Abstract
Keywords
Introduction
Total knee replacement (TKR) is considered as a safe and effective treatment against knee osteoarthritis over the last few decades. The longevity of TKR has increased with the advancement of implant design and surgical techniques. Yet, there is a tendency towards the application of unicompartmental knee replacement (UKR) in cases with unicompartmental pathology. Many studies have shown superior clinical outcomes in UKR including shorter hospital stays, reduced blood loss, shorter operative time, reduced knee pain and faster return to the normal daily routine. The survivorship of UKR is up to 97.2% and 94.2% at 5 and 10 years, respectively.1,2
With good patient selection and techniques, 3 the clinical outcome is satisfactory. However, an early study in France in 2012 4 which looked at 418 failed mobile and fixed bearing UKRs, aseptic loosening accounts for 44% of these cases, with a mean failure time of 6 years. Tibial implant loosening accounts for 24% of loosening cases. Identifiable causes include malpositioned and defective implant, insufficiency of subchondral support either due to fracture or osteolysis and malcorrection of the mechanical axis.4,5 Although failure rates have slowly reduced over time with better design and surgical techniques, complications from UKR should be prevented due to difficult revisions, especially in cases with significant bone loss.
Medial tibial bone loss (MTBL) is a common phenomenon after replacement surgery. Different studies in TKR demonstrated MTBL range from 13% to 45%.6–8 while in UKR studies, MTBL range from 4% to 29%.9–12 Bone loss is most prominent in the first 2 years and the effect plateaued. MTBL can potentially lead to premature implant loosening and therefore early revisions. For mechanical theory, stress shielding is widely accepted as the major contribution to MTBL. Once the bearing load is reduced and transferred to the tibial implant after deformity correction, the periprosthetic bone undergoes remodelling into an osteopenic state following Wolf's law. 13 The MTBL difference in TKA and UKR can possibly be explained by a smaller tibial implant and less deformity correction in UKR. For physiological theory, that is, the bone resorbs in response to trauma from surgery, that is, bony resection as proposed by Lee et al. 12 The smaller MTBL in UKR could be explained by a smaller area of bone resection.
Due to the aforementioned difference in MTBL in TKA and UKR, this study was conducted to compare and identify any significant MTBL difference. We hypothesized that there is more medial tibial bone resorption in TKR than in UKR.
Materials
The study was conducted in a retrospective cohort manner with data collected from a local joint centre registry. Ethical approval was obtained from the local institutional review ethics board. Two groups of subjects are recruited and categorised into UKR and TKR. Zimmer® Unicompartmental High Flex Knee (ZUK) system, fixed bearing implant was chosen for UKR cases, while Triathlon Total Knee System was chosen for TKR cases. These replacement surgeries were carried out by a senior joint consultant between January 2015 and December 2016. Data collected were further stratified according to the inclusion and exclusion criteria. Inclusion criteria include patient age older than 18 years old, varus knee deformity on preoperative standing film, and radiological evidence of Kellgren-Lawrencegrade two osteoarthritis or osteonecrosis. Exclusion criteria include post-traumatic or post-infection knee deformity, revision cases, intra-operative complications such as iatrogenic fracture, patient with a follow-up period of less than 36 months, and missing data for analysis.
Initially, there are 82 cases of TKR and 48 UKR cases recruited for analysis. After stratification, 48 cases of UKRs and 66 cases of TKR were collected for the study (a reference to the study flowchart).
These patients were monitored with post-operative X-ray films at 3 or 6 months, 1, 2 and 3 years in the clinic. The medial tibial bone density was measured from an X-ray film at the corresponding time period as the primary outcome.
Patient demographics, including age, sex, body mass index (BMI), preoperative knee range of motion (ROM), mechanical femoral–tibial alignment (MFTA), and Knee Society Scores (KSS) were collected. Post-operative (at 6 months) ROM, MFTA, and KSS were collected as secondary outcomes.
Method
Radiographs were taken according to the local centre follow-up protocol and studied in a retrospective cohort manner. Long-standing radiographs were taken preoperatively and post-operatively (at 3 months) to measure the mechanical femoral–tibial alignment. Standard anterior–posterior (AP) radiographs were then further taken immediately after the operation, 3 or 6 months, first year, second year and third year accordingly. Medial tibial bone densities were measured on serial radiographs. AP radiographs were considered standard if the posterior femoral condyles were not visible, partial overlapping of the fibular head with lateral tibia condyle and the superior position of the patella.
