Abstract
The metastatic dissemination and spread of malignant circulating tumor cells (CTCs) accounts for more than 90% of cancer-related deaths. CTCs detach from a primary tumor, travel through the circulatory system, and then invade and proliferate in distant organs. The detection of CTCs from blood has been established for prognostic monitoring and is predictive of patient outcome. Analysis of CTCs could enable the means for early detection and screening in cancer, as well as provide diagnostic access to tumor tissues in a minimally invasive way. The fundamental challenge with analyzing CTCs is the fact that they occur at extremely low concentrations in blood, on the order of one out of a billion cells. Various technologies have been proposed to isolate CTCs for enrichment. Here we focus on antigen-independent approaches that are not limited by specific capture antibodies. Intrinsic physical properties of CTCs, including cell size, deformability, and electrical properties, are reviewed, and technologies developed to exploit them for enrichment from blood are summarized. Physical enrichment technologies are of particular interest as they have the potential to increase yield and enable the analysis of rare CTC phenotypes that may not be otherwise obtained.
Introduction: Clinical Needs and Biology of Circulating Tumor Cells
Cancer metastasis involves the spread of cancer cells from an initial site to form distant secondary tumors and is the main cause of death in cancer patients. 1 It is thought that primary tumor cells undergo the process of metastasis in the following schematic steps: (1) localized invasion, whereby the tumor cells detach from the primary tumor and breach the basement membrane (which makes the tumor malignant); (2) intravasation into blood or lymphatic circulation systems, which allows for transport via circulation and interactions with blood components; (3) arrest in microvessels of various organs; (4) extravasation and migration into the distant tissue followed by colonization to form micrometastases; and (5) stimulation of angiogenesis, leading to growth into macrometastases (metastatic tumors) ( Fig. 1 ). 2 However, this process is highly inefficient, and less than 0.01% of circulating tumor cells (CTCs) will seed metastases.3,4 The fact that CTCs occur at extremely low concentrations and are obscured by billions of cells in peripheral blood has hindered the understanding of their mechanism of action, as well as their clinical importance. 5
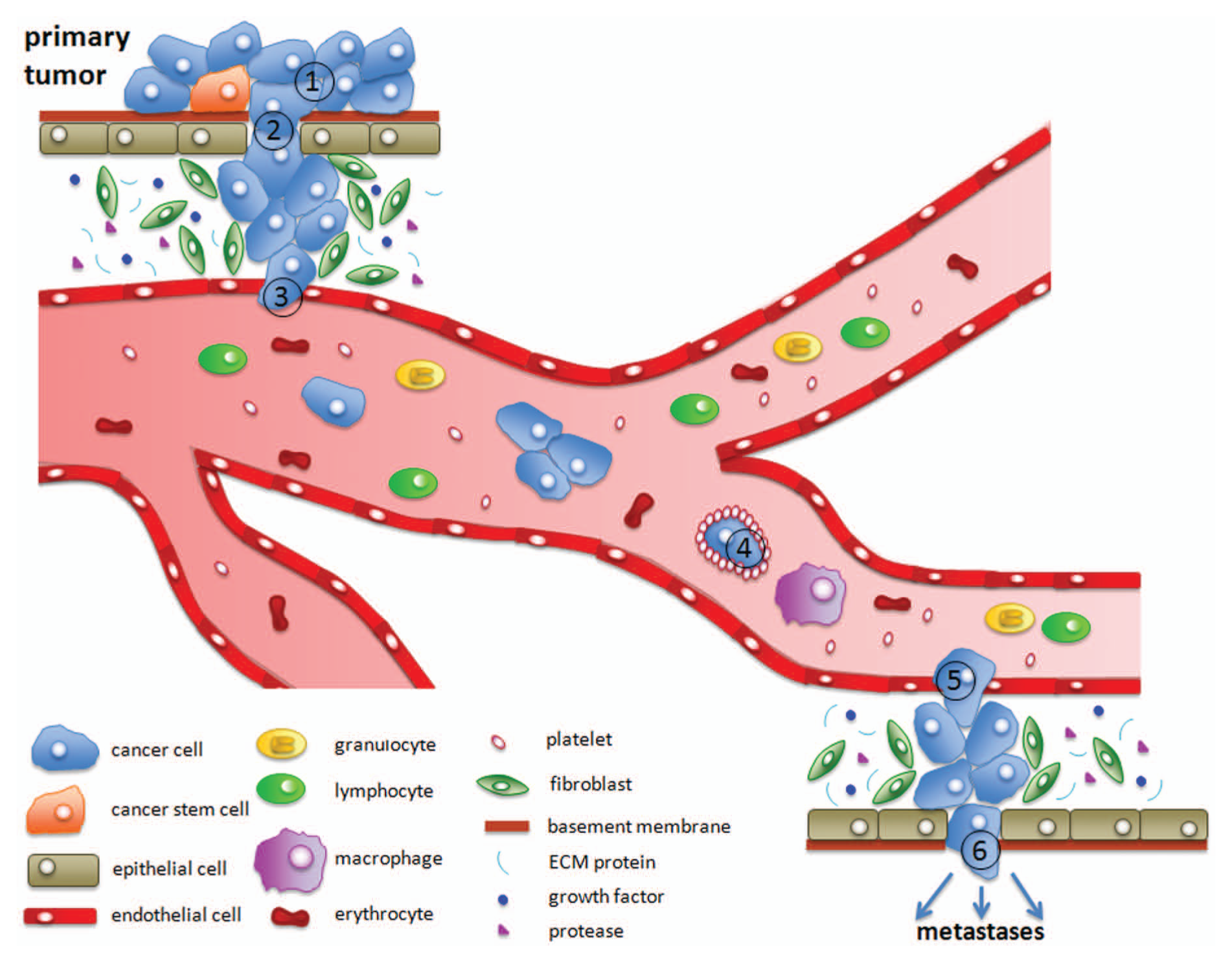
Overview of the process of metastasis: Progression from a primary epithelial cancer cell to an invasive, metastatic cell involves several steps. First, cancer cells undergo an epithelial to mesenchymal transition to (1) reduce adhesion to neighboring cells and (2) dissolve the basement membrane through the secretion of extracellular matrix metalloproteases. (3) Intravasation, or the entry of a cancer cell into the bloodstream, is achieved by the release of molecules, such as vascular endothelial growth factor, that stimulate angiogenesis. In the bloodstream, cancer cells can interact with platelets (4), which protect the cancer cell from the immune system. After reaching the secondary site, cancer cells can exit the bloodstream (5) by inducing endothelial cell retraction or death. Last, the cancer cells undergo a mesenchymal to epithelial transition (6) and continue to proliferate at the metastatic site. 157
Conventional cancer treatments elicit only a transient response in patients with metastatic disease, and as a result, these patients often relapse within 12 to 24 months of therapeutic intervention.6–8 Although quality of life may improve, the increase in survival rates has thus far been minimal. It has been long known that the presence of CTCs is indicative of shorter survival times.9–12 Detecting, isolating, and analyzing CTCs has the potential to improve diagnosis, allow prognostic monitoring, and enable targeted treatment strategies that are based on the metastatic cells most responsible for cancer mortality. CTCs may be sampled repeatedly in a minimally invasive way to monitor therapeutic efficacy and to account for constantly evolving tumor phenotypes.
