Abstract
Purpose:
High Volume Distension Injection (HVDI) is a treatment adjunct in chronic mid-portion Achilles tendinopathy. This study analysed whether HVDI is safe and effective in an outpatient clinic setting.
Methods:
Retrospective service evaluation of 18 participants over 18 months. Ultrasound-guided saline, corticosteroid and bupivacaine was injected to disrupt vasculo-neural ingrowth, with post-procedure eccentric exercises. Mean follow-up was 8 weeks. Outcomes used were Numeric rating Scale (NRS) of pain, tendon thickness and Modified Ohberg score (MOS) of neovascularisation measured on Ultrasound and Doppler imaging.
Results:
Mean reduction in NRS was 5.30 ± 2.53 (P < 0.001). Mean tendon thickness reduction was 0.21 ± 0.17 cm (P < 0.001). Median MOS improved from 2.00 ± 2.00 to 1.00 ± 3.00 (P = 0.009). No complications occurred. 11 participants had successful primary HVDI, 3 responded to a second injection. Two non-responders were referred for surgery and two referred for alternative therapy by patient preference.
Conclusion:
Significant reduction in pain, tendon thickness and neovascularity was observed in 78% of patients. Recurrence in 39%. HVDI with eccentric training is safe and effective intervention in outpatient clinic setting.
Background
The Achilles tendon is the mechanical connection between the calf muscles (gastrocnemius and soleus) and the calcaneus bone. It is anatomically named the Tendocalcaneus. If the tendon is chronically overloaded this can cause tendon disrepair resulting in degenerative tendinopathy. 1 Achilles Tendinopathy (AT) classically results in: pain, stiffness and impaired performance. Clinically, thickening of the tendon is associated with degenerative tendinopathy, this is due to recurrent tendon injury resulting in collagen disarray and an increase in glycosaminoglycans. These polysaccharides cause an increase in oedema resulting in a palpable swelling. 2 This condition can impact physical and daily activities, having both health and social implications if prolonged or untreated. The pathological process of tendinopathy is summarised below (Figure 1).
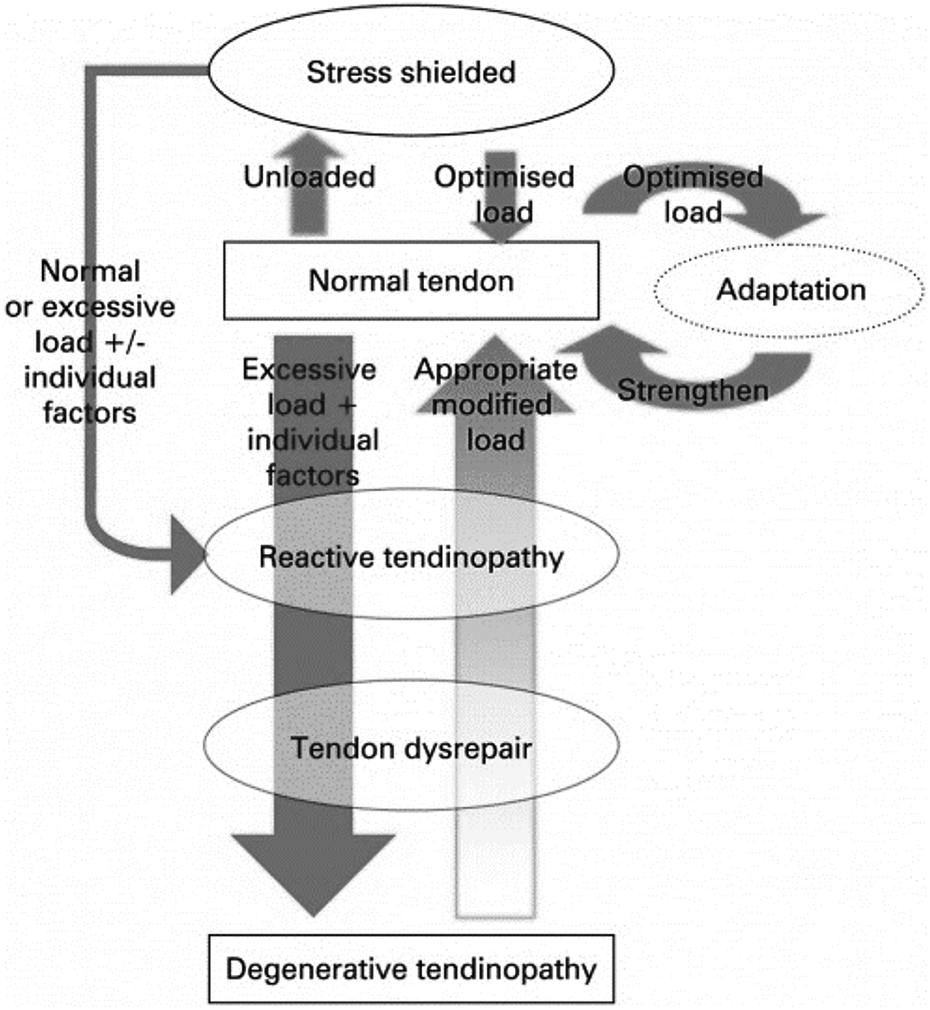
The pathological process of deterioration of normal tendon to degenerative tendinopathy and the reversibility of this in the early stages. 2
The incidence of AT among people aged 21–60 years old is 2.35 cases per 1,000 people 3 with approximately 150,000 cases per year in the UK. 4 AT commonly affects physically active individuals who do training or sport that involves running and jumping 5 as these activities can result in repetitive, excessive tendon loading. With increased popularity of sports and endurance events, incidence of AT is increasing. 6,7 It is also strongly related to metabolic disorders such as Diabetes Mellitus, Thyroid dysfunction and Dyslipidaemia 8 these causes may be more commonly seen in a community Musculoskeletal (MSK) service.
