Abstract
The increasing mortality from ovarian and lung cancers worldwide is largely due to late diagnosis and therapeutic resistance. Conventional biomarkers such as CA125 and CYFRA 21-1 lack the sensitivity or the specificity for monitoring real time disease progression or therapeutic response. This has prompted researchers to work on the development of relevant biomarkers that are minimally invasive for cancer diagnosis, prognosis and for therapeutic stratification. As such, liquid biopsies, which analyze tumor-derived components circulating in the blood including circulating tumor cells (CTCs), which are shed from primary or metastatic tumors into the bloodstream have emerged as a promising approach for detecting such cancers. This review critically analyzes the diagnostic and prognostic significance of CTCs in ovarian and lung cancers, highlighting technological advances, molecular characterization, and translational challenges associated with their clinical application. It briefly discusses the clinical utility of some biomarkers from liquid biopsies used in the diagnosis, prognosis as well as monitoring of ovarian and lung cancers with their detection methods. It highlights some of the relevant studies conducted in the field and finally it provides insights on the future of liquid biopsies. An extensive literature review analyzing papers between 2008 and 2026 was conducted using PubMed, Scopus, and Web of Science databases, that evaluated CTC detection methods, molecular profiling, and clinical outcomes in ovarian and lung cancer patients. Current literature reports that CTC enumeration and molecular characterization can provide significant insights into disease progression, therapeutic resistance and predict survival outcomes. Whilst the enrichment technologies such as CellSearch®, ISET, and microfluidic platforms have enhanced detection sensitivity, variability in assay performance and lack of a clinical standardization impede proper implementation. CTCs represent a powerful liquid biopsy tool with potential to revolutionize cancer diagnostics and precision medicine. Integration of multi-omics profiling, CTC-derived organoids, and AI-driven analytical models may further enhance their clinical utility in guiding personalized treatment strategies for ovarian and lung cancer patients.
Keywords
Introduction
Ovarian cancer is a significant contributor to global cancer morbidity and mortality, with an estimated 324 603 new cases and 206 956 deaths worldwide in 2022 (GLOBOCAN, 2022), corresponding to an age-standardized incidence rate (ASR) of 6.7 per 100 000 and an ASR mortality of 4.0 per 100 000. 1 The 5 principal histological types of epithelial ovarian cancers include low grade serous carcinomas, high grade serous carcinomas, clear cell, endometrioid and mucinous tumors. 2
Low grade serous carcinomas (LGSCs) are generally diagnosed at an early age and have better prognosis compared to High grade serous carcinomas (HGSCs), which comprises 70% of cases and leads to death within 10 years of diagnosis in 75% of the cases.
3
Studies showed a high frequency of
On the other hand, lung cancer remains the most lethal cancer globally. GLOBOCAN 2022 estimates show ~2.48 million new lung cancer cases (12.4% of all new cancers) and ~1.82 million deaths (18.7% of all cancer deaths). 1 There are mainly two broad types of lung cancers: Non-Small Cell Lung Cancer (NSCLC) and Small Cell Lung Cancer (SCLC). 6
The most prevalent genetic mutations linked to lung cancer development include
Both these malignancies, however, are often asymptomatic initially or diagnosed during advanced stages and demonstrate high recurrence rates. As a result, 5-year survival in the advanced stages remains quite low.8,9
Methodology
A literature search was conducted using PubMed, Scopus, and Web of Science databases to identify relevant studies published up to 2025. Search terms included “circulating tumor cells,” “ovarian cancer,” “lung cancer,” “liquid biopsy,” “multi-omics,” “organoids,” and “artificial intelligence.” Articles were selected based on relevance to translational and clinical applications.
