Abstract
Background
This study aimed to investigate the effect of topical administration of TXA on blood loss and hematologic parameters following RSA.
Methods
This retrospective study included 111 patients who underwent RSA mainly for cuff tear arthropathy and massive rotator cuff tears between November 2014 and September 2021, excluding patients with proximal humerus fractures, of whom 52 received TXA (TXA group) and 59 did not receive TXA (non-TXA group). In the TXA group, 1 g of TXA was administrated topically in the subacromial space after skin closure. Hematological parameters were assessed preoperatively and at postoperative days 3, 7, and 14. Intraoperative blood loss, surgical drain volume, calculated total blood loss, blood transfusion, and venous thromboembolic events were compared.
Results
There was no significant difference in intraoperative blood loss between the groups. Calculated blood loss and total surgical drain volume were significantly lower in the TXA group than the non-TXA group (843 ± 334 mL vs 1051 ± 391 mL and 213 ± 142 mL vs 389 ± 164 mL,p < 0.01 and p < 0.01, respectively). In the non-TXA group, hemoglobin (Hb) and hematocrit (Hct) values decreased until postoperative day 7 before increasing at postoperative day 14; in the TXA group, these values decreased at postoperative day 3 and then increased up to postoperative day 14. No thromboembolic event occurred in either group.
Conclusions
The present results showed that topical administration of TXA in RSA reduced blood loss without adverse events and promoted the early recovery of hematologic parameters.
Keywords
Introduction
The volume of perioperative blood loss is critical in shoulder arthroplasty surgery. Patients who have major bleeding requiring blood transfusion frequently have complications that can include postoperative infection, hemolytic reactions, immunomodulation, acute lung injury, and disease transmission. 1 Previous studies have reported allogenic blood transfusion rates after shoulder arthroplasty in the range 4.3%–43%.2–5 Risk factors for blood transfusion after shoulder arthroplasty include advanced age, female sex, preoperative anemia, ischemic heart disease, and reverse shoulder arthroplasty (RSA) technique.2–5 In addition, the risk of blood transfusion after surgery is higher for RSA than for anatomical total shoulder arthroplasty (TSA).6–8
Tranexamic acid (TXA) is a synthetic antifibrinolytic agent that inhibits the conversion of plasminogen to plasmin through competitive inhibition of the lysine binding site on plasminogen.9,10 TXA has been used to reduce perioperative blood loss and blood transfusion in lower extremity arthroplasties such as total hip arthroplasty (THA) and total knee arthroplasty (TKA), as well as spinal surgery.11–18 Several studies have demonstrated the effect of TXA on perioperative blood loss with minimal risk of thromboembolic or cardiac events.16,19 Several studies have investigated the effect of TXA on perioperative blood loss in shoulder arthroplasty.20–22 These studies have shown that TXA reduces postoperative blood loss; however, its effect on transfusion requirements remains controversial. Furthermore, the impact of topical TXA on postoperative hematologic parameters after RSA has not been clarified.
The purpose of this study was to investigate the effect of topical administration of TXA on blood loss and hematologic parameters following RSA. Our hypotheses were that (1) topical administration of TXA would reduce postoperative blood loss, and that (2) hematologic parameters would begin to recover from the early postoperative period in patients underwent RSA with topical administration of TXA.
Materials and methods
Study design
All studies were performed in accordance with the guidelines of the institutional review board of our institution. This retrospective study included 128 patients who underwent RSA between November 2014 and September 2021. The inclusion criteria were symptomatic patients with irreparable rotator cuff tears, with or without glenohumeral osteoarthritis, as well as patients with rheumatoid arthritis and neurogenic arthropathy such as Charcot joint. Indications for surgery included failure of nonoperative treatment. Patients with proximal humerus fractures were excluded because of the potential influence on intraoperative bleeding.
52 patients who underwent surgery between November 2014 and June 2019 were classified as the No-TXA group, and 59 patients who underwent surgery between July 2019 and September 2021 were classified as the TXA group. The following patient characteristics were recorded: age at surgery, sex, height, weight, body mass index (BMI), antiplatelet and anticoagulant drugs, previous history of shoulder surgery, operative time, and intraoperative blood loss. Intraoperative blood loss was recorded based on the weight of surgical gauze and the volume of suction drainage.