Initial management of mid-portion Achilles tendinopathy should include tendon loading and eccentric training. 9 However, in chronic mid-portion AT symptoms continue despite these exercises. Therefore, several different treatment adjuncts have been proposed. Eccentric exercises should be continued alongside these adjuncts to improve both pain and muscular strength. 10 Some alternative treatment adjuncts include: extracorporeal shock wave therapy (ESWT), high volume distension injection (HVDI), platelet-rich plasma (PRP) injection, autologous blood injection, sclerosing injection, corticosteroid therapy and operative treatment. 11 There is limited evidence published to support the use of injectable therapies 11,12 and the literature calls for further investigation of these procedures.
One implicated mechanism in AT is vasculo-neural ingrowth. It was postulated that neovascularisation on the ventral aspect of the tendon was accompanied by the formation of new nerves which could be the source of pain. 13 Therefore, injecting local anaesthetic ventral to the tendon in a cohort of affected individuals led to pain being temporarily relieved in all affected individuals while undertaking a load baring challenge. 13 Consequently, interrupting vasculo-neural ingrowth was deemed a viable treatment adjunct. This formed the rationale of the HVDI procedure. It is thought that an injection of high volume has a mechanical effect upon vasculo-neural ingrowth. In theory, disrupting these structures will result in reduced pain. 14 This is a relatively recent addition to the management options and there is currently no consensus on what should be injected and in what proportions. 15
In this study HVDI refers to an injection of local anaesthetic, 0.9% sodium chloride (saline) and corticosteroid with a total volume of roughly 40 mls. Kager’s fat pad lies anteriorly to the Achilles tendon, vasculo-neural ingrowth originates from here and so the high volume of fluids is injected into this area. Care is taken to avoid intra-tendon injection of corticosteroid and the procedure is performed under direct visualisation using ultrasound imaging.
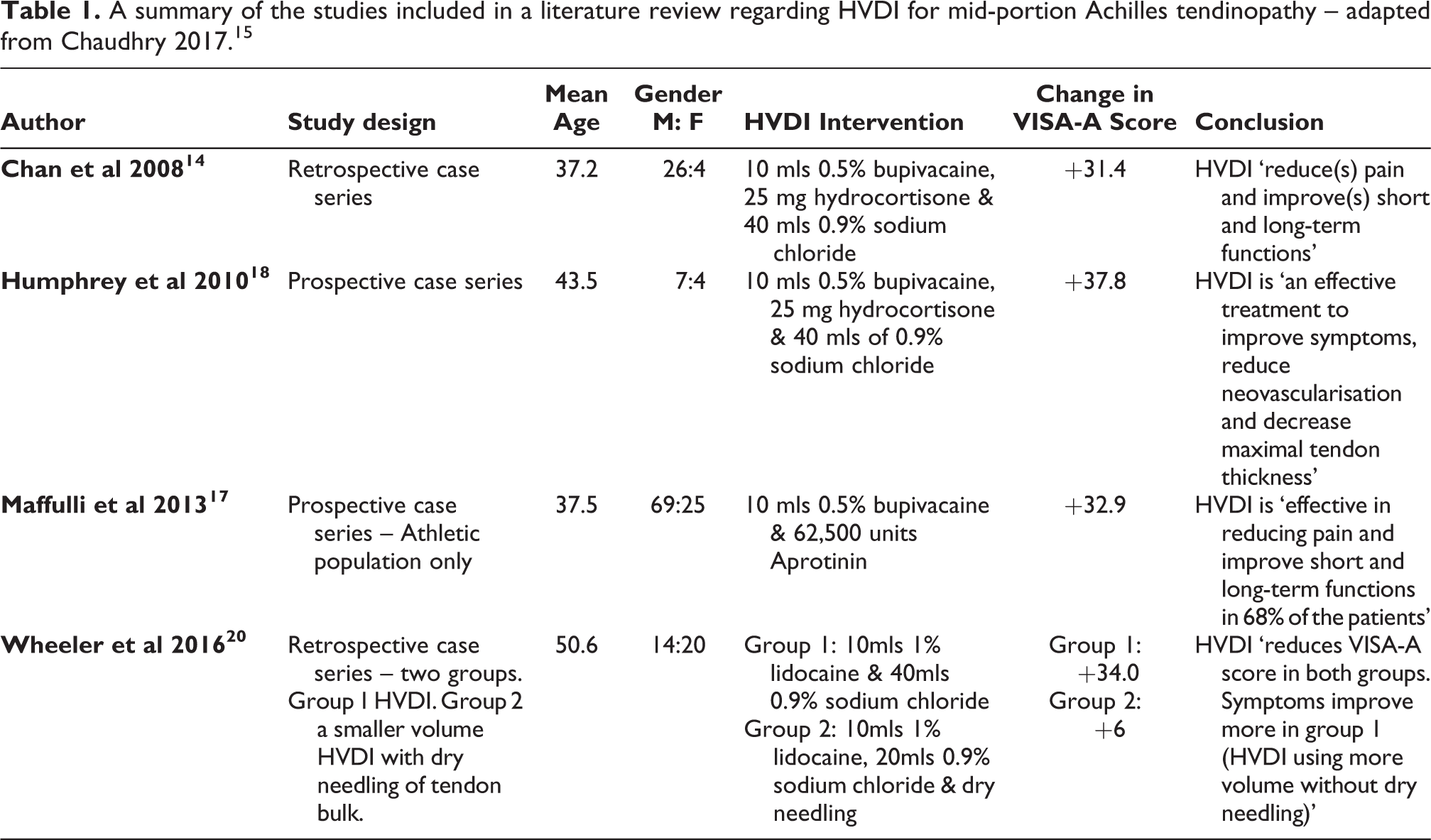