Conventional Biomarkers and Their Limitations
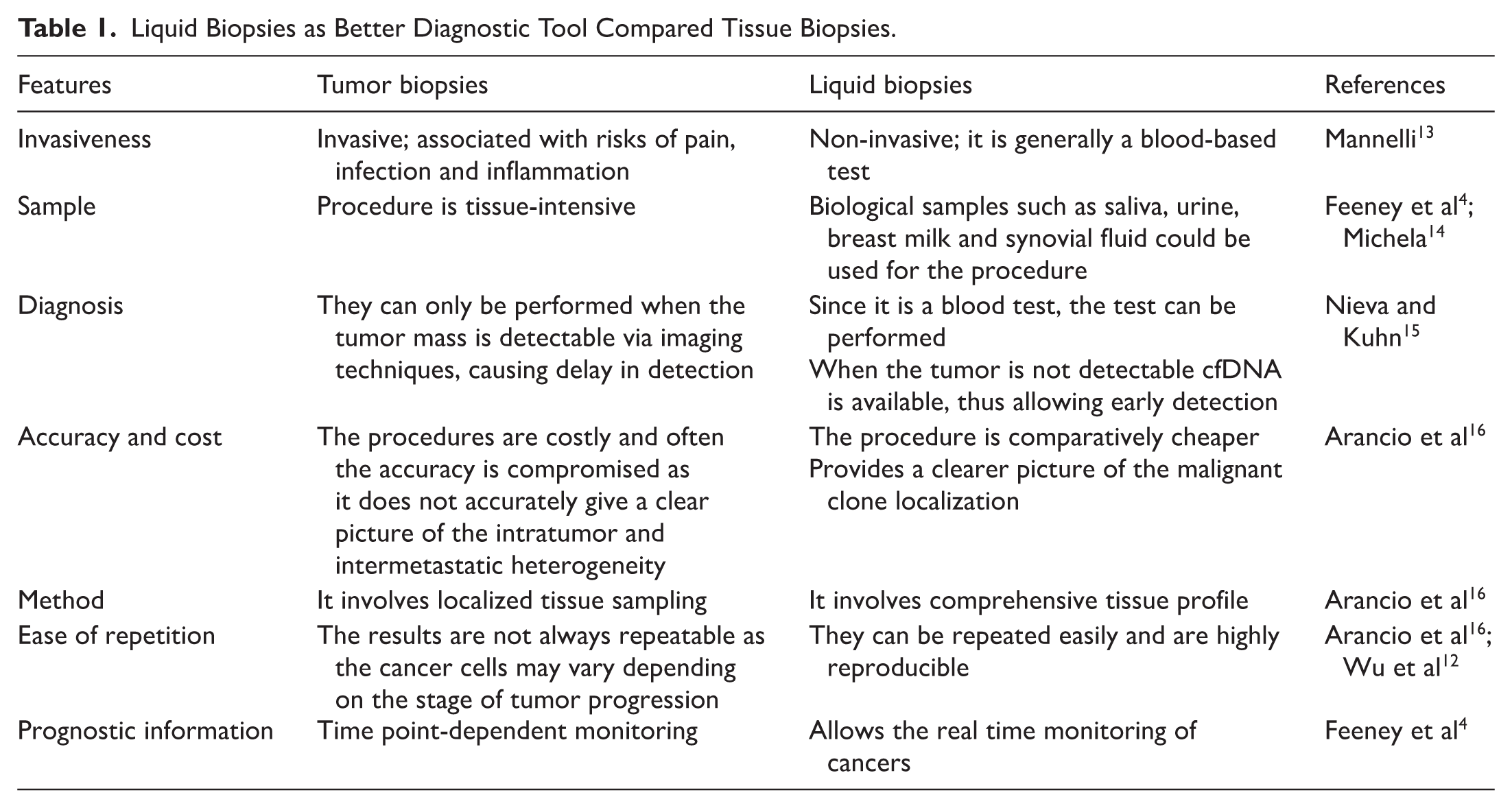
The diagnostic and prognostic tools for these cancers remain limited. In ovarian cancer, the serum biomarkers CA-125 and HE4 (human epididymis protein 4) are routinely used; however, CA-125, an antigen and a standard biomarker for the monitoring of ovarian cancer, 10 particularly demonstrates low sensitivity in early disease and can be elevated in other benign gynecological conditions, such as endometriosis, while HE4 offers higher specificity but still lacks validation.8,11 In contrast, biomarkers used for lung cancers, such as CEA (carcinoembryonic antigen), CYFRA 21-1 (cytokeratin 19), and NSE (neuron-specific enolase) do not reliably support early detection or real-time monitoring of therapeutic response. Tissue biopsy, while a diagnostic gold standard, 12 is invasive, may not fully represent tumor heterogeneity, and is poorly suited for longitudinal assessment. Together, these limitations underscore the urgent need for minimally invasive biomarkers capable of capturing tumor evolution and treatment dynamics (Table 1).
