Abstract
Objective
While most geriatric patients with hip fracture benefit from definitive operative treatment, pre-operative pain management is often overlooked. Conventional oral analgesics may provide suboptimal pain control, resulting in poor morbidity outcomes. Fascia iliaca compartment block (FICB) provides a more consistent analgesic effect. This study aims to provide high level local evidence on the effect of FICB in pre-operative pain control for these patients in Hong Kong.
Materials and Methods
This study was a double-blinded randomized controlled trial. Mentally competent geriatric patients with acute fracture hip, admitted to United Christian Hospital, were recruited to receive pre-operative FICB. The procedure was carried out by landmark approach with addition of ultrasonography-guidance. They were randomized into intervention group (receiving 0.25% levobupivacaine), and control group (receiving normal saline as placebo). Numeric Rating Scale of pain score (at rest, and on gentle movement) at different time intervals, timing of injection, systemic analgesic use, complications, and demographics were compared.
Results
Total 80 patients were recruited from July 2019 to September 2021 and randomized into intervention and control groups. Two patients in intervention group and 1 in control group were withdrawn due to development of delirium. Demographics of both groups were comparable. Pain score at rest was significantly lower at 12 h after injection in intervention group (41.4% reduction, p = 0.03). Pain score upon gentle movement was significantly lower in intervention group at 1 h (p = 0.01), 12 h (28.9% reduction, p = 0.003) and 24 h after injection (p = 0.01). There was no complication from the procedure documented. Total systemic analgesic use was similar in both groups.
Conclusion
FICB is an effective choice of pre-operative analgesia for geriatric hip fracture patients with good safety profile, especially in controlling pain induced by movement. It shall be considered as a routine practice for suitable patients.
Keywords
Introduction
Facing an aging population, the Hong Kong medical system is overloaded by a rising incidence of geriatric hip fractures. 1 Most geriatric patients with hip fracture benefit from definitive operative treatment for pain control and early mobilization. 2 Pre-operative pain management, however, is often overlooked in everyday hectic in-patient care. Minority, down to 2%, of elderly patients with limb fractures received adequate pain relief. 3 Untreated pain from hip fracture may result in development of delirium, poor mobilization and its sequelae.4,5 Multiple studies echoed that adequate peri-operative analgesia in hip fracture may prevent the development of delirium up to 9 times. 6 Polypharmacy and multiple medical comorbidities in geriatric patients may expose this population to undesirable side effects from conventional systemic analgesics, including opioids and non-steroidal anti-inflammatory drugs. 7 Fascia Iliaca Compartment Block (FICB), since first introduced by Dalens et al., 8 has been shown to be a more superior alternative analgesic for hip fractures with a more significant reduction of peri-operative pain score, and a reduced use of systemic opioids.3,9,10 FICB could be performed safely under anatomical landmark approach or under ultrasonography guidance, having a high success rate up to 96%3,11,12,13,14 in both techniques.
Despite there is a rising number of evidence analyzing the efficacy of FICB over the world, 15 FICB is yet not commonly performed in geriatric hip fracture patients in Hong Kong. This study aimed to provide high-level local evidence to prove the effectiveness of FICB, compared to placebo, in pre-operative pain control for this population.
Materials and methods
A prospective, double-blinded, randomized controlled trial was carried out in United Christian Hospital, a local acute general hospital in Hong Kong. Geriatric patients with acute hip fractures admitted to the hospital were screened and recruited as study subjects according to inclusion and exclusion criteria.
Inclusion criteria were:
Age 60 or above Radiographically confirmed unilateral fracture hip Indicated for hip fracture surgery FICB administered within 24 h after admission Mentally incompetent (Abbreviated Mental Test score <6)16,17 Presence of more than one fracture Patient's refusal to participate Contra-indications to FICB
Known true allergy or previous anaphylactic reaction to local anaesthetic Previous femoral-bypass surgery, or near a graft site Previous surgery over the site of injection Inflammation, infection, or hernia over the site of injection INR > 1.4 Platelet count < 100 × 109/L Patient on heparin / low molecular weight heparin / fondaparinux / rivaroxaban / dabigatran / ticlopidine / clopidogrel / prasugrel / cilostazol
Exclusion criteria were:
Sample size
Sample size was estimated using data from previous study by Candal-Couto et al. 18 It was based on two-sided repeated measures analysis of variance comparison of the VAS scores between groups. 19 The assumptions of 5% type I error rate and 80% power were considered. If total 80 subjects, 40 subjects in each arm of intervention group and control group, were recruited, it would be adequate to demonstrate a significant 30% difference in pain score on movement according to previous study by Matot et al. 20
Procedure
Two equal sets of enclosed envelopes containing information on group allocation were prepared by a member of clinical staff not involved in the analysis of the study. Randomization of the envelopes and allocation to consecutive recruited subjects were carried out by that person.
