Abstract
Angiogenesis, the formation of new blood vessels in the vasculature, is a major research topic in biology with implications in development, cancer, tissue engineering, and regenerative medicine. Although much knowledge has been acquired over many decades through application of various angiogenesis assays, these methods have various drawbacks that limit their overall utility. Given the importance of angiogenesis in our understanding of numerous biological processes and its potential as a therapeutic target in cancer and other diseases, there is need to develop useful tools with improved physiological relevance, accessibility, robustness, and throughput over existing assays. Recent developments in microfluidics have demonstrated enormous potential of microscale cell culture systems for biology studies, especially angiogenesis. This area is advancing rapidly, and it is important to remain up to date with the state of the art in technology and evaluate its current and future impact on angiogenesis research. This review examines the latest advances in microfluidic angiogenesis systems. Design and methodology of microscale systems are discussed, and biological insights obtained from the systems are examined. Importantly, physiological relevance, accessibility, and data output of microfluidic angiogenesis systems are compared with traditional angiogenesis assays, and next challenges facing researchers are presented with consideration of the potential integration of automated systems.
Keywords
Introduction
Microfluidic systems have become an increasingly useful technology for creating in vitro cellular and tissue microenvironments for applications in biology research. Using microengineering methods that have gained popularity over the past decade, complex microchannel geometries and networks can be fabricated, various cell types and matrix components can be cultured and organized at defined locations, and various chemical and physical stimuli can be applied to recapitulate important aspects of the in vivo microenvironment with precise spatiotemporal control. Steady progress toward the fundamental understanding of cell culture at the microscale has led to increased complexity and functionality within microfluidic cell culture systems,1–3 with important developments in the application of these systems in various biology research fields, including endothelial and cardiovascular biology,4,5 neuroscience, 6 stem cells,7,8 immunology, 9 and cancer research.10–12 Most recently, we have also seen the emergence of “organ-on-chip” microdevices, which can incorporate chemical and physical stimuli on in vitro tissue interfaces to demonstrate organ-level functions.13,14 These advances demonstrate that the level of interest to integrate microfluidics with cell biology continues to grow rapidly within the life sciences research community.
One research area of major development in the past 5 years is the application of microfluidic cell culture systems for angiogenesis and capillary morphogenesis. Because of the spatial and temporal scales of the angiogenesis process, microfluidic cell culture systems are particularly well suited for studying the dynamic events of angiogenesis. The purpose of this review is to summarize the latest developments in the application of microfluidic cell culture systems for the study of angiogenesis and to evaluate these new tools by comparing them directly with existing angiogenesis assays. The review focuses on the key designs and developments in the past 5 years and highlights the relevant features and important enabling qualities of microfluidic angiogenesis systems not available in traditional assays. Importantly, an evaluation will be presented on the usefulness of microfluidics for angiogenesis research, with a discussion on the issues of physiological relevance, accessibility, and data output, including the potential for laboratory automation and high-throughput applications.
Angiogenesis: A Brief Overview
Angiogenesis, or the formation and extension of new blood vessels from existing vasculature, derives from the need to supply oxygen and nutrients to avascular and ischemic regions within the body and is therefore essential to both normal development and many pathological processes. The basic sequence of events that comprise angiogenesis has been known to involve dissociation of the basement membrane, as well as the nearby extracellular matrix (ECM), that supports the endothelium; migration into the matrix by leading endothelial “tip” cells toward a stimulus; proliferation of trailing endothelial “stalk” cells connecting the tip cell to the original vessel; lumen or tubule formation of the endothelial cells into sprouts; and subsequent branching (intussusception) or joining (anastomosis) of angiogenic sprouts into complex microvascular networks.15,16 The coordinated events of this angiogenic cascade play critical roles during normal physiological processes such as development, reproduction, and wound healing, as well as during the development of diseases such as diabetic retinopathy, 17 asthma, 18 cardiovascular diseases, 19 and most notably cancer. 20 Indeed, the modern field of angiogenesis research is often traced to the classic study by Folkman, 21 in which he proposed the dependence of tumor growth on neovascularization. In the four decades since that hypothesis, the field has witnessed significant progress and growth, culminating in key milestones such as the approval of the first ever antiangiogenic drug (bevacizumab) by the U.S. Food and Drug Administration (FDA) and declaration of antiangiogenic therapy as a major modality of cancer treatment. 22 While numerous preclinical studies and clinical trials are under way with hopes of discovering novel therapeutic agents for the advancement of antiangiogenesis therapy, there also remain many challenges for antiangiogenesis research, including the need to overcome low patient response rates, address cases of acquired drug resistance, and ultimately improve clinical survival outcomes. 23 It is clear that much work remains in both basic biology and clinical research to fully elucidate the mechanisms of angiogenesis, facilitate the discovery of new and efficacious antiangiogenesis drugs, and enable the effective application of antiangiogenesis therapies for better patient outcomes.
