Abstract
Background/Purpose:
Osteoarthritis (OA) is the main orthopedic disease to cause pain and edema in humans. This study evaluated the influence of complementary medicine on the hyperalgesia and edema induced by exercise in an animal model of OA as well as possible role for interleukin (IL)-1β to these outcomes.
Methods:
Mice (25–35 g) were subjected to intraplantar injection of Freund’s complete adjuvant and were subjected to exercise and oral treatment with ethanolic crude extract of Casearia sylvestris (ECE-CS) or vehicle (alone or in combination) and evaluated through behavioral and biochemical tests.
Results:
At day 5, exercise and ECE-CS alone did not reduce hyperalgesia, while the combination of both enhanced this effect. In muscle and skin tissues from the treated paw of mice, both treatments alone or in combination reduced in similar extent the levels of IL-1β in relation to the control group.
Conclusion:
Association of both complementary therapies may bring benefits on pain associated with OA in humans.
Keywords
Introduction
The pain has a protective character and comprises an alert mechanism with the purpose of reestablishing the homeostasis and acting in the defense of the organism. In this way, pain is defined as being an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage; consequently, pain is subjective in nature and is understood according to individual experiences. 1
Although it is important to protect the individual when they signal the presence of a threat, chronic pain can persist beyond this end, has pathogenic traits, and is considered maladaptive. 2 In this way, chronic pain gradually emerged as a distinct phenomenon compared to acute pain that persists beyond the normal healing time. 1 In the United States, the financial burden of chronic pain in adults is estimated by the Institute of Medicine 2 for health care costs and lost labor productivity is around $600 billion per year. In addition, pain affects the individual, his/her family, and social environment. 3
Among the chronic pain conditions, osteoarthritis (OA) stands out, as it is a chronic progressive condition that can trigger an integral involvement of the joint tissues and generate incapacity of the functions of this region, being considered one of the most prevalent chronic diseases and one of the main causes of pain and disability in affected individuals. 4 Worldwide, it affects 9.6% of the population over 60 years old, causing limitation of range of motion, impairment of daily living activities, and decreased quality of life. 5 Associated with physical impairment, OA is also deleterious to mental health since it is associated with a higher risk of depression, suicide, memory loss, sleeping deficit, and mood changes. 6
The treatment of OA patients is aimed at minimizing pain and other symptoms as well as increasing functional capacity. To that end, there is the involvement of both a pharmacological approach 7 and a multidisciplinary intervention. In the first case, different classes of anti-inflammatory drugs targeting pro-inflammatory mediators, such as interleukin (IL)-1β (which presents a pivotal role in the inflammatory nociceptive pathways) can be used; on the other hand, multidisciplinary interventions encompassing nutritional support, 8 physiotherapy, 9 and alternative therapies 10 are also of great benefit for patients. However, despite the large number of drugs belonging to different pharmacological classes available for the symptomatic treatment of OA, these drugs are generally used for pain relief. Still, since OA is a chronic and degenerative condition, the tendency for patients is to need drugs in increasingly larger doses or in more potent classes, which greatly increases the chance of adverse effects related to them.
Thus, complementary therapies that promote pain reduction, the main symptom of OA, can be a benefit in the treatment of patients with this condition. In this sense, the use of medicinal plants for the treatment of symptoms of acute or chronic diseases is widespread. In this sense, the plant Casearia Sylvestris (CS), a species of the family Salicaceae and popularly known in Brazil as “Guaçatonga,” has been topically and orally used to inflammatory conditions. 11 Popular uses are based on its pharmacological properties, such as inhibition of the activity of phospholipase A2 12 or antihyperalgesic action for its hydroalcoholic crude extract. 13 In particular, the analgesic effect of the plant was confirmed in different models of nociceptive, inflammatory, and immunological pain. 13,14
Another complementary therapy approach for the management of physical pain is physical exercise with experimental studies, indicating antihyperalgesic effect after its practice. Recently, it has been demonstrated that a high-intensity swimming protocol (HISP) reduced pain through the activation of the opioidergic, serotonergic, and cannabinoidergic systems. 15,16 In addition, it was demonstrated that this protocol produced an antihyperalgesic effect in the animal model of peripheral chronic pain and OA, induced by the Freund’s complete adjuvant as inflammatory agent. 16 In addition, the use of physical exercise as a possible therapy to reduce the symptom of pain seems particularly important in the case of OA since it could also contribute to reduce the physical impairment associated with the disease.
