Abstract
Background:
In this study, we want to search the effectiveness of Duloxetine on the severity of pain and quality of life in patients with chronic low back pain who had posterior spinal fixation.
Methods:
In this randomized, placebo-controlled trial done in 6 months 50 patients who had CLBP and were candidates for PSF surgery selected and divided into two groups (drug and placebo). They filled the VAS, SF-36, and Hamilton questionnaires before surgery and after 6 weeks from using 30 mg of duloxetine or placebo.
Results:
Significant differences were evidenced among groups for the Visual Analogue Scale (P = 0.005) and Verbal Analogue Scale (p = 0.003). Patients in the Duloxetine group have more visual and verbal pain scores than the placebo group. In the quality of life, there was a significant difference between the two groups before the intervention. Also, significant differences were evidenced among groups for the Hamilton Anxiety Rating Scale (p = 0.17). After the intervention, only the Hamilton Anxiety Rating Scale (p = 0.001) and ‘bodily pain’ and ‘general health’ subscales of quality of life (p = 0.008, 0.004, respectively) have a significant difference between the two groups. There was a significant difference between pre and post-intervention in the Hamilton Anxiety Rating Scale only in the duloxetine group. Also, in terms of quality of life, the subscales of ‘physical role’, ‘emotional role’, ‘physical pain’ and ‘total score of quality of life’ in the duloxetine and placebo groups were significantly different between pre and post-intervention. However, the subscales of ‘physical function’ and ‘general health’ were significantly different only in the duloxetine group between pre and post-intervention.
Conclusion:
The results suggest that the use of duloxetine in patients who had spinal surgery can help to better control back pain, on the other hand, it can cause a better psychological condition that affects the quality of life.
Introduction
Low back pain is a usual disorder with high dysfunction in physical and mental health, especially if prolonged for more than 3 months. 1, 2 The lifetime prevalence of low back pain is about 84% and if prolonged more than 3 months is considered chronic low back pain (CLBP) which prevalence is about 23%. 3 –5 CLBP can cause high disability, economic burden, and low quality of life and the patients with CLBP have significantly greater levels of depression, anxiety, and sleep problems. 1,4,6
Although the definition is clear the population with CLBP is heterogeneous and is different in demographics, quality, and manifestations of pain and radiological findings. 2,7
For the treatment of low back pain, pharmacological agents like nonsteroidal anti-inflammatory drugs (NSAIDs) are first lines then non-pharmacological therapies like spinal manipulation are used. 1,2,8 If low back pain is acute in many cases show rapid improvement within 1 month or gradual improvement in 3 months after onset. 4,6,8 Treatment with NSAIDs is effective for acute low back pain but in CLBP long-term use can cause different side effects like gastrointestinal, cardiac, or renal adverse effects. 4,6,9
Duloxetine is an analgesic agent that inhibits serotonin and norepinephrine reuptake (SNRI) with an affinity for both transporters. 10,11 In several studies show that duloxetine can help to decrease pain in chronic pain disorders like CLBP. 2,12
Although the exact mechanisms of duloxetine are unknown with increasing synaptic levels of serotonin and norepinephrine in spinal and supraspinal pathways produces an analgesic effect.3,13 With this effect, duloxetine has been approved for diabetic neuropathic pain and fibromyalgia. 2,9 On the other hand, duloxetine is a viable medication drug in the treatment of major depressive disorder and generalized anxiety disorder. 5,9 In some studies, have shown that a lot of those with CLBP have depression or anxiety that exacerbate pain, 5 therefore the use of duloxetine with treatment effect on depression and anxiety maybe can help to reduce CLBP. 2,8 The number needed to treat (NNT) for at least 50% pain relief at 12 to 13 weeks with duloxetine 60 mg versus placebo (1211 patients in the total comparison) was 5.8 (95% Cl 4.5 to 8.4) and for duloxetine 120 mg (1410 patients) was 5.7 (4.5 to 5.7).
In this study, we want to search the effectiveness of duloxetine on the severity of pain and quality of life in patients with CLBP who had a posterior spinal fixation.