There is currently only one US Food and Drug Administration (FDA)–cleared technology for CTC enrichment, CellSearch (Veridex, LLC, Raritan, NJ). Enumeration of CTCs enriched with this technology has been established as a prognostic marker and predictor of patient outcome in metastatic breast, 13 prostate, 14 and colon cancers. 15 CellSearch is based on immunomagnetic enrichment, employing antibody-coated magnetic beads to isolate cells that express the epithelial cell adhesion molecule (EpCAM). CTC identification criteria include (1) positive expression of monoclonal antibodies targeting cytokeratins (CK), a class of intermediate filaments present in epithelial cells; (2) negative expression of a leukocyte-specific antibody targeting the leukocyte common antigen, CD45; and (3) positive expression of a nuclear stain, DAPI. In addition, a cell must have a diameter of at least four microns to be identified as a CTC. 16 Nagrath et al. 17 designed a microfluidic chip consisting of an array of silicon microposts coated with EpCAM to improve CTC enrichment. This CTC-chip captured CTCs at a high purity of 50%, with a capture efficiency of 65% and a throughput of 2.5 mL/h. Various other immunoafinity-based technologies have been developed to enrich CTCs using capture antibodies that target EpCAM, including a microvortex-generating herringbone chip, 18 a magnetic sweeper device, 19 nanostructured silicon substrates, 20 selectin-coated microtubes, 21 and a surface functionalized medical wire that is injected in vivo for continuous sampling. 22 Other approaches have employed antibodies against cytokeratins, 23 prostate-specific antibodies,24,25 and a cocktail of various antibodies.26,27 In general, immunoaffinity-based approaches for positive enrichment of CTCs result in high cell purity, depending on the specificity of the antibody. Negative enrichment techniques have been demonstrated to enrich CTCs using antibodies that select and remove hematopoietic cells that express CD45 but with a purity that is orders of magnitude lower.28–30
One major limitation of immunoaffinity methods is their reduced efficiency with capturing tumor cells with low expression of EpCAM and other epithelial antigens. 31 When CTCs detach from the original tumor and intravasate into blood circulation, it has been hypothesized that phenotypic changes occur. One proposed change is an epithelial-to-mesenchymal transition (EMT) by which epithelial tumors cells overcome the physical constraints of cell-to-cell adhesions and acquire the motility and invasive characteristics of mesenchymal cells. EMT is known to be essential for cell and tissue remodeling during embryogenesis 32 and wound healing. 33 During EMT, the expression of epithelial proteins such as E-cadherin is downregulated, 34 while mesenchymal markers such as N-cadherin are upregulated. 35 Mani et al. 35 discovered that EMT generates cells with many of the properties of self-renewing stem cells. A reverse process of mesenchymal-to-epithelial transition (MET) during extravasation, and colonization has also been proposed. Currently, the EMT/MET model is gaining more evidentiary support in cell line studies, animal models, and clinical investigations. However, it is still unclear how widely this model applies to all metastatic diseases.
The recent discovery of the presence of cancer stem cells (CSCs) is another important concept in cancer metastasis. This rare subpopulation of cells within a tumor retains the ability to initiate and drive tumor cell expansion, as well as a capacity for self-renewal and to produce differentiated progeny. 36 The existence of CSCs was first proven in acute myelogenous leukemia37,38 and later verified in breast 39 and brain tumors.40–42 Recently, the identification of CSCs has also been reported in prostate, 43 ovarian, 44 and pancreatic 45 cancers based on the identification of specific cell surface markers. Conventional cancer treatments have been developed and tested based on their ability to kill the majority of the tumor population. However, CSCs can escape conventional therapy and have been shown in several tumor types to be more resistant to standard chemotherapeutic agents. 46 Approaches for CTC enrichment that are independent of expression of EpCAM or other cell surface antigens have been developed on the basis of intrinsic physical properties (e.g., size, deformability, density, and electrical properties). Expression of cell antigens can vary widely among different cancer types, and immunoaffinity-based enrichment is further complicated by the loss of epithelial markers during EMT and with rare CSC phenotypes. Physical enrichment strategies therefore have the potential for greater CTC yield, and they enable the analysis and evaluation of clinical relevance of new CTC subpopulations that do not fit within currently established definitions. There have been numerous reviews on biological and clinical studies involving CTCs5,47–56 and technological developments for CTC enrichment and detection.52,57–61 Readers are urged to consult those references for more general information, as we will focus specifically on CTC enrichment based on physical properties.