Following the admission of the patients with hip fractures to the Orthopaedic and Traumatology ward, the authors were informed and they reviewed the medical records of the patients. According to the inclusion and exclusion criteria, subjects were recruited by the authors within 24 h after admission. The allocated enclosed envelope was opened by the authors, without the subjects’ acknowledgement of the allocation. A syringe of either 0.25% (2.5 mg/ml) levobupivacaine or placebo (normal saline), depending on the allocation to the intervention or the control group, was prepared. 30 ml of injection was prepared for subjects with an estimated body weight of less than 50 kg, whereas 40 ml was prepared for those with an estimated body weight of 50 kg or more. FICB was performed by the orthopaedic surgeons, among the authors, using a 21G needle on the affected hip under landmark approach with ultrasonography guidance and aseptic technique (Figure 1, Figure 2). Correct placement of the injection was confirmed with ultrasonography (Figure 3). Close observation of subjects’ condition and vital signs will be performed by their respective nurse-in-charge, who had no knowledge on the group allocation and was not involved in the study analysis, in 10-min interval till 30 min after injection. The nurse-in-charge subsequently conducted the pain score assessment and filled in the case report form.
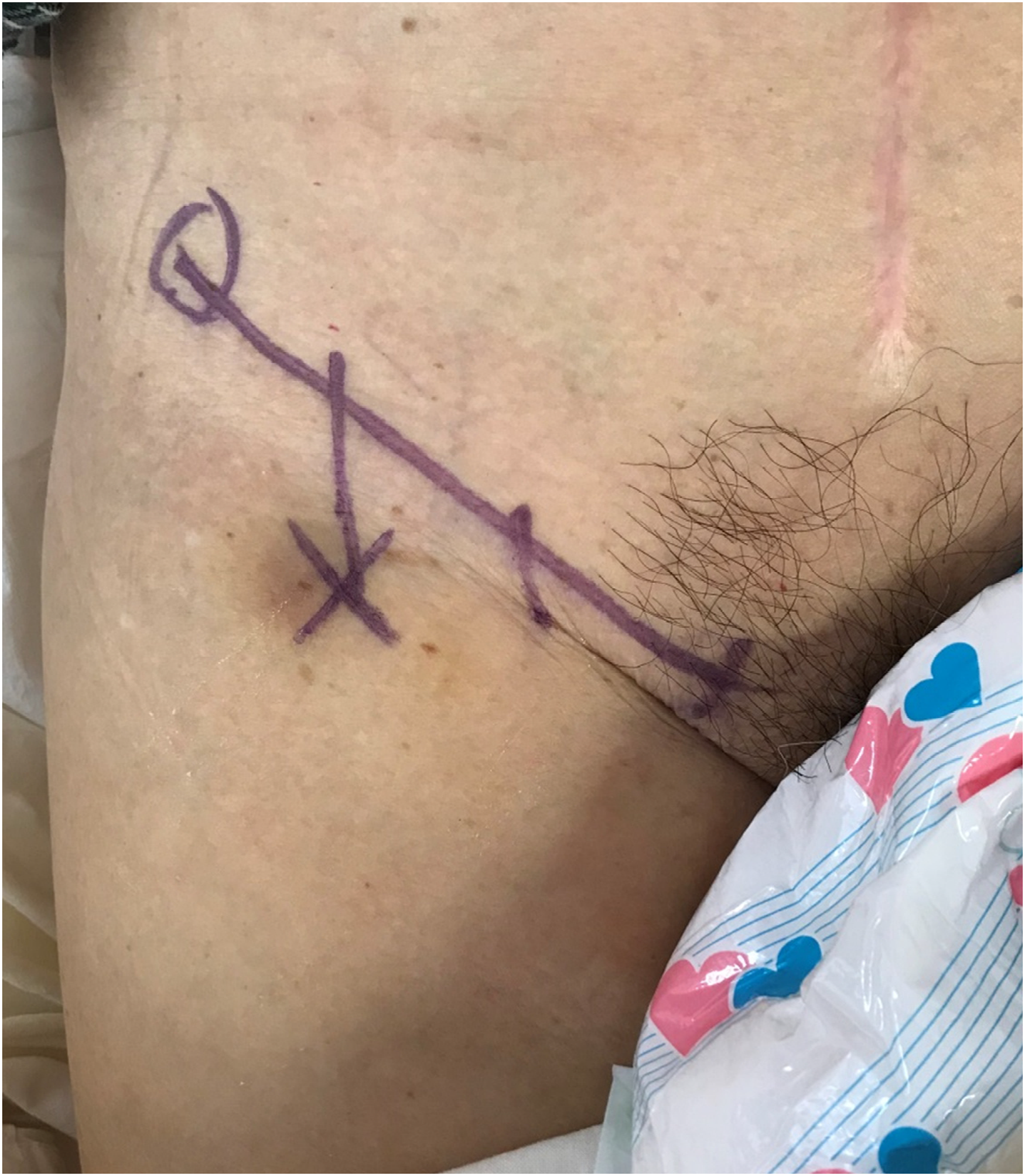
Injection site marked as “X” using landmark approach. (2 cm distal the junction of lateral 1/3 and medial 2/3 of distance between the anterior superior iliac spine and the pubic tubercle).