Materials and methods
This randomized, prospective, placebo-controlled trial (IRCT20191210045685N1) was conducted in the neurosurgery department of Rasoul-e Akram hospital in Tehran for 6 months (started from 2018/09/01). In this period, the patients admitted to the clinic and had chronic low back pain (CLBP more than 3 months) and candidate for posterior spinal fixation (PSF) if met inclusion criteria were randomized divided into two groups (duloxetine or placebo).
In the first step, all patients filled the VAS and SF-36 and Hamilton questionnaires. Then all of them had PSF surgery and on first day post operation when the patient could take oral drugs the drug group received 30 mg of duloxetine and the placebo group received a placebo for 6 weeks. After this time all the patients again filled the questionnaires. 70 cases were seen and 30 cases were excluded because of exclusion criteria (like depression, drug abuse, using antidepressants, severe hypertension and convulsion).
From total 40 cases who were included to the study and divided randomly in duloxetine and placebo groups, three patients from drug group and two from placebo dropped out (because of drug adverse effects) and four cases (two from each group) didn’t want to continue the study.
Informed consent was obtained from all patients before the trial starting and the protocol was approved by the ethics committee (IR.IUMS.FMD.REC.1398.016).
Inclusion and exclusion criteria
Patients older than 18 years old and younger than 80 with CLBP (more than 3 months’ duration) who were candidates for PSF surgery. All patients had to discontinue any medication that could interfere with their pain such as nonopioid or opioid drugs, antidepressants, and anticonvulsants at least 6 months before surgery.
Non-pharmacological pain-relieving procedures such as acupuncture or physical therapies were not allowed during the study.
Exclusion criteria were as follows: prior use of opioids, depression, and use of antidepressants, drug abuse, pregnancy and breastfeeding, severe coexisting diseases such as heart failure, severe hypertension, convulsion, and kidney dysfunction.
Treatment protocol and instruments
Patients included in this study randomly divided into two blinded groups (Simple or unrestricted randomization by coin tossing method was used and based on the sample size, the same number of coins were tossed and individuals were randomly divided into two equal groups) and after PSF surgery was treated with once-daily 30 mg oral duloxetine or placebo for 6 weeks. The VAS, Hamilton and SF-36 questionnaires filled two times first before the surgery and second at the end of the trial. If the patients had adverse effects of drugs were excluded from the study.
VAS questionnaire
The visual analogue scale or visual analogue scale (VAS) is a scale used for the subjective response for indicating the level of agreement to a statement by showing in a continuous line between two. VAS is the most common pain scale. Although VAS and numerical rating scales (NRS) are the best in pain measurement in clinical researches VAS is suggested.
Short-form-36 Health Survey
This scale is used for quality-of-life assessment and reported by the patient. The following eight domains are vitality, physical functioning, bodily pain, general health perceptions, physical role functioning, emotional role functioning, social role functioning, and mental health.
Hamilton Anxiety Rating Scale
The Hamilton Anxiety Rating Scale (HAM-A) is a questionnaire that shows the severity of anxiety. The reasons for anxiety can be different things such as ‘a mental state a drive a response to a particular situation a personality trait and a psychiatric disorder’. It was originally published by Max Hamilton in 1959 and it is widely used in the clinic till now.
The scale consists of 14 items designed to assess the severity of a patient’s anxiety. Each of the 14 items contains several symptoms, and each group of symptoms is rated on a scale of zero to four, with four being the most severe. All of these scores are used to compute an overarching score that indicates a person’s anxiety severity. The Hamilton Anxiety Rating scale has been considered a valuable scale for many years, but the ever-changing definition of anxiety, new technology, and newly perceived usefulness. As a result, there have been changes, and challenges, to the original version of the scale over time.
Results
Demographic characteristics are represented separately per each group in Table 1. Groups did not differ for gender distribution, age, education, and marital status (P > 0.05). According to the Kolmogorov–Smirnov test, distribution of the data was nonparametric, so the Mann–Whitney U test was used to compare the duloxetine and placebo groups and the Wilcoxon signed-rank test was used to compare the results before and after the intervention.
Demographic characteristics, Visual Analogue Scale, Verbal Analogue Scale, Hamilton Anxiety Rating Scale, and quality of life of the participants before intervention P < 0.05.
