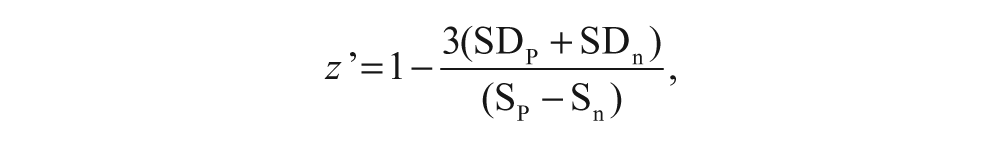Abstract
In this study, we have generated a high-throughput screening (HTS)–compatible 3D cell culture platform by chemically “welding” polystyrene scaffolds into standard 2D polystyrene 96-well plates. The variability of scaffolds was minimized by introducing automation into the fabrication process. The fabricated 3D cell culture plates were compared with several commercially available 3D cell culture platforms with light and scanning electron microscopy. Voltage-gated calcium channel functionality was used to access the Z′ factors of all plates, including a 2D standard plate control. It was found that with the No-Wash Fluo-4 calcium assay and neural progenitor cells, all plates display acceptable Z′ factors for use in HTS. The plates with “welded” polystyrene scaffolds have several advantages, such as being versatile and economical, and are ready to use off the shelf. These characteristics are especially desired in HTS preclinical drug discovery applications.
Keywords
Introduction
Traditionally, flat surface 2D-based cell cultures are widely used in preclinical drug discovery studies. However, recent studies have shown that 3D cell cultures provide more physiologically relevant results and should be the preferred choice for implementation in high-throughput screening (HTS) platforms. Currently, there are several commercially available, “ready-to-use” 3D cell culture systems for HTS. A fraction of these systems are based on natural or synthetic structures designed to provide some elements found in the in vivo extracellular matrix (ECM). 1 Examples include Extracel and HyStem hyaluronic acid–based hydrogels from Glycosan BioSystems (Alameda, CA), as well as HydroMatrix Peptide Hydrogel and MaxGel Human ECM, both marketed by Sigma-Aldrich (St. Louis, MO). Another fraction comprises structures that purely provide 3D microspaces. Examples include Micro-Space Cell Culture plate from Kuraray (Houston, TX), 3D Insert from 3D Biotek (Brunswick, NJ), and Algimatrix from Invitrogen. Also, a few systems are scaffold free, where other conditions (e.g., gravity) are provided to promote the formation of microtissues without a need for scaffolding. The most well-known example is the recently introduced Perfecta 3D hanging drop plates marketed by 3D Biomatrix (Ann Arbor, MI).
In comparison to standard polystyrene 2D multiwall plates, 3D plates can cost between 5- and 165-fold. At the same time, there is relentless pursuit to lower the screening cost per compound in the drug discovery HTS labs. In response to this need, we developed a patent-pending process to convert a standard 2D polystyrene plate into 3D plates, by chemically “welding” polystyrene scaffolds into standard 96-well plates 2 (hereafter referred to as porous polystyrene 3D plates). A similar idea has been independently developed by Knight et al. 3 and is being marketed as Alvetex (Durham, UK); however, no HTS-compatible products have been provided. The first porous polystyrene 3D plate prototypes in our lab revealed unacceptable well-to-well variability, and we have addressed this problem by using an automated fluid handling robot (Beckman-Coulter Biomek NP, Brea, CA) for key operations in the fabrication process. In this article, we have evaluated the performance of the polystyrene plates with a high K+ voltage-gated calcium channel (VGGC) gating assay with neural stem cells, and we have compared the performance with a 2D plate equivalent as well as a few commercially available plates. Although the results show a lower Z′ factor in comparison to 2D, the porous polystyrene 3D plate performance is comparable to that of the commercially available plates.
Materials and Methods
Materials and Reagents
Neural progenitor cells were obtained from the Regenerative Bioscience Center at the University of Georgia as well as Millipore (ENStem-ATM, Billerica, MA). Polystyrene, chloroform, ammonium bicarbonate, glutaraldehyde, and sodium cacodylate buffer were obtained from Sigma-Aldrich. Neural basal medium, penicillin/streptomycin, L-glutamine, recombinant human leukemia inhibitory factor (hLIF), basic fibroblast growth factor (bFGF), B-27 supplement, and phosphate buffered saline (PBS) were obtained from Gibco (Gaithersburg, MD). Fluo-4 No-Wash (NW) calcium assay kit and Algimatrix cell culture plates were obtained from Invitrogen (Carlsbad, CA). Celltreat 3D Insert-PS cell culture plates were purchased from Chemglass Life Science (Vineland, NJ). The Extracel Hydrogel Kit was obtained from Glycosan BioSystems.