This study was approved by the Institutional Review Board of our hospital (#R-50-8J).
Surgical and postsurgical protocol
The surgeries were performed by six orthopedic surgeons, each with more than 10 years of experience in shoulder surgery.
All procedures were performed through a standard deltopectoral approach. The subscapularis tendon was peeled off the lesser tuberosity when it was attached, and the humeral head was resected at 20° of retroversion. The glenoid was exposed, reamed, and fitted with a baseplate and glenosphere. Humeral stems were implanted without cement when fixation was adequate and with cement when fixation was poor. The severed subscapularis tendon was repaired when feasible. Regarding intraoperative procedures, no vascular injuries or other complications that could have affected blood loss occurred in either group, suggesting that surgical technique had minimal influence on the results.
RSA was performed using the following systems: Comprehensive Reverse Shoulder System or Trabecular Metal Reverse Shoulder System (Zimmer Biomet, Warsaw, IN) in 47 shoulders, Medacta Shoulder System (Medacta International, Castel San Pietro, Switzerland) in 7, Aqualis Reversed (j67Stryker, Bloomington, MI) in 4, and Equinoxe Reverse System (Exactech, Gainesville, FL) in 53. Humeral stem fixation was press-fit in 103 shoulders and cemented in 8 shoulders. In the TXA group, 1 g of TXA was administered in the subacromial space after skin closure. The dose of TXA was set at 1 g according to our institutional protocol for total joint arthroplasty. In all patients, a hemovac drain was placed in the subdeltoid space and postoperative immobilization was achieved using an abduction pillow. Drain output was recorded by nursing staff and the drain was removed after the day unless output was >50 mL during 24 h. Passive shoulder motion exercise was initiated after removal of the drainage tube.
Blood transfusion
A standard care blood transfusion protocol was used in both groups. Transfusion was performed by allogenic red blood cell transfusion in all patients with Hb concentration <7 g/dL. In those with an Hb concentration between 7 and 8 g/dL, blood transfusion was performed if symptoms such as tachycardia and palpitations were present. Transfusion was not indicated in patients with an Hb concentration >8 g/dL.
DVT screening
Perioperative blood samples were obtained in all patients, and D-dimer levels were measured. Patients with D-dimer levels ≥10 μg/mL underwent further evaluation using contrast-enhanced CT or ultrasound.
Outcome measurement
Blood loss was evaluated as total blood loss and drain output after RSA. Total blood volume (L) was estimated using the formula of Nadler et al.,
23
as follows. Females: height (m)3 × 0.3561 + weight (kg) × 0.03308 + 0.1833 Males: height (m)3 × 0.3669 + weight (kg) × 0.03219 + 0.6041
Total blood loss was then estimated by total blood volume and Hct using the formula of Gross et al.,
24
as follows: Mean perioperative Hct (%) = [Preoperative Hct (%) + Post operative Hct (%)]/2
Total blood loss during surgery (mL) was estimated as follows: Blood volume (mL) × [preoperative Hct (%) − postoperative Hct (%)]/mean perioperative Hct (%).
Drain output was recorded on postoperative days 1 and 2. The hematologic parameters Hb and Hct were measured preoperatively and on postoperative days 1, 3, 7, and 14. The number of patients who received transfusion and experienced any adverse events such as deep venous thrombosis (DVT) were recorded.
Statistical analysis
All data were analyzed using Excel 2022 (Microsoft Corp, Redmond, WA) and JMP® software (v12.0, SAS Institute, Cary, NC). All variables were tested for normal distribution using Shapiro–Wilk test. Age, height, weight, BMI, operative time, and intraoperative blood loss were compared between the groups using an unpaired t-test. A χ2 analysis was performed to compare sex and use of antiplatelet and anticoagulant drugs. To evaluate the efficacy of topical TXA administration, total blood loss, drain output, and postoperative Hb and Hct were compared between the groups by an unpaired t-test. Hematologic parameters were compared before and after surgery using analysis of variance (ANOVA). For parameters identified as statistically significant, post hoc analysis was performed by Tukey’s honest significant difference test. p < 0.05 was considered to indicate statistical significance.