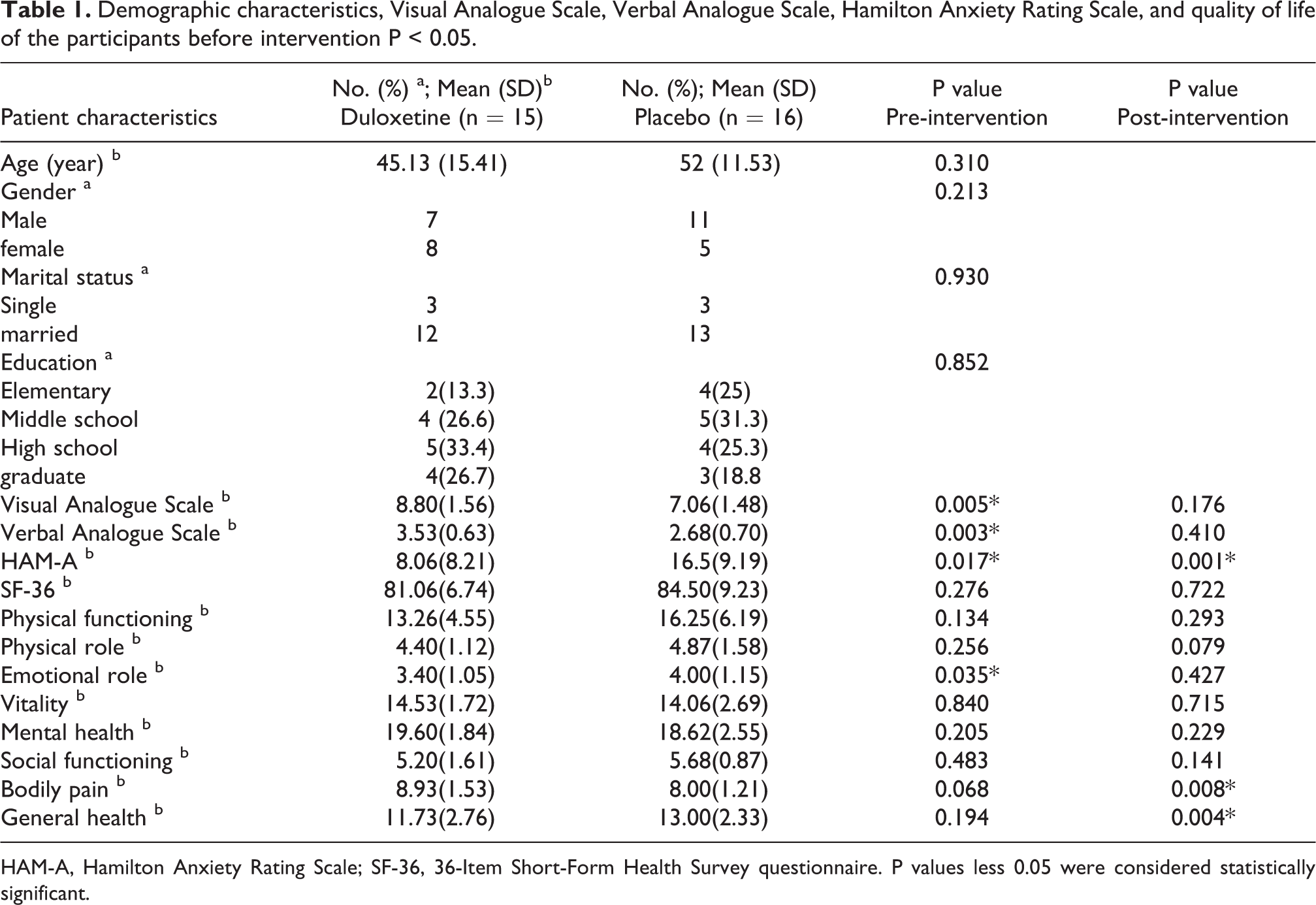
HAM-A, Hamilton Anxiety Rating Scale; SF-36, 36-Item Short-Form Health Survey questionnaire. P values less 0.05 were considered statistically significant.
As shown in Table 1, the following results were also obtained regarding the difference between the two groups before the intervention: Significant differences were evidenced among groups for the Visual Analogue Scale (P = 0.005) and Verbal Analogue Scale (p = 0.003). Patients in the Duloxetine group have more visual and verbal pain scores than the placebo group.