An important factor in the advancement of angiogenesis research over the past 40 years has been the development of experimental tools and assays that have enabled the experimentation, visualization, and quantification of the complex processes within angiogenesis. Existing angiogenesis assays span a wide range of methods, from in vitro assays conducted in microtiter plates (e.g., cell proliferation, cell migration, and tubule formation assays), to in vivo methods in small animals (e.g., mouse corneal micropocket assays, rabbit ear chamber assays), to ex vivo methods that rely on excision and culture of intact tissue samples. A comprehensive review of these traditional assays is available for interested readers. 24 Most of these assays are established methods in the laboratory that have provided numerous contributions to the field and continue to provide important benchmarks for the latest research discoveries. However, despite providing much of our current understanding of angiogenesis, many of these existing assays are known to be laborious, technically challenging, lacking in physiological relevance, or a combination of these shortcomings. Furthermore, many are also not amenable to automation, high spatial and temporal resolution, or high-throughput applications, thereby limiting the amount of data that can be obtained. To tackle challenging contemporary questions related to angiogenesis, new and innovative methods for studying this process need to be developed to overcome these limitations.
Studying Angiogenesis via Microfluidic Devices
Recent developments in microfluidic systems have started to fill an important need and bridge a previously large gap between traditional in vitro and in vivo angiogenesis assays. While traditional angiogenesis assays continue to serve our current (and future) needs, ongoing development of novel experimental methods is also vital because it creates new research opportunities and enables new classes of experiments that previously may have been inconceivable. The studies highlighted here were published within the past 5 years (earliest in 2008), demonstrating the enormous interest and growth in this field. Table 1 summarizes the key features of these microsystems. While there are unique aspects in each of the designs and methods, there are also similarities in general methodology—namely, that the overall procedure in all the studies consists of (1) the use of microfabrication via soft lithography and other techniques; (2) assembly of molds and substrates made of different materials; (3) injection of matrix components, proteins, or gels; (4) seeding of live cells; and (5) the application of chemical or physical stimuli, or both, in a spatially and temporally controlled manner.
Microfluidic Angiogenesis Systems.
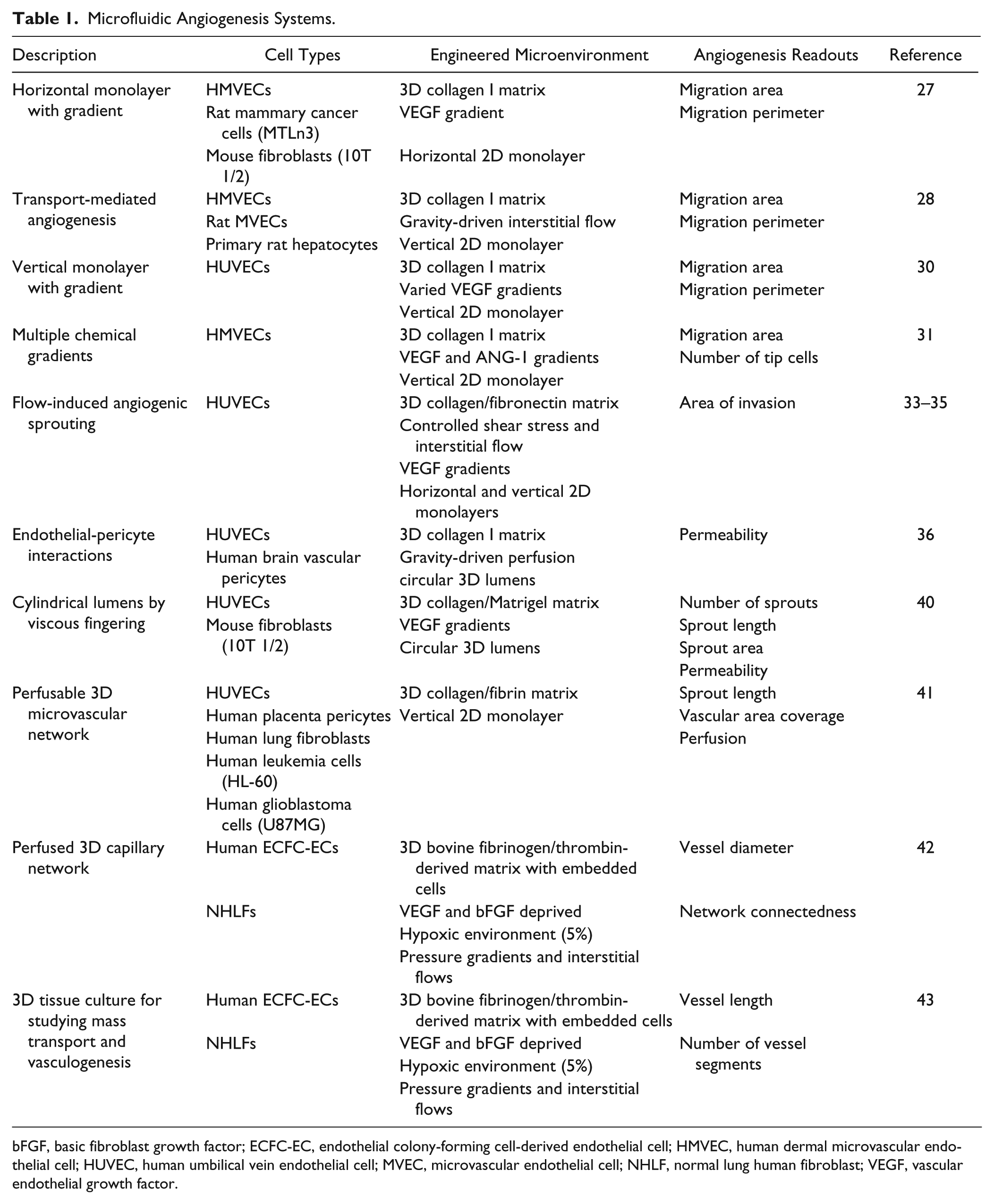
bFGF, basic fibroblast growth factor; ECFC-EC, endothelial colony-forming cell-derived endothelial cell; HMVEC, human dermal microvascular endothelial cell; HUVEC, human umbilical vein endothelial cell; MVEC, microvascular endothelial cell; NHLF, normal lung human fibroblast; VEGF, vascular endothelial growth factor.
