Abstract
Aims:
The combined (IV (intravenous) + topical) use of tranexamic acid (TXA) has been shown to be a safe method and more effective than single (IV or topical) application. The optimal administration method of TXA is still being investigated and safety, efficiency and cost are the three main crucial parameters in achieving the best administration method. We aimed to determine whether combined (oral + topical) use of TXA reduced blood loss and transfusion rates more than single (topical) administration in TKA and whether oral + topical use is as safe and efficient as the IV + topical use, in addition to the main advantage of relatively low cost.
Methods:
In this prospective, randomized study, 100 patients were randomly assigned to either the topical TXA group or the combined (oral + topical) TXA group. There were no significant differences between the groups in age, body mass index or gender. The haemoglobin and haematocrit levels of each patient were recorded preoperatively and on post-operative days 0, 1, 2 and 3. The post-operative suction drainage and blood transfusion volumes were also recorded.
Results:
There were statistically significant differences between the groups in haemoglobin and haematocrit levels on post-operative days 0, 1, 2 and 3 (p < 0.05) in favour of the combined group. The post-operative drainage amounts (p = 0.0001), measured blood loss volume (p = 0.003) and transfusion rates (p = 0.03) were lower in the combined (oral + topical) group compared to the topical group.
Conclusions:
Of the different methods of TXA administration, the combined use of oral and topical TXA is a safe, efficient and low-cost method in reducing blood loss and transfusion rates after TKA.
Introduction
Although the use of a tourniquet during total knee arthroplasty (TKA) results in minimum intraoperative bleeding, patients may have severe bleeding with significantly low blood values and a high amount of blood loss after the tourniquet release. 1 –4 Recent randomized controlled trials (RCTs) have proved that tranexamic acid (TXA) reduces blood loss and transfusion rates after TKA, and TXA is routinely used in TKA following the publication of studies with excellent results. 2,3,5,6 TXA has been shown to be safe in patients with severe comorbidities 7 and to be beneficial regardless of preoperative haemoglobin (Hgb) value 8 in total joint arthroplasties. In respect of the single use of TXA, topical TXA has been reported to be safe 1,9,10 and as equally effective as the IV (intravenous) regimen 1,4,11 in TKA. However, the combined administration of TXA (IV + topical) has been shown to more effective than the single regimen. 12,13
There is ongoing debate in current literature in respect of the most effective and optimal regimen of TXA administration. There is a cost-effective benefit as the use of TXA has been reported to reduce costs in hip and knee arthroplasty in addition to decreasing blood loss. 14 Oral TXA has been shown to decrease blood loss 2 and has similar efficiency as IV administration. 15 As the oral form of TXA is cheaper than the IV form, the financial benefit of oral TXA application has been reported as £2.04 (GB sterling) per patient. 16 Beside the relatively low cost of oral TXA, the simple application avoiding IV access is a further advantage reducing additional costs and avoiding expensive nursing care for IV application.
Safety, efficiency and cost are three main crucial parameters in achieving the best administration method for any drug. The present study aimed to take one step closer to determining the best administration method of TXA in the light of previous studies. The hypothesis of the present study was that the combined (oral + topical) use of TXA is safe and more efficient than the single use (topical), so combined use of TXA with oral form instead of IV form is the optimal method in reducing blood loss and transfusion rates in TKA with a relatively low cost.
Materials and methods
Approval for the study was granted by the Local Ethics Committee. The study was designed to include 100 patients with knee osteoarthrosis who underwent primary total knee replacement according to the inclusion and exclusion criteria described below. Patients were randomized before the operation by generating random numbers with Microsoft Excel 2007 (Microsoft Corporation, Seattle, Washington, USA). Topical TXA was applied to group I (n = 50) and oral + topical TXA to group II (n = 50). The inclusion criteria were primary knee osteoarthritis and a patient age of 55–85 years. The exclusion criteria were rheumatological joint diseases, allergy to TXA, previous knee surgery, anticoagulant therapy, preoperative anaemia and metabolic bone disease. None of the patients had any chronic hepatic or haematological disease, malignancy history, vascular disorders or cardiac operation history. During the TKA operations, a Vanguard® Complete Knee System prosthesis (Biomet Inc., Warsaw, Indiana, USA) was implanted using the same surgical technique in all patients. The tibial and femoral components were implanted with bone cement. The posterior cruciate ligament was retained and patellar surface arthroplasty was not performed in any case. The age, sex and body mass index distributions of the groups are shown in Table 1. There were no significant differences between the groups in respect of anthropometric and demographic data.