Neural Progenitor Cell Culture
Human neural stem cells or neural progenitors (NPs) were maintained as described in Cheng et al. 2 The growth medium for NP cells was neural basal medium (Invitrogen) supplemented with penicillin/streptomycin, L-glutamine, recombinant hLIF, bFGF, and B-27. The medium was half changed every 48 h. For subculturing, 90% confluent cells were aspirated by pipetting, and the subculture ratio was typically 1:2 to 1:3. Before cell seeding, both the 3D porous polystyrene and Insert-PS Cell Culture Plates and 2D 96-well plates were coated with poly-ornithine and laminin to promote better cell adhesion. The subculture of NP cells in Algimatrix and Extracel Hydrogel was done according to the manufacturer’s instructions. Where no specific seeding density was required by the manufacturer, we seeded the plate at 10 000 cells per well.
Multiwell Plate Fabrication with Biomek NP
The scaffold fabrication process is illustrated in Figure 1 . Briefly, a viscous polymer solution was prepared by dissolving polystyrene (PS) in chloroform. Sieved ammonium bicarbonate particles in the range of 40 to 60 µm were added to the polymer solution and mixed thoroughly. The paste mixture of polymer/salt/solvent was poured into a reservoir. The reservoir was immediately covered to minimize the evaporation of chloroform. The cover had 96 holes for easy access by the Biomek NP controlled pipette tips. Because ammonium bicarbonate particles tend to sink to the bottom of the reservoir, the mixture solution was pipetted up and down three times before transferring to plates to improve the uniformity. Then, 15 to 30 µL of the mixture was transferred to each well of the 96-well plates, and the plates were shaken at 200 rpm for 20 s both counterclockwise and clockwise to ensure even distribution of the mixture. The plates were then left to air dry. The above procedures were carried out by Beckman Coulter’s robotics Biomek NP to improve efficiency and consistency. After chloroform was completely evaporated under atmospheric condition as well as by vacuum drying overnight, the plates were immersed into 37 °C water overnight to either dissolve or decompose ammonium bicarbonate to water and carbon dioxide, creating porous polymer scaffolds. After fabrication, the scaffolds were subjected to oxygen plasma treatment with the PLASMOD (Concord, CA) oxygen plasma system for 60 s to increase the hydrophilicity of the material.
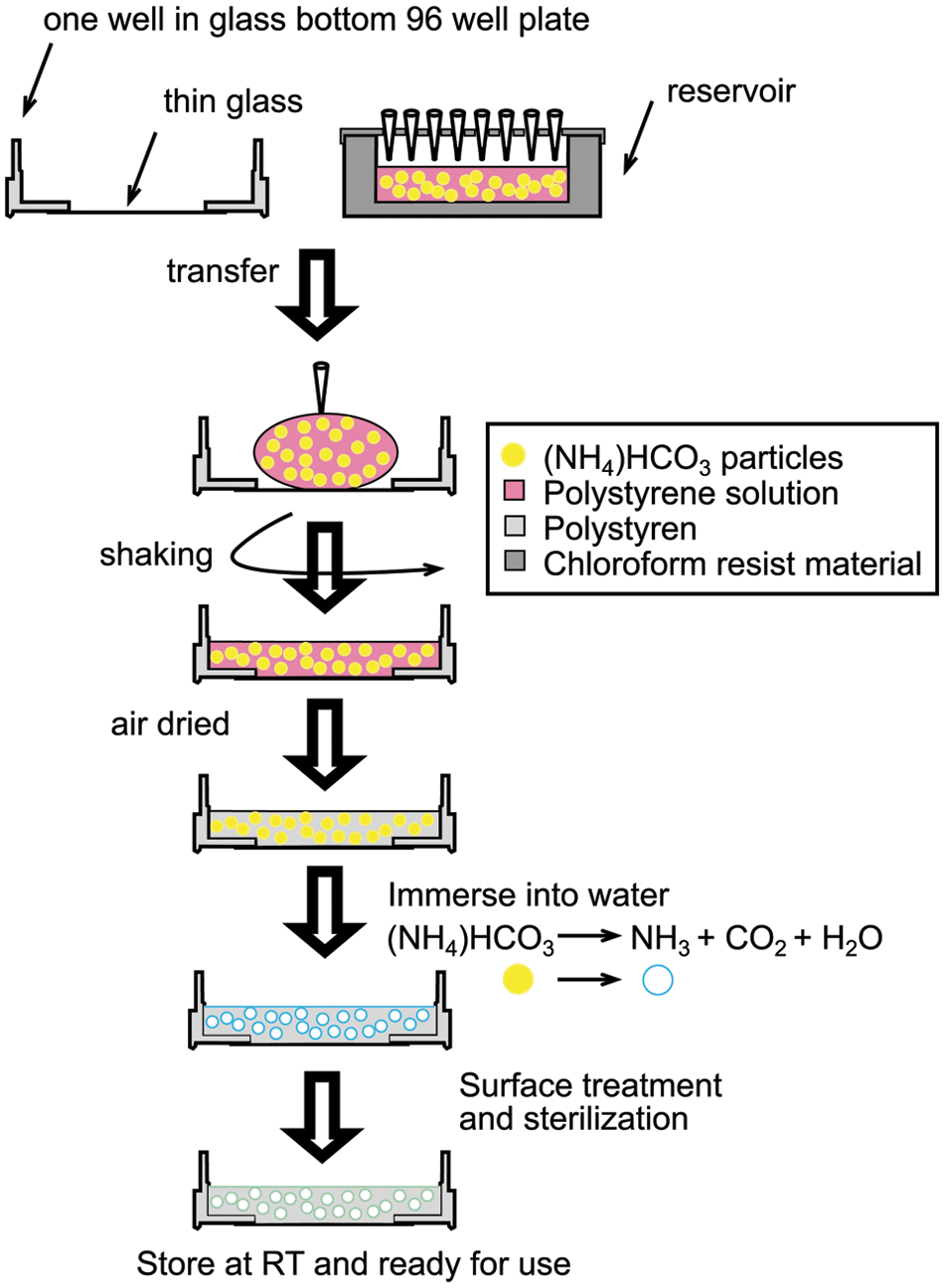
Fabrication of polystyrene 3D porous scaffold 96-well plates. Procedures up until air drying were accomplished with automation. RT, room temperature.
No-Wash (NW) Calcium Assay
In total, 10 mL NW Fluo-4 dye loading solution was prepared immediately before each experiment, according to the manufacturer’s protocol. The growth medium in the plates was removed and 100 µL of the dye-loading solution was quickly but carefully added into each well. The plates were then incubated for 45 min at 37 °C in a standard incubator. The fluorescence signals were monitored and recorded on the FlexStation (Molecular Devices, Sunnyvale, CA) every 3 s with an excitation wavelength of 485 nm and an emission wavelength of 538 nm. High potassium buffer was added to the plates at 34 s, and the calcium transient signals were recorded for 100 s.
The Z′ factor was calculated using the following formula:
where Sp is the signal recorded upon addition of high potassium buffer, Sn is the signal using normal buffer that serves as the negative control, and SD stands for standard deviation. Eight replicates were generated for each of these signals. The signal or “response magnitude” was calculated as (F – F0)/F0, where F is the peak value upon addition of high potassium buffer and F0 is the basal fluorescence signal value. For the negative control group, we chose the maximum fluorescence as F after buffer addition to calculate the response magnitude.