There is limited research regarding HVDI but recent studies suggest there is clinical promise. A literature review of four HVDI studies demonstrated improved patient symptom severity scores in the ‘short to medium term’ 15 (Table 1). All studies involved in this review used the 100 point scale of Victorian Institute of Sports Assessment-Achilles (VISA-A) questionnaire, a validated and reliable symptom severity tool. 16 Two of these studies used tendon thickness post-procedure as an outcome and showed significant reductions in tendon thickness, which is associated with improved function. 17,18 In 2017, a RCT showed HVDI had greater VISA-A and tendon thickness improvement than eccentric training alone. 19 However, the RCT only used a cohort of male subjects.
This study aims to analyse the safety and effectiveness of HVDI for chronic mid-portion Achilles tendinopathy in a Musculoskeletal (MSK) outpatient clinic setting.
A summary of the studies included in a literature review regarding HVDI for mid-portion Achilles tendinopathy – adapted from Chaudhry 2017. 15
Methodology
Study design
This retrospective service evaluation was performed in conjunction with University of Leeds and Leeds Community Healthcare National Health Service (NHS) trust. Ethical and Institutional R&D clearance were obtained. 26 HVDI procedures were performed by a single clinician (MS) in a period of 18 months of service provision. Clinical encounters were recorded in the computed based electronic patient record software ‘SystmOne’. SE extracted the data from the records and performed independent analysis without the bias of the clinician who performed the intervention (MS).
Subjects
All patients who had HVDI as a primary procedure performed by MS during the 18 months of retrospective service evaluation were included. A cohort of 18 patients (10 male and 8 female) had primary HVDI during this time frame. Individuals were excluded from the cohort if they were below the age of 18 or had a non-mid-portion Achilles tendinopathy.
Ultrasound imaging
Ultrasonography of the Achilles tendon was performed by the same clinician, MS, for every patient in the cohort. A GE Logiq E portable ultrasound with 12 MHz linear array musculoskeletal probe was used for diagnosis and guiding intervention.