In past literature, medial bone loss is defined as the change in distance from the medial tibial plateau to the medial edge of the tibial plate on coronal view. However, the change was too subtle for quantification on radiographs and was not representative of actual bone loss. Therefore, medial tibial bone density was used as a quantitative measurement. In normal circumstances, a dual energy X-ray absorptiometry (DEXA) scan is the gold standard for bone density measurement.14,15 However, the DEXA scan is not readily available in all outpatient centres. Radiographs were studied instead to estimate bone density. Radiographs were viewed and processed by the image-viewing software Centricity Enterprise Web 4.0 (GE Healthcare IT, USA). The method of digital radiological densitometry (DRD) was employed to measure the serial medial tibial bone density.
Each radiograph carries a pixel value, ranging from 0 to 4096 on each point of the image following a linear scale. After obtaining the pixel values, the values are further converted into grayscale (Gy) for comparisons. Grayscale is represented on a linear scale of 0–255 Gy.
DRD is a method to measure density based on historic radiographs. It is first described by Hernandez-Vaquero et al. 14 in 2005. In his study, he took standard radiographs of knees with a digital camera and analysed data with a computer program. DEXA scan for the same knees was also performed for comparison. It produced a comparable result with Cronbach alpha of 0.84–0.93 and intraclass correlation coefficient of 0.72–0.87. Details of the rationale of the method can also be found in previous studies on UKA and TKA using the DRD method by our institute.8,12
In order to standardise the calibration in a linear scale, we assume the femoral implant (Pf) has the maximum pixel value whereas the surrounding air (Pa) has the least. Then we measure the mean pixel value of the region of interest (Pr) which is the medial tibial bone plateau. We defined the area from 5 mm below the tibial baseplate to the metaphyseal region. The extent of the metaphyseal region is defined by the Heims system of the square. Pixel values are then converted to grayscale by the following formula. Mean medial tibial bone density = 255 × (Pr – Pa)/(Pf – Pa) (Figure 1).
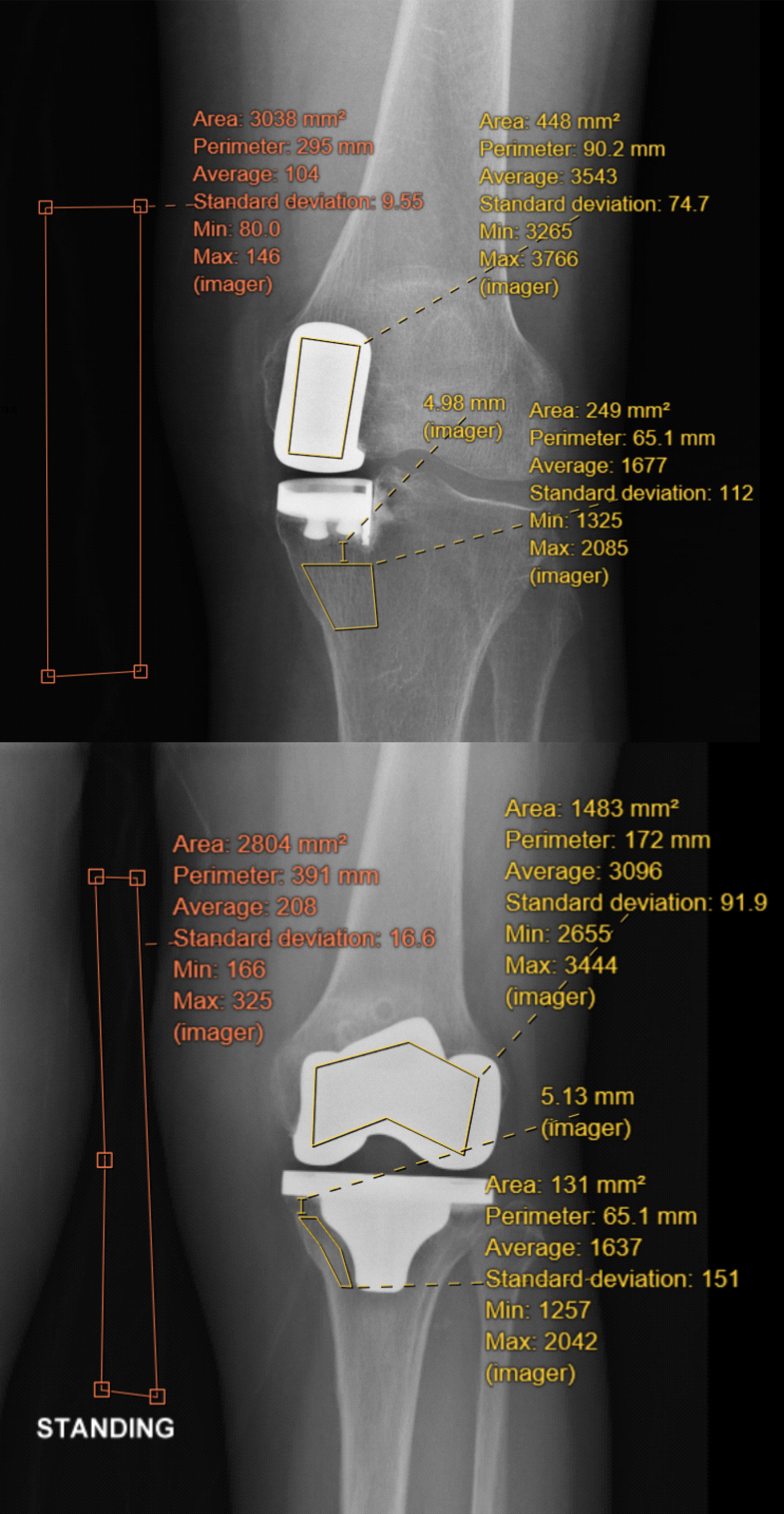
Demonstration of measurement of pixel values on X-ray for both unicompartmental and total knee replacement. Femoral implant (Pf), Region of interest (Pr) which is 5 mm under the tibial implant, air (Pa). Average values were used for calculation.