Biological Basis of Physical Enrichment Methods
Most metastatic solid tumors originate from epithelial cells. In addition to biochemical differences, there are distinct physical differences between these tumor cells and blood cells. The majority of physical methods for CTC enrichment exploit differences in size, deformability, and electrical (conductive and dielectric) properties between CTCs and blood cells. The physical properties of tumor cells are reviewed here.
Size of Tumor Cells
In general, caution should be taken when interpreting cell size measurements. The method of measurement, variation among samples, and sample preparation steps can result in different measured values even within the same cell population (e.g., a tumor cell line). Most published studies measure cell size by either optical microscopy or flow cytometry. With optical microscopy, the cell diameter or area is reported, whereas with flow cytometry, electrical impedance sensing (by Coulter counters) measures cell volume at DC or low frequency. In flow cytometry, optical diffraction and fluorescence measure the equivalent diffraction area or cross-sectional area, respectively, and both require calibration with standards (e.g., glass or plastic beads).62,63 There is a significant difference when cell size (diameter) is measured on a 2D surface by microscopy versus derived from cell volume measurement in suspension by flow cytometry. For example, on typical blood smears on a 2D surface, leukocytes are severely deformed into a pancake shape and normally reported as 7- to 20-µm diameter particles. 64 However, all leukocytes in isotonic suspension are measured to be less than 10 µm in diameter. 65 The relative importance of the techniques used to measure cell size depends on the CTC enrichment approach. For filtration-based methods, the cell diameter on a 2D surface is important, while for flow separation, the cell volume and 3D shape are likely to be more relevant. Ambient conditions can affect the results of a cell size measurement. For example, the tonicity of the measurement media will change the volume of viable cells, and fixation processes can also alter cell size. For cell lines, cell size is affected by the culture media formulation and in general by the health and status of the cells. Considering such complexity in cell size measurements, it is critical to compare tumor cell size with blood cells using the same measurement method under the same experimental conditions to facilitate size-based CTC separation.
Sizes of blood cells have been reported in many studies and textbooks ( Table 1 ). For example, erythrocytes in 2D blood smears have a diameter of 7.0 to 8.5 µm.64,66 The diameter of granulocytes has been measured to be 10 to 20 µm using microscopy of a blood smear,64,66 8.7 to 9.9 µm using flow cytometry, 66 and 7.3 to 13.2 µm using light microscopy of cell suspensions. 67 Lymphocytes are smaller than granulocytes. Their diameter is measured to be 6 to 18 µm in blood smears,64,66 7.1 to 10.5 µm using flow cytometry, 66 and 5.2 to 10.1 µm using light microscopy of cell suspensions. 67 Monocytes are among the largest of leukocytes, with a diameter of 12 to 20 µm in blood smears64,66 and 9.7 to 10.5 µm measured with flow cytometry. 66
Size (Diameter or Area) of Tumor Cells and Major Blood Cells.
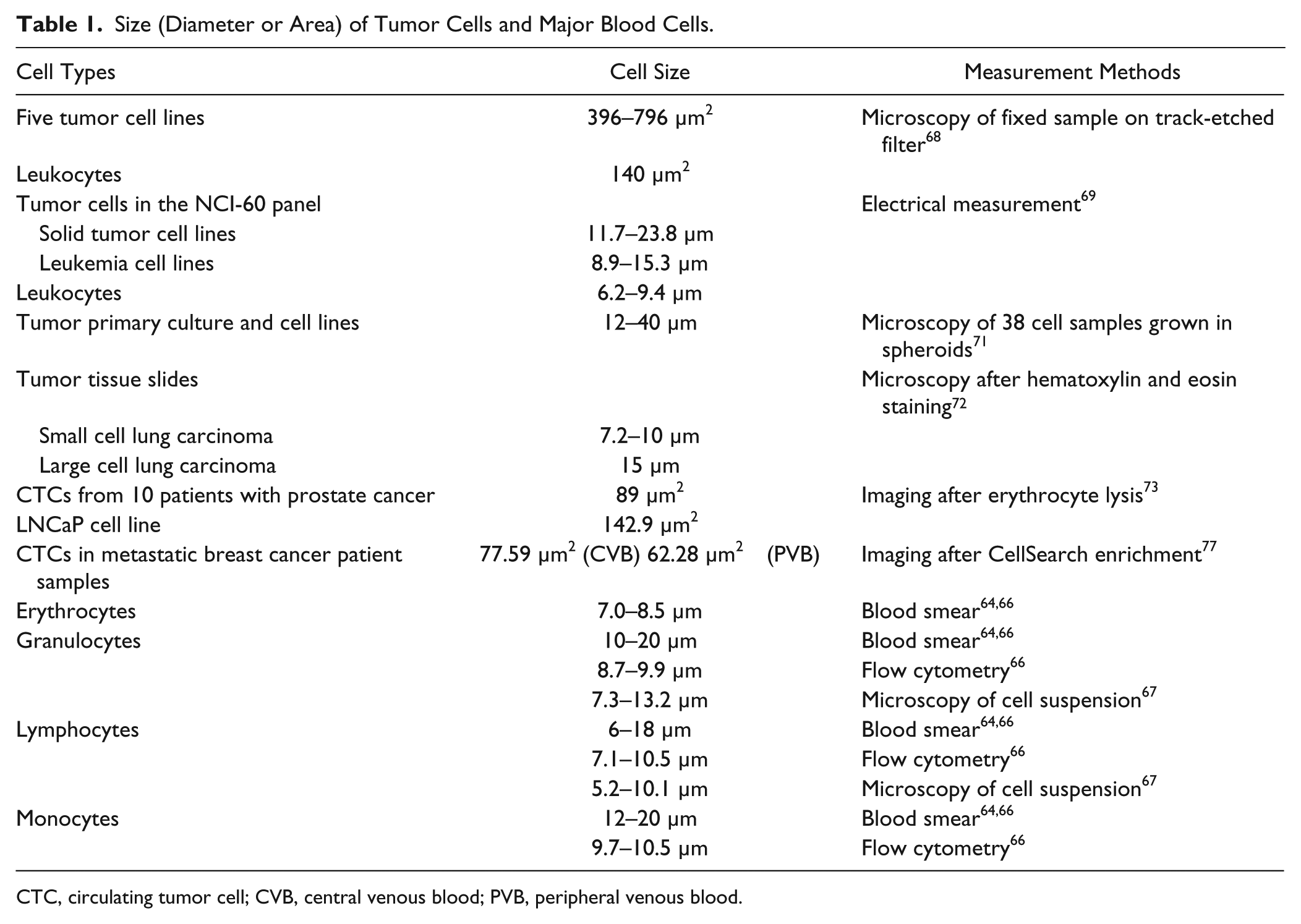
CTC, circulating tumor cell; CVB, central venous blood; PVB, peripheral venous blood.