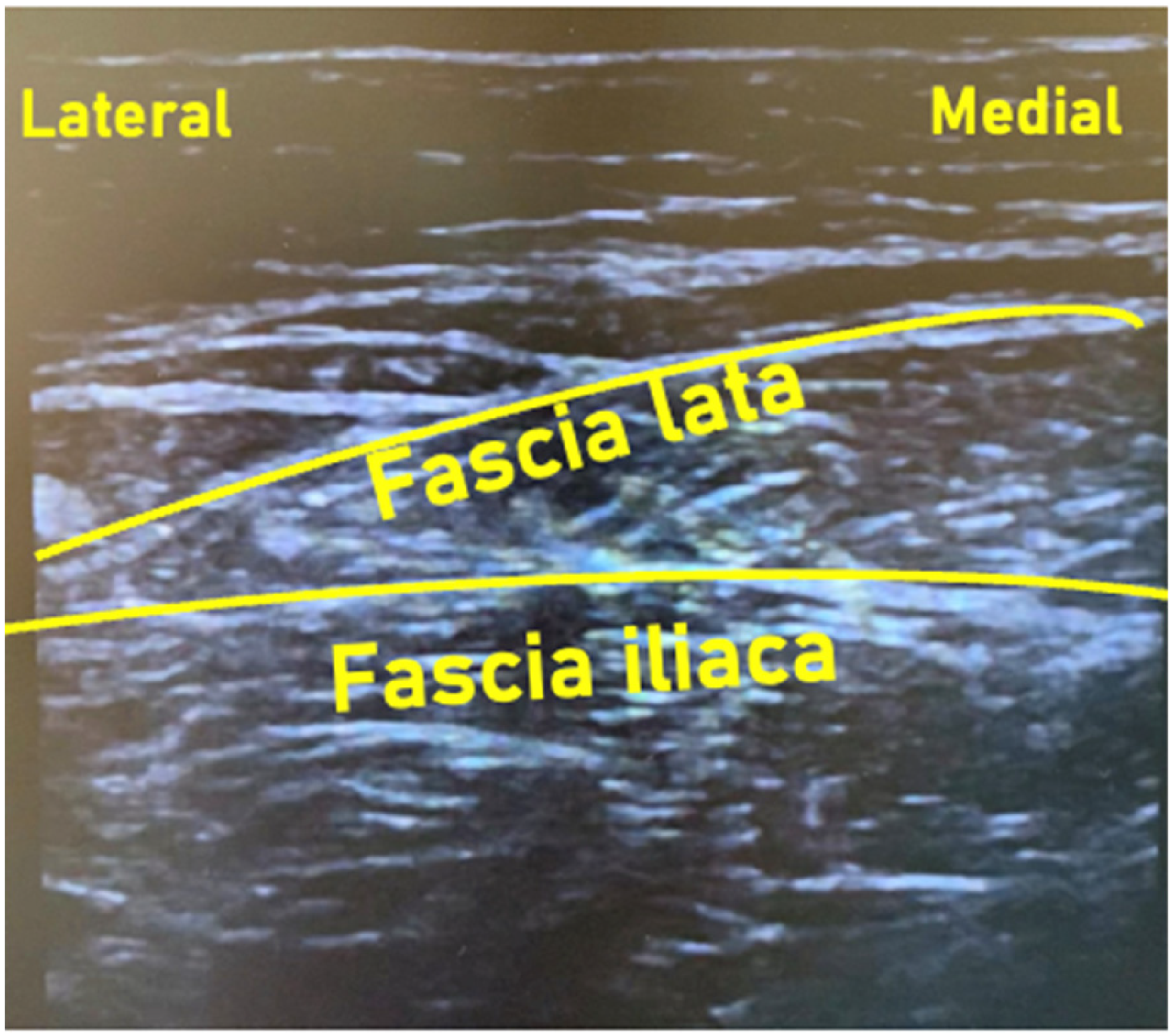
Ultrasonographic imaging at site of injection using a linear transducer.