Results
Patient demographics
The primary diagnosis was cuff tear arthropathy in 74 patients, massive rotator cuff tear in 19, sequelae of proximal humerus fracture in 6, osteoarthritis in 5, rheumatoid arthritis in 5, chronic shoulder dislocation in 1, and Charcot shoulder in 1.
Patient demographics.

TXA: tranexamic acid; BMI: body mass index; Hb: hemoglobin; Hct: hematocrit.
Blood loss
Blood loss according to group.

TXA: tranexamic acid.
Change in postoperative hematologic parameters
Preoperative and postoperative hematologic parameters.
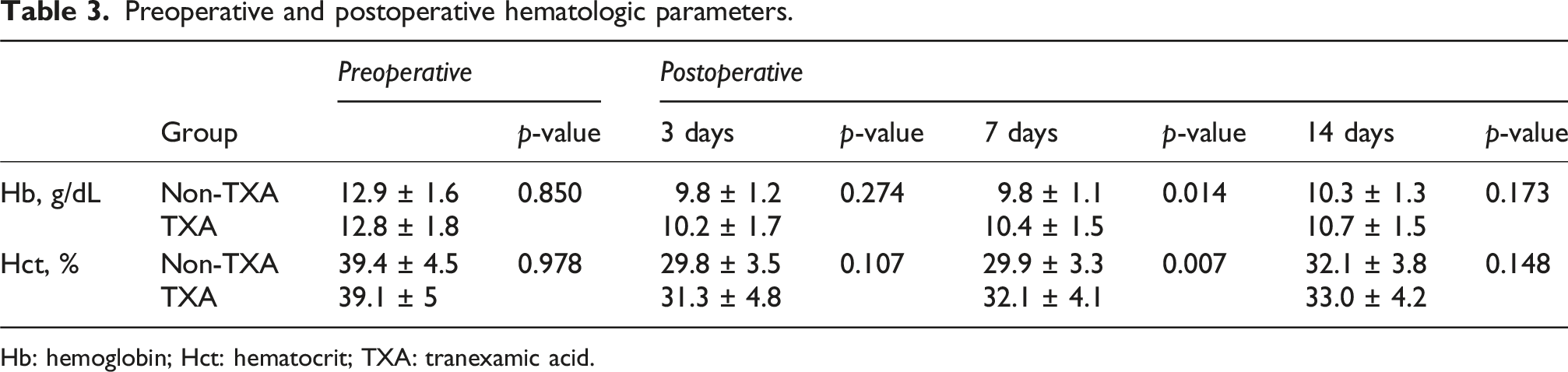
Hb: hemoglobin; Hct: hematocrit; TXA: tranexamic acid.
There was no significant difference between the groups in terms of Hb and Hct values on postoperative day 3 or 14 (p = 0.274 and 0.107, respectively, on postoperative day 3; p = 0.173 and 0.148, respectively, on postoperative day 14). Hb and Hct values were significantly higher in the TXA group than the No-TXA group on postoperative day 7 (p = 0.014 and p < 0.007, respectively). Compared with the postoperative change from the preoperative values, the postoperative change values in Hb and Hct in the No-TXA group were lower on postoperative day 7 and then increased at day 14 after surgery (p < 0.001 and p = 0.002, respectively) (Figure 1(a)). In the TXA group, the postoperative change values in Hb and Hct were lower on postoperative day 3 and then increased at day 14 (Figure 1(b)). Comparing postoperative change values in Hb and Hct between the groups, the change value in Hb was significantly greater in the No-TXA group than the TXA group on postoperative days 3, 7, and 14 (p = 0.018, < 0.001, and 0.024, respectively). The change value in Hct was significantly greater in the No-TXA group than in the TXA group on postoperative days 3, 7, and 14 (p = 0.008, <0.001, and 0.017, respectively). Change values in the hematologic parameters of hemoglobin (a) and hematocrit (b) at preoperative and postoperative day (POD) 3, POD7, and POD14 after RSA in the TXA and non-TXA groups. *p < 0.05.
