Abstract
Introduction
Geriatric hip fractures are one of the most common trauma cases requiring operative treatment. 1 Minimising blood loss during the operation is a challenge. The reported average operative blood loss for cemented hip hemiarthroplasties is approximately 500 mL. 2 Blood loss can originate from the fracture, during surgery, and concealed in the surgical site. 3 Post-operative anaemia and associated blood transfusion can lead to local and systemic complications, resulting in poor functional recovery and increased mortality. 4
Tranexamic acid (TXA) is an anti-fibrinolytic agent that competitively inhibits the fibrinolysis cascade by binding to plasminogen, allowing the formation of stable blood clots for haemostasis in injured tissue. The half-life of TXA is approximately 120 min and is excreted by the kidney. 5 There have been many studies on the use of intravenous (IV) TXA in geriatric fracture hips, with consistent evidence showing favourable clinical outcomes.6–9
Despite the evidence, a significant group of patients are relatively contraindicated for IV use of TXA due to its thromboembolic potential. Recent studies comparing topical and IV TXA use in total hip replacement have shown similar effects in reducing blood loss without an increased risk of complications.6,7 Yet, patients undergoing hip fracture surgery are typically older and have more medical comorbidities. Considerable differences in patient characteristics have raised concerns about the heightened risk of TXA use, particularly related to thromboembolic event. Until now, the utilisation of topical TXA in the treatment of neck of femur fractures (NOF) has remained unstudied in Hong Kong. Here, we aim to evaluate the effect of topical TXA on post-operative transfusion rate and haemoglobin (Hb) level in patients of Hong Kong following geriatric hip fractures requiring operative treatment.
Materials and methods
Design and setting
This study is a single-centre, retrospective cohort study conducted between June 2020 and June 2023 in a tertiary centre in Hong Kong. The trial was registered under the regulation set by the Hospital Authority Clinical Research Ethics Review (HA CRER) committee.
Inclusion and exclusion criteria
Patients over 65 years old with hip fractures, including intertrochanteric and NOF requiring surgery, and those who are also contraindicated to receiving IV TXA (Table 3), were screened for eligibility. Patients were excluded if they met any of the following exclusion criteria: hypersensitivity to TXA, pathological fracture, periprosthetic fracture or multiple fractures. Patient with serum creatinine <500 μmol/L were allocated to the case group receiving topical TXA, and otherwise to the control group. All patients provided informed consent. For mentally incompetent patients, consent was obtained from two medical officers after explaining to the patient's relative.
Using the sample size calculator 10 with a type-I error rate of 0.05, 80% study power, a 1-to-2 case-to-control ratio, 11 and our hypothesised 15% reduction in Hb mass drop with the use of topical TXA based on Jia, 6 an estimated 132 patients were required for the study.
Interventions
Patients with intertrochanteric hip fractures were treated with cephalo-medullary device, either proximal femoral nail anti-rotation or Gamma 3 nailing system. Neck of femur fractures were treated with hemiarthroplasty. All operations were either performed or supervised by senior surgeons with at least four years of experience. Intraoperative blood salvage was not used. Tranexamic acid dosage adjustment was titrated against the patient's creatinine level accordingly (Table 1). The calculated TXA powder was dissolved into 50 mL of 0.9% normal saline by the scrub nurse. The wound was closed in layers to achieve a watertight seal. For cephalon-medullary device cases, the solution was instilled subfascially via a blunt tip needle. For hemiarthroplasty cases, the solution was instilled through a drain placed subfascially. The drain was clamped for 30 min post-operatively.
Topical tranexamic acid (TXA) dosage adjustment.