Despite the above mentioned, a possible benefit for a possible synergistic antihyperalgesic action from the association of physical exercise and oral treatment with CS in animal models of OA has not been observed yet. This possibility is mainly important with plants for which analgesic activity has already been observed, as for CS 13 and Citrus reticulata. 17 Thus, aiming at the importance of complementary therapies in the control of pain, it is possible to consider that the association between these two modalities of treatment promotes the benefit of obtaining an analgesic effect in chronic condition. Based on this, the present study evaluated the influence in antihyperalgesic effect induced by exercise with the oral pretreatment of mice with ethanolic crude extract of CS (ECE-CS) on an animal model of OA and a possible role for IL-1β to this effect.
Material and methods
This is a preclinical study approved by the Ethics Committee for the Use of Animals (CEUA/UNISUL) under registration 17.006.4.09.IV and all experiments were carried out according to ARRIVE guidelines and ethical guide for experimental investigations of pain in conscious animals. 18
Animals
Thirty-two female Swiss mice (2–3 months old, 25–35 g) were maintained at 22 ± 2°C in a 12-h light/12-h dark cycle (6 a.m. to 6 p.m.) with free access to food and water. Before the experiment, they were acclimated to the laboratory conditions for at least 1 h. After OA induction, animals that presented low levels of response to mechanical stimulation (frequency less than 80%, which means the animals withdrew the paw at 8 of 10 stimulation) or autonomy behavior were excluded from the study. At the end of the experiment, animals were euthanized by intraperitoneal (i.p.) administration of excessive doses of ketamine + xylazine anesthetics and sample collections of skin and muscle from the treated paws were immediately performed.
Animal model of OA
Complete Freund's adjuvant (CFA)-induced arthritis in rodents is a well-established model through histological parameters. 19 For the induction of OA, animals were sent to a subcutaneous administration in the plantar surface of the right hindlimb (intraplantar (i.pl.)), containing 20 µL of CFA (80% solution) diluted in Tween 80 (20% solution). 20
Preparation and treatment with the ECE-CS
Plant was collected from the botanical garden in the campus of the University of Southern Santa Catarina, in the municipality of Tubarão, Santa Catarina, Brazil (28°28′00″ S and 49°00′25″ W) and identified by professor Jasper Zanco (voucher SRS-174, herbarium Laelia purpurata). Procedures were followed as previously described for the hydroalcoholic crude extract of the plant, and ECE-CS was dissolved in 5% ethanol (vehicle) to the desired concentration just before the use and dose were tested on previous studies in our laboratory. 13
Exercise protocol—HISP
In the present study, exercised animals were subjected to the HISP adapted from Mazzardo-Martins et al. 15 The mice were placed in a plastic box (540 × 390 × 325 mm3), segmented with acrylic, in eight compartments (170 × 110 mm2), containing approximately 35 L of water, which were heated to 35°C (Figure 1). To reduce the surface tensile strength of the water and thus the flotation behavior, 1 mL of liquid soap per compartment was added. At the end of each swimming session, the animals were placed in a box with shavings to dry.

Diagram showing the apparatus used for HISP protocol. HISP: high-intensity swimming protocol.
Initially, all animals were sent for 3 days to an adaptation to the aquatic environment, which was performed as follows: during the first 2 days, the animals swam for 2 min and on the third day for 5 min (Figure 2). At the end of this habituation stage, only the animals of the exercised group were sent to the exercise protocol per se, so, on the fourth day of the HISP, three 10-min series were performed with a 5-min interval between the series. On the fifth day and sixth day, two sets of 15 min were performed with a resting time of 5 min.