Scanning Electron Microscope Imaging
Cells in scaffolds were fixed with 2% glutaraldehyde in 0.1 M sodium cacodylate buffer (pH 7.2) for 1 h and then rinsed in cacodylate buffer three times (15 min each). The samples were then dehydrated in 35%, 50%, 70%, 80%, 95%, and 100% ethanol successively for 10 min each and dried in a SAMDRI-780A critical point drier (Tousimis Research Corporation, Rockville, MD). Scaffolds were sputter-coated with gold for 60 s to achieve a thickness of about 15.3 nm. Scanning electron microscope (SEM) images were captured with the LEO 982 scanning electron microscope (LEO Electronenmikroskopie GmbH Korporation, Oberkochen, Germany) with an acceleration voltage of 4 kV.
Results
Fabrication and Characterization
The fabrication process of the porous polystyrene 3D 96-well plates was modified from a process previously published by Cheng et al. 2 The Biomek NP liquid handler was used to implement automation into the fabrication process. Before use, the Biomek NP was calibrated according to the manufacturer’s instructions to ensure the best accuracy and precision. The fabrication process is illustrated in Figure 1 . The first part of the process was carried out by Biomek NP, and the second part was carried out manually outside of the Biomek NP. We first prepared a viscous polystyrene polymer solution by dissolving polystyrene in chloroform with ammonium bicarbonate particles. The ammonium bicarbonate particles were sieved to a size range of 40 to 60 µm. The paste mixture was poured into a Biomek NP compatible reservoir (Axygen, Union City, CA) so that 96 pipette tips could transfer the liquid from this reservoir simultaneously. The 96-well openings (5 mm in diameter) were drilled on the cover of the reservoir so that the evaporation of chloroform could be minimized without blocking the openings for easy access of the pipette tips. Then, 15 to 30 µL of the mixture was transferred to each well by the liquid handler and immediately followed by shaking with low speed to evenly spread the viscous mixture. The plates were covered to control the evaporation of the chloroform so it slightly dissolved the surface of the polystyrene in the plates to firmly fix or “weld” the scaffolds to the polystyrene plates. After chloroform was completely evaporated, the plates were collected and immersed into 37 °C water to ensure complete removal of the salt particles. The baking process described in Cheng et al. 2 was omitted for the 96-well plates because the adhesive used to glue the glass to the plates was sensitive to heat. The plates were sterilized by UV light in a biohazard laminar flow hood overnight. Although automation improved the consistency of the plates fabricated, as expected, in some wells, the scaffold was not well aligned with the bottom glass. Such wells were excluded from the follow-up studies. Further process improvements are needed to eliminate such wells from the plates. However, the plates are versatile as it is possible to change physical properties such as porosity, thickness, and light transmittance by altering the composition of the starting mixture, the amount of liquid, and even the material itself. In the rest of the experiments, we used salt/polymer weight of 15/1 (wt/wt) and 20 µL mixture for each well, which has been well tested in our previous studies. 2 Please note that this recipe can be changed according to the size of the cells studied or used.