Also, significant differences were evidenced among groups for the Hamilton Anxiety Rating Scale (p = 0.17). Patients in the placebo group have more anxiety scores than the duloxetine group.
In terms of quality of life, there was a significant difference between the two groups before the intervention on the ‘Emotional role’ subscale (p = 0.035).
After the intervention, only the Hamilton Anxiety Rating Scale (p = 0.001) and ‘bodily pain’ and ‘general health’ subscales of quality of life (p = 0.008, 0.004, respectively) have a significant difference between the two groups.
The results after the intervention in the two groups of duloxetine and placebo as well as the comparison of the results of each group before and after the intervention are shown in Table 2.
The results after the intervention in the duloxetine and placebo, and the comparison of the results of each group before and after the intervention.
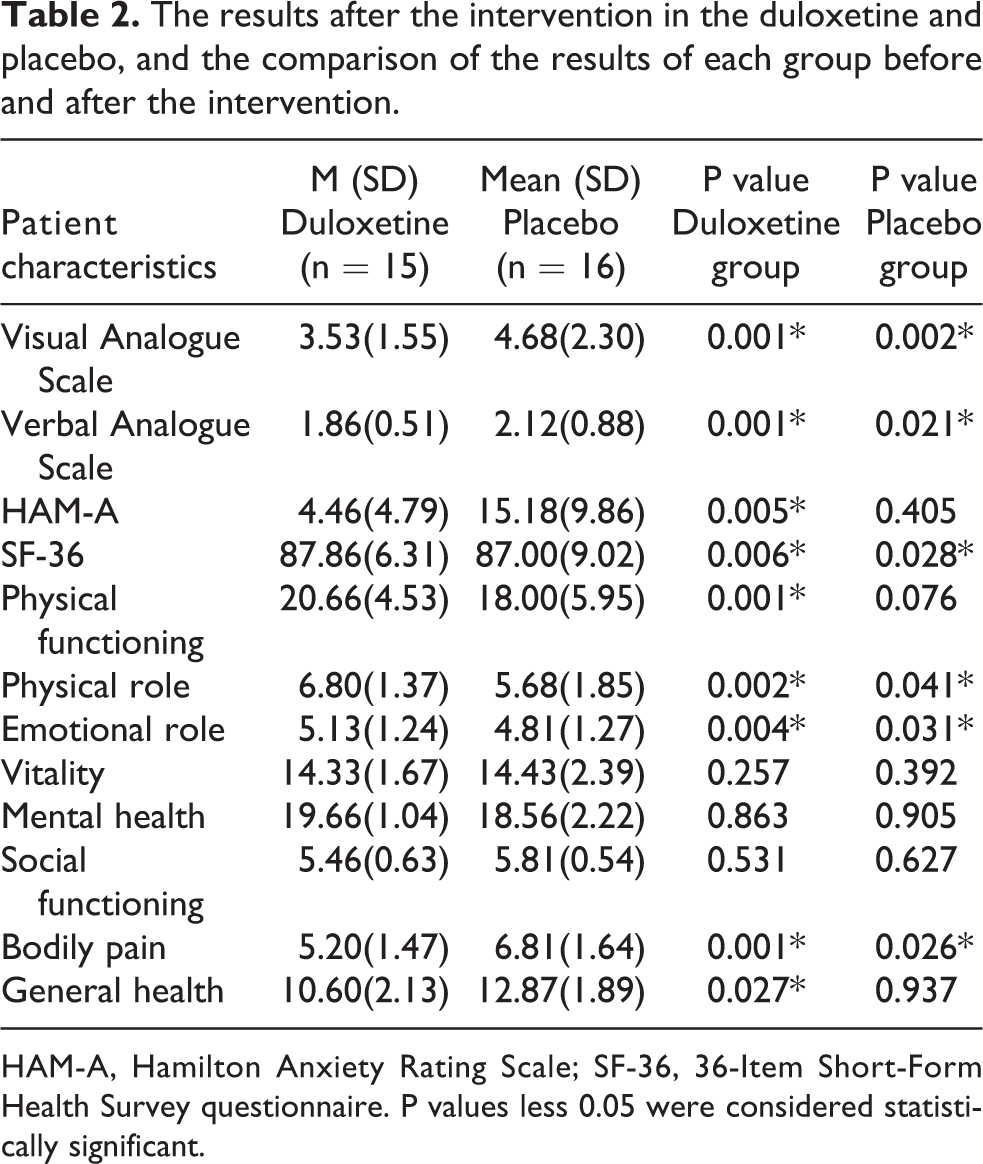
HAM-A, Hamilton Anxiety Rating Scale; SF-36, 36-Item Short-Form Health Survey questionnaire. P values less 0.05 were considered statistically significant.