The first body of work of note is from the group led by Kamm, who developed a microfluidic system for studying the dynamics of capillary morphogenesis and sprouting angiogenesis ( Fig. 1A ). 25 He first demonstrated the ability to culture endothelial cell (EC) monolayers on the vertical surface of a hydrogel and successfully induced formation of a capillary-like sprout. 26 The vertical orientation of the EC monolayer allowed the sprout to grow into the gel perpendicular to the original monolayer and be visualized in its full length in the horizontal imaging plane, circumventing a common problem with monolayer orientation in many in vitro assays. Kamm and coworkers subsequently showed the versatility of their system by introducing co-cultures with other cell types,27,28 effects of interstitial flow driven by gravity, 28 use of alginate beads to create capillary networks, 29 and, in related work conducted by the group led by Chung, the ability to control the spatial patterns and temporal dynamic of multiple chemical gradients.30,31 Besides improvements in visualization and control, these advancements also enabled new and interesting insights, such as the observation that tip cells disconnected from stalk cells in the presence of a vascular endothelial growth factor (VEGF) gradient, but connections between tip cells and stalk cells were recovered in the presence of cooperative VEGF and ANG-1 gradients. This represents a prime example of how microfluidic systems can offer flexibility in experimental design, allowing gradients and other stimuli to be added or removed as desired to elucidate cooperative or competitive effects. While this observation that tip and stalk cells may separate in the absence of ANG-1 gradients is novel in that it has not been previously reported, more research is necessary to confirm that this phenomenon can be observed in other systems, both in vitro and in vivo. Nevertheless, the utility of microfluidic systems for furthering our understanding of angiogenesis is clearly evident from this body of work.
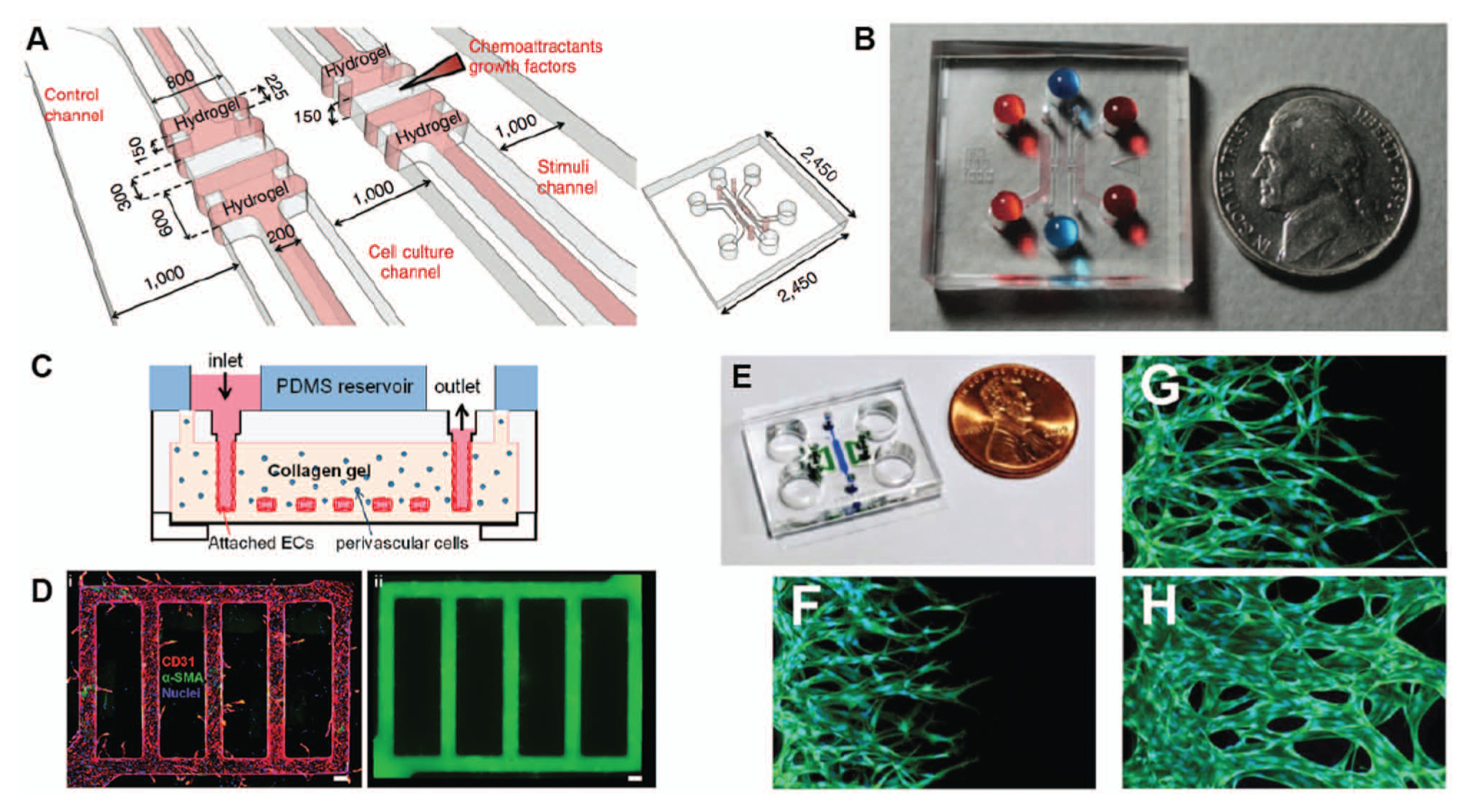
Recently developed microfluidic angiogenesis systems. (
Endothelial cells that line blood vessels are regulated by mechanical signals from the shear of flowing blood. 32 While numerous macroscale and microscale systems have been developed to study vascular ECs under shear flow,4,5 these systems have not been able to study in detail the effects of fluid flow on angiogenic sprouting. Recently, Song and Munn 33 developed a microfluidic system that applied physiological shear flow, in conjunction with VEGF gradients and interstitial flow, on human umbilical vein endothelial cells (HUVECs) cultured in a microfluidic design consisting of three parallel microchannels, where the center microchannel was filled with collagen I gel, and the two side microchannels were cultured with HUVEC monolayers. Syringe pumps were connected to the system to generate the desired shear and interstitial flow rates. These experiments demonstrated that fluid forces attenuated angiogenic sprouting in the presence of VEGF and showed that interstitial flow dictated the morphology of angiogenic sprouts in a manner dependent on the direction of the VEGF gradient. The system was recently applied to further examine dynamics of anastomosis in the presence of shear 34 and the role of RhoA in mediating flow-induced sprouting, 35 resulting in findings on flow-mediated angiogenesis that would not have been possible with existing assays.