Morphology of Different 3D 96-Well Plates
The NP cells used in this study are derived from human embryonic stem cells and are feeder free. These cells have been shown to be a good cell model to study the human neuronal cell functions.4,5 To test whether our plates are suitable for HTS and are comparable to the commercially available plates, we cultured NP cells on traditional 2D plates, our PS porous 3D plates, and three commercially available plates (Algimatrix, 3D Insert-PS cell culture plates, and Extracel Hydrogel Kit). Figure 2A – E shows the light microscopy images of NP cells cultured on 2D and 3D plates. It is clear that 2D cultured cells can be viewed individually by light microscopy easily, whereas in all the 3D plates, it is somewhat difficult to observe the detailed outlines of single cells. In Figure 2D , E , spheroids (identified by arrows) formed in the 3D culture vessels are visible. Because these spheroids were at different heights, we were not able to take one picture with all the spheroids in focus. For the Algimatrix, because there were bubbles released by the scaffolds after cell seeding, we were not able to see the spheroids very clearly either. According to the manufacturer’s manual, the bubbles disappear after several days of culturing. But for short-term culturing as in our study, the bubbles can be a problem for observation.
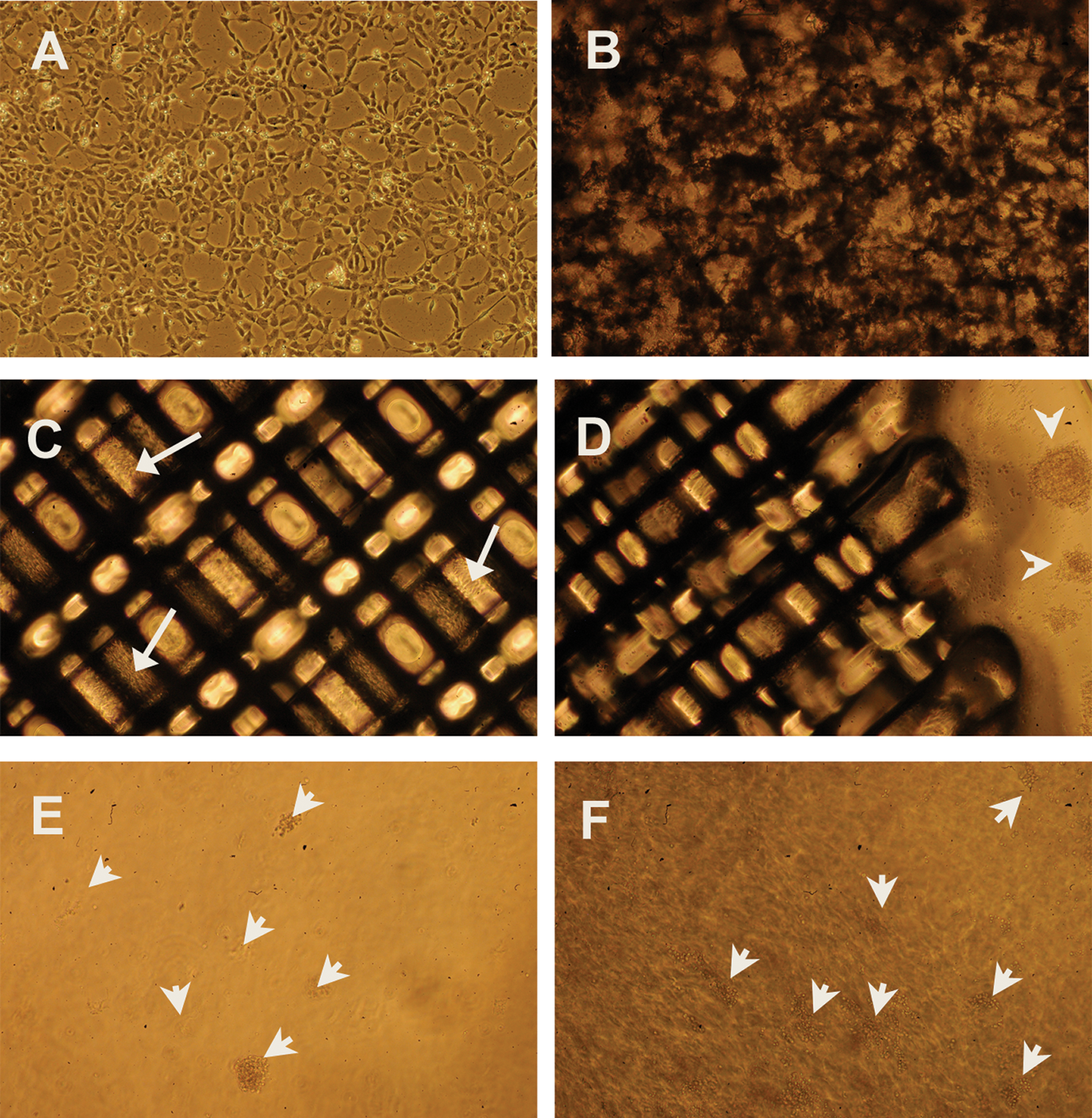
Light microscopy of neural progenitor (NP) cells cultured under different conditions. (
Our 3D porous polystyrene and the 3D Insert-PS cell culture plates were both not spheroid based, and the structure of the cells was further investigated by SEM, as shown in Figure 3A – I . Figure 3A – D shows NP cells on the porous polystyrene scaffolds that had been cultured for three days. A, B, and C are top views with different magnification. D is the side view showing cells that penetrated deep into the scaffolds. The cells did not form spheroids directly in our scaffolds. Instead, they scattered in the scaffolds with rounder shape and short neurites. After a longer period of culturing (e.g., 10 days), the cells formed spheroids in the cavities of the scaffolds ( Fig. 3E , F ). For the 3D Insert-PS cell culture plates, without coating of ECM proteins (e.g., laminin in our case), NP cells formed spheroids and were observed to loosely float in the culture medium and were easily washed away by pipetting. Thus, we coated the scaffolds with polyorithine and laminin as we did for the 2D polystyrene dishes. As shown in Figure 3G – I , NP cells attached to the polystyrene fibers, and most of the cells had a flattened shape just as they were on 2D surfaces.