It is generally agreed that cell lines originating from solid tumors are larger than blood cells. Using optical microscopy, the average diameter of 15 cancer cell lines commonly used in our laboratory is between 15 and 25 µm, whereas most leukocytes are less than 12 µm (unpublished results). After partial fixation and filtration through track-etched filters, the cross-sectional area of five tumor cell lines (MCF-7, Hep3B, HepG2, LNCaP, and Hela) has been measured to be 396 to 796 µm2 using microscopy, which is significantly larger than that of leukocytes (average of 140 µm2) measured under the same conditions. 68 In a recent study, the diameters of all cell lines in the NCI-60 panel were calculated from dielectrophoretic field-flow fractionation (DEP-FFF) and compared with those of various subtypes of blood cells. 69 Cell size extracted from the DEP data clearly shows the size difference of leukocytes (6.2–9.4 µm), leukemia cells (8.9–15.3 µm), and solid tumor cell lines (11.7–23.8 µm).
In examinations of routinely fixed and stained pathological tissue samples, viable epithelial cells (e.g., solid tumor cells) always have been larger than virtually all normal blood cells, including leukocytes.1,70 Measurement from 38 primary tumor tissue biopsy samples and cell lines grown in semisolid agar medium has resulted in tumor cell sizes in the range of 12 to 40 µm. 71 Measurement of 206 biopsy slides of small cell lung carcinoma (SCLC) and large cell lung carcinoma (LCLC) showed that these cells are on average 7.2 to 10 µm and 15 µm in diameter, respectively. 72
CTCs from clinical samples can be smaller than tumor cell lines. A study of imaging CTCs after erythrocytic lysis measured the cross-sectional area of CTCs from 10 patients with stage IV prostate cancer to be 89 µm2, whereas that of the LNCaP cell line was measured to be 142.9 µm2 under the same measurement conditions. 73 However, the clinical relevance of CTCs of varying sizes is unclear. One group studied the cytomorphology of CTCs from patient samples of various cancer types. Erythrocytes were lysed first, and then nucleated cells were plated on slides and CTCs detected by immunocytochemistry. A laser scanning imaging system was applied for high-definition imaging of CTCs. 74 In non–small lung cancer, smaller CTCs (4.3 vs. 15 mL−1 of normal CTCs) were not related to clinical features, survival, and treatment variables. 75 In a study of CTC detection on slides after erythrocytic lysis, CTC aggregates were found in 43% of 86 blood samples of breast, non–small cell lung, pancreatic, and prostate metastatic patients. 76 Area per cell for both single CTCs and CTCs in aggregates was twice that of leukocytes in the same field of view, although the length of CTCs in aggregates was smaller than that of single CTCs. It is interesting to note that in a CTC study using CellSearch, the average single-imaged CTC area from central venous blood (CVB) was larger than that measured from peripheral venous blood (PVB) of the same patient (77.59 vs. 62.28 µm2). 77
Smaller CTC-positive particles (defined as CK+CD45–DAPI+) were reported after immunological CTC enrichment. 18 CellSearch excludes CTC-positive particles smaller than 4 µm and regards them as cell fragments. 48 It has been reported that a high percentage of CTCs are in different stages of apoptosis and apoptotic cells are expected to have a smaller size, and thus it is possible that the smaller CTCs in clinical samples can be attributed to cell death. Mild apoptotic changes (e.g., cytoplasmic blebbing) are observed and acceptable for the CTC definition, whereas cells with apoptotic indicators in the nucleus are excluded from CTC counts with the assumption that these cells will not have clinical significance. 70 Another potential cause of CTC size variation is that CTCs may shift from an active state to a dormant state (cell cycle G0 like), which may increase their metastatic potential and thus indicate a poorer prognosis for patients.
Deformability of Tumor Cells
Various biomechanical tools have been developed to measure mechanical properties of living cells. 78 Local properties can be probed with atomic force microscopy (AFM) 79 and magnetic twisting cytometry (MTC), 80 while optical tweezers/laser traps, 81 a microplate stretcher, 82 micropipette aspiration, 83 and micropillar arrays 84 can study mechanical loading to whole cells. The advantages and drawbacks of these techniques have been reviewed in detail.85–87 In general, the measurement of cell deformability is affected by the method of choice as mentioned above, cell handing (e.g., culture media, ice/room temperature, dry/wet, fixed/unfixed), and the heterogeneity of cell mechanical properties (e.g., different regions of cells, cell cycle, cell differentiation, and aging). Although measurement of absolute Young’s modulus might not be reliable, comparing different cell populations under the same conditions will be able to provide qualitative conclusions.