Patients undergoing the procedure lay prone on the examination couch with support under the distal tibia. This allowed the foot to hang off the end of the bed in a neutral position. Ultrasound gel was applied and the tendon was visualised in both the longitudinal and transverse planes. Tendon thickness was measured at the point of maximum thickness in the longitudinal scan. Power Doppler is more sensitive than colour Doppler and allows better visualisation of small blood vessels 21 therefore this was used to manually assess the number of vessels anterior to the tendon, this was then recorded against the Modified Ohberg scale (MOS) a validated scale of neovascularisation quantification. 22
Procedure
The technique used was similar to that used in previous studies. 14,18,19 Each patient provided informed written consent prior to the procedure. Lidocaine 1% local anaesthetic was used to infiltrate the skin overlying the Achilles tendon, using aseptic technique. Under ultrasound guidance, using sterile transmission gel, a 21-gauge needle was inserted into the fat pad ventral to the tendon. The insertion site is defined by the area of principal discomfort and tendon thickening on ultrasound. A HVDI of 40mls comprising of 37.5 ml of 0.9% sodium chloride, 2mls of 0.25% bupivacaine (local anaesthetic) and 0.5 ml (20 mg) of Kenalog (corticosteroid) was then performed. Needle positioning was visualised in real-time under ultrasound guidance, with positional changes to ensure an even distribution of the treatment across the area adjacent to the affected tendon.
Rehabilitation protocol
All patients were recommended to use a standardised exercise protocol after a mandated 2-day rest period following the procedure. Starting on day three gentle seated and standing static stretching of the calf muscles and Achilles tendon were commenced. From day 7 onwards, progressive eccentric loading exercises were started. Eccentric exercises consisted of ‘heel-drop’ exercises where the Achilles tendon is actively stretched while under the load of body mass, this was typically performed while on the lowest step of a staircase with the forefoot positioned on the step and the heel lowered over the edge of the step gradually. This was initially performed bilaterally to reduce the load but eventually progressed on to a single leg heel-drop, with active heel elevation added in later. Patients were advised to perform these exercises as frequently as possible, even if pain was experienced, but to stop if the pain became disabling. In 2014 this ‘do-as-tolerated’ programme was shown to be as effective as the 180 repetitions a day that were instructed in the Alfredson protocol. 10,23 The volume of exercise was not formerly recorded but some patients informally reported their adherence.
Ethical considerations
This project was a retrospective service evaluation and therefore does not require patient consent per se. It does, however, include data that was related to patients. Therefore, ethical approval for original data collection was granted by NHS research and development for Leeds Community Healthcare NHS Trust (reference number: SE/0105). Ethical approval was also granted by the University of Leeds Faculty of Biological Sciences (reference number: BIOSCI 18-014). All HVDI procedures were performed prior to this project commencing.
Data collection
The data set was anonymised during data collection by SE. In order to find appropriate cases all of MS’ clinics during the 18 month period were reviewed. Patients identified to have mid-portion AT with HVDI treatment were included in the data collection. Initially all procedures were included in the data set resulting in 26 HVDI procedures performed in 18 months. However, there were five individuals who required repeated treatments, making up 8 of the 26 procedures performed. Therefore, the data set was streamlined to only include HVDI as a primary procedure, resulting in 18 individuals and 18 procedures.
Outcome measures
The primary clinical outcomes were measured at baseline prior to the procedure and then repeated at an average follow-up time of 8 weeks. Pain scores were measured using the Numeric Rating Scale (NRS) of pain (where patients rated their pain out of 10, with 10 being most severe pain ever experienced and 0 being no pain). NRS and the Visual Analogue Scale (VAS) have been shown to have similar sensitivity and are both preferred to the less sensitive Verbal Rating Scale. 24 An absolute reduction in NRS of greater than 2.5 is considered to be ‘most accurate in predicting a successful pain reduction after a given treatment’. 25 Tendon thickness was measured during ultrasound imaging of the affected Achilles tendon, Power Doppler imaging and MOS assessed neovascularisation as outlined below (Table 2). Complications, recurrent treatments and referrals for other treatments or surgery were clearly documented in the patient records.
Modified Ohberg Score (MOS) for Neovascularisation, adapted from Watson et al 2018. 22

Statistical analysis
Data analysis was performed using the IBM SSPS Statistics 25 analysis system.
As all the individuals underwent a standardised primary procedure the statistics were focused on comparing pre-procedure to post-procedure outcomes. The three clinical outcomes were tested for normality of data distribution using Shapiro-Wilk’s test. Paired samples T tests were used to test tendon thickness and NRS as they were normally distributed. These results were reported as mean ± SD; P < 0.05 was used to define statistical significance. Wilcoxon Signed-rank test was required for MOS scores due to non-parametric data, therefore this data was presented as median ± IQR.
Results
Numeric rating scale of pain (NRS)
15 individuals had accurately recorded pain scores both before and after HVDI. Pre-procedure the mean NRS of the cohort was 7.77 ± 1.70. Post-procedure this reduced to a mean of 2.47 ± 2.22, this is equivalent to a mean reduction in NRS of 5.30 ± 2.53 (Figure 2). This demonstrated statistical significance with (t(14) = 8.10, P < 0.001).
