Abstract
Conventional enzyme-linked immunosorbent assay (ELISA) is a gold standard for screening antibodies and testing for protein or antigen presence. A significant limitation of this assay resides in the fact that only one analyte can be assessed per microplate well. Here, we describe and investigate a new technology consisting of an automated ELISA system in which up to 10 analytes can be measured within one single well, thus improving productivity, accuracy, and repeatability by reducing the amount of human labor required. Another strength of the platform is that a user can load any necessary sets/subsets of beads to perform required assays, with improved flexibility compared to manufactured-loaded arrays for multiplex analysis. We also demonstrate that this system can be used to determine the pathogenicity (i.e., presence of Shiga toxins) and serotype (i.e.,
Conventional enzyme-linked immunosorbent assay (ELISA) is a gold standard for screening antibodies and testing for protein or antigen presence. A significant limitation of this assay resides in the fact that only one analyte can be assessed per microplate well. Here, we describe and investigate a new technology consisting of an automated ELISA system in which up to 10 analytes can be measured within one single well, thus improving productivity, accuracy, and repeatability by reducing the amount of human labor required. Another strength of the platform is that a user can load any necessary sets/subsets of beads to perform required assays, with improved flexibility compared to manufactured-loaded arrays for multiplex analysis. We also demonstrate that this system can be used to determine the pathogenicity (i.e., presence of Shiga toxins) and serotype (i.e.,
Plexarray Format
The system used is based on a novel multiplexing format (U.S. patent pending) developed by Dynex Technologies, Inc. (Chantilly, VA). Undyed polystyrene beads (2 mm diameter) were first coated/treated to enhance adsorption and then stored dry. To build the assay, we passively adsorbed capture antibodies onto the beads (see Immunoassay section below). Then the beads were pressed into cylindrical pockets in the bottom of a custom well to fit into a microplate-sized holder ( Fig. 1 ). This special well-plate was manufactured from black polypropylene and fitted into a rectangular 6 × 9 array in a standard microplate-sized holder ( Fig. 2 ). The construction allowed the rigid polystyrene beads to be forced tightly into the narrower pockets, distending them slightly to form a leak-proof seal. The colorless beads provided a good medium for luminescence, whereas the black color of the plates reduced reflectance and cross-talk between beads sharing the same well. Ten beads were fitted into the bottom of each well. Although the format allows for multiplex assay of 10 different analytes, in this proof-of-principle work, only three beads were loaded with capture antibody in each well ( Fig. 3 ). Each of these three beads was coated with a different antibody. The other seven positions were filled with negative control beads coated with an inert blocker. The identity of the bead coating, and thus the specificity of the assay for each bead, was determined by its physical position, as detailed below.

Cutaway close-up of a single well with beads.

Image of the plate used in the new Dynex system.
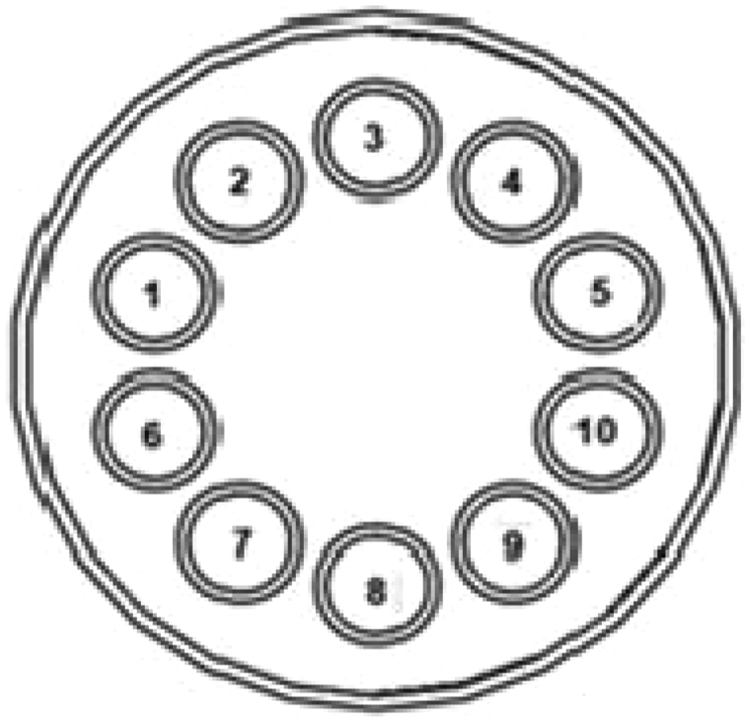
Diagram showing the physical position of the beads used in the current 3-Plex immunoassay. Beads in positions 1, 4, and 9 were coated with anti-Stx1, Stx2, or O157 antibodies, respectively. The remaining beads in positions 2, 3, 5, 6, 7, 8, and 10 were blocked with StabilGuard (SurModics, Eden Prairie, MN).