Transfusion and adverse events after RSA
Four patients in each of the TXA and no-TXA groups received blood transfusion after RSA (no significant difference, p = 0.95). Regarding adverse events after RSA, no venous thromboembolic event occurred in any patient.
Discussion
This study investigated the effect of topical TXA administration on postoperative blood loss and hematologic parameters following RSA. The major findings were that topical TXA administration decreased postoperative blood loss, especially on the first day after surgery; and that postoperative decrease in the hematologic parameters Hb and Hct recovered earlier in patients who received topical TXA than in those who did not. No venous thromboembolic events occurred in the TXA group, and there was no effect of TXA treatment on the rate of postoperative blood transfusion. These findings confirmed our hypothesis and suggest that topical TXA administration decreases postoperative blood loss and promotes early hematologic recovery following RSA.
Blood transfusion after shoulder arthroplasty is required more frequently in RSA than in anatomical TSA, and its frequency has been reported to range from 6.78% to 51.2%.6–8 Botros et al. reported that blood transfusion was required twice as often in RSA than anatomical TSA, 6 whereas Makhni et al. reported blood transfusion rates of 3.1% in anatomical TSA and 11.7% in RSA. 8 The subacromial dead space is generally greater in RSA than anatomical TSA because of rotator cuff removal and distalization of the humerus, enabling postoperative bleeding into the subacromial space that can continue for some time after surgery. Accordingly, blood loss after RSA may increase the risk of blood transfusion and affect the patient’s general condition after surgery. For this reason, it is desirable to decrease postoperative blood loss after RSA to the volumes typical in lower extremity arthroplasties such as THA and TKA.
Previous studies have reported risk factors for blood transfusion following shoulder arthroplasty as female sex, age >85 years, preoperative anemia, rheumatoid arthritis, and fresh trauma.2,5 Technical issues and the learning curve also affect perioperative bleeding in RSA. 25 Kim et al. reported that the blood transfusion rate after RSA was 2.5 times higher in patients with preoperative Hb of <13.5 g/dL, and 1.4 times higher in those with preoperative Hct of <40%, 7 which suggests a correlation of preoperative hematologic parameters with blood transfusion after shoulder arthroplasty. However, the change in hematologic parameters in the postoperative period after RSA relative to the preoperative values is unclear. The present results identified that Hb and Hct decreased on postoperative day 7 and then increased on postoperative day 14, suggesting that the postoperative hematologic parameters should be monitored carefully until postoperative day 7 in patients with risk factors for blood transfusion because postoperative anemia may progress.
Tranexamic acid is a synthetic antifibrinolytic agent that acts on the fibrinolysis system to inhibit conversion of plasminogen to plasmin through competitive inhibition of the lysine binding site on plasminogen.9,10 This system can promote hemostasis and is useful for the control of postoperative bleeding. Therefore, TXA has been applied in several orthopedic surgeries, mainly in lower extremity and spinal surgery to decrease postoperative blood loss.11–18 TXA is also widely used in TKA, THA, and spinal surgery to suppress perioperative bleeding. Gilbody et al. reported that use of TXA resulted in a reduction in transfusion rates from 19.3% to 0% for THA and from 13.1% to 0% for TKA compared with non-TXA groups. 26 Badeaux et al. reported in a systematic review that intravenous TXA significantly reduced intraoperative bleeding in patients undergoing spinal surgery. 27
Perioperative application of TXA in shoulder arthroplasty has been shown to reduce postoperative blood loss and transfusion requirements.20–22 In their double-blinded randomized study, Gillespie et al. found that topical administration of TXA in shoulder arthroplasty inhibited perioperative blood loss and decreased Hb compared with the placebo group. 22 Abildgaard et al. investigated the efficacy of intravenous TXA injection in 77 shoulders in anatomical TSA and 94 shoulders in RSA, 20 and clarified that intravenous TXA injection inhibited blood drainage volume and decreased Hb and Hct. Belay et al. reported that both intravenous and topical TXA were effective in reducing bleeding and the transfusion rate after TSA. These findings indicate the efficacy of both topical and intravenous administration of TXA to decrease blood loss and transfusion after TSA, and suggest that topical administration could be an alternative option for patients undergoing TSA in whom intravenous administration poses risks of pulmonary embolism and DVT. 21 Previous studies demonstrated that TXA administration reduced postoperative bleeding, but they did not report changes in hematologic parameters.