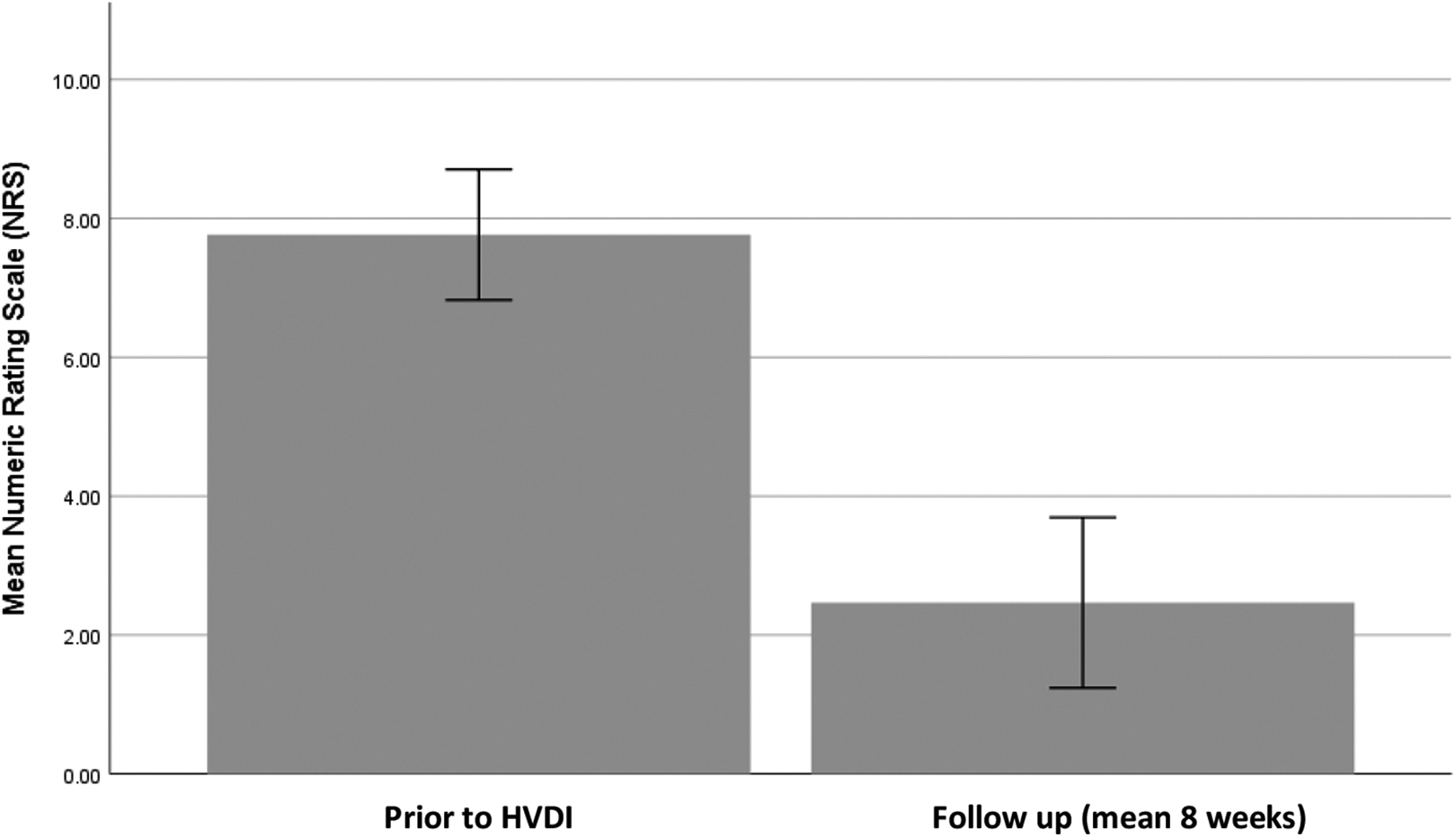
Bar chart comparing mean Numeric Rating Scale (NRS) of pain both pre-procedure and at follow-up. Error bars of 95% confidence interval.
Tendon thickness
17 participants had complete records with regard to tendon thickness. Pre-procedure a mean tendon thickness of 1.10 ± 0.25 cm was seen. After HVDI this showed a significant decrease to 0.89 ± 0.24 cm (t(16) = 5.01, P < 0.001), a mean reduction in tendon thickness of 0.21 ± 0.17 cm (Figure 3).