Demographic data of the patients.a
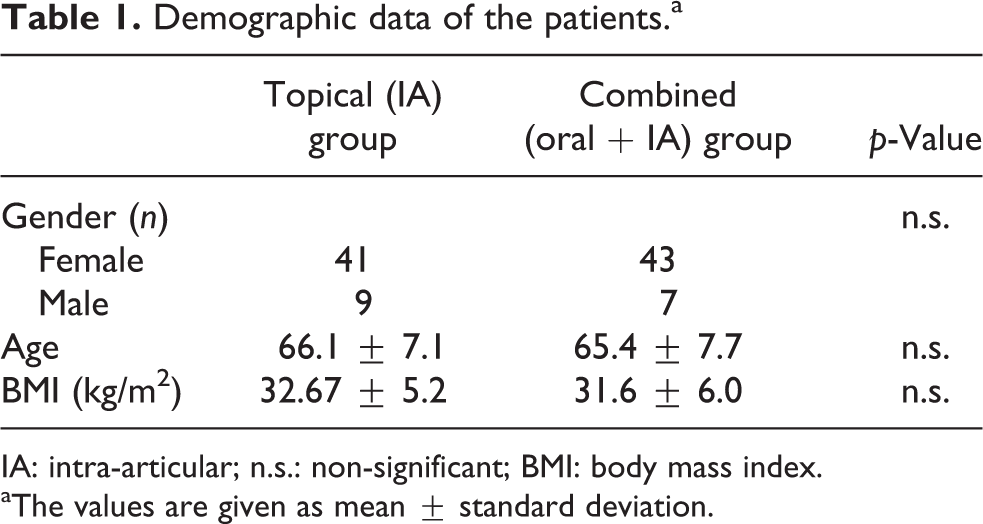
IA: intra-articular; n.s.: non-significant; BMI: body mass index.
aThe values are given as mean ± standard deviation.
A low dose of low-molecular-weight heparin was administered to all patients 12 h before surgery. All patients were normotensive during the perioperative period. A tourniquet was inflated to 300 mmHg pressure after spinal anaesthesia. For the combined group (oral + topical), oral TXA (Transamine, Actavis, Istanbul, Turkey) in tablet form (25 mg/kg; maximum 2 g) was given 2 h preoperatively. A straight, longitudinal midline skin incision and medial parapatellar arthrotomy were performed. The intramedullary guide hole inside the femur was closed by impaction of an autologous structural bone graft into the entry point in all patients. Both femoral and tibial prostheses were implanted with pressured bone cement. A suction drain was placed inside the knee capsule, and while overlying layers were closed in the anatomical planes, 1.5 g of TXA (Transamine, Actavis) in 50 cc of saline was administered directly into the joint cavity for combined and topical groups. Both groups had same surgical procedures and topical TXA doses. The tourniquet was deflated and a Jones bandage was applied. The total time of tourniquet application was recorded. Twenty-four hours after surgery, the Jones bandage and suction drain were removed. The patients were mobilized, and continuous passive motion was started just after removal of the drains.
No iron or erythropoietin was given before or after surgery. Cell savers and bipolar sealers were not used in any patient. Blood transfusion was performed in patients with a haemoglobin value of <8.5 g/dl or with a compromised clinical condition as evidenced by tachycardia, hypotension or symptoms of anaemia during or after surgery in consultation with the anaesthesia team. All blood transfusions were applied within 3 days post-operatively. The haemoglobin and haematocrit values were recorded at post-operative 12 h and days 1, 2 and 3. Post-operative blood drainage was recorded by pouring blood from the suction drain into a measuring jar. The external scale of measuring jar was read by the same two researchers for all patients. The total blood transfusion volume was also recorded for all patients. Blood loss was calculated with the formula defined by Gross. 17 In all patients, perioperative antibiotic prophylaxis using first-generation cephalosporin was administered to prevent infection and the same analgesic treatment to reduce pain was administered using patient-controlled analgesia.