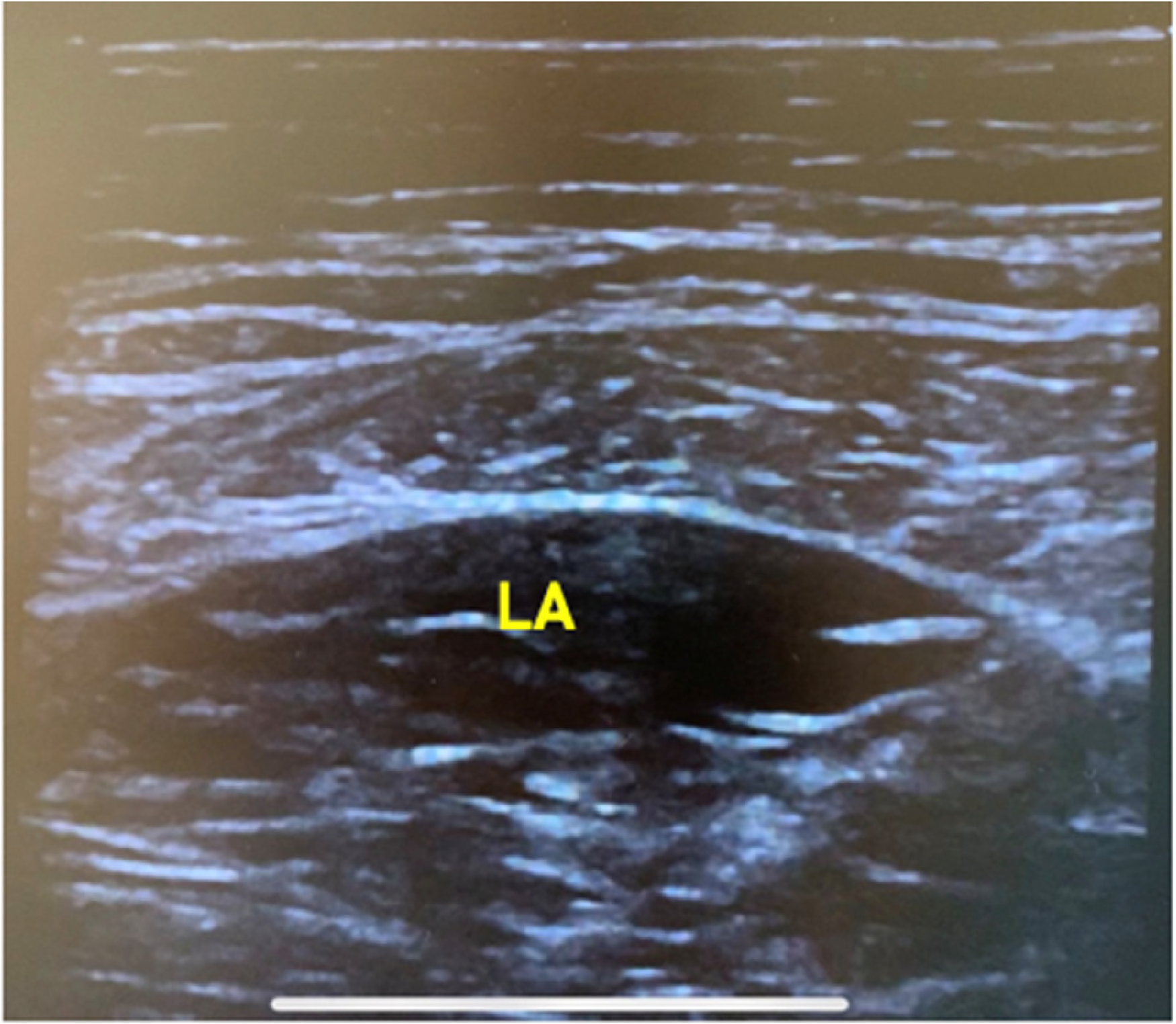
Ultrasonographic imaging at site of injection using linear transducer after injection, showing local anaesthetics (la) placed in correct compartment.
All the recruited subjects in both groups received the same routine in-patient nursing and medical care as other patients with acute hip fractures. A standardized prescription of oral analgesics of both paracetamol (500 mg po QID, 500 mg po Q4H prn) and tramadol (50 mg Q6H prn) was given.
The study and assessment ended when the subject underwent definitive surgery for the hip fracture, or at 48 h after the injection, whichever came first.
Measurements and outcomes
Primary outcome was the subject's reported pain score in terms of the Numeric Rating Scale (NRS) at rest and on gentle movement. By “gentle movement,” it was defined as gentle rotation movement of the affected lower limb beyond its neutral axis with knee extended. The NRS was assessed in different time intervals: on admission, just before injection, 1 h after injection, 12 h after injection, 24 h after injection, and 48 h after injection.
Secondary outcomes, including pre-operative total dosage of systemic analgesics use, and incidence of complications from the injection, were also measured. Baseline characteristics, including subjects’ demographics (age, gender, history of diabetes, American Society of Anesthesiologists (ASA) Physical Status Classification), timing of injection after admission, and hip fracture characteristics (traumatic or pathological, intracapsular or extracapsular), were also reported.
Statistics
Data analysis was performed using IBM® SPSS® Statistics 23. Continuous variables, presented as the mean, standard deviation, and range, were analyzed with T-test. Categorical variables, presented as percentages, were analyzed with Chi-squared test. Mean difference in pain score between the intervention group and the control group was analyzed. A P value < 0.05 was considered to be of statistical significance.
Ethical considerations
Ethical approval was obtained from Research Ethics Committee (Kowloon Central / Kowloon East), Hospital Authority, Hong Kong on 28th May 2019 (Reference: KC/KE-19-0033/FR-1). Only patients with the mental capacity to make an informed consent at the time of recruitment were included in the study. They were free to withdraw from the study at any juncture. The FICB was given as an adjunct in addition to the systemic analgesics, which were the same as the routine prescription for other usual patients with hip fracture admitted to the hospital. The procedure was reported to be of low risk. 10 All recruited subjects were monitored closely for side effect, complication, and adequacy of pain relief. Subjects would be withdrawn from the study if they lost mental competency after recruitment, or if they developed life-threatening complications, including but not limited to local anaesthetic toxicity.