For physical enrichment of CTCs from blood, it is important to compare the elastic properties of tumor cells and blood cells (especially leukocytes). In an AFM study, the apparent stiffness of neutrophils has been measured to be 156 Pa, 88 which is small compared with many other types of normal human cells as determined from other studies. 89 However, in another AFM study, the elastic moduli of the cell body part of neutrophils were measured to be 1.548 kPa, while the leading and trailing edges were significantly lower (686 Pa and 494 Pa). 90
Many studies have shown that tumor cells that have greater metastatic potential are more deformable. For example, one study applied AFM to measure elastic properties on single cells. The Young’s modulus of cancer or transfected cell lines (Hu456, T24, BC3726) was one order of magnitude lower than those of nonmalignant cell lines (Hu609 and HCV29). 91 In studies of breast cancer cell lines using AFM, benign MCF-10A cells were 1.4 to 1.8 times more rigid than the cancerous but nonmetastatic breast cancer cell line MCF7 (~0.5–1.1 kPa vs. 0.3–0.5 kPa). 92 Similar results were obtained with an optical cell stretcher.93,94 In another study, MCF7 cells were more rigid than the metastatic breast cancer cell line MDA-MB-436. 95 Similar measurements have been applied to clinical samples. Cross and colleagues 96 used AFM to measure the stiffness of live metastatic cancer cells taken from pleural fluids of patients with lung, breast, and pancreatic cancer. These metastatic cancer cells were 70% softer than the benign cells within the same pleural fluid samples (Young’s modulus 0.53 ± 0.10 kPa vs. 1.97 ± 0.07 kPa). Nevertheless, careful consideration should be applied when interpreting the Young’s modulus of living cells measured by AFM. 97 Most AFM studies have fit their results into the Hertz model, which assumes the measured material to be infinitely thick, purely elastic, isotropic, and homogeneous. Indentation depth, sample thickness, and contact point relevant to the center of the cell can all affect the measured results. For example, reducing ramp size from 2 µm to 500 nm and ramp velocity from 2 µm/s to 500 nm/s has increased the measured Young’s modulus by ~5 times for the same cell lines. 95 Microfluidic optical stretching is a method to measure single-cell deformability at a higher throughput. Measured with a microfluidic optical stretcher, primary oral squamous cell carcinomas cells have been 3.5 times more compliant than normal keratinocytes from healthy donors. 98 Similar results were obtained with normal and cancerous oral epithelial cell lines. Recently, one group used hydrodynamic shearing forces to measure the deformability of whole cells in microfluidic channels at a throughput of ~2000 single cells per second. They found that malignant cells obtained from pleural effusions of 11 carcinoma and mesothelioma patients were at least 18% more deformable than unactivated mononuclear leukocytes and granulocytes, 5% more deformable than activated granulocytes, and similar or more deformable than activated mononuclear leukocytes. 99 Microfiltration with track-etched pore filters has been used to study the deformability of tumor cells qualitatively. In a study of B16 melanoma cells and its variants, the time required to pass 50% of the tumor cells was used to correlate to cell deformability, and again it was found that metastatic cells are more deformable. 100 The elastic rigidity reduction during malignant transformation may be induced by a reduction of F-actin concentration 101 and a generally more fluid-like state of the cytoskeleton. 102
Electrical Properties of Tumor Cells
It has long been known that most mammalian cells have a net negative surface charge under physiological conditions.103,104 Moreover, cells contain a variety of polarizable particles/molecules, including proteins, peptides, and nucleic acids. Thus, cells can have complicated dielectric properties that are frequency dependent. At low frequencies, interfacial polarization dominates, which depends mainly on the conductive properties of polarizable particles and their surrounding medium. In general, the plasma membrane acts as an electrical insulator below 25 kHz, and viable cells behave like insulating objects. At high frequencies (10 MHz up to GHz range), dielectric properties (permittivity) become more important, and cellular features, such as nucleus-cytoplasm volume ratio and endoplasmic reticulum, play important roles. 105 While at intermediate frequencies, both conductive and dielectric properties can be significant. Cell size and shape, membrane integrity and morphology, and cytoskeletal structure can be explored by electrical measurements. Various techniques have been used to measure electrical properties of cells, including impedance spectroscopy, electrorotation (ROT), and dielectrophoresis (DEP). Impedance spectroscopy uses electrodes to scan the electrical impedance over a certain frequency range.106–110 It can be applied to either a cell population or single cells in a flow cytometry format. ROT can be used to measure single-cell dielectric properties.111,112 In ROT, normally four electrodes are arranged in a crisscross configuration, and the single cell is located at the center of the four electrodes. The ROT excitation signals are applied to the four electrodes in a 90-degree phase-shifted fashion. The cell rotates and the rotation speed is recorded. This speed depends on the difference between the electric polarity of the cell and its surrounding medium under the frequency of the ROT excitation signal. DEP requires applying a nonuniform electromagnetic field to the cell, with the cell responding to the DEP force by moving toward (positive DEP, pDEP) or away from (negative DEP, nDEP) the strong electromagnetic field.112–115 The DEP crossover frequency from pDEP to nDEP is a characteristic value for cell dielectric properties given the same surrounding medium.
Han and colleagues 116 measured whole-cell impedance spectroscopy of various breast cancer cell lines (MCF-10A, MCF-7, MDA-MB-231, and MDA-MB-435) using a microfabricated device. After fitting the data into a parallel RC circuit model, the equivalent membrane capacitance and resistance were calculated for each cell line at 100 kHz. The results show that both magnitude and phase of the whole-cell impedance of different cell lines are distinct.
ROT spectra (electrorotation speed vs. frequency) between 1 kHz and 10 MHz were measured for metastatic breast (SkBr3) and lung (A549) cancer cell lines along with different human leukocyte subtypes, including T lymphocytes, B lymphocytes, granulocytes, and monocytes in a low-conductivity medium. 117 After fitting into a single-shell dielectric model, the dielectric properties of cells, including unit membrane capacitance and cytoplasm conductivity, were extracted. The tumor cells were shown to have higher unit membrane capacitance and lower cytoplasm conductivity compared with leukocytes.