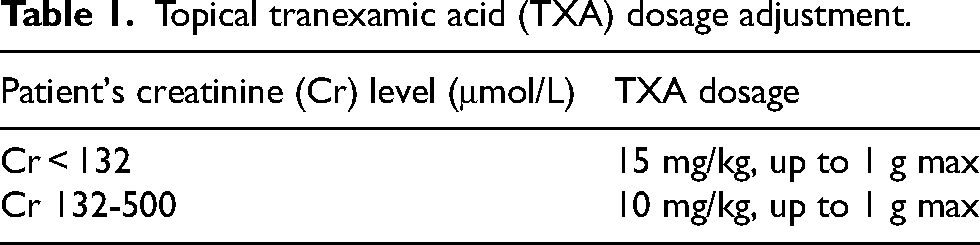
Before surgery, all patients receive mechanical venous thromboprophylaxis with a sequential compression device. For patients whose surgery was performed more than 48 h after admission, 1 mg/kg enoxaparin was administered. All clexane were withheld 24 h before operation. An antibiotic, either cefazolin or vancomycin, was given on induction according to the patient's weight. Haemoglobin levels were measured pre-operatively, on post-operative day (POD 0), and on POD 1.
Patients were stratified into four treatment groups based on their fracture type and subsequent treatment type, as well as if topical TXA was used. Patients with NOF were treated with hemiarthroplasty and were grouped into either group A or B if they had or had not administered topical TXA, respectively. Patients with basal NOF (bNOF) and intertrochanteric fracture (TOF) were treated with intramedullary devices and were grouped into either group C or D if they had or had not administered topical TXA, respectively.
Outcome and data collection
Data were collected by fragility nurses and the principal investigator. The baseline characteristics of patients were recorded, including age, gender, weight (kg), length of stay in hospital, creatinine clearance (eGFR), American Society of Anaesthesiology (ASA) score, use of enoxaparin, type of fracture, type and duration of operation, pre-operative and post-operative Hb level, clotting profile, platelet counts and estimated intravascular blood volume. 12 Patients with eGFR value over 90 were recorded to have 90. Subjects were labelled using the hospital-assigned person-specific medical record ID number (MRN). The MRN was used to ensure no repeat recruitment of subjects without the need to keep a record of any personal identifier, for example, HKID.
After surgery, all patients were monitored according to the standard monitoring and rehabilitation protocol outlined in the hospital fracture hip pathway. There were three primary outcomes: (a) total Hb mass loss, (b) the incidence of patients requiring transfusion and (c) the occurrence of thromboembolic event and mortality up to one month. Haemoglobin levels were measured pre-operatively, on POD 0, on POD 1, and after each transfusion of red blood cells. The total blood loss was calculated using the formula by Jaramillo et al. 13 , 14 (Figure 1). The incidence of patients required blood transfusion of at least 1 unit of packed red blood cell from the start of surgery till post-operative was recorded. The transfusion trigger point for asymptomatic and symptomatic patients was defined as when the patient's Hb level dropped below 8 g/dL and 10 g/dL, respectively. The transfusion trigger point for the high-risk patients, including those with acute coronary syndrome, severe left ventricle dysfunction or chronic respiratory failure, was when the patient's Hb level dropped below ≤10 g/dL. Thromboembolic events included deep vein thrombosis (confirmed by duplex ultrasound examination), pulmonary embolism (confirmed with computed tomography) and mortality within 30 days post-operation. Mortality was reviewed and confirmed using the electronic patient record database.

Patient's intravascular blood volume (mHBEBL) estimated based on the percentage difference of haemoglobin level before (Hb preop) and after operation (Hbnadir) multiplied by total blood volume (BV).
Statistical analysis
All summary statistics were generated using STATA IC/16 (StataCorp, 2019). 15 Based on the normality of data determined by the Shapiro–Wilk test, summary statistics were summarised and presented as either mean or median with an interquartile range (IQR).
Boxplots were used to illustrate the distributions of the described statistics. Student t-test was used to compare the defined statistics that were normally distributed. Kruskal–Wallis equality-of-populations rank test followed by Dunn's post hoc test were used to compare the defined statistics that were not normally distributed. P-values less than or equal to 0.05 were considered statistically significant.
