Abstract
Patients with Calreticulin (CALR) mutation positive essential thrombocythemia are often thought of as having a “low-risk” of thrombotic complications. This report examines a case of a patient with CALR-mutation positive essential thrombocythemia presenting with peripheral retinal ischemia and subsequent retinal neovascularization. This patient had a 2-year documented history of CALR-mutation positive essential thrombocythemia with a maximum platelet count of over 800,000 cells/µL. Fluorescein angiogram showed significant leakage in the areas of the vascular lesions with peripheral vascular nonperfusion consistent with neovascularization. The patient was treated with photocoagulation to the areas of avascular retina. At 6-month follow-up, some regression of neovascularization was noted with no signs of progression in the retinopathy. Furthermore, essential thrombocythemia patients can present with neovascularization secondary to retinal ischemia due to microvascular thrombotic events. Identification of these changes is important for guiding medical and procedural interventions in order to preserve vision.
Introduction
Essential thrombocythemia (ET) is a myeloproliferative neoplasm defined by a platelet count of over 450,000 cells/µL. 1 This condition leads to an increased risk of thrombotic and hemorrhagic events due to qualitative and quantitative changes in the patient’s platelets. Due to the neoplastic nature of the disease, platelet counts can increase drastically while their function may be diminished. Manifestations of this disorder may include headaches, visual disturbances, lightheadedness, dysesthesia, and erythromelalgia. 1 It is reported that thrombotic complications occur in 20%–50% of patients. 2 The thrombotic complications may cause both microvascular and large vessel thrombosis, usually affecting arteries more than veins.
In this condition, up to 90% of cases have been identified secondary to a mutation in JAK2, CALR, or MPL genes. 1 Over half of all ET patients harbor a JAK2 mutation with less than a quarter of patients expressing mutations in CALR and MPL, respectively. 1 The general consensus of previous research has supported the idea that patients with JAK2 mutations have a higher incidence of thrombo-hemorrhagic complications, while patients with CALR mutation are typically classified as low-risk with an unclear association. 3 Ocular thrombotic events are a rare phenomenon of ET described in a handful of case reports with findings of retinal arterial or venous occlusions and capillary nonperfusion.4,5 This is the first case-report describing findings of retinal neovascularization (NV) in a patient with CALR mutation.
Case report
A 49-year-old female with a past medical history of ET presented via referral to retina service for incidental findings of peripheral retinal vascular lesions. The patient was diagnosed with ET 2 years prior in the setting of progressive fatigue, new-onset headaches, erythromelalgia, and periorbital pain. The diagnosis was confirmed with an initial platelet count of 792,000 cells/µL, a bone marrow biopsy showing atypical megakaryocytes, and genetic testing confirming heterozygous CALR mutation. Over this time course, the patient has inconsistently taken hydroxyurea 1000 mg twice daily and aspirin 81 mg daily as a cytoreductive treatment regimen for ET. Platelet counts have ranged from 300,000 to 800,000 cells/µL over this time. She has had no other reported systemic complications secondary to ET.
On presentation, best-corrected visual acuity was 20/20 OD (right eye) and 20/20 OS (left eye). Intraocular pressure was 15 mmHg in each eye. Anterior segment examination was unremarkable in both eyes with no signs of iris NV in either eye. Dilated fundus exam revealed hypopigmented, elevated, retinal lesions with vascular engorgement in the far periphery of the retina (Figure 1). Wide-field fluorescein angiography (FA) demonstrated areas of capillary dropout and nonperfusion in the far temporal periphery, along with leakage in the areas of the elevated retinal lesions. These FA findings were consistent with peripheral ischemia associated with retinal NV in both eyes (Figure 2). Two sessions of sectoral panretinal photocoagulation were performed along the temporal periphery of the left eye in the areas of avascular retina. Since the ischemic changes in the right eye initially were not as pronounced as the left eye, it was elected to observe the right eye. After the second session of photocoagulation, the area of NV began to regress with associated vitreous hemorrhage. At 8-month follow-up, regression of the NV was noted in the left eye, with a stable exam with no signs of progression on NV in the right eye (Figure 3).