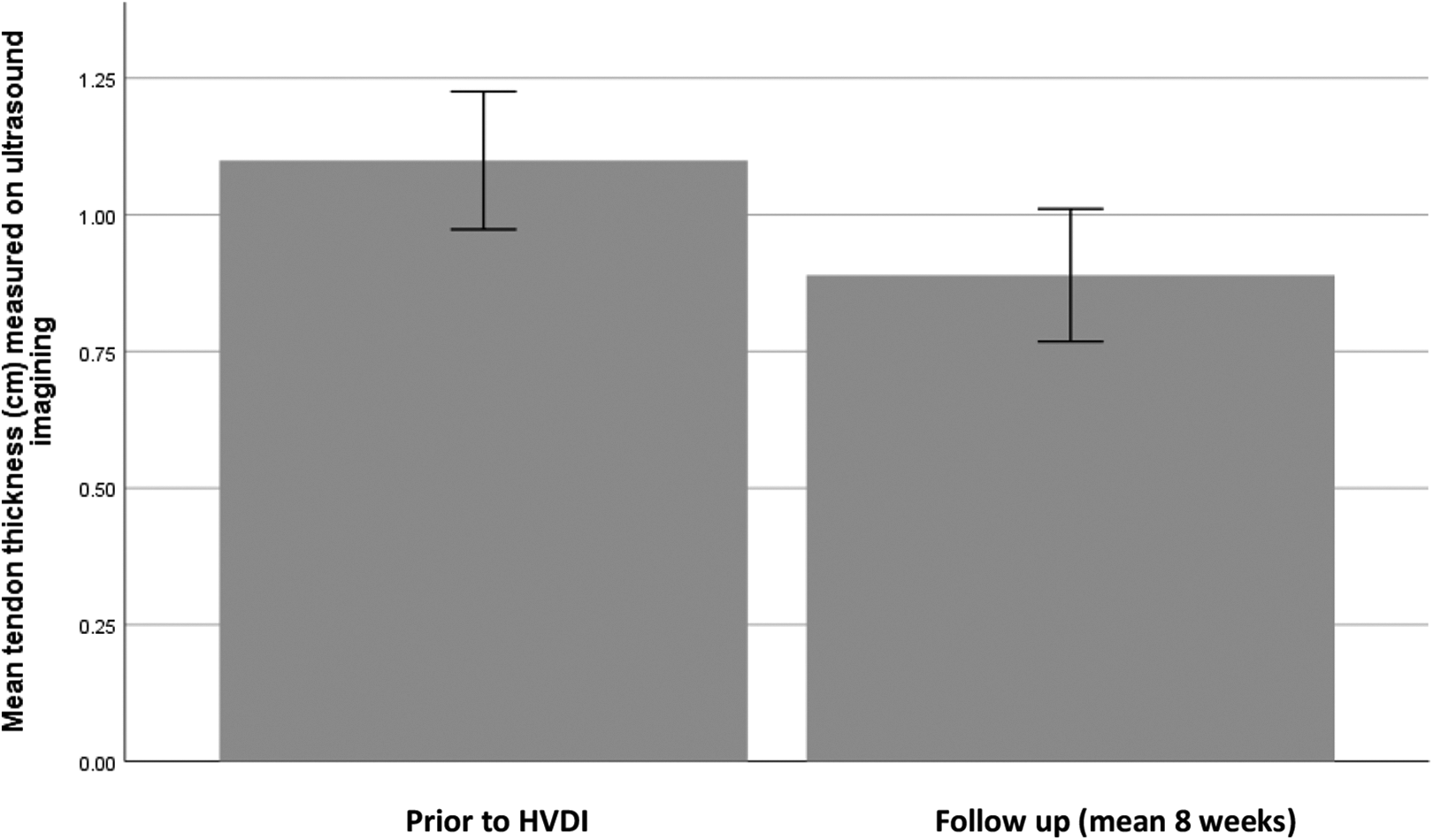
Bar chart comparing mean Achilles tendon thickness, as measured on ultrasound imaging, both pre-procedure and at follow-up. Error bars of 95% confidence interval.
Modified Ohberg score (MOS)
Accurate records of MOS were available for 16 participants. Median MOS values were significantly lower at post-HVDI follow-up 1.00 ± 3.00 when compared to pre-HVDI 2.00 ± 2.00 (t = 4.00, r = −0.41, P = 0.009) (Figure 4). Clinically, the change in median correlates with 1–2 fewer vessels seen on follow-up imaging.