Scanning electron microscope (SEM) images of selected 3D 96-well plates. (
NW Calcium Assay
We chose the calcium assay for this study because ion channels have emerged as an attractive drug discovery target class for modern drug discovery.6–8 Ion channels have been discovered to be associated with neuronal signal transduction, muscle contraction, T cell activation, cell proliferation, apoptosis, and other important cellular functions. Recent ion channel studies have expanded from targets for CNS and cardiovascular disorders to targets for other diseases such as cancer 9 and immune disorders. 10 Unlike some enzyme and receptor targets, which can be screened with high-throughput biochemical assays, ion channels rely heavily on cell-based assays for both primary and secondary screening. 11 Thus, the development of efficient, physiologically more relevant, HTS-compatible cell-based assays is essential for ion channel targeted drug discovery.
The cells were loaded with a membrane-permeable dye, NW Fluo-4, which has been specially designed for HTS application, with no wash steps. However, it still requires the complete removal of the culture medium. The intracellular calcium concentration was monitored by the fluorescence intensity changes with a FlexStation microplate reader for 100 s. At 34 s, a buffer with or without high potassium was added by the automatic liquid transferring function of the FlexStation. Typical time course fluorescence intensity changes are shown in Figure 4 . We observed that cells on the 2D surface had relatively higher peaks after the addition of high potassium than all the 3D plates tested. The cells cultured in 3D Insert-PS cell culture plates had relative higher peaks compared with the cells cultured on other 3D plates. The rest had comparable peak values. The Z′ factor values calculated are listed in Table 1 . Again, 2D plates had a higher Z′ factor than all the 3D plates. Our porous scaffolds yielded similar Z′ factor values as Invitrogen’s Algimatrix and Glycosan’s Extracel Hydrogel.