One group used DEP-FFF to study dielectric properties of cancer cells and applied it for CTC enrichment. A summary of the total cell electrical capacitance of seven subtypes of blood cells and nine cancer cell lines shows that the capacitances of cancer cells were significantly larger than those of blood cells. 118 All the data points show that the total cell capacitance scaled with the cube of cell diameter, which is consistent with the general conclusion that cancer cells are larger than blood cells. In a recent study, this group expanded on their previous work and compared more than 50 solid cancer cell lines in the NCI-60 panel with various blood cells and leukemia cells. 119 The dielectric and density properties of cells were measured by DEP-FFF. The DEP crossover frequencies of solid tumor cells were extracted from the measurements and were distinctly lower than for blood cells and leukemia cells. The cutoff frequency was 65 kHz. Under 1 MHz, the DEP crossover frequency of a spherical mammalian cell was inversely proportional to cell diameter and the capacitance per unit area of the cell plasma membrane. Both calculated cell diameter and the capacitance per unit area differed between cancer cells and blood cells. 69
Physical Enrichment Technologies
The rarity of CTCs in whole blood, on the order of one CTC per billions of blood cells or per millions of nucleated blood cells, poses a major challenge for enrichment. Heterogeneity among CTCs and the complexity of clinical blood samples present additional challenges. In general, to successfully evaluate technologies for CTC enrichment, multiple performance parameters (i.e., capture efficiency/recovery, enrichment against leukocytes, cell viability, processing speed, blood sample capacity, sample pre-processing requirements, cost of consumables and equipment, and repeatability and reliability) must be considered. The optimal enrichment solution may require a compromise among performance parameters, and it is likely to depend on the intended downstream application.
Various approaches have been used to exploit the differences in physical properties between tumor cells and blood cells to enrich and separate CTCs from blood samples. These technologies are summarized in Figure 2 and discussed in detail below.
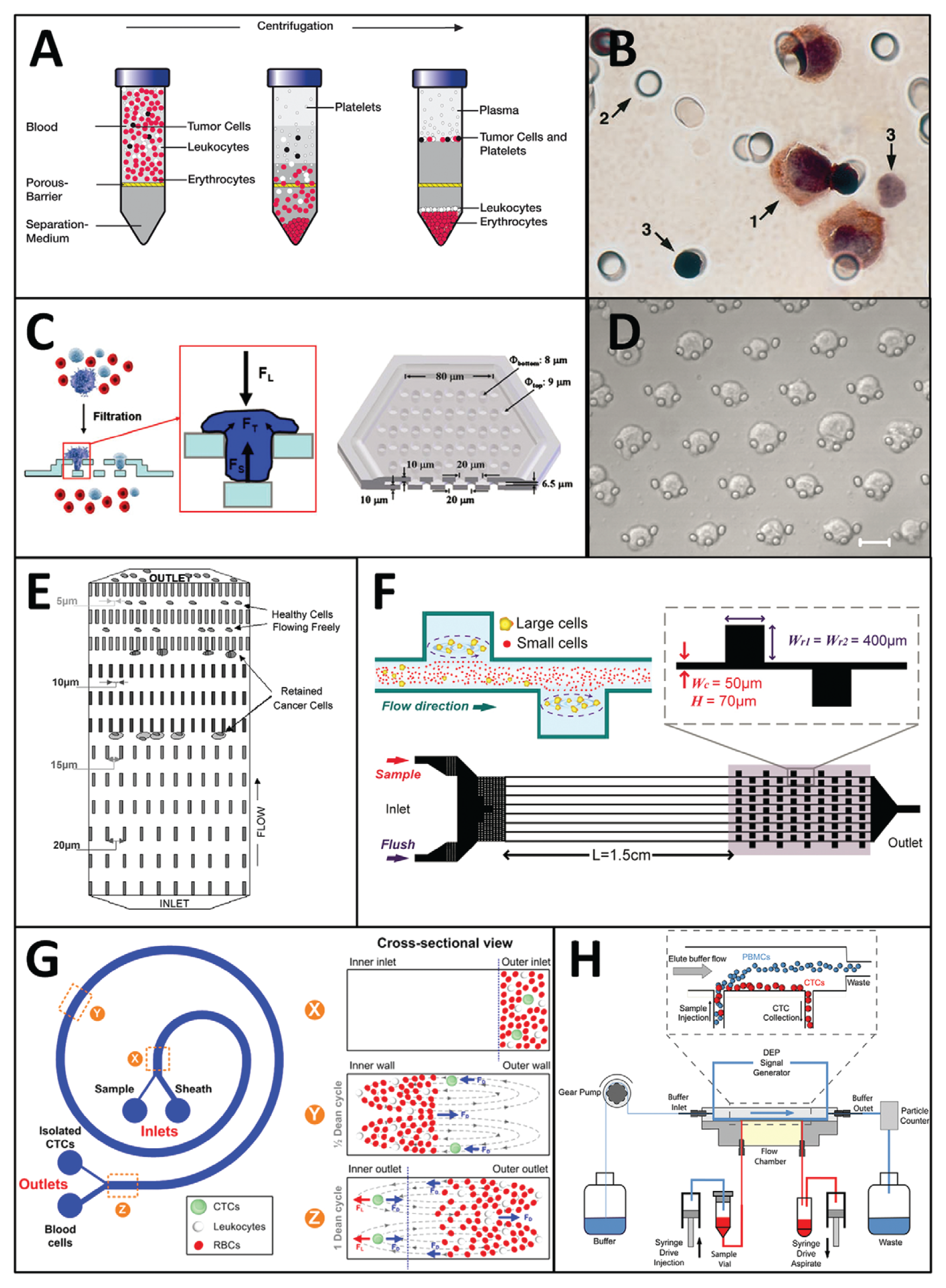
Technologies for circulating tumor cell (CTC) enrichment based on physical properties. (
Centrifugation
Centrifugation is a widely used technique for fractionating blood into its constituent components. Density-based gradient centrifugation segregates CTCs in the mononucleocyte fraction of blood away from the more dense cells present in the erythrocytic and granulocytic fractions. In 1959, Seal 120 developed a floatation method using blended silicone oils as a solution for CTC enrichment that was quantitative, inexpensive, and simple to perform. Using this technique, he successfully detected CTCs in 45% of patient samples obtained from a variety of cancers with cytological staining after a secondary size-based filtration step. A synthetic polysaccharide was later incorporated into a Ficoll-Paque (Pharmacia-Fine Chemicals, Uppsala, Sweden) solution, which became the gold standard for gradient centrifugation separation of mononuclear cells from blood and bone marrow aspirates. Ficoll-Paque was used to enrich CTCs that could be detected with a resolution of 1 cell/mL of blood in model systems using reverse transcription–PCR assays (RT-PCR). 98 This technique was used to detect CTCs in 41% of patients undergoing surgical resection in colorectal cancer. 98 OncoQuick (Greiner BioOne, Frickenhausen, Germany) is a novel technology incorporating a porous barrier that allows erythrocytes and some leukocytes to pass through while retaining CTCs in conjunction with density-based centrifugation to achieve more effective enrichment ( Fig. 2A ). Rosenberg et al. 121 report an enrichment ratio of 632-fold against leukocytes with OncoQuick compared with 3.8-fold with Ficoll-Paque–based centrifugation. The OncoQuick method was used to enrich CTCs in blood samples obtained from 30% of 37 patients with gastrointestinal cancer 121 and 40% of 63 patients with advanced breast cancer 122 with RT-PCR–based CTC detection. In a comparative study, CTCs were found in 23% of 61 patients using immunocytochemical detection on cytospins prepared after OncoQuick enrichment, as opposed to 54% with the CellSearch instrument. 123
As an inexpensive and dependable technique, centrifugation is widely employed for CTC enrichment. However, the elimination of contaminant leukocytes is limited, with even the most advanced centrifugation techniques achieving a purity of less than 1%. It is therefore commonly used as an initial step in combination with further enrichment techniques.
Microfiltration