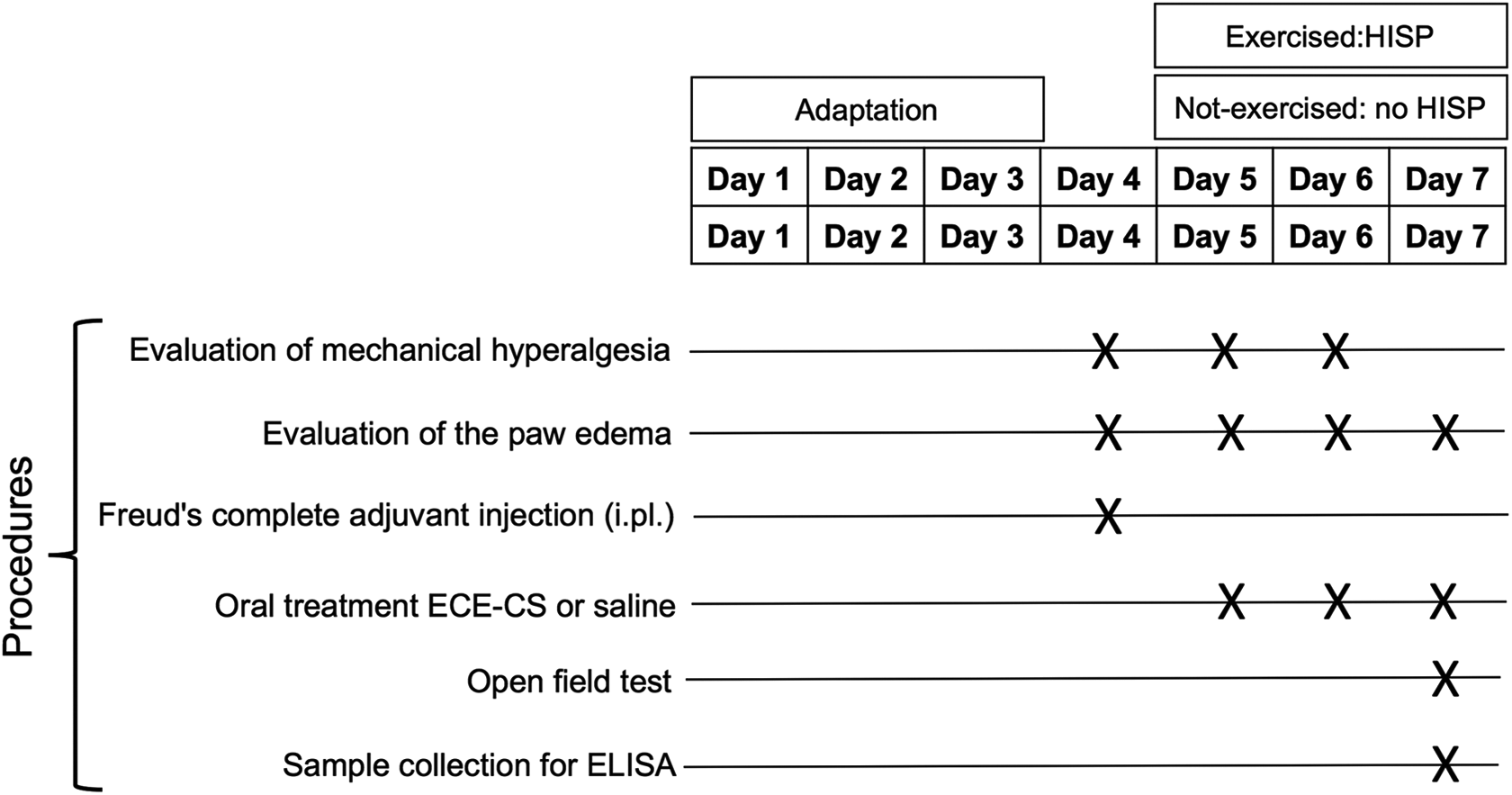
Schematic experimental protocol of the study.
Oral treatment with ECE-CS
The mice belonging to the orally treated groups (ECE-CS or vehicle) were sent to the previously described procedures, while between days 5 and 7 animals received, once a day, the plant treatment immediately before swimming (days 5 and 6) or immediately after the measurement of the basal values of edema (day 7). As mentioned earlier, the dose of 30 mg/kg of ECE-CS was selected based on the previous study, 13 and 1 h after ECE-CS treatment, mice were evaluated for edema and mechanical hyperalgesia evaluations.