Results
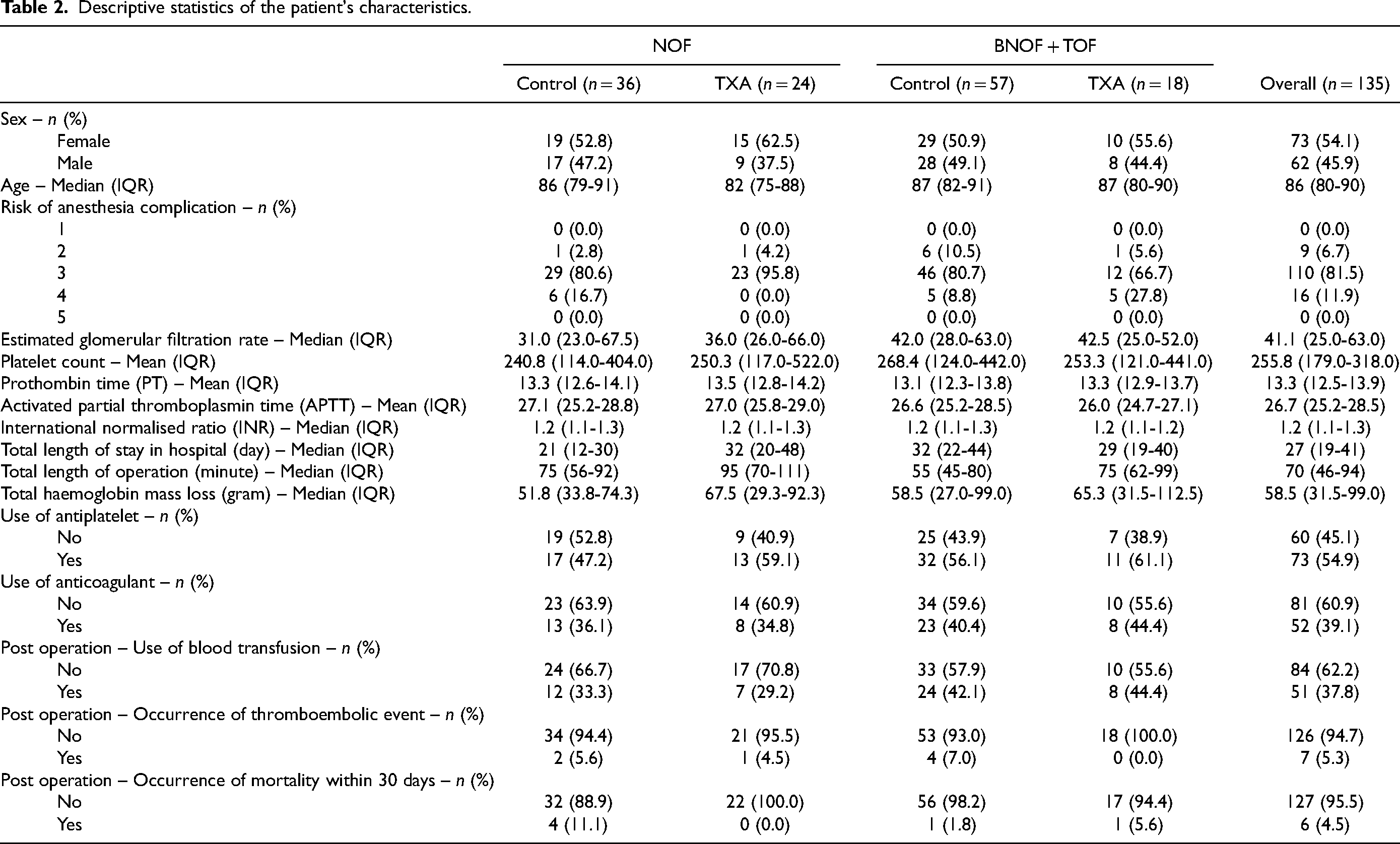
From June 2020 to June 2023, a total of 141 patients were eligible for the study, six of whom were excluded from the analysis due to missing data. The median age of the patients was 86 years old (IQR: 80–90), with 73 (54%) patients being male (Table 2). A total of 42 patients received topical TXA, with 24 in group A and 18 in group C (Table 2). The remaining 93 patients did not receive medication, with 36 in group B and 57 in group D (Table 2). There was a significantly longer operative time in the intervention group in both the NOF (p = 0.05) and bNOF + TOF subgroups (p = 0.03) when compared to the control group. No statistically significant differences were observed between the patient's groups regarding their age, gender, ASA grade, pre-operative clotting profile, platelet count and length of hospital stay.