While these two microsystems employed similar microchannel configurations that proved effective in both cases, the rectangular cross sections of the microchannels resulted in HUVEC monolayers with cross sections that did not resemble the cylindrical lumens in vivo. Zheng and coworkers 36 developed a novel injection molding technique to fabricate a type I collagen gel consisting of a network of microvessels that could be lined with HUVECs. While the microvessels initially had square cross sections, after 3 days in culture, the microvessel network transformed into elliptical cross sections that more closely resembled in vivo lumens ( Fig. 1B ). One enabling quality that was demonstrated with this particular design was the use of the collagen gel mold to encase perivascular cells in the matrix surrounding the entire microvessel network. While co-culture arrangements were also shown in other microfluidic systems, this particular arrangement was an advance in terms of mimicking the three-dimensionality of the in vivo microenvironment. Importantly, although pericytes were seeded into the bulk of the gel, pericytes were eventually found adjacent to the endothelium after culture, with a layer of basal lamina separating the cell types. This suggested that the combination of microenvironmental factors that were reassembled in the system enabled remodeling of the appropriate vessel architecture, including the supporting ECM.
A potential limitation of the technique by Zheng et al. 36 is the rather elaborate fabrication procedure and its limited scalability for increased throughput. Others have also reported methods of creating single, straight, cylindrical 3D lumen structures to mimic vessels,37,38 but these require interconnects and tubing for interfacing the device with accessory equipment. To circumvent the need for interconnects and tubing, and thereby simplify the process of creating lumens and lumen networks with cylindrical geometries in microfluidic systems, Bischel and coworkers 39 demonstrated a simple viscous fingering method that relied only on a micropipette. The method was recently advanced to include the culture of confluent HUVEC monolayers that lined the inner surface of the 3D lumens, including lumen networks. 40 The system demonstrated a combination of various features from the other systems, including the simple use of a micropipette for fluidic operations, the ability to set up VEGF gradients and induce angiogenic sprouting, and the flexibility to include stromal cells in the surrounding matrix to facilitate the study of heterotypic cell-cell interactions. While this system offers accessibility and user-friendliness, it has yet to demonstrate integration of shear and interstitial flows, which would likely require modifications on how interfacing is achieved on the platform.