We defined the area of the region of interest (Pr) as from 5 mm below the tibial baseplate to the metaphyseal region. This area is chosen because bone density dropped most dramatically over time in past studies, (6, 7, 10–12, 16) which makes it representable for comparisons. We aimed to reduce selection bias by measuring the mean value of a targeted area. Also, by measuring 5 mm below the implant, we eliminated the Uberschwinger artefact, where the black halos around the implant may be mistaken as loosening or bone loss. All the measurements were performed by a single physician.
The primary outcome is the estimated medial tibial bone densities of both TKA and UKA at immediate post-operation, 3 or 6 months, 1, 2 and 3 years, respectively. Percentage loss as compared to baseline, that is, immediate post-operation, will be calculated for interpretation. Secondary clinical outcomes including post-operative range of motion, alignment correction and KSSs were collected for comparison.
Data analysis
Based on the studies performed by our institution,8,12 we estimated the MTBL difference between TKA and UKA to be 15%. A sample size of at least 17 subjects per group is required to achieve 80% power to detect differences at a significant level of 5%. Means were calculated from the demographics and clinical outcomes. Student t-tests were performed to assess any significant mean differences between the two study groups. Multiple linear regression was performed to identify and adjust for the different confounding factors leading to changes in MTBL. Spearman correlation was also used to look for a relationship between bone density losses over time. IBM SPSS Statistics Version 26 was used to perform the above statistical analysis. Collected data were analysed by a single clinical statistician from our institution.
Results
A total of 66 TKR cases and 48 UKR cases were included in the study after following the selection criteria. During data collection, a total of five cases (two UKRs, three TKRs) were found to have a follow-up period of less than 36 months and there were 13 cases (two UKRs, 11 TKRs) with missing data including standardised X-ray films for analysis (i.e. non-applicable pixel reading for measurement, absence of X-ray films at follow-up). Therefore, only 44 UKR cases and 52 TKR cases were included in the data analysis. The number of study cases satisfies the sample size requirement for statistical analysis. Please refer to Figure 2 for the study flowchart.