Liquid Biopsies as Better Diagnostic Tool Compared Tissue Biopsies.
Circulating Tumor Cells as a Liquid Biopsy Tool
Liquid biopsies have emerged as a transformative approach in oncology, providing a minimally invasive means to obtain molecular insights into tumor biology through peripheral blood or other biological fluids. This technique enables real-time monitoring of tumor dynamics, offering valuable information on tumor heterogeneity, therapeutic response, and disease progression.14,17
Among the key analytes accessible through liquid biopsy are circulating tumor cells (CTCs), circulating tumor DNA (ctDNA), cell-free DNA (cfDNA), tumor-derived extracellular vesicles (tdEVs), cell-free microRNAs (cf-miRNAs) and tumor-educated platelets (TEPs).17,18 Each of these elements reflects a distinctive biological aspect of cancer, collectively forming a comprehensive snapshot of the tumor’s molecular and genetic landscape. 19
CTCs (Figure 1), in particular, are intact, viable tumor cells that shed from the primary or metastatic sites and circulate into the bloodstream.20,21 They often carry tumor-specific antigens or oncogenic genetic alterations, including gene amplifications, point mutations, chromosomal rearrangements, and copy number variations, which reflect those found in the primary tumor.22,23 These attributes primarily make CTCs a powerful tool for tracking disease progression through blood sampling and therapeutic decision-making.

Schematic representation of CTCs, ctDNA and cfDNA found in blood serum and plasma.
Whilst cell-free DNA represents fragmented genetic material released by apoptotic or necrotic tumor cells, it remains stable in plasma compared to free RNA, which rapidly degrades in circulation. Nevertheless, microRNAs enclosed in vesicles such as exosomes or platelets are also detectable in the serum or plasma of cancer patients and provide additional layers of transcriptomic information. 20 Analyzing the RNA in exosomes or platelets within exosomes or TEPs can provide important insights into tumor gene expression and intercellular signaling mechanisms.
Recent advances in high-sensitivity and high-specificity analytical methods have significantly enhanced the detection of circulating tumor-derived materials, hence revolutionizing early cancer diagnostics and disease monitoring. 14 These include microfluidic enrichment systems, immunomagnetic capture technologies, and next-generation sequencing (NGS)-based approaches that allow multiplexed genomic profiling from minute quantities of circulating material. 19
These developments, collectively, underscore the potential of liquid biopsy, primarily CTC analysis, as a minimally invasive, reproducible, and real-time monitoring tool for assessing both ovarian and lung cancers compared to conventional biopsies (Table 1). However, liquid biopsy currently lacks the spatial resolution needed to define tumor histotypes or accurately assess local tissue invasion, limitations that still need the conventional histopathological examination. 16 Continued refinement in detection technologies and multi-analyte integration will likely expand the clinical utility of CTC-based liquid biopsies in the near future.