Results
From July 2019 to September 2021, a total of 80 subjects were recruited, after screening 1637 patients for the inclusion and exclusion criteria. Most of excluded patients were mentally incompetent to make an informed consent to our study, while the remaining ones were excluded due to the block not possibly being done within 24 h after admission, refusal to the study, and bleeding tendency while taking the listed antiplatelet agent or anticoagulants. The recruited subjects were equally randomized into the intervention and the control group. However, three subjects, of which two were in the intervention group and one in the control group, were withdrawn from the analysis due to the development of delirium after the recruitment and before the end of study (Figure 4). The delirious status rendered them to be unreliable and inconsistent in their reported outcomes.
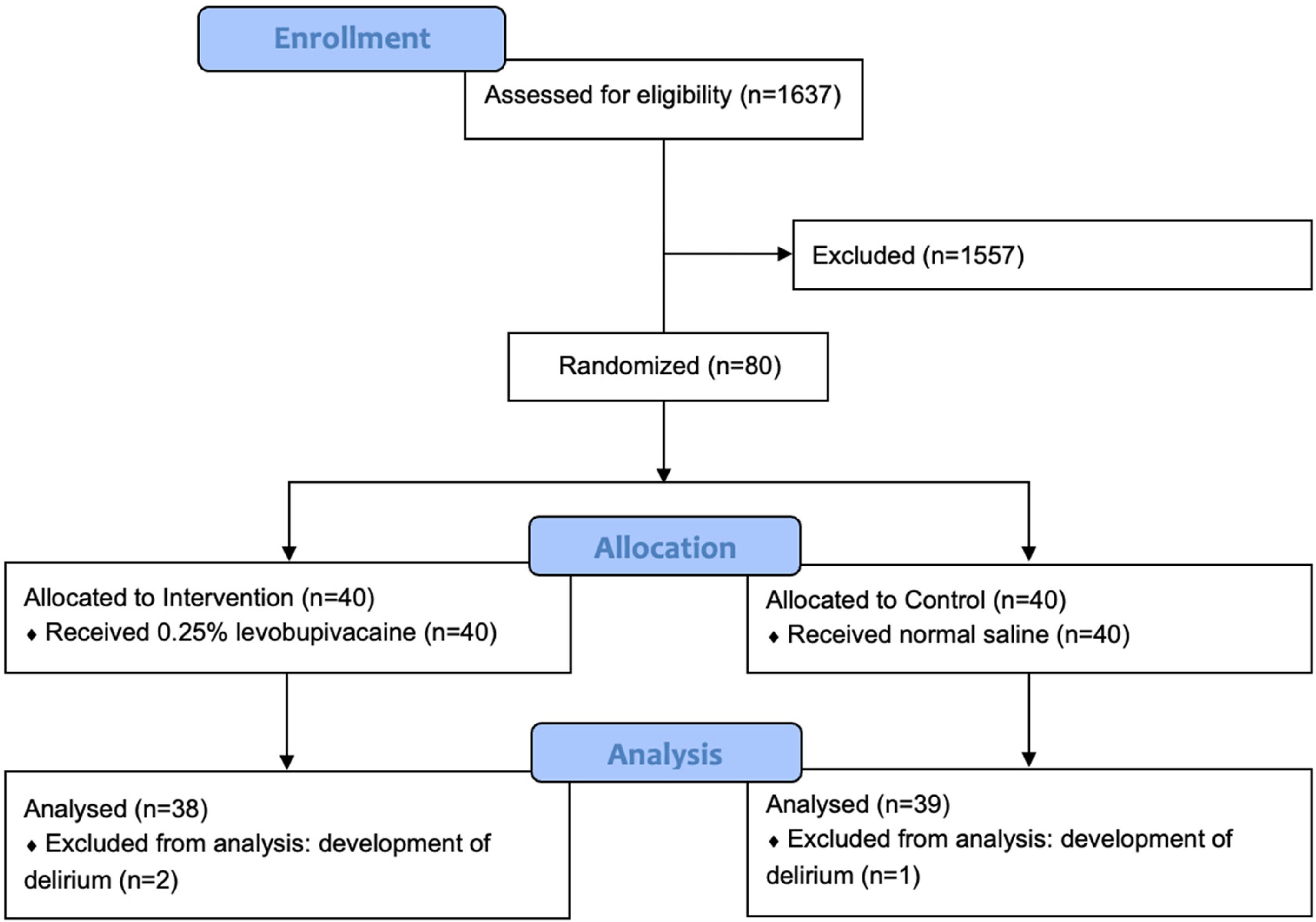
Flow diagram (CONSORT 2010) showing the recruitment and the analysis process of the RCT.
Demographics and baseline characteristics of both groups were summarized in Table 1. There were no significant differences in gender, age, medical history of diabetes mellitus, and Abbreviated Mental Test scores between the two groups. 57.9% (n = 22) were female in the intervention group, while 61.5% (n = 24) in the control group. The mean age was 81.3 in the intervention group, and 81.4 in the control group. Most of the subjects in both groups were diagnosed with hip fractures following a compatible trauma event (97.4% in the intervention group, 100% in the control group). The majority of the subjects had an estimated body weight greater or equal to 50 kg, and thus received FICB of 40 ml injection. The mean time from admission to injection was 17.6 h in the intervention group, and 16.7 h in the control group. There was a statistically significant difference (p = 0.02) in ASA class between the two groups.