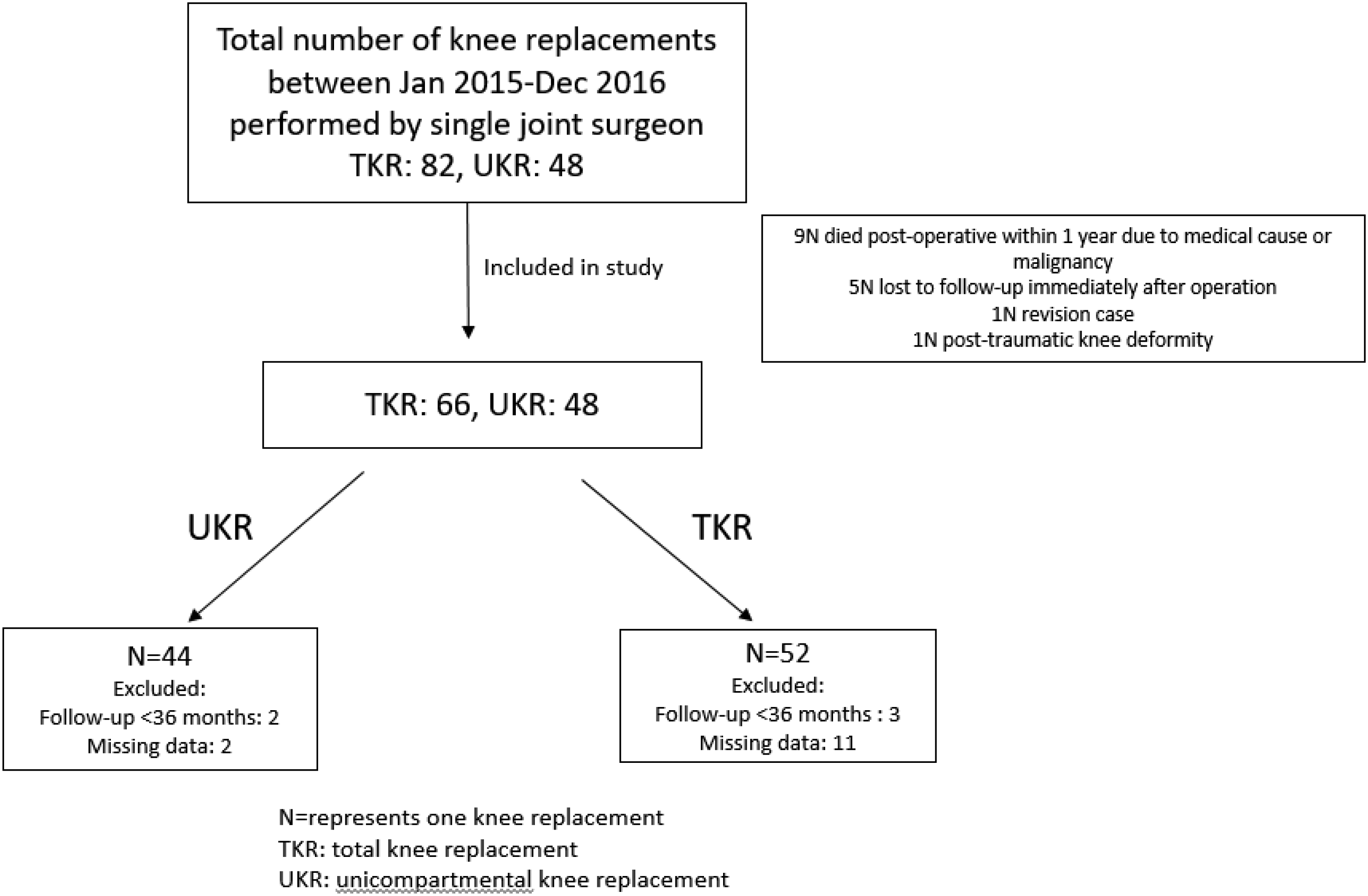
Study flowchart.
Demographics including age (p = 0.2), gender (p = 0.962) and pre-op mechanical femoral–tibial alignment were comparable (Table 1). However, BMI, pre-operative knee range of motion and pre-operative KSS showed significant differences between the two groups. Please refer to the table for reference.
Demographics of patient groups.
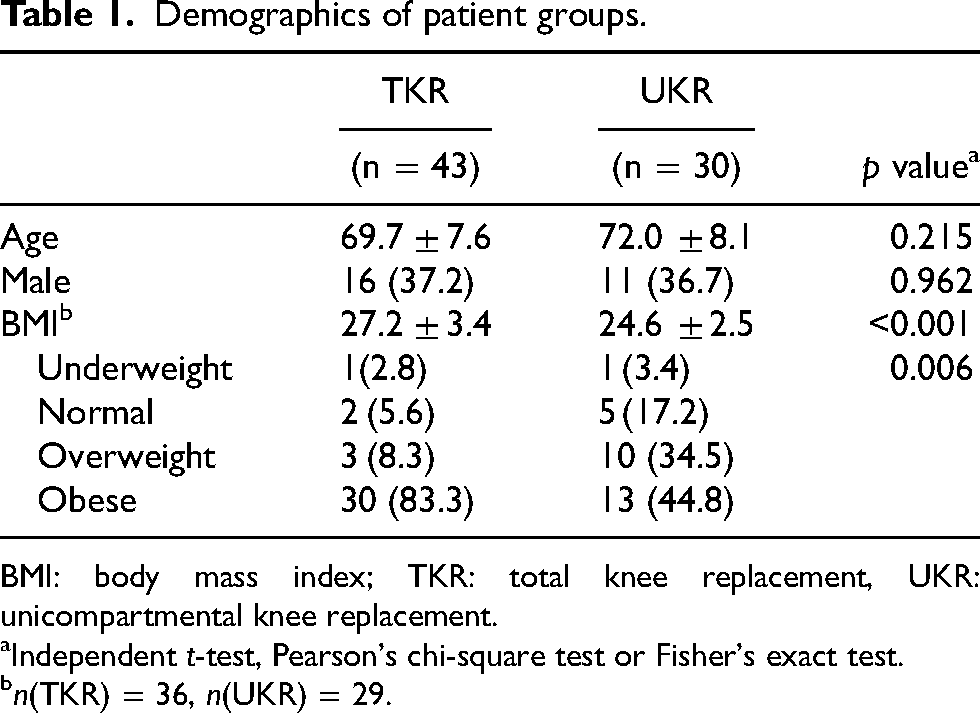
BMI: body mass index; TKR: total knee replacement, UKR: unicompartmental knee replacement.