Study design
As shown in Figure 2, initially, the animals were sent to adaptation to the aquatic environment for 3 days. In the fourth day (day 4), animals were evaluated for the basal thickness of the paw (in µm) and for the withdrawal reflex of the paw to the mechanical stimulus as hyperalgesic response, before the induction of OA through an i.pl. injection of CFA. After 24 h (day 5), the animals were homogeneously distributed in different groups. Group not-exercised/vehicle: not exercised—i.pl. CFA—oral treatment with saline 10 mL/kg; group not-exercised/ECE-CS: not exercised—i.pl. CFA—oral treatment with ECE-CS 30 mg/kg; group exercised/vehicle: exercised—i.pl. CFA—oral treatment with saline 10 mL/kg; group exercised/ECE-CS: exercised—i.pl. CFA—oral treatment with ECE-CS 30 mg/kg.
Evaluations of mechanical hyperalgesia and edema were assessed after oral administration of plant extract and performing exercise, as described in the following. On the fifth day (first day of ECE-CS treatment), a temporal evaluation was performed for the antihyperalgesic and antiedematogenic effect of treatments, while on the sixth day and seventh day (for edema), animals were evaluated only at the time in which the best antihyperalgesic effect was observed in the temporal evaluation on day 4, which means 1 h after ECE-CS treatment. This schedule was planned to evaluate a possible cumulative antihyperalgesic effect from ECE-CS treatment given on day 5 for the basal values obtained on day 6 of the protocol. One last handling on day 7 was performed in these animals to assess a possible effect of the treatments on edema and paw tissues levels of IL-1β. Finally, aiming to exclude a possible influence of the plant extract, administered for three consecutive days, on locomotor activity (which would mean an unspecific effect of the plant on behavioral analysis of mechanical hyperalgesia), two different groups of animals were treated as not-exercised/vehicle or not-exercised/ECE-CS (vehicle 10 mL/10 g or ECE-CS 30 mg/kg, respectively), as described earlier, on day 7, they were sent (1 h after treatment) to the field test.
Finally, on the seventh day, animals from the not-exercised/vehicle or not-exercised/ECE-CS were treated (vehicle 10 mL/10 g or ECE-CS 30 mg/kg, respectively) and sent (1 h after treatment) for the paw evaluation and open field test, followed by euthanization and sample collections of the paws.
Evaluation of mechanical hyperalgesia
For the evaluation of mechanical hyperalgesia on days 5 and 6 after the beginning of the experiment, the mice were individually observed, as described earlier. 13 The hyperalgesic response was recorded as the frequency of paw withdrawal for 10 applications of the von Frey monofilament 0.6 g. Data were presented as a percentage of response to 10 stimulations. A significant increase in the frequency of response of animals after administration of CFA, relative to the basal values, was interpreted as a hyperalgesic response. In this experiment, animals from not-exercised/ECE-CS, exercised/vehicle, and exercised/ECE-CS groups were compared to not-exercised/vehicle group, in which animals presented the higher frequency of response to mechanical stimulation due to the induction of OA; a sham group was not employed in accordance to rational use of animal guidelines, since other reports from our laboratory have consistently demonstrated that in this group, the frequency of animals response is around 20% 21,22 and statistically lower than that in the not-exercised/vehicle group.
Open field test
To exclude unspecific effects of the ECE-CS on the central nervous system, such as sedation, locomotor and exploratory activity of the not-exercised animals were evaluated on the open-field test at 1 h after ECE-CS or vehicle treatment, as described earlier. 14 Each animal was individually assessed for the number of squares crossed with all paws (number of crossings) and time each animal remained on the hind paws (rearing, in seconds).
Evaluation of the paw edema
To investigate paw edema, a digital micrometer was used to measure the variance between basal thickness of the right paw (central region) from that observed after CFA injection as well as after treatments. Results were expressed by the difference (▵ in µm) between a baseline and subsequent measurements. In this experiment, animals from not-exercised/ECE-CS groups, exercised/vehicle, and exercised/ECE-CS were compared to not-exercised/vehicle group for the same reasons discussed in “Evaluation of mechanical hyperalgesia” section but related to the paw edema.