Demographics and baseline characteristics.
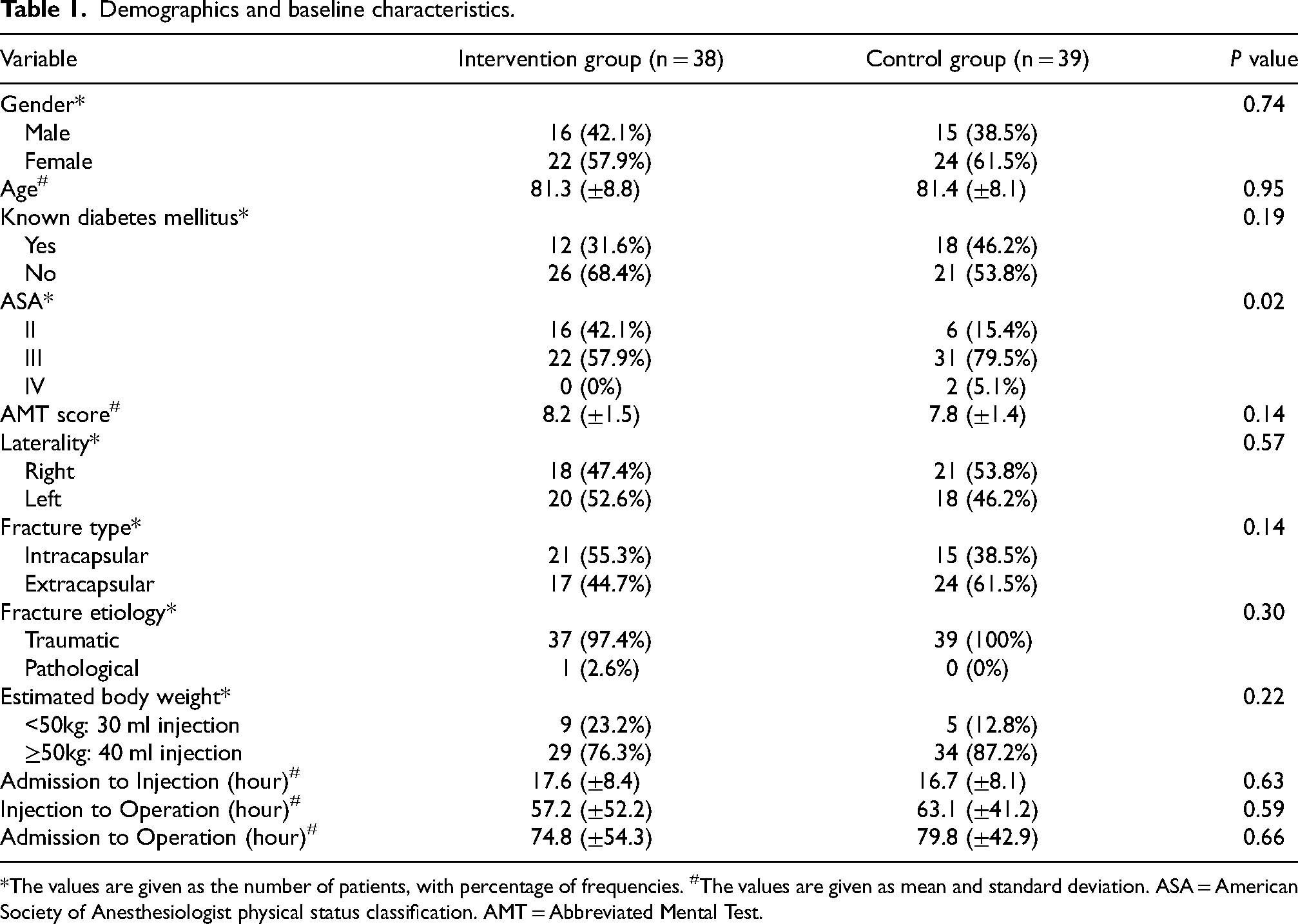
*The values are given as the number of patients, with percentage of frequencies. #The values are given as mean and standard deviation. ASA = American Society of Anesthesiologist physical status classification. AMT = Abbreviated Mental Test.
Results of pain scores at rest were shown in Table 2. There were similar pain scores between the two groups on admission, just before injection and 1 h after injection. The pain at rest was significant lower at 12 h after injection (mean score 1.7 in the intervention group, 3.1 in the control group, P = 0.03). This represented a 41.4% reduction of the mean pain score from 2.9 to 1.7 in the intervention group. The differences at 24 h after injection and 48 h after injection were not statistically significant.
Pain score at rest.