Independent t-test, Pearson's chi-square test or Fisher's exact test.
n(TKR) = 36, n(UKR) = 29.
Primary outcome (medial tibial bone density)
Both groups showed a progressive drop in medial tibial bone density following 3 years after the index operation. The cumulated drop in 3 years was 33.7% ± 12.9 in TKR and 23.3% ± 13.1 in UKR, respectively. The percentage drop was compared to the immediate post-operative film at each time interval. The cumulative drop between the two groups at 12 months (p = 0.009) and 36 months (p = 0.002) were significantly different (Table 2, Figure 3). The drop was most dramatic in the first year in both groups and the drop plateaued in the later 2 years. Bone densities drop over time (Spearsman correlation in TKR: 0.282, UKR: 0.154).
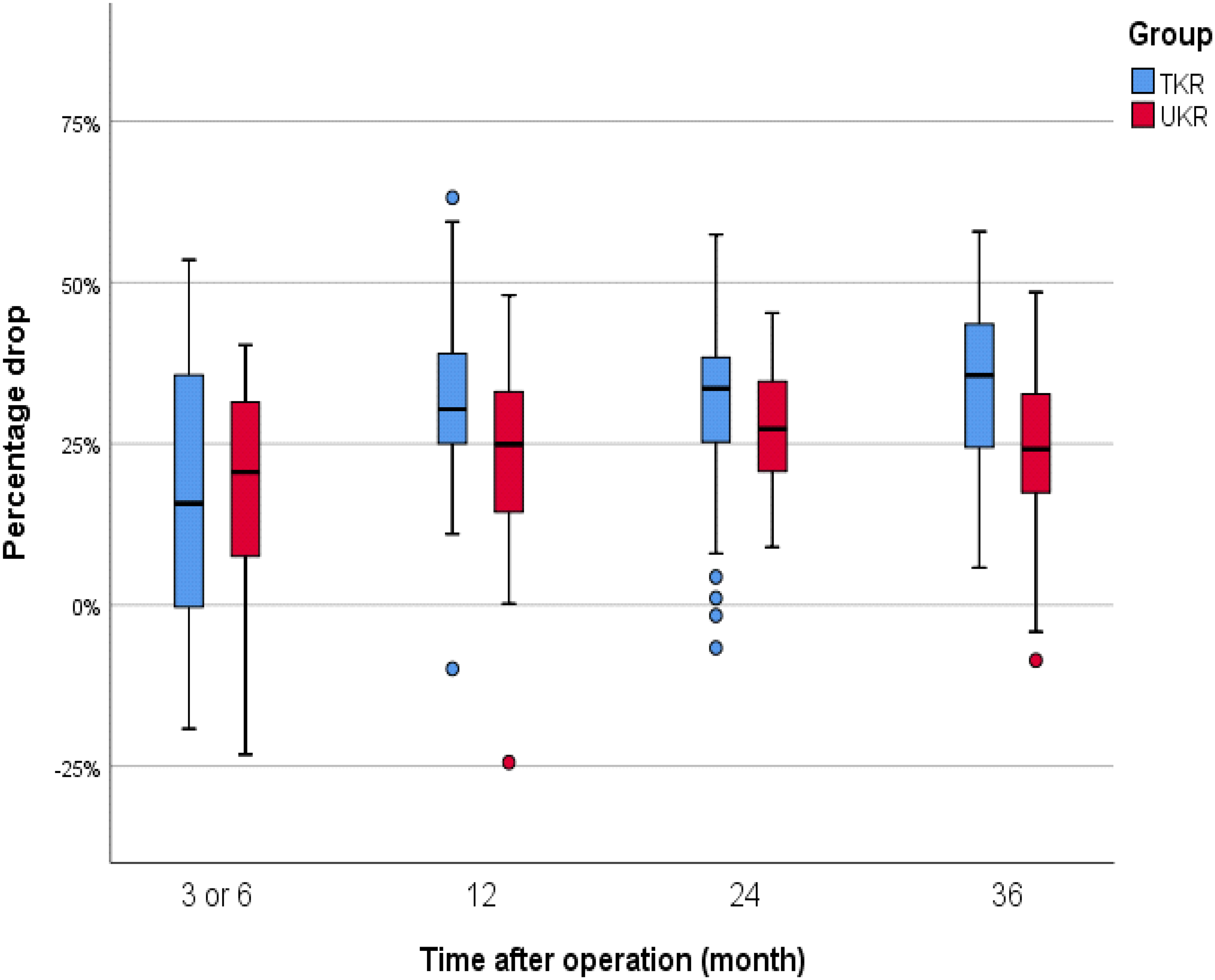
Boxplot diagram showing percentage (%) drop in bone density between TKR and UKR.