Finally, the most recent development involves multiple microfluidic chips that can support the culture of 3D microvascular networks consisting of patent perfusable vessels.41–43 To demonstrate that perfusion was achieved, fluorescent microbeads were introduced into the microvascular network of these platforms and were found to either localize within the lumen structures 41 or travel from the arterial to the venular side of the microdevice. 42 Other cell types, including pericytes, cancer cells, and leukocytes, were inoculated and cultured in one case to show the robustness of the system for enabling the study of heterotypic endothelial cell–stromal cell interactions. 41 Furthermore, morphological and biochemical characterization of the microvessel network were carefully performed to demonstrate proper development of cell-cell junctions consisting of VE-cadherin, β-catenin, and ZO-1 tight junction proteins. The application of such platforms for both angiogenesis and vasculogenesis studies in the future may lead to interesting insights on the functional characteristics of perfusion and vessel permeability.
Evaluating Microfluidic Angiogenesis Assays
Microfluidic cell culture systems have offered new ways of testing and measuring the dynamic process of angiogenesis and have yielded novel insights that would be challenging to obtain with existing methods. In particular, two major advantages of microfluidic angiogenesis systems that deserve special mention are (1) their ability to tailor the structural and biological aspects of the microenvironment to improve physiological relevance and (2) their ability to fine-tune the spatiotemporal control of chemical and physical stimuli in the microenvironment to exert increased regulation over experimental conditions. First, microfabrication techniques, from basic to advanced methods, enable design and fabrication of microchannel networks with precise dimensions, complex geometries, and distinct compartments that allow excellent spatial organization of different materials and biological components. Complex cellular interactions and more realistic 3D structures can be created within these microsystems to better mimic the in vivo microenvironment. This is clearly important given the essential roles of cell-cell communication and dimensionality in the regulation of cell morphology, behavior, and function. Second, because of how microfluidic systems are designed and fabricated, they are inherently flexible in allowing different combinations of chemical and physical cues to act on the engineered in vitro microenvironments. The researcher has precise control over experimental parameters such as magnitude, direction, duration, and the inclusion or exclusion of various stimuli, including chemical gradients, drug cocktails, shear flow, and interstitial flow. This ability to turn stimuli on or off on demand is simply not possible for in vivo angiogenesis assays in animal models, which by their very nature must provide all the various intrinsic factors at physiologic levels (unless the animals have been genetically engineered to do otherwise). While some existing in vitro systems have been designed to offer additional flexibility (e.g., parallel plate flow chambers add shear flow, Transwell inserts enable chemotactic gradients), these systems typically present other shortcomings, such as lack of proper dimensionality or inappropriate diffusion time scales, which lower their biological relevance and reduce the fidelity of the results obtained. Thus, microfluidic cell culture systems have clearly provided an alternative approach to studying angiogenesis with many advantages that can be exploited to tackle previously inconceivable biological questions. But how do microfluidic cell culture systems measure up against the other popular angiogenesis assays that have been established over the past 40 years?
A rather constructive method I have found for assessing the utility of microfluidic systems for angiogenesis research is to graphically represent this comparison between microfluidic and conventional angiogenesis assays, as depicted in Figure 2 , across three major evaluation metrics: physiological relevance, accessibility, and data output. The remaining discussion revolves around the key features and trends illustrated by this graphical representation pertaining to these three metrics.