Liquid Handler
Operation of the assay was greatly facilitated through the use of a liquid handler robot. The Dynex DS-2 Automated ELISA Processing System workstation was used ( Fig. 4 ), which includes all necessary hardware and software for automated sample dilution, reagent addition, temperature-controlled incubation, shaking, and washing of microplate wells. Since 2007, this instrument has been used in clinical diagnostics and other applications, where it is widely used for routine ELISA assays using standard microplate format (not beads). After loading the wash and reagent reservoirs on the DS-2, up to two assay microplates containing beads were placed on the instrument. The instrument was then controlled by computer through a USB serial communication protocol using DS-Matrix software. After the ELISA reaction was completed, the plate was manually removed and mounted into the CCD (charge-coupled device) camera reader.
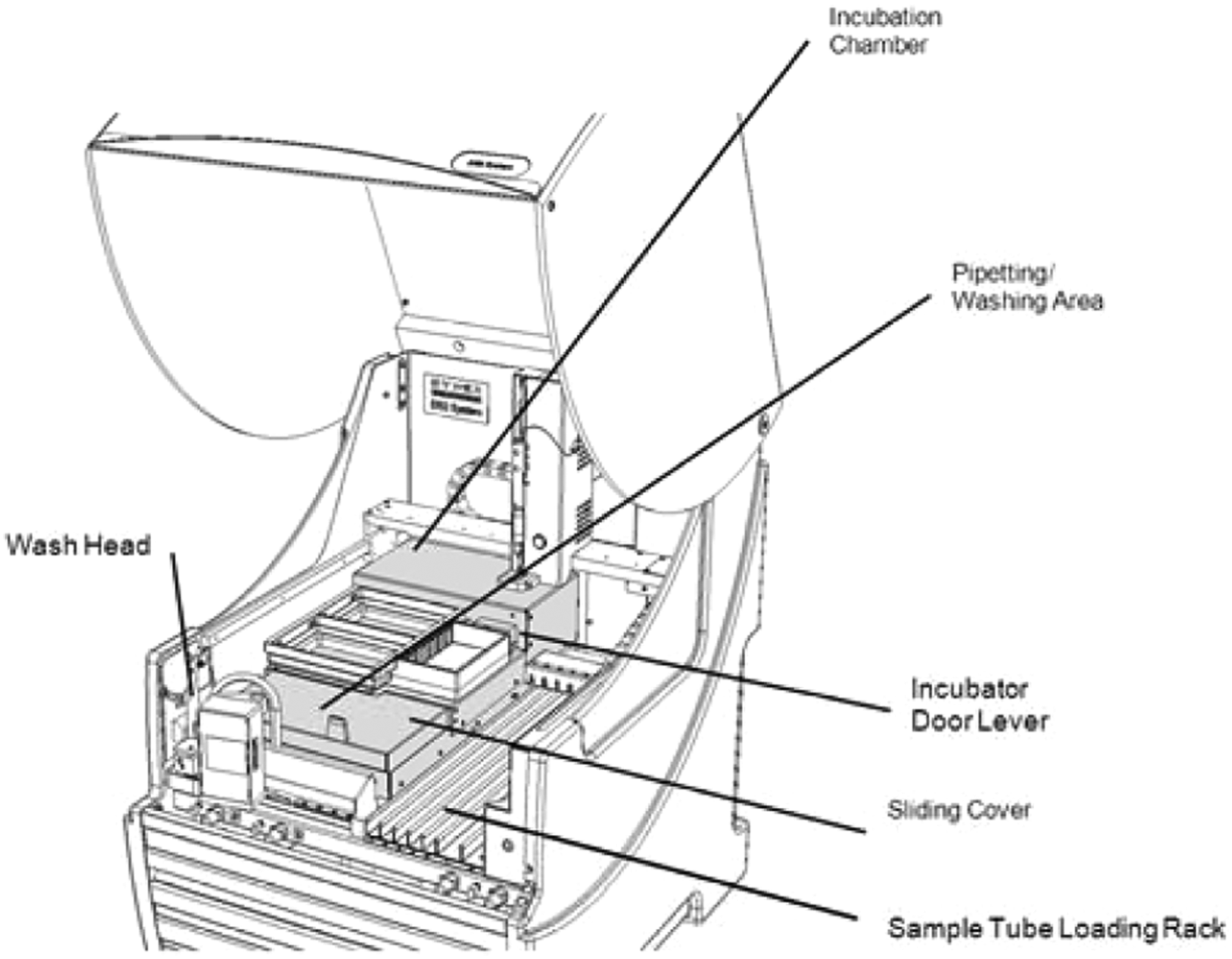
DS-2 Automated ELISA Processing System.