Primary outcomes: Absolute and percentage (%) bone density loss over a three year period.
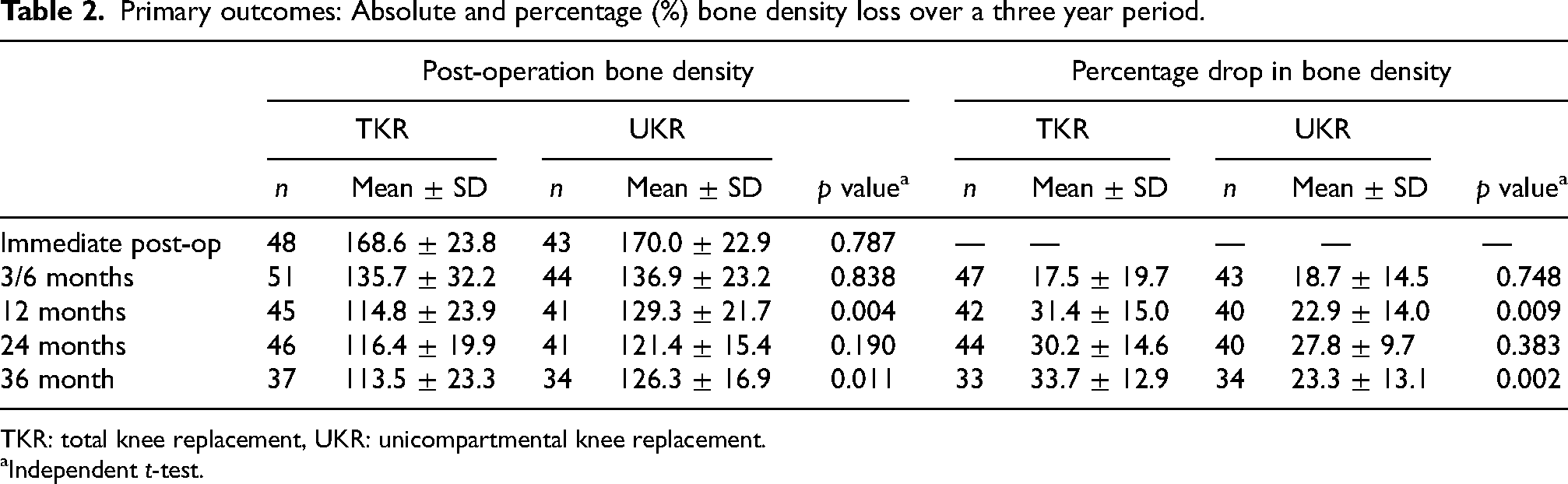
TKR: total knee replacement, UKR: unicompartmental knee replacement.
Independent t-test.
Secondary outcomes
Post-operative knee range of motion (TKR: 124.6° ± 10°, UKR: 131.6° ± 8.4°), alignment correction (TKR: 8.8° ± 7.8, UKR 5.5° ± 3.3) and post-operative KSSs (TKR: 93.1 ± 6.1, UKR: 88.3 ± 3.4) were significantly different between two groups (p < 0.001). Values are also significant after adjustment to pre-op measurement (Table 3).
Secondary outcomes: clinical outcomes between TKR and UKR.