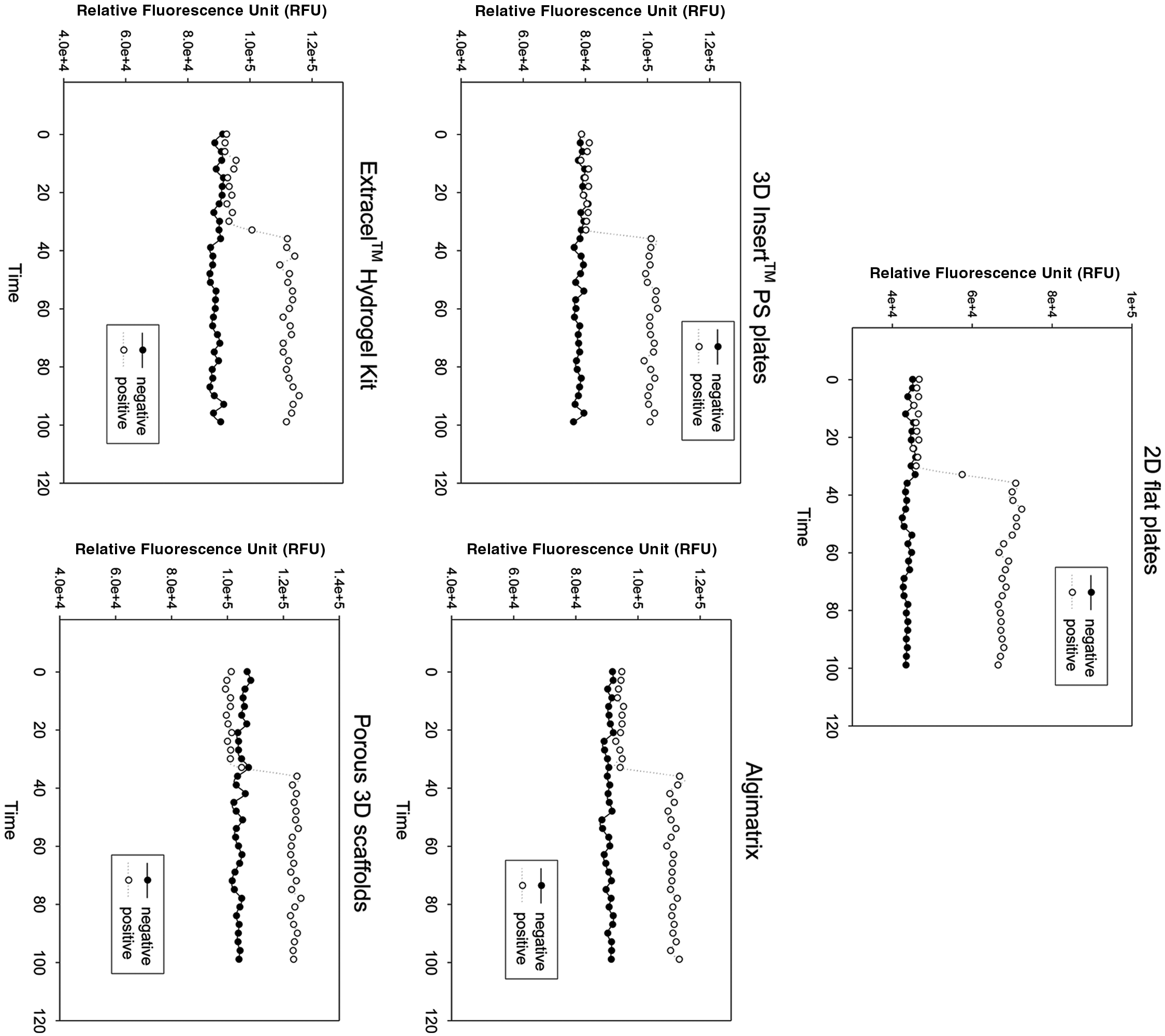
Representative No-Wash (NW) Fluo-4 fluorescence time course measured with the FlexStation.
Z′ Factors for the No-Wash (NW) Calcium Assay
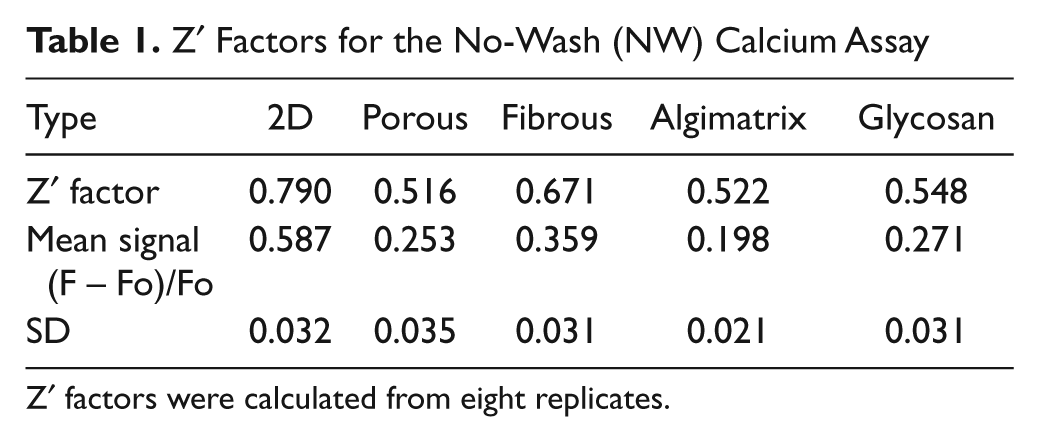
Z′ factors were calculated from eight replicates.