CCD Camera
For chemiluminescent readout of the multiplex assay format, we used a Photometrics (Tucson, AZ) CoolSNAP HQ2 CCD camera ( Fig. 5 ). The camera was mounted within an enclosure designed to exclude extraneous light and maintain proper focus length for detection of light emanating from beads within the microplate. A light-tight door near the bottom of the enclosure allowed insertion of microplates. Microplates were loaded and unloaded manually, one at a time. Images were captured across the entire plate simultaneously, comprising chemiluminescent emission directly related to the presence of analyte. The images were then digitized via Array-Pro Analyzer Software (Media Cybernetics, Bethesda, MD), and the reduction of the raw intensity data was carried out with Microsoft Excel (Microsoft, Redmond, WA).

CCD (charge-coupled device) and camera enclosure.
Macroplexing Beads
The coating of Dynex macroplexing beads is similar to coating a polystyrene microplate with antibodies. In this study, the beads were coated with anti-Stx1 (Shiga toxin 1; Sifin GmbH, Berlin, Germany), anti-Stx2 (Shiga toxin 2; Sifin GmbH), or anti–
Bacterial Preparation
A total of 38
Bacterial Strains Used
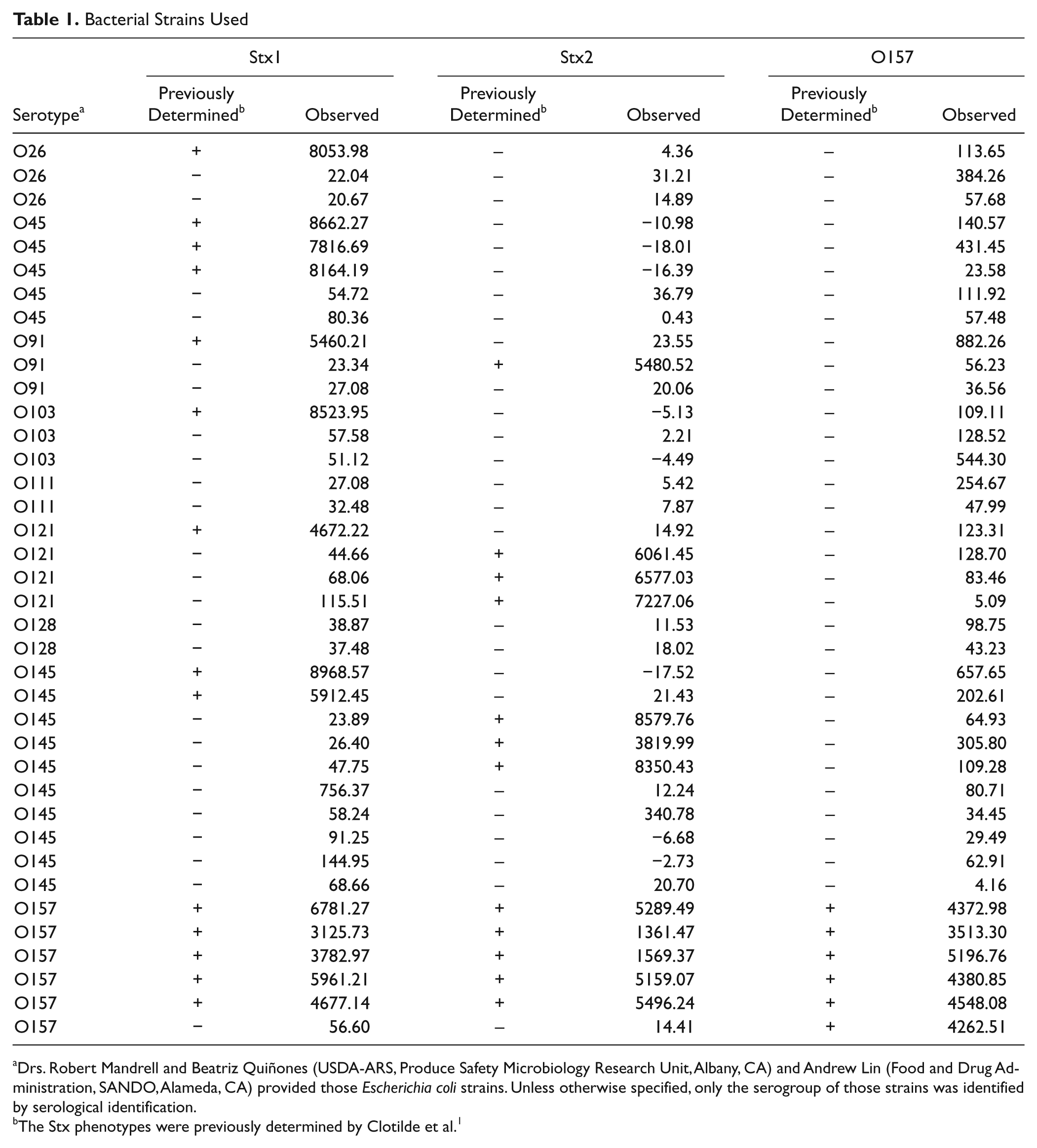
Drs. Robert Mandrell and Beatriz Quiñones (USDA-ARS, Produce Safety Microbiology Research Unit, Albany, CA) and Andrew Lin (Food and Drug Administration, SANDO, Alameda, CA) provided those
The Stx phenotypes were previously determined by Clotilde et al. 1
Immunoassay
Tween-20 was purchased from Sigma Chemicals Co. (St Louis, MO), PBS from Fisher Scientific (Pittsburgh, PA), IgG-free/protease-free bovine serum albumin (BSA) from Jackson ImmunoResearch (West Grove, PA), and streptavidin-horseradish peroxidase (SA-HRP) conjugate from Invitrogen (Carlsbad, CA). The bead-loaded plates were blocked with 500 µL/well of StabilGuard for 30 min at 20 °C with constant shaking. After five washes with 700 µL/well