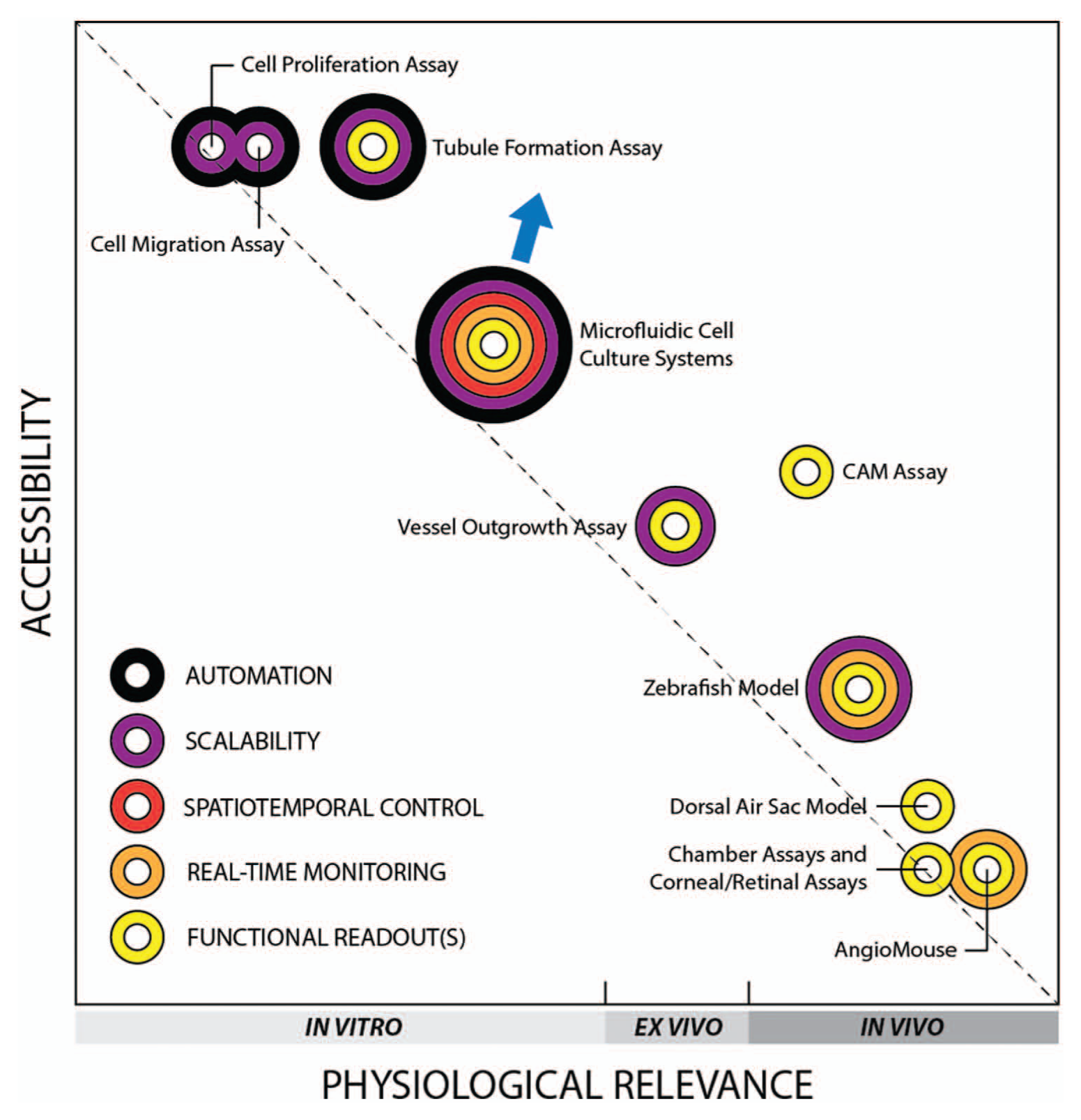
Angiogenesis assays. Graphical representation compares assays in terms of physiological relevance, accessibility, and data output and illustrates the inherent compromise between physiological relevance and accessibility across the spectrum. Concentric rings were assigned to the assays to illustrate the range of data that can be extracted (described in text). CAM, chorioallantoic membrane. (Note: the author selected weighted criteria for physiological relevance and accessibility and scored each assay to obtain overall scores that helped position each assay in the graph.)
Physiological Relevance
Physiological relevance in this discussion not only refers to the basic in vitro, ex vivo, and in vivo classifications of an assay but also encompasses aspects of dimensionality, composition and morphology of the surrounding structure, presence of neighboring cell types, and the model species used. The most frequently cited limitation of existing in vitro assays, despite their recognized utility thus far in angiogenesis research, is their overall poor physiological context due to the use of non-microvascular endothelial cells (bovine aortic endothelial cells and HUVECs are the two most commonly used types); the isolation of these ECs from their natural, complex tissue environments; and their subsequent culture on stiff plastic 2D substrates, which is significantly different from the stiffness and three-dimensionality of native tissues. 44 Migration assays that employ Transwell inserts to enable co-cultures and tubule formation assays performed in 3D gels are two examples of improvements in physiological context, but these modifications are relatively incremental. Ex vivo vessel outgrowth assays, such as the aortic ring and aortic arch assays, 45 were considered major improvements over in vitro assays with respect to physiological relevance because native tissue structures, matrix components, and cellular arrangements were preserved. Because of the challenges associated with excising and handling small vessels, however, these assays have conventionally been performed only with large vessels such as aortas, venae cavae, and saphenous veins, which lowers their physiological relevance. The majority of fundamental studies in angiogenesis research continues to be conducted with classic in vivo assays, such as the chorioallantoic membrane (CAM) assay, various chamber assays (e.g., rabbit ear chamber, dorsal window, and cranial window), and assays involving the eye (e.g., corneal micropocket assay, retinal angiogenesis assay), largely because these model systems remain the most faithful representations of the complex and dynamic angiogenic processes that occur within a living organism. Besides the need for laborious surgical procedures, the only other drawbacks are (1) the species used, ranging from mammals such as mice, rats, and rabbits, to nonmammalian chick embryos and zebrafish, and (2) the highly varied locations of the blood vessels used for experimentation in relation to the true location of the blood vessels with respect to the problem of interest. In this latter case, the development of the AngioMouse is an additional step toward improved relevance for tumor studies because of the orthotopic location of the tumor vessels visualized (indirectly) by green fluorescent protein in the mouse. 46 Thus, in terms of physiological relevance, given this backdrop of conventional assays, in vitro microfluidic systems have made a significant contribution by pushing the in vitro envelope and filling a significant gap along the spectrum of physiological relevance ( Fig. 2 ).