Determination of the IL-1β levels in the paw
In the present study, the possible influence of exercise, alone or in combination with oral ECE-CS treatment, on IL-1β levels of muscle and skin tissues from the treated paw of mice was analyzed as a possible mechanism of action for these therapies, according to that registered by Hilel et al. 17 using ELISA kits (R&D Systems, Minneapolis, MN, USA), in an ELISA plate reader at 450 nm. Briefly, the treated paw of each animal was dissected right after they were subjected to euthanasia on day 7 of the protocol. This time-point was selected to better understand if the influence on the levels of this inflammatory mediator could explain the mechanism of antihyperalgesic action of different treatments after 3 days of oral administration of ECE-CS.
After paw removal, both skin and muscle (from ventral surface of the right paw) of each animal were detached from the bones, homogenized in buffered saline solution, and centrifuged at 6000 ×g for 20 min at 4°C. The total protein content was measured in the supernatant using the Bradford method, while other aliquot of supernatants was used to determine the level of IL-1β in each group, according to the manufacturer’s instructions. Cytokine levels were estimated by interpolation from a standard curve by colorimetry (correction wavelength 450 nm) on a spectrophotometer. The results were expressed in pg/mg of protein.
In this experiment, animals from groups described in “Study design” section were compared to a sham group since several observations at our laboratory have shown that basal levels of cytokines, measured by biochemical assays, are subjected to light variations related to factors that cannot be reasonably explained.
Statistical analysis
Using the GraphPad Prism 5.0 program, the Shapiro–Wilk test was used to confirm normality of the data within the groups (n = 8). Comparisons between two groups were performed by unpaired t-test. For comparisons between three or more groups, one-way or two-way analysis of variance (ANOVA) was used, followed by the Bonferroni test. Results were presented as mean ± standard error of the mean; p values less than 0.05 were considered statistically significant.