In this study, topical TXA administration reduced postoperative bleeding without affecting transfusion rates. Moreover, our analysis clarified the effect of topical TXA on postoperative hematologic recovery after RSA. In the TXA group, Hb and Hct values decreased by postoperative day 3 and began to recover by day 7. In contrast, in the non-TXA group, Hb and Hct continued to decline until postoperative day 7 and recovered only by day 14. These findings indicate that topical TXA administration promotes earlier recovery of hematologic parameters. Such effects on hemostasis may also contribute to improved recovery of the patient’s condition and potentially shorter hospitalization after RSA.
Patel et al. reported no significant difference between in terms of perioperative change in Hb level and total drain output in TKA. 28 In a meta-analysis, Chen et al. reported that topical TXA was as effective as intravenous TXA in reducing bleeding and transfusion rates after TKA and THA. 29 Despite the similar effects of intravenous and topical TXA administration, the safety of TXA intravenous administration without adapting anticoagulant therapy after RSA is not clear. Therefore, we investigated topical TXA administration in this study, and found that topical TXA administration decreased blood loss and total surgical drainage volume after RSA. We consider that topical TXA affects hemostasis at the local site by its antiplasmin action.
A possible concern regarding TXA administration is the theoretical risk of DVT or thromboembolic events19,20,22 However, Yue et al. reported that topical TXA effectively reduced bleeding and the transfusion rate in TKA without increasing the risk of deep venous thrombosis and pulmonary embolism. 30 Although no study has compared intravenous and topical administration of TXA in RSA in terms of safety, TXA was topically administered in this study. No venous thrombosis occurred after RSA and the topical TXA acted to minimize local blood loss and the total surgical drainage volume after RSA, leading to early recovery of postoperative hematologic parameters. These results suggest that topical administration of TXA should be considered in RSA to reduce postoperative blood loss.
There are several limitations in this study. First, the study design was retrospective and the patients were not standardized, which may have affected bleeding. It is necessary to conduct a prospective randomized study to further clarify the role of topical TXA in shoulder arthroplasty. Second, the optimal dose of TXA in the shoulder joint is unclear, and its effect might be dose-dependent. In this study, all patients received the same dose of TXA, which was effective for reducing blood loss after RSA. Third, as we did not obtain daily blood samples, change in hematologic parameters was not assessed in continuous variables. The accuracy of the results would be improved by comparison of daily values obtained over the postoperative period. Fourth, the use of a drain may have influenced the amount of bleeding. Bartosz et al. reported that the use of a drain paradoxically lead to a higher volume of blood loss, the need for blood transfusion, and the potential for the introduction of infection in TKA and THA. 31 In the future, we should investigate the effect of topical administration of TXA without the use of a drain on blood loss and hematologic parameters in RSA. Fifth, disease heterogeneity may have influenced intraoperative blood loss and hematologic parameters. The cohort included patients with various diagnoses, such as rotator cuff tear arthropathy, massive rotator cuff tears, fracture sequelae, osteoarthritis, and rheumatoid arthritis. Differences in surgical complexity and bleeding risk among these conditions may have affected the outcomes. However, subgroup results did not differ substantially, suggesting that the overall impact of disease heterogeneity in this study was minimal. Sixth, the influence of the surgeon’s learning curve might have affected the results because the cases were allocated chronologically. However, there was no significant difference in operative time between the TXA and No-TXA groups, suggesting that the impact of the learning curve was minimal.
Conclusion
The present results showed that topical administration of TXA in RSA reduced blood loss without adverse events and promoted the early recovery of hematologic parameters. These findings suggest that topical TXA administration could be beneficial in RSA.
Footnotes
Ethical considerations
This study was performed in line with the principles of the Declaration of Helsinki. This study was approved by the Institutional Review Board of our hospital (#R-50-8J).
Consent to participate
Consent to participate was obtained from all patients who participated in this study.
Consent for publication
Consent to publication was obtained from all patients who participated in this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used and/or analyzed during this study are available from the corresponding author on reasonable request.