Low-molecular-weight heparin was administered for 4 weeks post-operatively to prevent deep vein thrombosis. A daily treatment of 40 mg enoxaparin sodium (Clexane; Aventis Intercontinental, France) was administered subcutaneously to all patients. Dynamic compression socks were applied to both lower extremities on post-operative day 2 in addition to the prophylactic enoxaparin treatment. All patients were in regular follow-up at 1, 5, 3, 6 and 12 months post-operatively. Complications such as deep vein thrombosis were evaluated and recorded during regular follow-up.
Sample size estimation was performed using the amount of drainage as a primary effect variable. Using the data of a previous study, 18 the group sample sizes of 50 and 50 achieved a power of 0.90 with a significance level (α) of 0.05 using a two-sided, two-sample test. All data were calculated as mean and standard deviation. The Student’s t-test was used for statistical analysis of the patient data. Statistical calculations were performed with SPSS 20.0 (SPSS Inc., Chicago, Illinois, USA). A value of p < 0.05 was considered statistically significant.
Results
No statistically significant differences were determined between groups I and II in respect of the ASA (American Society of Anesthesiologists) score of the patients and operation time (Table 2). The preoperative haemoglobin level, haematocrit level and platelet count were within the normal range in all patients. The preoperative mean haemoglobin level of groups I and II was 13.1 and 13.3 g/dl, respectively (n.s.). The preoperative haematocrit level of groups I and II was 39.4% and 39.9%, respectively (n.s.). The mean tourniquet time for groups I and II was 80.4 ± 15.5 min and 82.9 ± 14.2 min, respectively. The average post-operative drainage volume within 24 h after surgery in groups I and II was 128 ± 62 ml and 81 ± 38 ml, respectively (p = 0.0001).
Surgical data, drainage and calculated blood loss (mL).a

IA: intra-articular; n.s.: non-significant.
aThe values are given as the mean ± standard deviation.
The calculated blood loss for groups I and II was 731 ± 180 ml and 628 ± 157 ml, respectively (p = 0.003). There were statistically significant differences in the mean suction drainage and calculated blood loss volumes in favour of the combined group. The mean suction drainage and calculated blood loss volumes are shown in Table 2. In the topical TXA group, three patients received a blood transfusion whereas in the combined group, none of the patients required blood transfusion. The post-operative transfusion rates were significantly lower in the combined group (p = 0.03). The haemoglobin and haematocrit levels were significantly lower in the combined group (p = 0.01 and p = 0.02, respectively) on the day of surgery. The haemoglobin and haematocrit levels on post-operative days 1, 2 and 3 were also significantly lower in the combined group (Tables 3 and 4).
Preoperative and post-operative haemoglobin values.a

IA: intra-articular.
aThe values are given as the mean ± standard deviation.
Preoperative and post-operative haematocrit values.a

IA: intra-articular; n.s.: non-significant.
aThe values are given as the mean ± standard deviation.
During the regular follow-up, one patient in topical group had symptomatic thromboembolic event. Additionally, there was suspicion of deep vein thrombosis in one patient in this group, and this patient received venous ultrasound study that reported negative result. In combined group, there was no symptomatic thromboembolic event and three patients received venous ultrasound studies with negative results after suspicion of deep vein thrombosis.