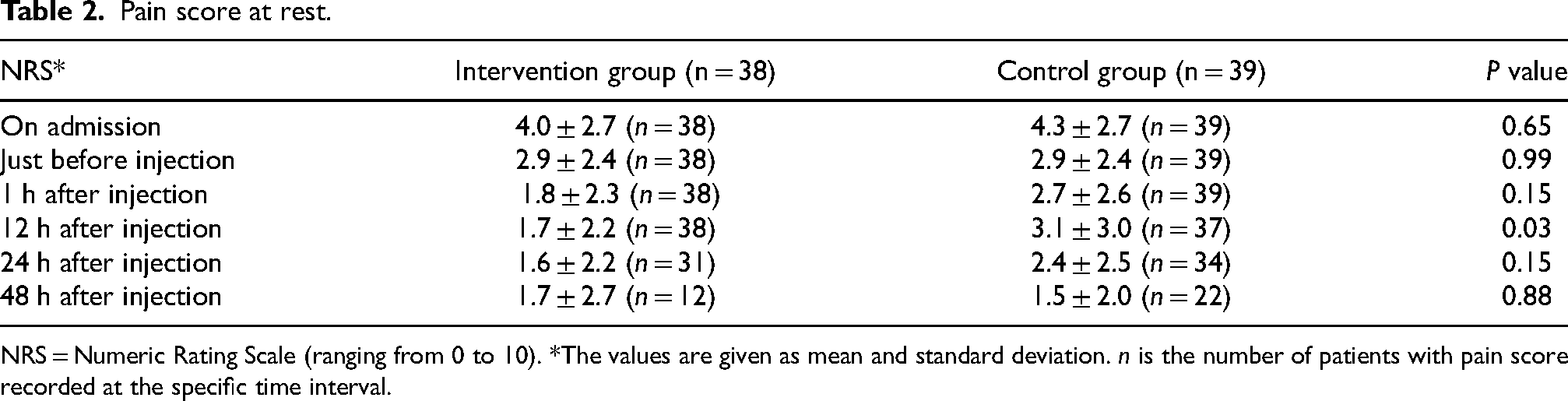
NRS = Numeric Rating Scale (ranging from 0 to 10). *The values are given as mean and standard deviation. n is the number of patients with pain score recorded at the specific time interval.
Results of the pain scores on gentle movement were shown in Table 3. The mean pain scores in the intervention group were significantly lower at 1 h after injection (mean score 5.6, P = 0.01), 12 h after injection (mean score 5.4, P = 0.003), and 24 h after injection (mean score 5.9, P = 0.01). At 12 h after injection, there was a 28.9% reduction in the mean pain score (from 7.6 to 5.4) in the intervention group. However, the difference became insignificant at 48 h after injection.
Pain score on gentle movement.
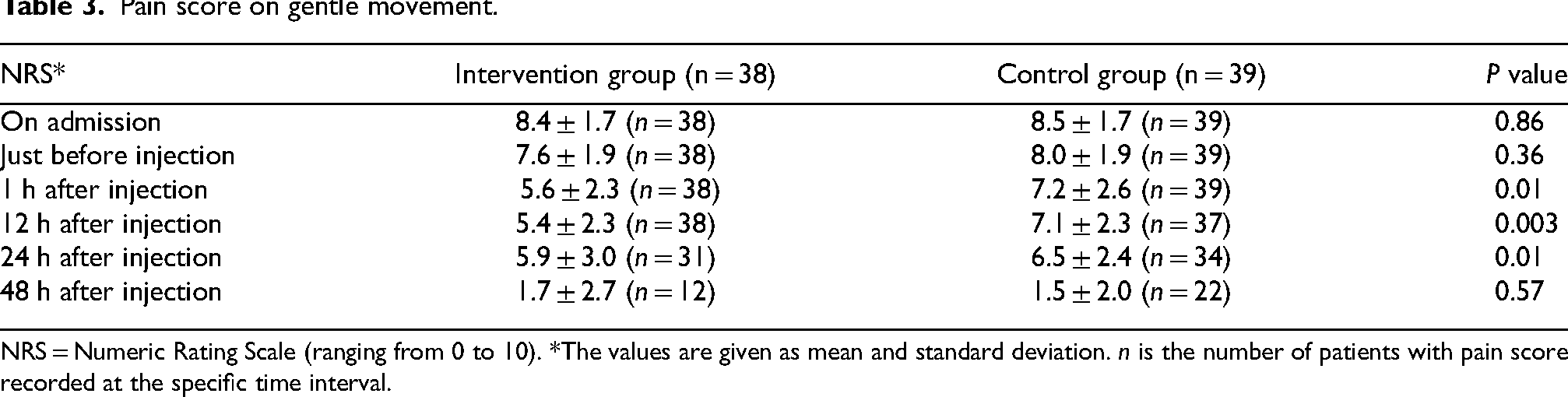
NRS = Numeric Rating Scale (ranging from 0 to 10). *The values are given as mean and standard deviation. n is the number of patients with pain score recorded at the specific time interval.
The total consumption of oral analgesics in both groups from the time of injection to operation and from admission to operation was summarized in Table 4. No significant difference was found in the use of paracetamol or tramadol after the injection.
Use of oral analgesics.

*The values are given as mean of total dosage given and standard deviation.
There were 2 subjects in the intervention group (5.3%) and 1 in the control group (2.6%) who developed delirium after the recruitment and before the end of study. However, the difference was not statistically significant (P = 0.51). No other complications or side effects attributable to the injection were noted during the study period.