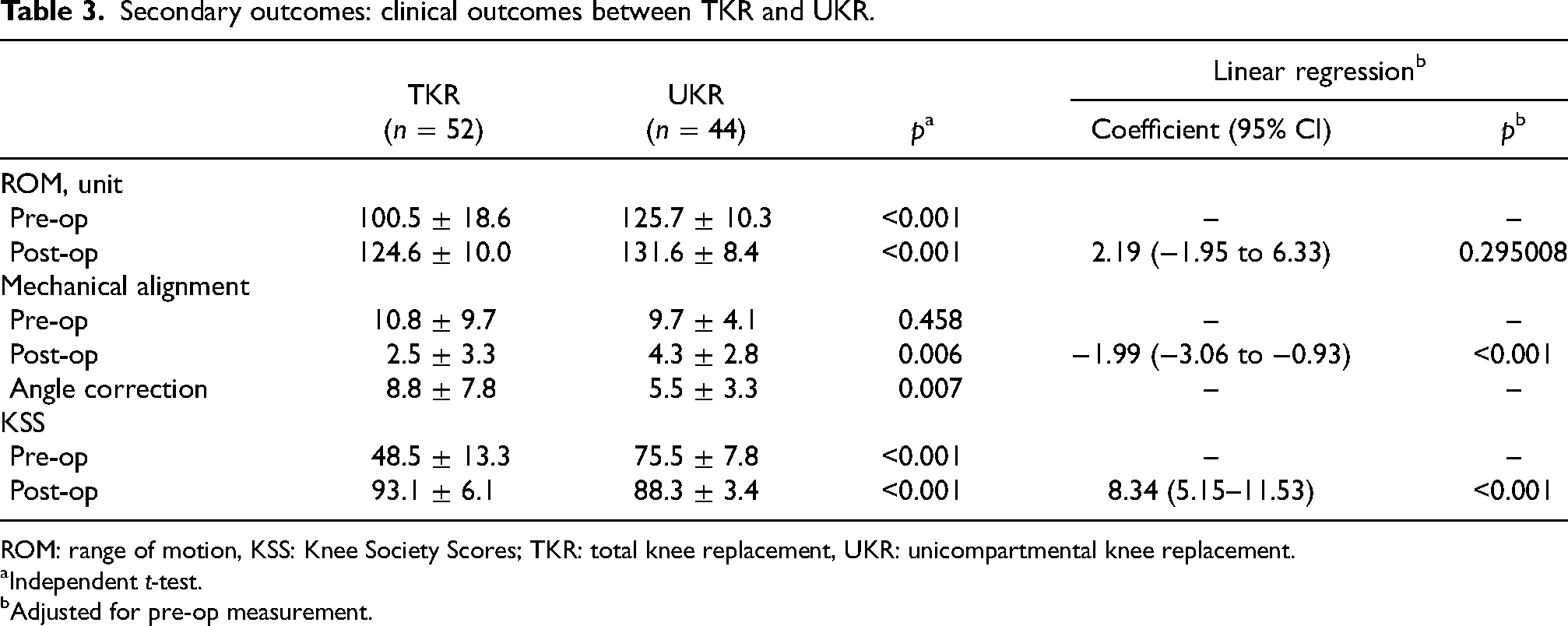
ROM: range of motion, KSS: Knee Society Scores; TKR: total knee replacement, UKR: unicompartmental knee replacement.
Independent t-test.
Adjusted for pre-op measurement.
Taking into account the significant difference in UKR and TKR MTBL, possible confounding factors including BMI, pre and post-operative knee ROM, pre and post-operative KSS, as well as angle correction were adjusted into multiple linear regressions. Only BMI showed a coefficient of 1.38 (p: 0.05) (Table 4).
Association with % drop in bone loss by multiple linear regression.
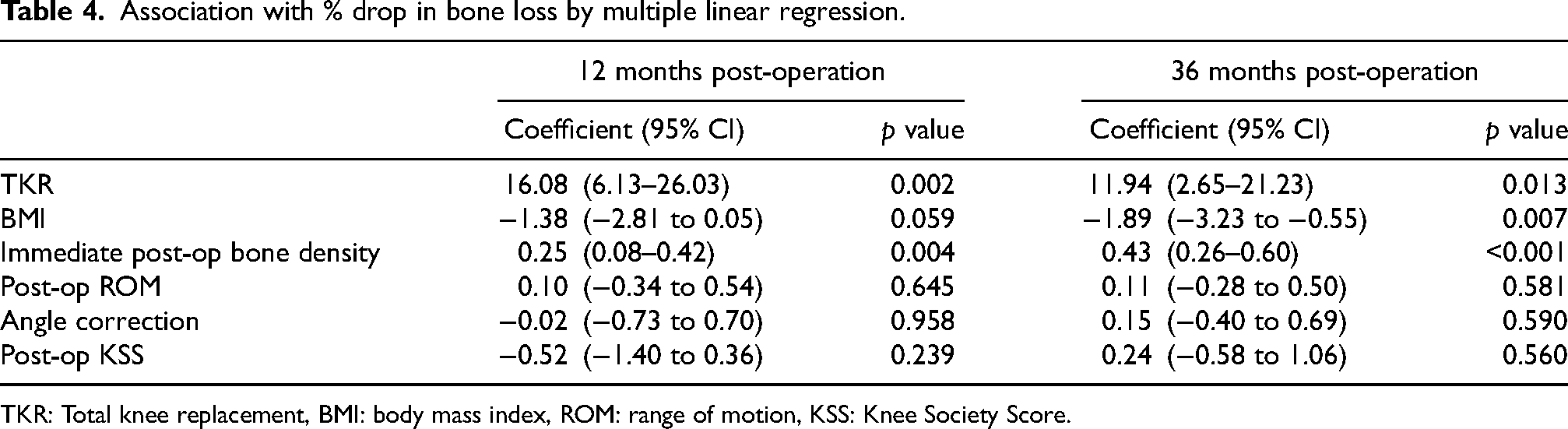
TKR: Total knee replacement, BMI: body mass index, ROM: range of motion, KSS: Knee Society Score.