Results
Antihiperalgesic effect of HISP or ECE-CS
The results shown in Figure 3 demonstrate that the i.pl. injection of CFA caused a significant development of mechanical hyperalgesia (mimicking pain symptom observed in patients with OA), indicated by the increase in the basal frequency (before treatment) of withdrawal response of the animals on day 5 (day 5, B) in comparison to values before CFA (day 4, B: before CFA). However, a significant reduction in the values of frequency of response was observed when the animals were sent to HISP (maximum effect at 1 h before, exercised/vehicle median: 60.0 ± 11.3% frequency of response) or oral treatment with ECE-CS (not-exercised/ECE-CS: 40.0 ± 7.5% frequency of response). More importantly, it is still possible to observe that ECE-CS administration prior to exercise significantly augmented the antihyperalgesic effect for ECE-CS or exercise alone (exercised/ECE-CS: 22.9 ± 8.1% frequency of response) at the same time.
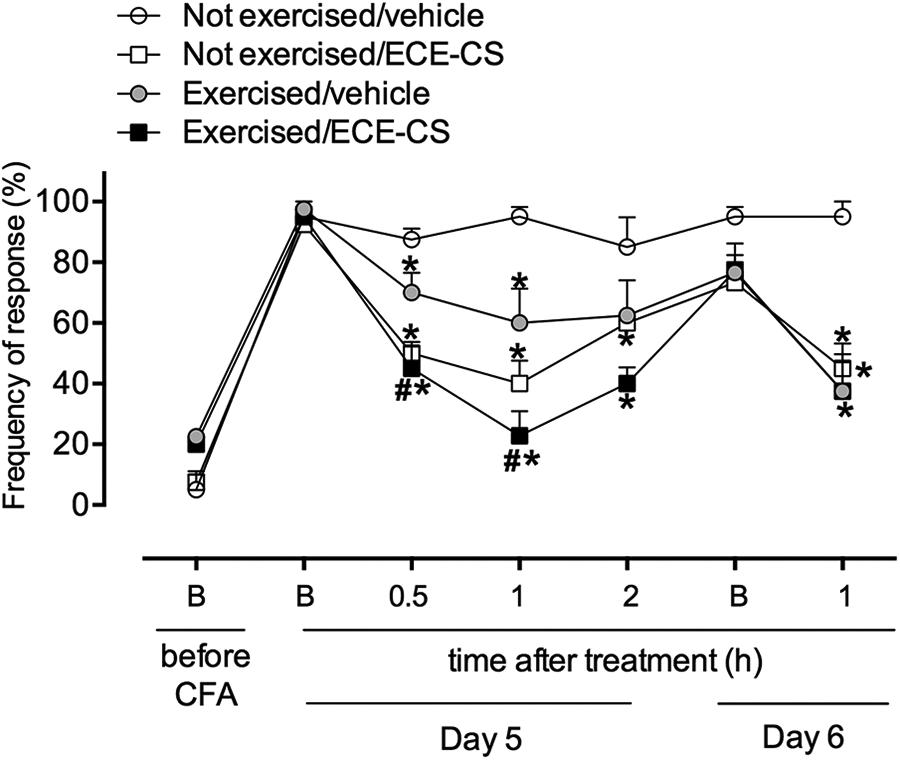
Influence of exercise and ECE-CS 30 mg/kg, alone or in association on mechanical hyperalgesia observed in an animal model of OA. Mechanical hyperalgesia was measured as the reaction elicited by 10 consecutive applications of von Frey stimulus (0.6 g), with positive response recorded when mice presented a sharp withdrawal of the stimulated paw from the mesh. Data are presented as mean ± SEM, n = 8 animals. The p value ≤0.05 in relation to not-exercised/vehicle group (*) or in relation to exercised/vehicle group (#). Two-way ANOVA followed by the Bonferroni test. ANOVA: analysis of variance; OA: osteoarthritis; SEM: standard error of the mean; ECE-CS: ethanolic crude extract of Casearia sylvestris.
Influence of the ECE-CS on locomotor activity
Treatment with ECE-CS did not influence the behavioral aspects of crossings (vehicle: 98.0 ± 18.7 s, ECE-CS: 104.7 ± 34.7 s; p = 0.257) or rearing (vehicle: 47.2 ± 3.6 s, ECE-CS: 37.8 ± 8.7 s; p = 0.085) in animals exposed to the open-field test 1 h after their treatment (Figure 4).