Descriptive statistics of the patient's characteristics.
Contraindications to the use of intravenous tranexamic acid under department protocol.

The median Hb mass loss in patients ranged between 51.8 and 67.5 (Table 2 and Figure 2). Within the groups of patients that were diagnosed with NOF fracture, there were no significant differences in the total Hb mass loss whether TXA was used (p = 0.3531). Similarly, within the groups of patients diagnosed with bNOF + TOF fracture, there were also no significant differences in the total Hb mass loss whether TXA were used (p = 0.7005).
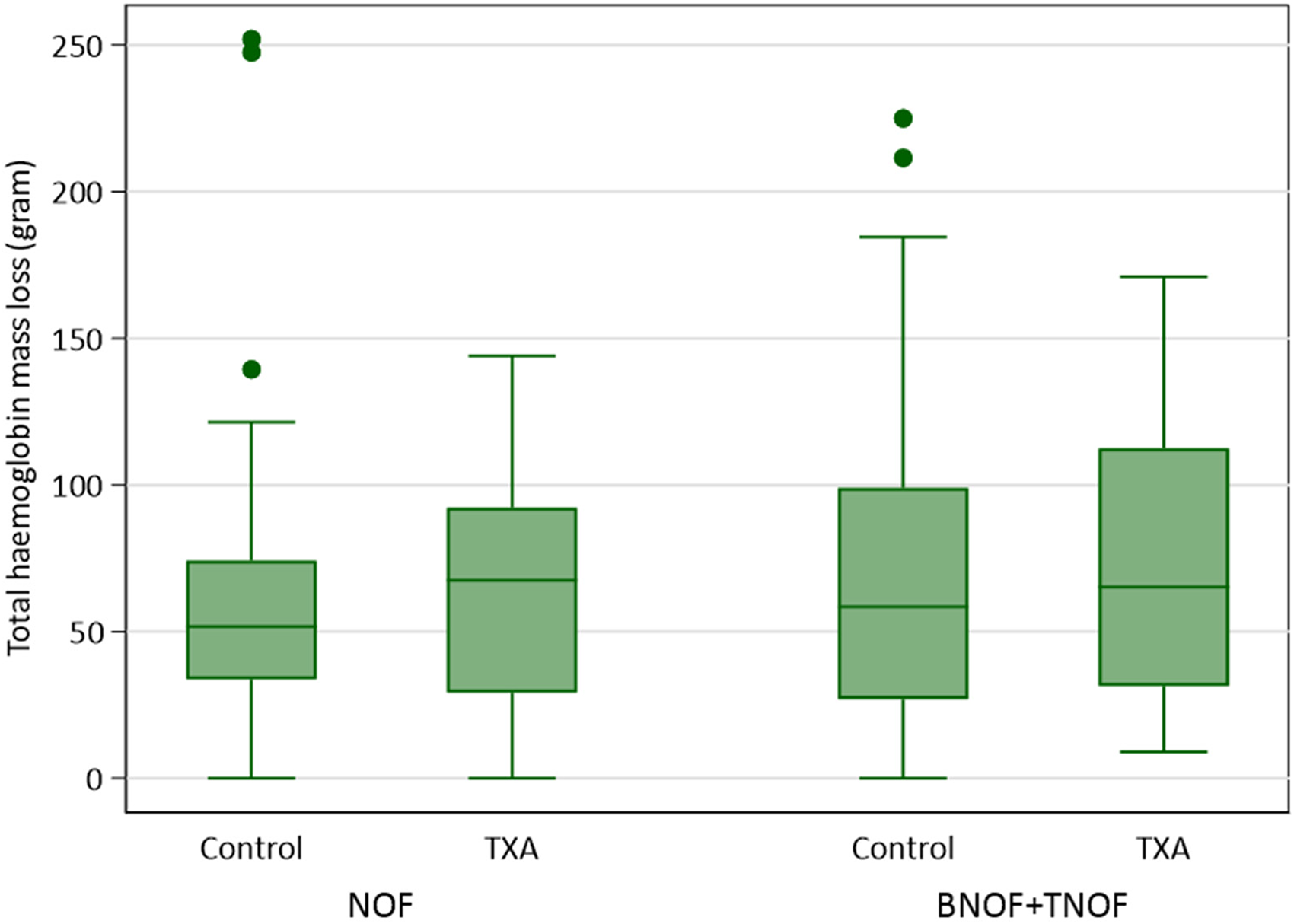
Boxplots of the total haemoglobin mass loss (gram) in patients post operation between June 2020 and June 2023, stratified by the treatment group of TXA and diagnosis.