Complications
No case of loosening of implants or periprosthetic infections was documented in both groups. In UKR, only one case required conversion to TKR due to the progression of lateral knee osteoarthritis 3 years later.
Discussion
In conclusion, the UKR group is generally superior in the preservation of bone stock under the medial tibial tray than the TKR group. Our study has shown a cumulative drop of 33% and 23% in TKR and UKR, respectively. The figures were comparable to findings from previous studies. Compared to TKR, UKR has a 10% reduction in medial tibial bone loss. The difference was statistically significant at 1 year (p = 0.009) and three-year time (p = 0.002). The drop progressed over time in a non-linear fashion was also consistent with other studies’ findings.
From past studies, the pathophysiology behind MTBL was believed to be bone remodelling after deformity correction in TKA. On the contrary, we did not find a correlation between alignment correction and MTBL (0.02, p: 0.95). Alignment correction was believed to play an important role in stress shielding, according to Zhang et al. 13 Interestingly, a recent finding from Lee et al., 12 found that the pattern of bone density loss in UKR is actually greatest at medial (26%) and anterior (29%) region which corresponded to the area of surgical resection. He proposed that it is the bone resection that causes physiological bone remodelling and therefore contributed to major bone resorption in the early post-operative period. As the area of bony resection in TKR is higher, it accounts for the major difference in MTBL between UKR and TKR in our study.
Although there is no concrete evidence to show medial tibial bone loss has direct effect on aseptic implant loosening, most studies only observe short post-operative bone density changes which may fail to identify possible long-term loosening. 16 From the study of Epinette, 4 we learnt that the mean failure time can be up to a six-year period. By extending the observation period, we can study the correlation between MTBL and aseptic loosening in TKR and UKR. Implant stability and survival may be jeopardised if peri-prosthetic bone loss is significant in the long term. It will have a clinical impact on the choice of replacement in patients with unicompartmental knee pathology.
Nevertheless, there are other potential contributing factors leading to MTBL. Implant factors such as implant material, design, thickness, and positioning are studied but the results are inconclusive.17–19 A study from our institution (8) concluded that the thickness of the tibial implant did not have an effect on MTBL.
There are a few limitations of the study. First, as discussed above, the study period is not long enough to identify any implant loosening and therefore we cannot draw a conclusion on the relationship between MTBL and loosening. As a retrospective cohort study, interobserver biases during X-ray film analysis were inevitable, despite strictly following the guidelines. Point bias was minimised by selecting a mean pixel through the computer software over the region of interest for calculation instead of a specific point of interest Thirdly, there were missing X-rays or X-rays that were not amenable for analysis over the study period. Taking this into consideration, we have adjusted the statistical analysis and are able to generate the same regression result. In our study, BMI in the TKR group was significantly higher. Out of the 43 cases, 30 cases reached the obese level, that is, ≥ 27 according to the Asian BMI scale. There are two ways in how BMI can interfere with bone mineral density. Obese patients create a greater load and the stress shielding effect becomes greater when the load is shared by the implant. Obesity is also predisposed to osteoporosis, therefore they have a higher bone turnover rate. Either way, BMI is not a favourable confounder in our study. Last but not least, mobile bearing UKR was not included in the study since the knee biomechanics are different. This minimises confounders.
Conclusion
Both TKR and fixed-bearing UKR showed a cumulative reduction in medial tibial bone loss 3 years after surgery. TKR produced 10% greater medial tibial bone loss than UKR. The reduction of bone loss in both replacements was most dramatic in the first year post-operative period. Angle correction does not correlate with bone loss. This further supported the role of physiological response to surgery with reference to Lee's finding. 12 More data is required to study the relationship between MTBL and implant loosening which have a clinical implication on long-term implant stability and therefore choice of replacement in unicompartmental knee arthritis.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