Expanding Applications of CTC Analysis in High-Mortality Solid Tumors
Ovarian and lung cancers share key oncogenic hallmarks, including activation of PI3K/AKT/mTOR, MAPK, and epithelial–mesenchymal transition (EMT) programs that drive tumor progression, metastatic dissemination, and therapeutic resistance.25,26 Recurrent driver alterations such as
Across multiple clinical cohorts, CTC enumeration has demonstrated a strong correlation with advanced stage and inferior survival outcomes. In ovarian cancer, elevated baseline CTC counts predict platinum resistance and shorter PFS and OS.16,28 In lung cancer, dynamic changes in CTC number and phenotype anticipate resistance to EGFR tyrosine kinase inhibitors and immunotherapy.29,30 Beyond enumeration, molecular characterization of CTCs, including detection of EGFR mutations and assessment of PD-L1 expression, provides clinically actionable information aligned with precision oncology frameworks.27,30
Integration of CTC profiling with circulating tumor DNA (ctDNA) and other circulating analytes enhances detection sensitivity and improves early identification of resistance mechanisms, supporting a multidimensional liquid-biopsy strategy.17,19
Origin and Circulation of CTCs in Blood
CTCs arise when tumor cells detach from primary or metastatic sites and enter the bloodstream through EMT-associated processes (Figure 2). In ovarian cancer, loss of epithelial markers such as E-cadherin and induction of mesenchymal markers including vimentin facilitate dissemination and hematogenous spread.
16
In lung cancer, early vascular invasion and activation of oncogenic drivers such as

Fate of CTCs in circulation.
Once in circulation, CTCs encounter shear stress and immune surveillance. Survival is supported by stemness-associated markers (CD44, ALDH1) in ovarian cancer and immune-evasive phenotypes such as PD-L1 expression and PI3K/AKT activation in lung cancer. Notably, CTC clusters demonstrate substantially higher metastatic potential than single CTCs. 32
CTCs display marked phenotypic and molecular heterogeneity, encompassing epithelial, mesenchymal, and hybrid states. Clinically relevant genomic alterations, including
Role of CTCs in the Circulation for the Diagnosis and Prognosis of Ovarian and Lung Cancers
Ovarian Cancer
CTCs are detectable in a substantial proportion of patients with epithelial ovarian cancer, particularly in advanced-stage disease, where their presence correlates with tumor burden and adverse outcomes. 28 Elevated baseline CTC counts are consistently associated with shorter PFS, reduced OS, and platinum resistance.16,28 Importantly, dynamic monitoring of CTC counts during chemotherapy has demonstrated predictive utility, with rising CTC levels often preceding radiological progression. 34
Beyond enumeration, molecular characterization of ovarian CTCs provides clinically actionable information. Recurrent alterations in BRCA1/2, PIK3CA, and TP53 are detectable in CTCs, reflecting tumor genomic evolution. EMT-associated transcripts and stemness markers (eg, CD44, ALDH1) are enriched in platinum-resistant disease, supporting their role in relapse biology.10,35 Detection of resistance-associated genes such as ERCC1 further suggests potential utility in predicting chemotherapy response.35,36
CTC clusters have also been identified in HGSC and are associated with more aggressive phenotypes. 37 Collectively, integration of CTC analysis with CA-125, imaging, and homologous recombination deficiency status may refine prognostic stratification and support real-time therapeutic decision-making. 38
Lung Cancer
In lung cancer, particularly NSCLC, CTCs are detectable even in early-stage disease and correlate strongly with TNM stage, metastatic burden, and inferior survival outcomes. 30 In small-cell lung cancer (SCLC), extremely high CTC counts have diagnostic and prognostic significance. 39
A major advantage of CTCs in lung cancer lies in their capacity to capture tumor evolution and emerging resistance. Molecular profiling of CTCs enables real-time detection of actionable alterations including
PD-L1 expression on CTCs and the presence of circulating tumor microemboli further contribute prognostic and predictive value, particularly in patients receiving immunotherapy.39,42 Together, these findings support the role of CTCs as integrative biomarkers that complement tissue genotyping and imaging in lung cancer management.