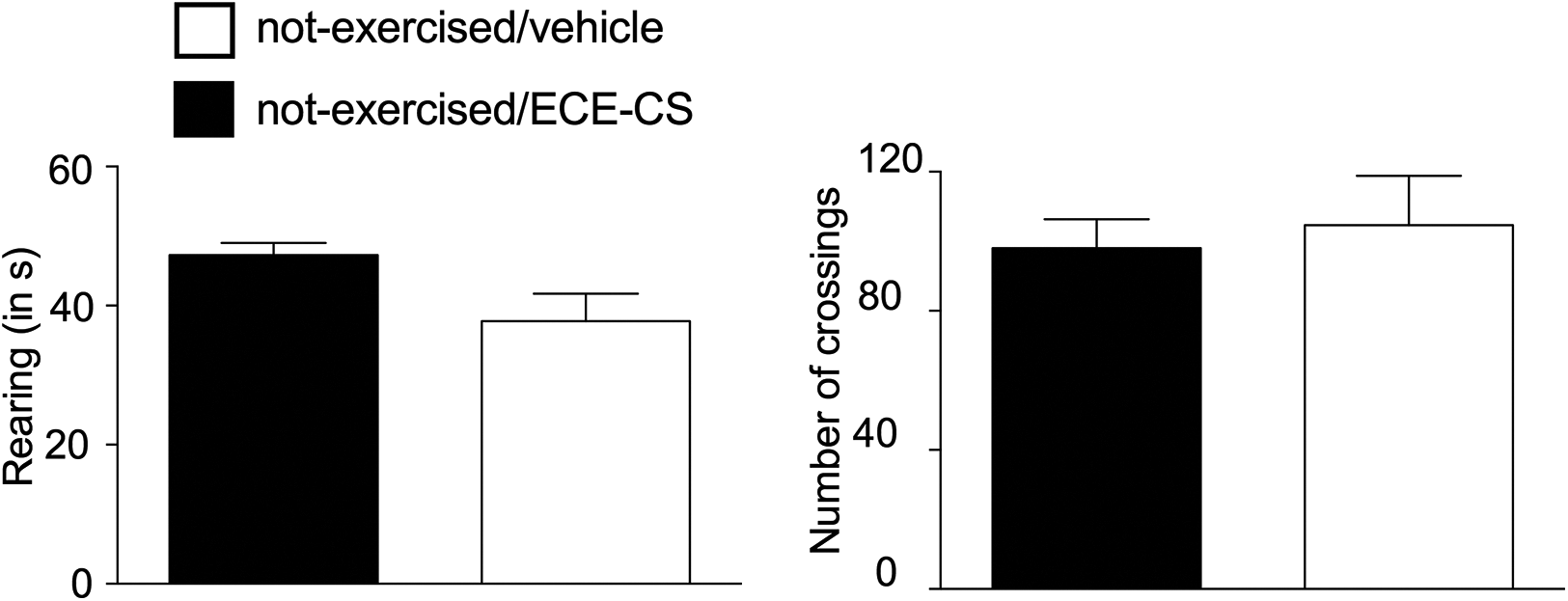
(a, b) Influence of the ECE-CS 30 mg/kg on locomotor activity of the mice in the open-field test, 1 h after treatment. Data are presented as mean ± SEM, n = 8 animals. Unpaired t test; no differences were observed between groups. SEM: standard error of the mean; ECE-CS: ethanolic crude extract of Casearia sylvestris.
Effect of HISP and oral ECE-CS on paw edema
The results shown in Figure 5 demonstrated that 24 h after the beginning of the experiment, there was an increase in the paw thickness of all the animals (day 5, B). In addition, it also showed that since the first day of treatment, 1 h after animals were sent to HISP alone (exercised/vehicle 2011 ± 100.8 µm) or in combination with ECE-CS (exercised/ECE-CS: 1749 ± 106.1 µm), a reduction in the edema was observed in relation to control animals (not-exercised/vehicle: 2468 ± 114.2 µm). Besides this, on the third day of treatment (day 7), all groups of animals presented a reduction of paw edema in relation to the control group (not-exercised/vehicle: 1484 ± 151.2 µm) even at the basal values (exercised/vehicle: 1113 ± 93.8 µm; exercised/ECE-CS: 960.0 ± 859 µm; not-exercised/ECE-CS: 971.8 ± 135.7 µm). However, differently from that observed for antihyperalgesic effect, no influence for ECE-CS administration on exercise (exercised/ECE-CS) was observed for the edema at all time-points evaluated in the present study.
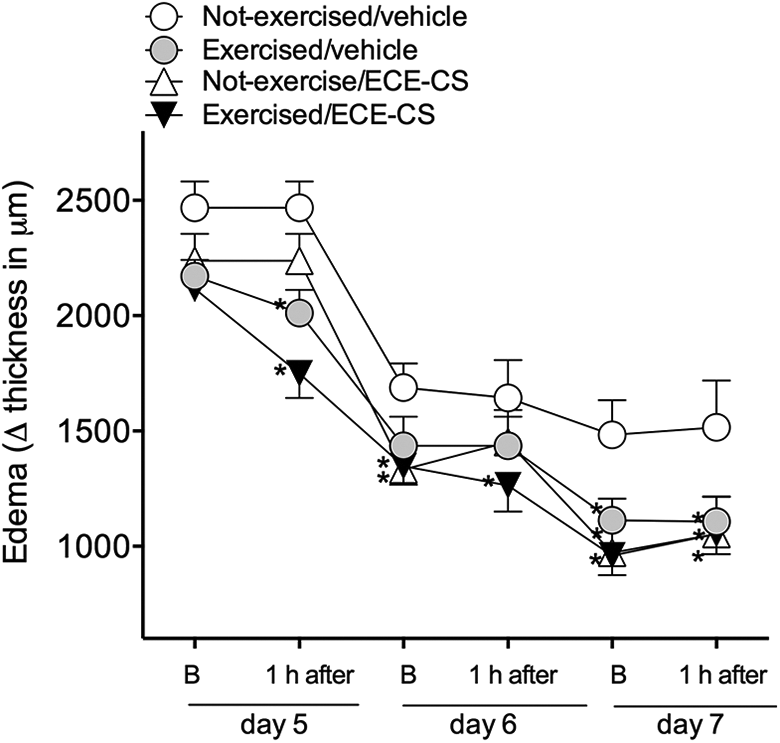
Influence of exercise and ECE-CS 30 mg/kg, alone or in association on edema observed in an animal model of OA. Edema was measured as the paw thickness (in μm) between basal values and that observed at several times after treatment. Data are presented as mean ± SEM, n = 8 animals. The p value ≤0.05 in relation to not-exercised/vehicle group (*). Two-way ANOVA followed by the Bonferroni test. ANOVA: analysis of variance; OA: osteoarthritis; SEM: standard error of the mean; ECE-CS: ethanolic crude extract of Casearia sylvestris.