Discussion
Although the necessity to use a more complex in vitro model (e.g., 3D cultured cells) for drug discovery has been demonstrated in the past decade, it is not easy to implement the 3D cell cultures in practical HTS applications. The Z′ factor, after being developed by Zhang et al., 12 has become a popular tool in HTS assay development for comparison and evaluation of the quality of assays. A useful HTS assay should have a Z′ factor score between 0.5 and 1.0. As observed in our study, 2D plates have an excellent Z′ factor of 0.790, which is close to the reported NW Fluo-4 calcium assay’s Z′ factor (0.842 with Jurkat cells and 0.827 with HEK293 cells) by Invitrogen. 13 The differences between our 2D assay’s Z′ factor and the company’s reported ones may result from the use of different cells, stimulant drugs, and instrument. All the 3D plates have lower but acceptable Z′ factors, with 3D Insert-PS cell culture plates having the highest score of 0.671. As described under Material and Methods, to get a higher Z′ factor, the assay should have an overall low well-to-well variability (to lower the standard deviation) and high difference between positive and negative response magnitudes (to increase the (Sp – Sn)). As shown in Table 1 , all assays have a small standard deviation and near-zero negative control’s response magnitude, which indicates that the main difference in Z′ factors is due to the positive response magnitude. We have previously reported that 2D cells have exaggerated the VGCC calcium transient signal when compared with 3D cells. 2 Exaggerated VGCC calcium transient signal generates high positive response magnitude, which may be why 2D has a better Z′ factor compared with all the 3D plates. The exaggerated cellular responses may then place a high confidence in the assay, as reflected by its high Z′ factor, which may not be warranted from the in vivo physiology context.
Among the four 3D plates, Insert-PS cell culture plates exhibited a better Z′ factor of 0.671 and a high average positive response magnitude. As shown in Figure 3G – I , the cells cultured on Insert-PS cell culture plates attached to the surface of polystyrene fibers and had a morphology similar to 2D cultured cells. The reason for the better Z′ factor score for Insert-PS cell culture plates may result from the fact that the cells on this scaffold were actually a mixture of 2D and 3D cells. In our hands, the 3D Insert-PS plates were not “characteristically” 3D after coating with ECM proteins. According to the manual, the plates are normally used without coating. However, as the application of stem cells in drug discovery grows, it is relatively difficult to use a culture vessel without introducing the ECM proteins. Stem cells are much more difficult to adhere to noncoated surfaces, and they would more likely stick to each other and float in the medium than stay in the openings of the scaffolds, especially when forces are applied to them, such as in washing steps or buffer changing steps. We observed floating cell spheroids around the 3D insert when no coating was introduced. When the plates were coated with laminin, cells attached to the fibers and formed flat morphology similar to that of 2D flat surfaces. There may have been spheroids entrapped in between fibers because we observed increased cell spheroid numbers around the insert after medium change. But after so many wash steps required in SEM sample preparation, we were not able to observe the spheroids in SEM pictures.
The porous polystyrene scaffold plate generated similar Z′ factors as Algimatrix by Invitrogen and Extracel Hydrogel by Glycosan. However, the porous polystyrene scaffold plate offers several advantages. The key feature comparisons of the tested plates are summarized in Table 2 . First, our plates are versatile. We can adjust the parameter (e.g., pore size, porosity) easily during fabrication to tailor to different applications. We can use the plates with or without coating of ECM proteins, which makes introducing 3D chemical cues on the surface easy. We can also seed one plate partially and still be able to use the rest of the plate later. Algimatrix plates are very sensitive to moisture. If moisture is introduced before cell seeding, the morphology of the scaffold will change and cell seeding will be more difficult. Thus, Algimatrix plates cannot be coated with ECM proteins, and the whole plate has to be used at one time. Second, our plate is easy to store and transport. Because the main component of our plates is polystyrene, just like the regular 2D plates, they can be treated as those polystyrene 2D plates and stored at room temperature. The other plates more or less require some special treatment during storage and transportation (e.g., Algimatrix plates have to be packaged with desiccant and Extracel Hydrogel requires low temperature). Last but not the least, our plates are expected to be very economical. With the major raw material being polystyrene, sodium bicarbonate, and chloroform, the cost to produce our plates can be kept to the minimum.
