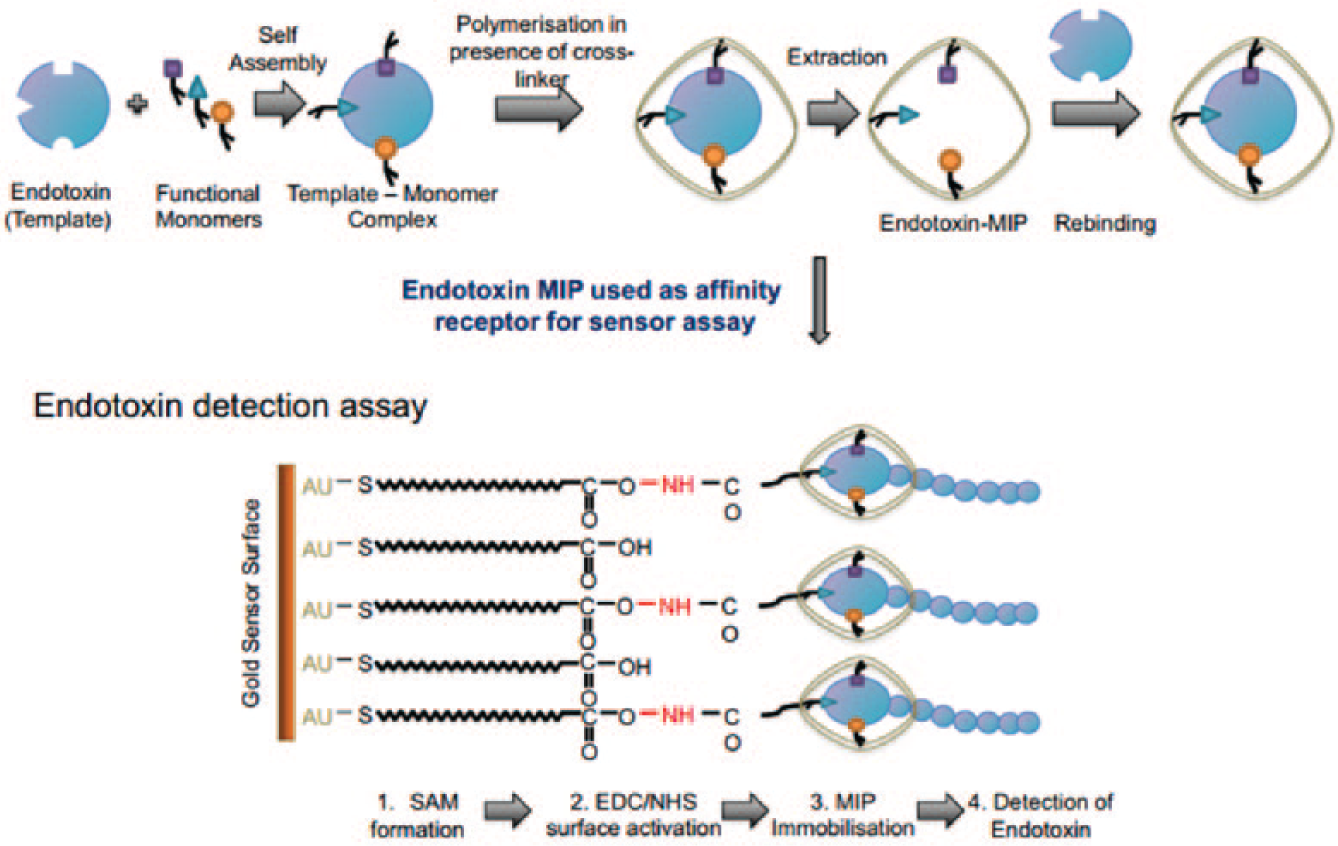
Whole process from endotoxin–molecular imprinted polymer production to endotoxin detection assay. Adapted from reference 105 with permission from Elsevier.
Critical standards of endotoxin detection have been established in many countries to regulate endotoxin limits in the fields of drugs, medical devices, and biological products. Research focused on endotoxin binding, determination, and removal has attracted the interest and infinite efforts of scientists. Many new materials and emerging techniques51,58,106 have been applied to improve the traditional LAL test method, such as SPR, piezoelectric, and so forth. Meanwhile, a novel detection mechanism beyond LAL enzymatic reactions has been explored so as to set up advanced endotoxin detection systems. Among these detection techniques, biosensors based on endotoxin affinity components assuredly bespeak the future trend of endotoxin detection techniques. In particular, the tremendous progress in microfabrication and nanomaterials must create a flourish of miniaturized and integrated biochip processing, contributing to the point-of-care test in a personalized endotoxin assay.107,108 Despite current achievements, there are numerous difficulties ahead, not only in the search for new LPS recognition elements but also in the design of unique detection systems and strategies, to realize the rapid, accurate, and reliable detection of endotoxin with a commercial biochip device. Elimination of these barriers relies on the comprehensive development of chemistry, biology, materials, electronics, and machine science.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (81301293) and National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (2014ZX09507008).