Table 2 provides a comparative overview of the diagnostic, prognostic, and molecular characteristics of CTCs across ovarian and lung cancers.
Comparison of the Diagnostic, Prognostic, and Molecular Characteristics of CTCs in Ovarian and Lung Cancers.

Detection and Analysis of CTCs
Among the most widely used methods for detection and isolation of CTCs are immune-based approaches, in which antibodies selectively bind to tumor cell surface antigens. 45 Tumor-specific markers allow separation of CTCs from circulating blood cells. Figure 3 depicts the principal CTC detection and analysis techniques used in clinical applications. CTCs can be isolated using label-dependent or label-free approaches.

CTC detection and analysis techniques.
CTCs are isolated from blood samples by label dependent or label free detection. Imaging, flow cytometry and artificial intelligence are generally used for CTC enumeration. Molecular characterization is performed by methods such as immunostaining, Fluorescence in situ hybridization (FISH), sequencing, qRT-PCR, expression analysis and cell culture. Therefore, CTCs can be used for diagnosis, prognosis, treatment selection, therapy monitoring and to identify drug resistance mechanisms. These could then be utilized for patient stratification, design personalized medications and therefore help with cancer management.
Enumeration is typically performed using imaging systems or flow cytometry, while molecular characterization may involve immunostaining, fluorescence in situ hybridization (FISH), sequencing, qRT-PCR, and other expression analyses. These methods enable CTC-based applications in diagnosis, prognosis, treatment selection, therapy monitoring, and identification of resistance mechanisms. CTCs commonly express epithelial markers such as EpCAM and cytokeratins, while lacking CD45, a marker of white blood cells. Affinity-based isolation strategies frequently use antibodies conjugated to magnetic particles or microfluidic platforms to enable positive enrichment of CTCs or negative depletion of leukocytes. 46 However, heterogeneity in EpCAM expression and epithelial–mesenchymal transition EMT-associated downregulation of epithelial markers may limit detection sensitivity, particularly in aggressive ovarian and lung cancers.
Most studies in ovarian cancer have relied on EpCAM as a principal surface antigen for detection.47,48 EpCAM is often used in combination with additional epithelial markers, including CK8, CK18, CK19, HER2, and others to enhance specificity. 49 While these approaches improve epithelial CTC capture, they may underrepresent mesenchymal or hybrid phenotypes.
Advanced imaging flow cytometry platforms, such as ImageStream®, have enabled simultaneous phenotypic and transcriptional characterization of CTCs in ovarian cancer cohorts. 50 Similarly, studies in lung cancer have integrated CTC enumeration with molecular analyses to assess perioperative dynamics and treatment-related changes. 21 Emerging technologies including Raman spectroscopy and copy number instability scoring have also been explored, though further validation in larger cohorts is required. 20
Currently, CellSearch®, an EpCAM-dependent platform, remains the only FDA-approved system for CTC enumeration in selected malignancies. 50 However, no platform has yet achieved standardized validation for routine application in ovarian or lung cancers. Continued development of multimarker and EpCAM-independent strategies may improve sensitivity and broaden clinical applicability.
Table 3 provides an overview of emerging technologies currently used for the detection of CTCs in ovarian and lung cancers.
Current Liquid Biopsy Technologies Used for Diagnosis and Prognosis of Ovarian and Lung Cancers.

Table 4 summarizes the principal CTC detection technologies, highlighting their mechanisms, strengths, and clinical limitations.
Major CTC Detection Technologies.