Using the Wisconsin test, it was shown that there was a significant difference between the visual and verbal VAS scores in both duloxetine and placebo groups between pre and post-intervention. Data analysis showed that there was a significant difference between pre and post-intervention in the Hamilton Anxiety Rating Scale only in the duloxetine group.
Also, in terms of quality of life, the subscales of ‘physical role’, ‘emotional role’, ‘physical pain’ and ‘total score of quality of life’ in the duloxetine and placebo groups were significantly different between pre and post-intervention. However, the subscales of ‘physical function’ and ‘general health’ were significantly different only in the duloxetine group between pre and post-intervention.
Discussion
Spinal fixation is normally a successful treatment for breaks, deformations, or precariousness in the spine. 3,13 Be that as it may, study results are progressively blended when the reason for the back or neck torment is hazy. 1,4,6 As a rule, spinal fixation is not any more powerful than nonsurgical medications for vague back pain. 1,2,8 Spinal fusion is surgery to permanently connect two or more vertebrae in the spine, eliminating motion between them. 2,8 Spinal fusion includes procedures intended to emulate the typical recuperating procedure of broken bones. During spinal fusion, the surgeon places bone or a bonelike material within the space between two spinal vertebrae. Metal plates, screws, and bars might be utilized to hold the vertebrae together, so they can mend into one strong unit. 10,14
Spinal systems are for the most part connected with extreme agony in the postoperative period, particularly for the underlying not many days. Sufficient torment of the board in this period has been believed to relate well with the improved utilitarian result, early ambulation, early release, and forestalling the advancement of constant torment. A different cluster of pharmacological choices exists for the successful improvement of post-spinal medical procedure torment. 15 –17 Every one of these medications has natural focal points and inconveniences which limits their widespread materialness.
The tricyclic antidepressants are an intricate gathering of medications that have focal and fringe anticholinergic impacts and narcotic impacts. 17,18 They affect pain transmission, and they obstruct the dynamic reuptake of norepinephrine and serotonin. 11,17
A review diagram audit was led in a subset of patients with ceaseless low back pain and lumbar spinal stenosis made do with low portion tricyclic antidepressants. Of 26 patients, 20 detailed improvements in back pain. Most of the patients revealed improvement with an underlying portion of 10 mg of either amitriptyline or nortriptyline and stayed on this portion. 17,18 Patients with both leg and back pain revealed improvement in a more prominent extent than patients with back pain alone. 17,18 As indicated by this investigation tricyclic antidepressants have all the earmarks of being powerful in controlling lumbar spinal stenosis side effects in this patient populace. 16,19
As of late, antidepressants, for example, duloxetine, a specific serotonin and norepinephrine reuptake inhibitor (SSNRI), have achieved help with discomfort in steady and incessant pain as in fibromyalgia, postherpetic neuralgia, diabetic neuropathy, osteoarthritis, and musculoskeletal agony.10,11,17 The analgesic effect of duloxetine is attributed to its ability to enhance both serotonin and norepinephrine neurotransmission in descending inhibitory pain pathways. 15,16 Moreover, some studies have promoted its use to improve the quality of recovery after surgery and reduce the acute postoperative pain after knee replacement surgery, mastectomy, hysterectomy, and after spine surgery.15,16,20 Likewise, it can improve the postoperative nature of recuperation through temperament improvement that can be useful in the postoperative period.15,16
In one randomized clinical trial that done to evaluate the effect of Duloxetine on Pain, Function, and Quality of Life Among Patients With Chemotherapy-Induced Painful Peripheral Neuropathy in 230 cases at eight national cancer institute between 2008 and 2011 showed that at the end of the treatment patients in the duloxetine group reported a larger decrease in average pain (mean change score, 1.06; 95% CI, 0.72–1.40). In the patients treated with duloxetine, 59% reported any decrease in pain vs. 38% in the placebo group. 15,21
At the end of the treatment when compared with placebo, patients treated with duloxetine reported a greater decrease in the amount that pain had interfered with daily functioning. 15,16 About pain-related QOL improved to a greater degree for those treated with duloxetine than the placebo group. 13,21