Discussion
Reducing blood loss and the need for blood transfusions during orthopaedic surgery remains a major concern in clinical practice, and many interventions such as hypotensive anaesthesia and various blood salvage techniques have been developed. 3,4,8,11,15,19,20 TXA has been shown to reduce blood loss and transfusion rates after TKA 1,3,5,9,12 as well as in other surgical procedures such as rhinoplasty. 21 The use of TXA was shown not to cause any extra complication such as thromboembolic events and renal dysfunction in a study using the data of 872,416 patients, 19 and it was also reported to be safe for patients with severe comorbidities in elective arthroplasty surgeries. 7 TXA has been shown to reduce transfusion rates in patients with a haemoglobin level higher than 15 g/dl, so it should be considered in all total joint arthroplasty patients independent of preoperative haemoglobin level. 8 Despite an increased drug cost with the use of TXA in healthy patients, concomitant reductions in operating room, blood/laboratory and room costs have been shown to contribute to nearly $900 (USA Dollar) cost savings. 22
TXA has been in routine use in total joint arthroplasty during the last decade with the benefits of decreasing blood loss, transfusion rates and hospital costs without any increase in complication rates. However, there is no final consensus regarding the optimal regimen, and it remains a very widely investigated topic in recent RCTs. Topical (intra-articular) and systemic (IV) TXA as the most routinely used single dose regimens have been shown to be safe and effective. 4,5,9,10 Recent RCTs have generally reported that IV and topical regimens had similar efficiency, 1,4,11 whereas a recent study concluded that the topical regimen seemed to be more effective. 6 After many studies evaluating and comparing the single regimens, combined regimens (IV + topical) started to be investigated with the aim of achieving the optimal regimen. The combined regimen has been shown to be more effective than the single regimen of topical TXA 13 and the single regimen of IV TXA 23 without any increase in complications. Various combination methods of TXA administration have also been demonstrated to be more efficient compared to the single dose regimen. 12
Aiming for the optimal regimen of the TXA administration, the combined regimen is one more step with its safety and effectiveness. Beside safety and efficiency, cost is another crucial parameter in achieving the best administration method for any drug. Oral TXA is another administration method of TXA, although its usage has not attracted so much attention in recent literature compared to the IV and topical regimens. A recent RCT evaluating the effectiveness of oral TXA administration reported a 224 ml decline in blood loss and a lesser haematocrit decline after oral TXA use. Oral TXA has also been evaluated in other surgical and clinical practices such as craniofacial surgery and gynaecology. In a recent RCT, 1 g oral TXA was used as the same dose of the present study, and it was shown to decrease blood loss without any significant side effect. 21 In studies to reduce menstrual bleeding, it has been concluded that oral TXA is as efficacious as combined oral contraceptives in reducing menstrual bleeding and improving quality of life. 24 Perioperative use of IV and oral regimens has been compared and no significant difference in blood sparing has been reported among IV and oral groups 15 with no additional complications in either treatment group compared to the control group.
Recent studies have mainly focused on the safety and efficiency of TXA and its different methods of administration. However, in addition to effective and efficient health care, orthopaedists are under increasing pressure to minimize costs yet maintain quality care. 14 Only considering the transfusion cost alone after accounting for the cost of TXA, an institution in the United States reported $83.73 saving per patient and concluded that TXA increased home dispositions and reduced cost. 14 Another study from the United States also considered the overall hospital cost of the patient and reported nearly $900 total cost saving. With regard to the systemic administration of TXA, using the oral form of TXA instead of the IV form may also decrease the cost as the oral form of TXA is cheaper than the IV form and the financial benefit of oral TXA application has been reported as £2.04 (GB sterling) per patient. 16 Beside its relatively low cost, oral TXA is advantageous with its simple application avoiding IV access, and thus, additional costs are reduced as there is no requirement for expensive nursing care for IV application. The combination of systemic (IV) and topical TXA seems to be the optimal regimen in recent RCTs. Therefore, in the current study, it was aimed to determine the optimal administration method of TXA (systemic + topical) in the light of previous studies. The value of the present study is to describe a new regimen (oral + topical) considering not only safety and effectiveness, as previous studies have done, but also in consideration of the cost of the regimen in achieving the optimal regimen of TXA administration in reducing blood loss and transfusion rates after TKA.
There are some limitations of the present study. First, this study was not a double-blind study as the evaluator was not blinded during the evaluation process. Different doses of TXA were not compared in the present study. Patients were not evaluated regarding post-operative functional recovery after administration of this regimen. The patients in the present study had primary knee osteoarthritis, and cases of knee osteoarthritis secondary to other disorders such as rheumatological disorders were not evaluated. The use of an additional blood preserving system such as bipolar sealer and iron therapy was not included in the present study. Clinical evaluations only were used and no deep vein thrombosis test was performed routinely in follow-up to detect any thromboembolic events. Finally, this study did not evaluate the results of TKA without a tourniquet or with the use of a tourniquet at different pressures.
Conclusion
The best administration method of TXA is still a matter of debate in literature. When safety, efficiency and cost are considered as the main crucial parameters during the evaluation of TXA administration methods, the combined use of oral and topical TXA is a safe, efficient and low-cost method in reducing blood loss and transfusion rates after TKA. Therefore, the combined use of oral and topical TXA seems to be the best TXA administration method of the previously described methods, and it takes a further step towards achieving the best administration method of TXA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