Accessibility
A number of important factors contribute to the level of adoption of an assay within the research community. These factors include cost, ease of preparation and experimentation, need for special training, and animal care requirements, which can be collectively referred to as the overall accessibility of an assay. All these factors add more time, money, and resources to research projects that are typically already under strict financial constraints. Nevertheless, the classic in vivo assays that require complex surgical procedures by well-trained technicians remain popular despite their low accessibility, likely because of their high physiological relevance and their reputation as standard and traditional assays that were employed in the early years of the field. The importance of these factors sometimes takes precedence over accessibility, particularly when the resources are available or have otherwise been established. At the other end of the spectrum, the high accessibility of in vitro proliferation and migration assays enables many researchers, even from other disciplines, to engage in angiogenesis research without the need to perform technically challenging in vivo assays. This applies to medicinal chemists and pharmacologists involved in drug discovery and development, where in vitro assays are attractive because of their amenability to high-throughput screening applications and automated laboratory processes. In this regard, microfluidic systems have a significant advantage over in vivo assays because of their potential for parallelization and multiplexing. The microfluidic systems described clearly require some engineering setup and are thus not as simple as the microtiter plates used for proliferation, migration, and tubule formation assays, but microfluidic systems certainly fill a major gap between the current in vitro and in vivo options.
Data Output
The third evaluation metric is concerned with data output, which encompasses the quality and quantity of end-point measurements and the manner and efficiency in which these measurements are acquired. Quantifiable functional data are extremely valuable to our ability to advance our understanding of complex multicellular systems and processes, and our ability to optimize the extraction of this information will therefore add significantly to the value of the assay. This value increases further when the increase in information output can be obtained without a proportional increase in time, effort, or resources. Figure 2 illustrates the value and potential impact of data output in the form of five concentric rings that represent specific aspects of this metric. The use of concentric rings was derived from a need to display how each additional ring amplifies the potential quantitative power of the assay.
Functional readouts refer here to measures of sprout length, sprout area, number of sprouts, and other metrics that are more directly related to angiogenic behavior than a nonspecific readout such as cell proliferation, which may be associated with processes other than angiogenesis. Real-time monitoring, made possible by the continuing advances in time-lapse microscopy, offers high temporal resolution and visualization of dynamic events, which have potential to offer deeper insights that may be overlooked if only occasional time points are recorded. While some in vivo assays may be adapted to achieve this level of temporal resolution (e.g., imaging of AngioMouse), some cannot (e.g., chamber assays), so to be fair, assays were credited with real-time monitoring capabilities only if this feature was built into the platform. Spatiotemporal control has been thoroughly demonstrated by microfluidic angiogenesis systems, which offer control over vessel size, shape, and network complexity; number and unique types of stromal and perivascular cells to be included; inclusion of mechanical forces via shear and interstitial flow; and control of biochemical gradients. This level of control of microenvironmental factors is simply not available in any other category (only microfluidic systems in Fig. 2 ). For our purposes, an assay is considered truly scalable when there are economic benefits associated with parallelization of the assay. Clearly, in vitro assays conducted in microtiter well plates belong to this category, but it may also be argued that aortic ring assays and zebrafish embryos deserve mention because each excised blood vessel can result in up to 30 independent ring slices, and a weekly culture of zebrafish usually means hundreds of embryos. 24 While in vivo assays are obviously not amenable to automation, the potential for in vitro assays to be automated (e.g., microtiter well plates interfaced to robotic liquid handlers) allows a significant increase in data output while reducing manual labor and minimizing human errors, thereby adding direct and indirect economic benefits to the assay. The potential for automation continues to be a major benefit of microfluidics technology, and although results thus far have not met the high expectations, some examples of automatable microfluidic systems have demonstrated commercial viability (e.g., Fluidigm, Bellbrook Labs). Regarding the current discussion, the passive pumping-based microsystem by Bischel et al. 40 offers an immediate example of a microfluidic angiogenesis assay that has potential for integration with automated systems. Automation may be challenging for some of the other microfluidic systems that were designed for other specific applications, but given how other engineering challenges in microfluidics have been overcome before, 47 it certainly remains conceivable.