Blood products were transfused to seven patients (29.2%) from group A and 12 (33.3%) from group B. Blood products were transfused to eight patients (44.4%) from group C and 24 (42.1%) in group D. Within the groups of patients that were diagnosed with NOF fracture, there was no evidence of an association between the use of TXA and the use of blood transfusion post operation (p = 0.734) with an OR of 0.82 (95% confidence interval [CI]: 0.27–2.52). Within the groups of patients that were diagnosed with bNOF + TOF fracture, there was also no evidence of an association between the use of TXA and the post-operative blood transfusion (z = 0.17, p = 0.861) with an OR of 1.1 (95% CI: 0.38–3.20).
A small number of thromboembolic events were recorded in groups A, B and D (Table 2). No thromboembolic event occurred in patients from group C (Table 2). Within the groups of patients that were diagnosed with NOF fracture, there was no evidence of an association between the use of TXA and the occurrence of thromboembolic event post operation (p = 0.866) with an OR of 0.81 (95% CI: 0.07–9.49). Within the groups of patients diagnosed with bNOF + TOF fracture, there was also no evidence of an association between the use of TXA and the occurrence of thromboembolic event post operation (p = 1) with an OR of 1. One patient from group B with deep vein thrombosis was complicated with pulmonary embolism confirmed with computed tomography pulmonary angiography.
There were a total of six patients who passed away within the 30-day post-operative period, with four from group B (11.1%), one from group C (1.8%) and one from group D (5.6%) (Table 2). Within the groups of patients diagnosed with NOF fracture, there was no evidence of an association between the use of TXA and the occurrence of mortality within 30 days post-operation (p = 1) with an OR of 1. Within the groups of patients diagnosed with BNOF + TOF, there was also no evidence of an association between the use of TXA and the occurrence of mortality within 30 days post-operation (p = 0.408).
Discussion and conclusion
Due to its anti-fibrinolytic properties, topical TXA has gained attention as a potential adjunct to hip fracture surgery. To the best of our knowledge, this is the first study conducted in Hong Kong on the use of topical TXA for NOF. In our study, topical TXA was not found to reduce the median Hb mass loss or influence the need for transfusion in patients. There was also no difference in the occurrence of thromboembolic events or 30-day mortality. These results appeared to be consistent with current evidence that topical TXA were not associated with mortality nor operation complication in elderly patients.
The results of this study further reinforce the existing research findings that suggest TXA did not influence the occurrence of thromboembolic events and mortality for patients with geriatric hip fractures undergoing surgical treatment. Over the past few decades, TXA has gained popularity due to its potential to reduce perioperative blood loss, the need for transfusion and its associated morbidities.3,4,6–9 A systematic review study by Sukeik et al. 12 analysed a total of 11 randomised controlled trials on the safety and efficacy on reducing blood loss using TXA in total hip replacement. They concluded that the use of TXA significantly reduced both the patient's blood loss and the need for transfusion without increasing the risk of adverse events. 12 These benefits could be particularly significant in patients with comorbidities or those at higher risk of complications from transfusion. Another study by Jia 6 also evaluated the effectiveness of combined IV and topical TXA use in patients aged over 70 years old undergoing total hip arthroplasty. Their study demonstrated a significant reduction in the total blood loss (p = 0.015) and Hb drop (p < 0.001) without increasing the risk of thromboembolic event when compared to stand-alone topical or IV TXA use. 6 The author highlighted the significance of ongoing bleeding from raw areas of dissection, which may accumulate and result in significant blood loss. 6 Compared to IV TXA use, topical TXA is easy to administer. Moreover, topical administration of TXA has a lower rate of systemic absorption when compared to those administered intravenously, 16 reducing the potential risk of thromboembolic events.