of PBS-T, 300 µL/well of diluted bacterial media was added to the plate and incubated for 30 min at 20 °C with constant shaking. After five washes with 700 µL/well of PBS-T, 300 µL/well of a 0.25-µg/mL solution in StabilGuard of detector mAbs was added to each well and incubated for 30 min at 20 °C with constant shaking. In this assay, the same anti-Stx1 and anti–
Results and Discussion
The specificity of the mAbs used has been tested previously by microplate sandwich ELISA and microbead-based immunoassay.
1
Our multiplex assay is a qualitative assay constructed around antibody-based detection of the bacterial biomarker antigens O157 LPS, Stx1, and Stx2. With the Dynex macrobeads, as with the previous assay formats, none of the Stx mAbs cross-reacted with any of the 17 non-Stx producing
Here we described a new automated platform capable of running an immunoassay testing for the expression Stx as well as the presence of
Our newly developed bead-based immunoassay is capable of detecting Stx1, Stx2, and
Footnotes
Acknowledgements
We thank Drs. Robert Mandrell and Beatriz Quiñones (USDA-ARS, Produce Safety Microbiology Research Unit, Albany, CA) and Andrew Lin (FDA, SANDO, Alameda, CA) for providing STEC strains. We also thank Dr. Julian Duncan (London, UK) for his help in data analysis and assay optimization.
Declaration of Conflicting Interests
Coauthors DES, AK, and AF are employees of the Dynex Technologies, currently developing the assay platform described herein as a commercial product. Their role in this work was limited to design and construction of the instrument. Coauthors LMC, CB, and JMC developed and performed the assay and declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Use of trade names and commercial products in this article is solely for the purpose of providing scientific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity employer.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