Comparison of Assays
The graphical representation of these metrics was created to illuminate three notable features within the broad spectrum of angiogenesis assays. First, the trade-off between physiological relevance and accessibility becomes strikingly clear in this illustration because all the assays lie along a diagonal that stretches from in vitro assays that are highly accessible but biologically inaccurate to classic in vivo assays that have high physiological relevance but involve animals and challenging technical procedures. An assay that lies above this “trade-off diagonal” possesses certain features that, when compared with others in their class, offers some advantage in one metric without sacrificing its standards in the other metric. This applies in particular to the tubule formation assay as well as the widely adopted CAM assay, both of which have remained popular choices within their respective classes. Note that the recently developed zebrafish model also possesses a unique combination of accessibility and physiological relevance that has helped its popularity. Importantly, the described microfluidic systems offer significant achievements in physiological relevance compared with other in vitro assays, with the only cost being the need for fabrication and operation of engineered microfluidic devices. Because microfabrication techniques are becoming increasingly economical and accessible,48,49 microfluidic systems will likely continue to evolve and improve, providing further advancements in both assay accessibility and physiological relevance (arrow, Fig. 2 ) that will be difficult to replicate by the other more established angiogenesis assays.
Second, microfluidic systems have filled a noticeable gap that previously separated in vitro assays from in vivo assays. These conventional assays cluster near the ends of the spectrum, with the ex vivo outgrowth assays providing the most balanced option by offering moderate levels of both accessibility and physiological relevance. With the development of microfluidic systems, however, the range of available in vitro assays has broadened, and the standards of physiological context for in vitro assays have been elevated significantly. Similar to the outgrowth assays, microfluidic systems appear to have created their own niche within the overall spectrum of angiogenesis assays.
Third, microfluidic systems offer a full set of data output elements that is unmatched in breadth by any of the other traditional angiogenesis assays. Traditional in vitro assays only provide basic readouts with limited functional information, and often across only limited time points, while in vivo assays using animals cannot be automated or reasonably scaled. In these respects, microfluidic systems have a distinct advantage because they offer a unique combination of functionality, real-time monitoring, scalability, and potential integration with automated systems. Furthermore, the microfluidic systems described here demonstrated a level of spatiotemporal control that enables novel insights into the coordinated and competitive effects of various physical and chemical stimuli, which cannot currently be acquired through any other angiogenesis assay. The potential impact of this powerful combination of data output elements is reflected in the illustration through the overall coverage of the concentric rings for microfluidic systems.
Conclusions and Outlook
In summary, microfluidic angiogenesis systems offer an exceptional combination of physiological relevance, accessibility, and data output that positions them uniquely on the spectrum of experimental angiogenesis assays. Essentially, any combination of stromal cell types, chemical gradients, and shear flow–induced physical forces may be included or excluded in a given microenvironment, depending on the application. Because of these features, microfluidic angiogenesis systems enable us to address new and unconventional questions in biology and to design biological experiments in a manner that may be significantly different from when only conventional assays were available.
While there is little doubt that microfluidic cell culture systems will continue to evolve and contribute to angiogenesis research, as well as to experimental biology in general, it is imperative to recognize the need for continued research and development in the area of microfluidics technology. Many aspects of the microsystems described here can be improved to further increase physiological relevance, accessibility, and data output. For example, all the microfluidic angiogenesis assays presented used HUVECs, which is understandable given their wide availability, but this warrants the same criticism given to the other in vitro assays that only employ cell lines. Moving forward, researchers need to also consider how to isolate, incorporate, and maintain orthotopic endothelial cells (and other cell types) into their microfluidic systems to create microenvironments with improved physiologic context. Indeed, future studies will also likely involve the use of primary human cells in microsystems, if the promise of assays for personalized tests is to be realized. In addition, while some microfluidics researchers are beginning to design accessibility directly into the platform, more research is needed to further simplify integration and interfacing for many of the other microfluidic systems. Biologists will be much more likely to adopt microfluidic systems if the methods are user-friendly, are economical, and do not require extensive technical training and expertise. It is hoped that published protocols that involve the use of accessible microfluidic systems to study angiogenesis will help to accelerate adoption. 25 Finally, for microfluidic systems to realize their utility for high-throughput applications such as drug screening, they should be tested with throughput similar to other in vitro assays, and the results should be validated and compared with those from the other in vitro assays. The platform by Bischel, for example, is amenable to being arrayed and automated, and thus can be immediately tested for “hits” to identify candidate compounds that may have been overlooked by proliferation, migration, and tubule formation readouts alone. Furthermore, such validation experiments may also reveal compounds that were previously considered positive hits but in fact would have been identified as negatives if more functional and physiologically relevant assays had been available.
Microfluidic cell culture systems have emerged on the scene of angiogenesis research over the past 5 years, and interest appears to be rapidly growing. More research is clearly necessary to further advance these microsystems to achieve improved physiological relevance, accessibility, and data output. Nevertheless, based on the comparisons presented here between traditional assays and microfluidic systems along the spectrum of experimental methods, it is already evident that microfluidic systems have begun to bridge a significant gap in our experimental repertoire and will likely contribute significantly to various areas within angiogenesis research, ranging from basic science to drug discovery applications.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