Clinical Use and Translational Challenges
CTCs have emerged as a powerful liquid biopsy modality for real-time monitoring of tumor biology in ovarian and lung cancers. Numerous studies have demonstrated the clinical utility of CTC enumeration and molecular characterization for monitoring treatment response, predicting survival outcomes, and identifying actionable genomic alterations. 72
In ovarian cancer, where late-stage diagnosis and high recurrence rates remain critical challenges, CTCs represent a promising minimally invasive biomarker for disease monitoring and prognostic stratification 73 . Elevated CTC counts in ovarian cancer correlate strongly with platinum resistance, early relapse, and poor PFS, particularly in high grade serous tumors. 74
In lung cancer, liquid biopsy has already influenced clinical decision-making, particularly through cfDNA-based mutation profiling in non-small cell lung cancer (NSCLC). However, CTCs provide intact cellular material that enables comprehensive genomic and phenotypic analyses, facilitating assessment of tumor heterogeneity, metastatic potential, and mechanisms of therapeutic resistance.
73
CTC-based genotyping in lung cancer can reliably detect resistance mechanisms such as
Despite strong biological and prognostic validity, CTC implementation in routine oncology practice remains limited. Translation from biomarker association to demonstrated clinical utility requires prospective, biomarker-driven trials showing improved patient outcomes when therapeutic decisions are guided by CTC results.71,72 Unlike cfDNA assays, which have been incorporated into international NSCLC guidelines for molecular testing, CTC-based strategies lack standardized detection thresholds, harmonized analytical platforms, and randomized validation studies demonstrating outcome benefit.73,76
Establishing analytical validity (reproducibility, sensitivity), clinical validity (prognostic and predictive accuracy), and ultimately clinical utility (impact on treatment decisions and survival) is essential for regulatory qualification and reimbursement.72,77
There are several barriers limiting broader application. Biological challenges include CTC rarity, particularly in early-stage disease, and phenotypic plasticity driven by EMT, which can reduce detectability using EpCAM-dependent platforms.77,78
Technical challenges arise from heterogeneity among isolation technologies, resulting in variable sensitivity and limited cross-study reproducibility. Lack of standardized operating procedures and consensus reporting criteria further complicates inter-laboratory comparison. 77 Clinical challenges include absence of validated cut-off values for clinically meaningful CTC counts and limited integration into evidence-based treatment algorithms. Cost-effectiveness, assay harmonization, and qualification as companion diagnostics, also influence adoption into routine oncology workflows.72,76
Multi-Omics Profiling for Precision Stratification
Integrated multi-omics profiling has fundamentally reshaped molecular stratification in ovarian and lung cancers by linking genomic alterations to downstream functional signaling states. In HGSOC, proteogenomic analyses conducted by the Clinical Proteomic Tumor Analysis Consortium (CPTAC) demonstrated that DNA copy-number alterations and transcriptomic signatures often fail to accurately predict protein abundance and phosphoprotein activity, underscoring the necessity of protein-level interrogation for clinically actionable pathway assessment 79 . These studies identified biologically and therapeutically relevant signaling programs associated with homologous recombination deficiency, immune modulation, and metabolic rewiring, thereby refining patient stratification beyond genomic alterations alone.80,81
Similarly, in lung adenocarcinoma, CPTAC-driven multi-omics integration revealed proteomic and phosphoproteomic network states that extend beyond canonical genomic drivers such as
CTC-Derived Organoids: Functional Precision Oncology
While molecular profiling provides mechanistic insight, functional validation remains essential to demonstrate therapeutic relevance. CTC-derived organoids offer a minimally invasive platform for ex vivo drug testing and dynamic modeling of resistance evolution. Proof-of-concept was first demonstrated in small-cell lung cancer, where CTC-derived tumor models recapitulated genomic features and treatment responses. 83 Subsequent studies have confirmed the feasibility of organoid-based drug screening in lung cancer. 84
In ovarian cancer, tissue and ascites-derived organoids are more established, CTC-derived systems may enable longitudinal monitoring without repeated invasive biopsies. 85 Although challenges such as CTC rarity, selection bias during culture, and scalability remain significant, successful implementation could provide functional evidence of drug sensitivity, thereby strengthening the clinical utility of CTC-guided strategies
AI as the Integrative Translational Layer
The clinical deployment of multi-omics and functional platforms increasingly depends on artificial intelligence (AI)-driven analytical models capable of integrating high-dimensional molecular and phenotypic datasets into clinically actionable outputs.