Feature Comparisons of the Tested 3D Cell Culture Platforms
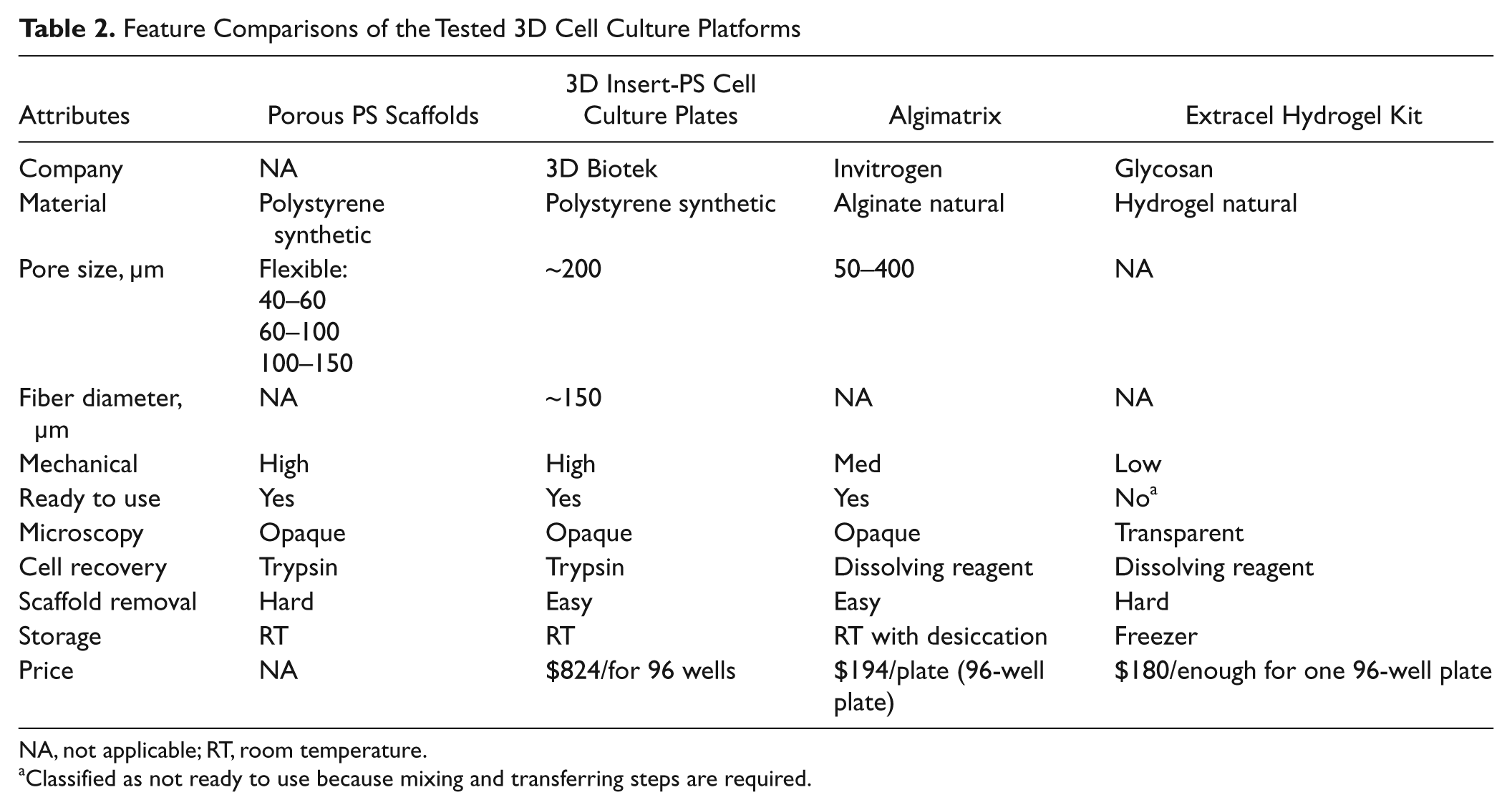
NA, not applicable; RT, room temperature.
Classified as not ready to use because mixing and transferring steps are required.
In conclusion, cells cultured on 2D surfaces have exaggerated calcium signals compared with those in 3D culture systems. It may be better to use the more physiologically relevant 3D culture-based assay to more effectively eliminate false-positive hits and improve the overall yield from drug screening campaigns. Compared with the commercially available 3D plates tested, our plate exhibited a similar Z′ factor with several advantages: (1) The properties of the scaffolds can be easily adjusted in the product fabrication process, (2) it can accommodate any ECM coating, (3) it has potential to be relatively more economical—polystyrene is not expensive, and (4) it is ready to use off the shelf and as such easier to store.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.