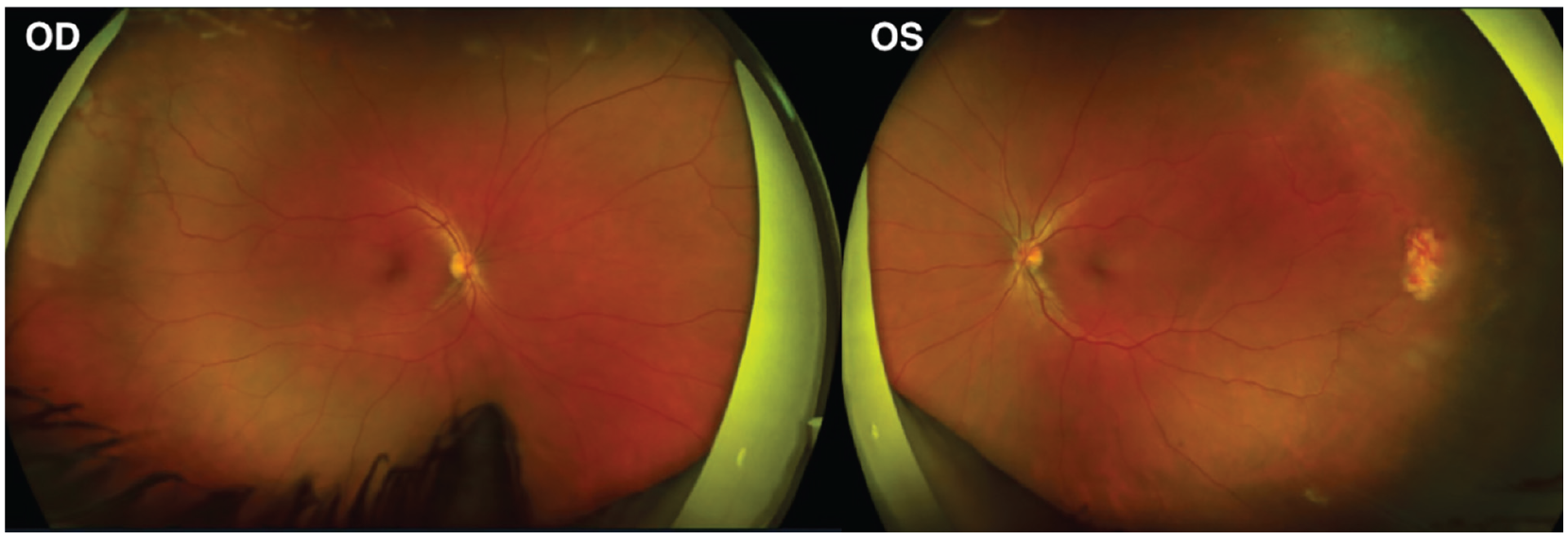
Peripheral vascular ischemia with temporal neovascularization in both eyes.

Early and late phase fluorescein angiography of the left eye showing capillary nonperfusion, arteriovenous anastomosis, and temporal neovascularization. (Bottom Left: Right eye late phase).

Regression of neovascularization in the left (treated) eye seen at 8 months follow-up. Right (untreated) eye showing stable lesion with peripheral avascularity and the appearance of sea-fan neovascularization.
Discussion
ET commonly leads to a hypercoagulable state due to an impaired hemostatic system. The risk of thrombotic events is 1%–3% per patient-year and is primarily seen in patients with a JAK2-mutation. JAK2 is a tyrosine kinase that functions in multiple complex intracellular signaling pathways that regulate hematopoiesis and thrombosis. In mouse models, JAK2 mutations have shown increased clonal platelet production with more frequent thrombosis, due to increased reactivity to pro-thrombotic factors, such as collagen-related peptide and thrombin. 2 The combination of increased platelet numbers and increased clotting tendency is proposed as the pathophysiology behind increased vascular events in patients with JAK2 mutation. 2 Meta-analysis has shown that ET patients with JAK2 mutation have a risk of thrombosis about 2–3 times higher than ET patients without a JAK2 mutation. 6 Reports of thrombotic events that have identified the patient’s genetic disposition have overwhelmingly presented as cases with JAK2 mutation. In the case of the patient presenting with central retinal artery occlusion, he was found to be JAK2 V617F positive. 4 At this point in time, there have been no reports of patients with CALR or MPL mutation with ocular thrombotic events.
For patients with CALR mutation, like our patient, the mechanisms leading to hemostatic dysfunction are less clear. Patients are typically younger and have higher platelet counts than those with JAK2 mutations. Early research suggests that this is due to the CALR mutation leading to dysregulated apoptotic pathways through overexpression of anti-apoptotic proteins. 7 Apoptotic dysfunction may lead to significantly increased platelet numbers. However, it is important to recognize the products of this mutation do not function individually and are part of a complex signaling network.