Discussion
Geriatric patients with acute hip fractures often suffer significant pain, and poor pain control can result in a multitude of complications.4,5 Pain management in these patients in most of the hospitals in Hong Kong is largely reliant on oral systemic analgesic. 21 Despite the growing body of evidence demonstrating its benefits, FICB is not commonly used as a pre-operative analgesic adjunct in our region.3,9,10,22
To our knowledge, this study is the first double-blinded randomized controlled trial on the effect of FICB conducted in Hong Kong. Our study aimed to evaluate the effect of FICB as a pre-operative pain control to our local patients. The randomization successfully made the two groups comparable in terms of demographics and baseline characteristics, despite a significant difference in the distribution of patients’ ASA class, with a larger proportion of ASA II included in the intervention group. Nevertheless, to our knowledge, there is no proven correlation between ASA class and analgesia effect or requirement.
Apart from the inherent pain due to the fracture itself, pain could be inevitably induced by myriads of pre-operative patient care activities, including regular turning for pressure injury prevention, changing diapers, and transferring to radiographic examinations or to operations. Therefore, both pain score at rest and on gentle movement were chosen to be our outcome measurements. The baseline pain scores on admission and before the injection in the two groups were similar and comparable, making the changes in pain scores after the injection more representative. Our study demonstrated significant differences in pain scores both at rest and on gentle movement after the injection. In the intervention group, the mean pain score at rest was significantly reduced from 2.9 to 1.7 at 12 h after the injection, a 41.4% reduction. Similarly, in the intervention group, the mean pain score on gentle movement was significantly reduced from 7.7 to 5.6 at 1 h after injection, to 5.4 at 12 h after injection, and to 5.9 at 24 h after injection. There was up to 28.9% reduction at 12 h after injection. In other words, the reduction in pain may last up to 12 h after injection at rest, and up to 24 h on gentle movement. The more long-lasting reduction of pain on gentle movement demonstrated the effectiveness of FICB in controlling pain induced by routine nursing care. Our results echoed some of the previous studies. Candal-Couto et al. reported significant improvements in 18 patients’ visual analogue scores from 7.2 to 4.6 (36% reduction). 18 Hanna et al. reported a significant pain reduction up to 8 h after FICB. 3
However, the pain reduction effect demonstrated in our study may not be as significant as some other previous studies. Fujihara et al. reported a 60% reduction in pain scores at 12 h after FICB. 9 From the study of Godoy Monzon et al., a single administration of FICB provided a 48%–73% reduction in pain scores. 23 Nonetheless, the actual pain relief effect in these studies may be inherently biased due to their unblinded and non-randomized study designs.
A double-blinded randomized trial by Foss et al. reported a low success rate of 67% for blocks using the landmark approach under double-blinded setting. 11 The author proposed that previous unblinded studies potentially tended to overestimate the success rates of the block. One unique aspect of our study was the use of ultrasonographic guidance for FICB, in addition to the conventional landmark approach. The correct placement of the anesthetic agent was assured, and therefore the safety and blockade success rate were enhanced. As a result, our study findings reflected the true effect of FICB.
Our study experienced some patient drop-out at 24 and 48 h after injection due to the earlier termination of the study when patients were sent for definitive hip fracture surgery. This may have resulted in the insignificant difference in pain scores between the two groups at these later time intervals.
Contrary to our postulation, our study could not conclude a significant difference in the pre-operative total consumption of systemic analgesics, especially opioids, between the two groups. Foss et al.'s results showed significantly less morphine consumption in the FICB group. 11 This discrepancy could have several explanations. First, part of our oral analgesic protocol involved pro re nata administration of paracetamol and tramadol. The total consumption may be influenced by the discretion of nursing staff who assessed the pain relief adequacy and administered pro re nata analgesics. Second, the mean time from injection to operation in the intervention group and the control group was 57.2 h and 63.1 h respectively, which were relatively longer than the proposed effective time of FICB in our study (up to 24 h). The effect of FICB on less consumption of oral analgesics may be neutralized by the longer time from injection to operation.
No procedure-related side effects or complications were observed in our study. Respectively 5.3% and 2.6% of patients in the intervention group and the control group developed delirium before end of study. Delirium is a known common complication of hip fracture, with a reported incidence of 16% in patients with hip fractures by Morrison et al. 6
There were several limitations in our study. Patients with dementia or being delirious at the time of admission were excluded from our recruitment for ethical considerations and to ensure more reliable patient-reported outcomes. This could limit generalizability of our findings, as it may underrepresent the actual population of geriatric hip fractures. Furthermore, our study was limited in its power to correlate the timing of block with the pain relief effect and pre-operative use of opioids. A more recent study by Garlich et al. concluded that a faster time-to-block (less than 8.5 h from emergency department arrival to block) resulted in less pre-operative opioid use and a lower pain score on post-operative day 1, but not pre-operative pain score. 24
In conclusion, our study demonstrated that FICB was effective in pre-operative pain relief in geriatric patients with hip fractures in our locality. The procedure is considered safe and practical in the setting of an acute orthopaedic ward. We suggest FICB be routinely performed, if not contraindicated, to improve the quality of pain management in addition to the existing protocols for these patients, and hence to improve outcomes. While the effect of reduced consumption of oral analgesics has not been proven, we recommend further high-quality research with more controlled factors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