In ovarian cancer, machine learning models integrating genomic, transcriptomic, and immune microenvironment features have demonstrated improved prediction of therapeutic response and survival outcomes compared with single-modality biomarkers. 84 Deep learning-based quantification of organoid phenotypes further enables scalable and objective drug-response profiling, converting complex imaging data into reproducible therapeutic metrics.
In lung cancer, AI frameworks integrating histopathology, molecular alterations, and clinical variables have shown enhanced predictive accuracy for immune checkpoint inhibitor response, outperforming PD-L1 expression alone. 86 These advances position AI as a critical integration layer, facilitating convergence between multi-omics profiling and functional tumor models, and accelerating their translation into deployable clinical decision-support systems for personalized cancer management.
Conclusion and Future Direction
CTCs represent a clinically meaningful, minimally invasive biomarker with significant potential to transform disease monitoring and therapeutic stratification in ovarian and lung cancers. Accumulating evidence supports their role in prognostication, dynamic treatment response assessment, and identification of actionable molecular alterations.31,73 Nevertheless, substantial methodological challenges, including biological heterogeneity, low CTC abundance, variability in isolation platforms, and lack of standardized clinical validation, continue to limit their routine implementation.
Future progress will depend on moving beyond enumeration toward integrative and functional frameworks. Single-cell multi-omics profiling of CTCs offers unprecedented resolution of intrapatient heterogeneity and metastatic evolution, 78 while proteogenomic network analyses in ovarian and lung cancers underscore the importance of pathway-level interrogation over single-gene biomarkers.79,82 Complementing molecular characterization, CTC-derived organoid systems provide functional validation of therapeutic vulnerabilities and resistance mechanisms, supporting personalized drug testing strategies. 83
Equally critical is the integration of AI-driven analytical models capable of synthesizing high-dimensional molecular and phenotypic datasets into clinically actionable predictions. 87 The convergence of CTC biology, multi-omics profiling, functional organoid platforms, and AI-based decision systems marks a paradigm shift from static biomarker assessment toward adaptive, real-time precision oncology. Embedding CTC analysis within a multi-analyte liquid biopsy framework, including ctDNA, extracellular vesicles, immune phenotyping, and CTC clusters, may further enhance predictive accuracy and longitudinal monitoring capacity. 88 Prospective, biomarker-driven clinical trials will be essential to validate composite CTC-based signatures and establish standardized clinical workflows. Finally, the true clinical power of CTCs will emerge not as standalone biomarkers, but as integral components of multidimensional, systems-level platforms that translate tumor biology into dynamic therapeutic intelligence for ovarian and lung cancer management.
Footnotes
Acknowledgements
Not applicable.
List of abbreviations
• CA125 –
• CYFRA 21-1 –
• BRAF –
• ERBB2 –
• KRAS –
• LGSCs –
• HGSCs –
• TP53 –
• BRCA1 –
• BRCA2 –
• CTNNB1 –
• PTEN – Phosphatase and Tensin Homolog
• TEPs – Tumor-Educated Platelets
• EpCAM – Epithelial Cell Adhesion Molecule
• PI3K –
• mTOR –
• MAPK –
• EMT –
• EGFR – Epidermal Growth Factor Receptor
• PIK3CA – Phosphatidylinositol-4,5-Bisphosphate 3-Kinase Catalytic Subunit Alpha
• ALK – Anaplastic Lymphoma Kinase
• CTCs – Circulating Tumor Cells
• EOC –
• aEOC –
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The information in this correspondence article is publicly accessible and not confidential in nature. Therefore, the data is available for use and not classified.