Microfiltration technology has demonstrated the greatest potential for achieving high-throughput continuous processing of large volumes of blood. Since track-etched polymer filters were first invented in the 1960s,124,125 they have been widely used in biological research and clinical practice for cell enrichment. In 1964, Seal 126 once again made a pioneering contribution to the field when he applied these filters to attempt to capitalize on his stated observations that CTCs isolated with his silicone flotation method were generally larger and more rigid than blood cells. Decades later, track-etched polycarbonate filters with 8-µm diameter pores were effectively used for CTC enrichment and cytological detection from fixed blood samples using the isolation by size of epithelial tumor cells technique (ISET) developed by Vona et al. 68 ( Fig. 2B ). Track-etched filters have been successfully used to enrich and analyze CTCs in liver cancer, 127 melanoma, 76 lung cancer,128,129 prostate cancer, 130 and other various cancers. 131 In a comparative study, ISET detected CTCs in 95% of 60 patients with metastatic breast, prostate, and lung cancer compared with 70% with CellSearch, although CellSearch did occasionally detect CTCs in greater quantities. 102 Zheng et al. 132 used deterministic photolithography to develop an improved pore-shaped microfilter that was fabricated out of a single 10-µm-thick layer of parylene, reporting a capture efficiency of ~90%. This device was used in a blind comparison study to identify CTCs in 89% of 57 patients with cancer as opposed to 46% with the CellSearch. 133 Similar porous membranes have been fabricated from electroformed nickel 134 and silicon substrates. 135 Although successful for CTC enumeration, these microfiltration techniques apply concentrated stresses that affect the viability of enriched CTCs.136,137 To address this issue and enable viable CTC enrichment, a 3D microfilter was designed out of two layers of parylene to incorporate support structures that mitigate cell damage ( Fig. 2C ). 137 Track-etched filters were also adapted for viable cell capture using the ScreenCell (ScreenCell, Paris, France) system, which reported 74% to 91% recovery and 85% viability. 138 Xu et al. 139 used a parylene filter designed by Tai’s group 158 with slot-shaped pores to detect telomerase activity from viable enriched CTCs filtered from Ficoll-Paque–isolated buffy coats of patients with metastatic prostate cancer.
The speed and simplicity of microfiltration allows for rapid CTC enrichment from large volumes of clinical blood in minutes with minimal processing. Although high capture efficiencies on the order of 90% and greater can be reliably achieved, the final purity is typically on the order of 10% or less, which may require further processing for some downstream applications.
Microfluidics
Microfluidics allows unparalleled control and ability to manipulate fluids in miniscule volumes. The past decade has seen many novel technologies proposed for biological cell sorting and analysis on microchips. Mohamed et al.91,140 used arrays with pillars of varying geometries to fractionate cells in blood and capture tumor cells ( Fig. 2E ). They demonstrated the capture of neuroblastoma and other cancer cell lines from 1:10 diluted blood samples with a processing rate of 1 mL/h. Tan et al. 141 incorporated crescent-shaped trap arrays with a fixed 5-µm gap width within microfluidic chambers to enrich CTCs from whole blood without preprocessing, reporting a capture rate of greater than 80% and a purity of more than 80%. This device was used to successfully detect CTCs in 1- to 3-mL blood samples obtained from patients with metastatic lung cancer. 142 Tumor cell separation based on size and deformability can also be achieved through cross-flow filtration within a serpentine microfluidic channel. 136
Inertial flow fractionation enables microfluidic enrichment of tumor cells by exploiting hydrodynamic forces to select for cells of different sizes. Microfluidic channels incorporating contraction and expansion reservoirs were developed for pinch alignment of tumor cells by Bhagat et al. 143 and tumor cell trapping in micro-scale vortices by Hur et al. 144 ( Fig. 2F ). These devices allow a significantly higher throughput compared with previous microfluidic approaches, but with potential reductions to cell recovery rate and enrichment against leukocytes. A similar approach was used to classify cells based on deformability. 145 Sun et al.146,147 developed a double-spiral microfluidic channel to hydrodynamically separate tumor cells using drag forces, reporting a recovery rate of 88.5% from diluted blood. Hou and colleagues 148 incorporated a spiral microfluidic channel to successfully enrich CTCs and microclusters in 20 blood samples from patients with metastatic lung cancer ( Fig. 2G ).