In addition to the above-mentioned study, a recent study by Yee et al. 17 also demonstrated similar results to our study, where they assessed the safety and effectiveness of topical TXA in geriatric patients with intertrochanteric fracture and found no difference in their total blood loss, transfusion rate, nor the frequency of occurrence of thromboembolic events regardless of the use of topical TXA. 17 Our results suggested that patients with or without topical TXA showed no differences in (a) total Hb mass loss, (b) the incidence of patients requiring transfusion and (c) the occurrence of thromboembolic event and mortality up to one month.
In our centre, patients admitted for hip fracture requiring operative treatment received a dose of on-induction IV TXA, if not contraindicated, to reduce intra-operative and post-operative blood loss. Patients who are contraindicated to IV TXA are generally frailer with comorbidity and hence less tolerant to the systemic side effects of the medication. We estimated around 30% of our patients who are contraindicated to IV TXA could benefit from the topical route due to a lack of systemic absorption.
There are several limitations in our study. First, some patients do not have a pre-operative Hb level taken within 24 h of operation. This could mask the effect of topical TXA, potentially resulting in a false negative result in the reduction of total Hb mass loss. Secondly, we noticed a statistically significant difference in the operative time between the control and treatment groups, which could potentially offset the effect of topical TXA and lead to similar false negative findings. We postulate that this difference may be due to a collective effect contributed by the operative surgeon, who may perform a more meticulous soft tissue closure in patients with the use of topical TXA, as well as administrative reasons, in the surgical theatre. Finally, our study only included deep vein thrombosis and pulmonary embolism as a measurement of thromboembolic events. Nevertheless, we have no record of patients suffering from other possible thromboembolic events, such as acute coronary syndrome and acute cerebrovascular accident. Generally, a 1-g TXA dosage regime is generally recommended. 7 Yet, a high dose of TXA has shown to increase the risk of seizures due to its dose-dependent antagonistic effect against GABAa in the brain. 18 To ensure the safety of our patients, we had opted to administer the range of 10–15 mg/kg, a more conservative dosing for topical TXA. This may have not allowed us to fully investigate the anti-fibrinolytic effect of topical TXA. Nevertheless, the results of this study suggested that a low dosage level between 10 and 15 mg/kg appeared to not influence total Hb mass loss, transfusion need, the occurrence of thromboembolic events nor mortality within 30 days post operation. Future studies should explore the option of administering a higher dosage of TXA for the potential anti-fibrinolytic effect of topical TXA in geriatric patients with hip fractures. Lastly, there may be potential selection bias introduced during the selection of patients into control groups. Patients with creatinine levels above 500 μmol/L were allocated to the control group due to the concerns regarding the potential complication of systemic toxicity from TXA in patients approaching end-stage renal failure. The cut-off for creatinine was decided after literature review with the anaesthesiology department for safety consideration to prevent systemic toxicity of TXA in patients approaching end-stage renal failure. Future studies will need to explore the optimal dosage of TXA in patients approaching end-stage renal failure without inducing systemic toxicity.
Despite the above-mentioned limitations, the findings of this study support the notion that topical TXA is a safe medication to be considered in managing geriatric hip fracture. The administration of 10–15 mg/kg of topical TXA did not result in a reduction of total Hb mass loss, nor did it affect transfusion requirements or increase the risk of thromboembolic events or 30-day mortality.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