Another Randomized, Double-blind, Placebo-controlled Crossover Trial that conducted at the outpatient clinic of Anaesthesia and Pain Therapy at the Medical University of Vienna, Austria in 120 cases for evaluating the Efficacy of Duloxetine in Chronic Low Back Pain with a Neuropathic Component demonstrated that duloxetine was efficacious in the treatment of CLBP with a clear radicular neuropathic component.7,10,14
The primary outcome parameter VAS was significantly lower in the duloxetine phase compared with the placebo phase. Although the threshold for an ‘important improvement’ in the individual patient was usually set at a reduction of 20 mm on the VAS scale, it was recognized that group differences between placebo and study medication tend to be smaller. 10,14
The presence of a neuropathic component in CLBP was associated with higher pain intensity, lower quality of life, and higher healthcare costs compared with CLBP without neuropathic pain component.7,10 Therefore, duloxetine could help because of the neuropathic component of pain. 15,16
A randomized, double-blind, placebo-controlled trials on the efficacy of antineuropathic medication in CLBP with a neuropathic component were sparse, interpretation of these results by comparison with other treatment options was challenging. 14,20
A crossover, randomized controlled trial of morphine, nortriptyline, or their combination versus placebo in patients with chronic lumbar root pain did not find a statistically significant difference between placebo and verum agents in the primary outcome parameter, which was average leg pain during the maintenance phase. 20
Another Randomized Double-blind, Placebo-controlled Trial of Duloxetine Monotherapy in Japanese world Patients with Chronic Low Back Pain that conducted in 58 medical institutions in Japan from May 2013 to July 2014 in 240 patients found that duloxetine was superior to placebo which was consistent with the findings of previous studies conducted. 4,10 Besides, regarding pain reduction, a reduction of 2 points or at least 30% in the Numeric Rating Scale was generally considered as a clinically significant change.
A greater response among patients with multiple painful sites was consistent with duloxetine acting on chronic pain mechanisms to modulate descending pain pathways. 16 The presence of multiple painful sites, including the low back, was thought to result from changes in the central nervous system, particularly reduced activity of descending inhibitory pathways, which amplify their pain perception. 7,14,19 The analysis suggested that the number of agonizing body locales might be an indicator of reaction to duloxetine. 16,19
In the recent study, we decided to evaluate the effect of duloxetine in controlling pain after surgery and its effect on the quality of life and anxiety.
According to our study pain in the duloxetine-treated group showed a greater reduction in compared with the placebo-treated group. Also, the anxiety level in the duloxetine group showed a significant decrease. The quality of life scales of physical performance, general health, and overall quality of life score in the duloxetine group was significantly improved.
So the effectiveness of surgery on pain is different case by case and sometimes the surgery can’t improve the pain completely. In this case, maybe the addition of a useful drug can help so much. In this condition, the drugs which can relieve neuropathic pain like duloxetine are good choices.
The limitations of our study included small sample size and short duration of treatment. Because of these limitations, we cannot rule out that the statistically significant effect might be lost at a later time point. On the other hand, because of the short period of study, we couldn’t increase the dosage of duloxetine to the maximum dose and maybe in upper doses, the result of the study could change. In this study, we couldn’t rule out all baseline spine problems so this limitation can affect the severity of pain and quality of life scores.
Finally, the results from this trial and related studies suggest that the use of duloxetine in patients who had spinal surgery can help to better control back pain, on the other hand, can cause a better psychological condition that affects the quality of life. So this group of patients has a better lifestyle and less psychological problems like anxiety and depression. But for assessment of long-term efficacy, further trials with larger sample sizes and longer treatment durations are needed.
Footnotes
Acknowledgements
The authors would like to thank all of 50 patients who accepted to participate in this study and neurosurgery ward of Rasoul-e Akram hospital that helped us to accomplish this study. Although we want to thank Dr Abidi Pharmaceuticals that provided our drug and placebo for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