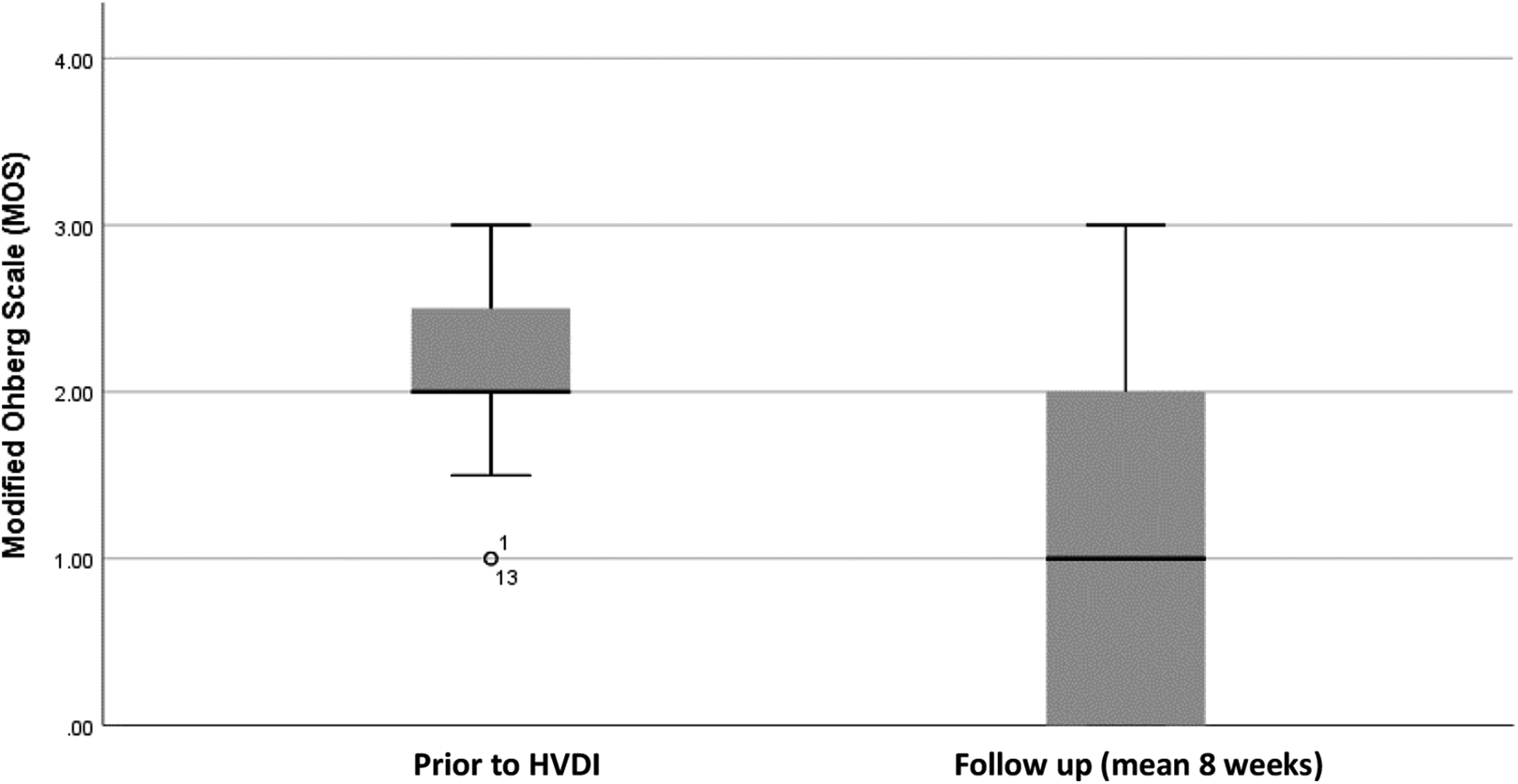
A Boxplot demonstrating difference in MOS prior to HVDI and at follow-up. Median, range and interquartile range is depicted with outliers also identified.
Change in these three key clinical outcomes showed a significant positive correlation to one another. Change in NRS and MOS showed the strongest positive correlation (r = 0.81, P < 0.001). MOS and tendon thickness had the weakest positive correlation (r = 0.52, P = 0.037). Tendon thickness and NRS also showed positive correlation (r = 0.61, P = 0.016).
Safety outcomes
Possible complications associated with the procedure include: infection, tendon rupture and haematoma formation. No complications occurred. A total of 14 patients (78%) were successfully discharged after 2 or fewer HVDI procedures. A breakdown of the different patient outcomes is given below (Table 3).
Patient outcomes of HVDI across the cohort of 18 patients.

Discussion
This retrospective service evaluation was observing a cohort of individuals who have suffered from chronic AT for a minimum of 6 months with minimal response to initial conservative therapy. Following two or fewer HVDIs 78% of this group showed significant clinical improvement and were discharged from the service. After primary HVDI, for most of the cohort, significant improvements were seen at short term follow-up in all three key clinical outcomes: pain score, tendon thickness and neovascularity. This study demonstrates a symptom recurrence rate of 39% after the primary procedure. There were no reported complications due to HDVI. 17% of patients in this study were referred for a surgical opinion.