Carefully applied microfluidic approaches are capable of achieving excellent purity of greater than 80% and high capture rates with little disturbance to CTCs. However, this comes at the expense of throughput, requiring either reduced sample volumes or several hours to process a full tube of blood. Recently developed hydrodynamic flow-sorting approaches can achieve a throughput of up to 3 mL/h for microfluidic systems, but with a reduction in sample purity to below 10%.
Dielectrophoresis
The phenomenon of dielectrophoresis has inspired novel approaches for the separation of cells based on their electrical properties. Becker et al. 149 fabricated interdigitated gold electrodes and used them to separate leukemia and breast cancer cell lines 63 from healthy blood cells. Tumor cells were attracted toward the electric field generated by the electrodes by pDEP, while other cells were flushed away. The electric field was then turned off and the cells were released for collection with a recovery rate of 95%. Building on this approach, Huang et al. 150 proposed DEP-FFF as a continuous cell fractionation process that did not require intermittent activation and deactivation of an electrical field. Gupta et al. 62 presented ApoStream, the first commercial instrument for continuous-flow DEP-FFF enrichment of CTCs ( Fig. 2H ). They reported a capture efficiency of more than 70% and viability greater than 97% from cell lines spiked in 7.5 mL of whole blood after an initial Ficoll-Paque gradient centrifugation step. Preliminary efforts have been undertaken to demonstrate the application of DEP-FFF to clinical patient samples. 119
The unique application of DEP-FFF to cell electrical properties may enable the capture of a population of CTCs that would not be obtained by other physical enrichment approaches. Testing with cell lines has demonstrated excellent viability and minimal disruption to captured cells. However, an initial centrifugation enrichment step is required, and whole blood cannot be processed directly. One hour is required to process mononuclear cells obtained from 7.5 mL of blood after Ficoll-Paque enrichment. The capture purity is expected to be less than 1%, although this can be significantly improved with additional enrichment stages at the risk of reduced capture efficiency.
Perspectives
Any enrichment technique applied to CTCs will be biased according to its principle of enrichment. It is therefore likely that different approaches will result in the enrichment of different CTCs. These CTC populations might not completely overlap. There is currently no universal marker that may be used to enrich or detect all CTCs. There is a need for comparative studies between technologies to learn more about the variety of cells being captured with the different enrichment methods.
It is expected that other proposed CTC enrichment and detection systems will apply the same criteria used by the current gold standard, CellSearch, for comparison. However, this is complicated by the fact that there are no standardized antibodies for CTC detection. CK consists of many different types and isoforms, and their expression levels vary widely among different cancer types. 151 Moreover, given the variable expression of markers during EMT and within CSCs, the current definition of a CTC in terms of immunocytochemistry may not include all clinically relevant tumor cells. For certain cancer types, there are known tissue- or organ-specific markers, such as prostate-specific antigen for prostate cancer, carcinoembryonic antigen for colon cancer, cancer antigen-125 for ovarian cancer, and human epidermal growth factor receptor 2 for breast cancer. These may be used to identify CTCs of that specific cancer type.
It is important not to lose sight of the overall clinical purpose of CTC enrichment. Since the ultimate goal of CTC analysis is to improve cancer diagnosis and treatment monitoring, it is important to demonstrate a correlation with disease progression and patient outcome. Promising new approaches such as enrichment by acoustophoresis152,153 and other emerging technologies must be evaluated with patient samples to determine clinical relevance. The clinical relevance of any enriched CTC subpopulation must eventually be established through large-scale clinical trials. To date, this has only been achieved with the US FDA clearance of CellSearch in breast, prostate, and colorectal cancers.
It is desirable to obtain CTCs that are not disturbed by the enrichment process and that maintain their viability. Enrichment approaches that allow rapid isolation in a single step could avoid phenotypic alterations to CTCs that would complicate RNA and protein expression analyses. The proliferation and primary culture of viable CTCs has the potential to significantly advance our understanding of CTC biology, as well as enable the evaluation of therapeutic efficacies ex vivo.
One of the most promising CTC applications is treatment monitoring, where patient blood samples are obtained periodically before, during, and after cancer treatment for CTC detection and analysis to determine if the treatment is effective. Conventional treatment monitoring is performed mainly with medical imaging techniques. However, CTC analysis might be a more sensitive and accurate technology and potentially provide more useful information.13,154 So far, CTC enrichment and detection has been mostly limited to metastatic solid tumors. However, there is recent evidence that CTCs at even lower concentrations might exist in early stage cancer patients.155,156 If the sensitivity and sample processing capability of CTC analysis can be improved, it is possible that CTC technologies can be used for early cancer detection.
Recent studies have confirmed a high degree of heterogeneity among CTCs. The resolution of CTC analysis must improve beyond enumeration and analysis of CTCs as a population and should allow for evaluation on a single cell level. This will allow the characterization of clinically important rare cell phenotypes, including CSCs and CTCs in various stages of EMT.
Footnotes
Acknowledgements
We thank the Penn State Materials Research Institute, Nanofabrication laboratory and Microscopy and Cytometry facility, and Penn State Hershey Cancer Institute for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is partially supported by the Pennsylvania State University start-up fund and the National Cancer Institute of the National Institutes of Health under award numbers R21CA161835 and DP2CA174508. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