The primary intervention for patients with ET is to control platelet counts with cytoreductive therapeutic agents including either hydroxyurea or interferon-alpha. The decision of what agent to use is based on a rigorous clinical assessment and consideration of factors such as history of thrombosis, age, and mutation status.1,2 Our patient self-reported inconsistent use of cytoreductive therapy. This was supported by highly variable platelet counts. Our hypothesis is that our patient, whose platelet count neared 800,000, underwent partial vascular thrombosis leading to retinal ischemia and subsequent NV.
Our patient presented with findings of bilateral retinal vascular lesions that were associated with peripheral retinal ischemia and retinal NV. There were no ischemic changes in the macula or in the midperiphery and there were no retinal hemorrhages in either eye. In attributing the patient’s retinal findings to ET, we considered a wide differential of conditions that can cause peripheral avascularity or large vascular lesions like NV or vascular tumors. Peripheral retina avascularity is non-specific and may be caused by a variety of conditions including retinopathy of prematurity, sickle cell retinopathy (SCR), diabetic retinopathy, and familial exudative vitreoretinopathy (FEVR). All of these conditions were considered and excluded. Our patient was not born prematurely and had no history or other symptoms of sickle cell disorder or diabetes. When considering FEVR, our patient has no family history of disease, no macular/vascular dragging and no v-shaped area of avascularity in the temporal retina. Von Hippel Lindau (VHL) was also considered as the large vascular lesion in the left eye appeared to have a feeding vessel, though the lesion itself was not a typical hemangioblastoma. This was excluded as well as the patient has had multiple brain MRIs and other systemic work-up with no systemic findings of VHL.
Two prior case reports described the association of ET with findings of capillary nonperfusion in the temporal periphery. In these reports, the causal genes were not reported. In one case, a 77-year-old man presented with decreased visual acuity in his left eye and with macular edema, dilated and tortuous veins, and scattered blot hemorrhages consistent with impending central retinal vein occlusion. 3 The patient’s right eye had no loss of visual acuity (20/20) but did have an area of capillary nonperfusion with dye leakage in the temporal periphery seen on angiography. The patient was noted to have an elevated platelet count at this time. The proposed pathological mechanism was increased venous pressure caused by incomplete thrombosis. The authors suggested a causal relationship between retinal vascular occlusion and ET due to the unusual presence of bilateral vascular occlusions seen in their patient. A second case described similar findings as our patient. A 24-year-old presented with vitreous hemorrhage and areas of peripheral nonperfusion associated with sea-fan NV as presenting signs of ET. 8 This patient was further treated with multiple rounds of photocoagulation and started on aspirin.
We suspect that retinal changes in our patient occurred secondary to thrombogenic vaso-occlusion and localized ischemia similar to SCR. Both ET and SCR have been shown to lead to peripapillary retinal nerve fiber layer thinning secondary to ischemia in patients even without pre-existing glaucoma.9,10 A recent article exploring retinal microvascular abnormalities in patients with sickle cell disease (using OCT [optical coherence tomography] angiography) discovered that sickled blood cells within retinal capillaries created intermittent vascular stasis and subsequent thrombus formation. 11 This thrombus formation may lead to impaired capillary perfusion and retinal ischemia. Thus, the NV seen in SCR is likely secondary to vaso-occlusive events due to thrombosis. Given similar retinal findings in our patient, we suspect vaso-occlusion resulting in retinal ischemia with subsequent NV as a consequence of a dysregulated thrombogenic system seen in patients with ET.
Given these findings and reports of other vaso-occlusive events seen in patients with ET, we believe that yearly screening may be warranted for these patients with dilated fundoscopic examinations. Similar to patients with sickle cell, screening would be a way to decrease the morbidity of this condition and avoid the severe visual consequences that may exist through early detection.
Conclusions
In summary, we describe findings of bilateral peripheral retinal ischemia and peripheral NV in a patient with CALR-mutation positive ET. This was likely secondary to the patient’s elevated platelet count and increased thrombotic potential. The areas of NV were stopped from progressing after intervention with photocoagulation.
Footnotes
Author contributions
S.P.H., M.R.A., and A.L.W. contributed to the conception of the idea, coordination, article preparation, and article review. S.P.H. contributed with the writing of the original draft. All authors read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was performed at UT Southwestern Medical Center and was supported by NEI Core Grant P30EY030413.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
The patient has consented to publication of this case. Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