This study demonstrates similar results to the case series studies outlined earlier (Table 1). The success rate in this paper is greater than a previous seminal study who found 70% of their patients were responders. 14 One study quoted a rate of repeated procedures at 45 of 94 cases (47%) 17 compared to the 39% seen in this study. Wheeler et al quote a percentage of 16.67% requiring a ‘surgical conversion’ from their HVDI cohort, 20 while another study suggests that 28% of all AT cases require surgical management. 26 Comparatively, this study shows the same or lower rates of recurrence and surgical intervention than the earlier literature. This study provides further evidence that HVDI has a short-term benefit in the management of chronic mid-portion AT.
We used a similar technique to previous studies. 14,18,19 This differs to other studies where Aprotinin 17 or no steroid was injected. 20 These studies all show significant improvements in symptoms. A future avenue for research could be a RCT to assess for the difference between HVDI with and without steroid. Significantly better outcomes were seen with a larger volume of injection compared to a smaller injection with dry needling 20 and so this supports the use of higher volumes in this study.
Symptom recurrence is an issue related to mid-portion AT. From the medical records it was noted that those individuals who relapsed after primary HVDI often admitted to poor compliance with the exercise regimen. Three of the seven patients who had a second HVDI went on to have a successful outcome. This is thought to be due to education from injecting clinician (MS) and better exercise compliance after repeat HVDI. This study did not use a structured rehabilitation programme for patients to follow and it could be argued that this may have reduced the number of participants who relapsed. A future RCT could assess the difference in outcome seen across three groups; a structured exercise programme without HVDI, the second group using HVDI alone and a third cohort having HVDI and a structured exercise programme. This would identify if HVDI is an adequate treatment or only for use as an adjunct alongside exercise.
Non-responders within this cohort received a large quantity of injections with minimal clinical benefit and all had to be referred on for alternative therapy. Although the sample size is small, it could be considered that if after two injections there is no response then further injections are unlikely to be of benefit. This will need further study with a larger cohort but could provide guidelines with regard to the use of recurrent treatments in the future.
This study provides clear evidence of HVDI being a safe and effective intervention in an outpatient clinic setting. 78% of patients responded positively to HVDI, could be discharged after follow-up and potentially avoided surgical decompression of tendon. HVDI in outpatient clinics approximately costs £280 compared to around £2500 for surgical intervention.
Limitations
This study has some limitations. A retrospective service evaluation relies upon the accuracy of the medical records and does not have the advantage of prospective intentional completion of all outcomes. This study focuses on the results of only 18 participants which is a relatively small cohort when compared to other studies (Table 1). A small cohort can lead to an increased ‘chance of assuming as true, a false premise’ 27 while a small cohort may not raise complications that may be evident in a larger study.
There is no control group for comparison in this study, again related to the retrospective nature of the study. All the cases included in this study had undergone a period of at least 3 months of regular eccentric exercise prior to being offered HVDI. Therefore, it can be argued that they had already failed conservative management. A control group may have demonstrated no significant improvements with continued conservative management. However, without a control this conclusion cannot be assumed.
The clinical follow-up in this study reflects the realities of working within the National Health Service (NHS). Across the 18 participants there was a range of 3–12 weeks for follow-up, this varied due to patient, clinician or clinic availability. This affects interpretation of results as it is not possible to know if an individual seen at 12 weeks had seen a response at 6 weeks or vice versa. However, with the information available from the patient records and clear documentation of patient discharge from the service with no further re-referral it was inferred that this follow-up period was representative of adequate short-term follow-up.
The lack of a structured monitored exercise programme meant it was very difficult to assess rehabilitation adherence. It may be that individuals who were more compliant with their exercises had better outcomes but this data is not available in this study. As discussed above this should be an area considered in future research.
VISA-A scores are a validated measure of functional outcome and pain in AT patients, hence why they have been used in the studies outlined in the recent literature review. 15 This study did not utilise VISA-A scores but instead used NRS pain score, while this allows an understanding of improvement in pain levels, it does not give any information regarding change in functional status. In a condition that affects a key weight baring structure, functional outcome is crucial for patient quality of life.
This study observes the performance of a single clinician, in a single centre which is not representative of the whole population. Outcomes were recorded by an unblinded single clinician who also performed the HVDI which could, in theory, result in bias.
Conclusion
HVDI is a safe and effective treatment adjunct in the management of chronic mid-portion Achilles tendinopathy, which has failed initial conservative management, in an outpatient clinic setting.
This retrospective service evaluation study shows significant reduction in pain, tendon thickness and neovascularity in most patients after a single procedure. Recurrence was the main challenge in 39% and this could be related to compliance with exercise protocols. Several areas for future research have been highlighted.
Footnotes
Acknowledgements
The authors acknowledge the support provided by University of Leeds Faculty of Biological Sciences and Leeds Community Healthcare National Health Service Trust.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