Effect of HISP and oral ECE-CS tissue levels of IL-1β
As observed in Figure 6, on day 7 of the experiment (third day of treatment), IL-1β levels were raised in paw tissues from CFA control group (not-exercised/vehicle: 113.7 ± 12.5 pg/mg of protein) in comparison to the sham group (57.9 ± 3.0 pg/mg of protein); besides this, levels of IL-1β were reduced in animals from all groups of treatment (exercised/vehicle: 84.3 ± 1.9 pg/mg of protein; exercised/ECE-CS: 72.4 ± 9.6 pg/mg of protein; not-exercised/ECE-CS: 74.4 ± 7.6 pg/mg of protein).
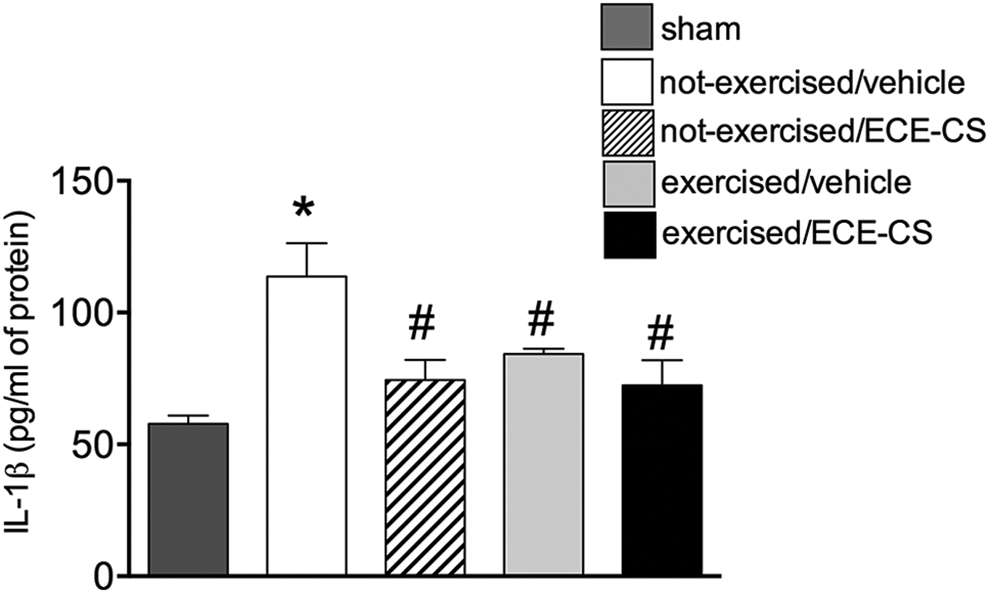
Level of IL-1β in paw tissues of mice under different treatments. Levels of cytokine were measured in pg/mg of protein, 1 h after treatment. Data are presented as mean ± SEM, n = 8 animals. The p value ≤0.05 in relation to sham (*) or in relation to not-exercised/vehicle group (#). One-way ANOVA followed by Bonferroni. ANOVA: analysis of variance; OA: osteoarthritis; SEM: standard error of the mean; ECE-CS: ethanolic crude extract of Casearia sylvestris.
Discussion
Preclinical studies demonstrating benefits from the association between different modalities of complementary therapies on OA can lead a venue for new therapeutic approaches for human patients suffering with this disease. The present work demonstrates that oral treatment of mice with ECE-CS augmented antihyperalgesic effect observed for HISP, suggesting that the association of both therapies (exercise + phytotherapy) can be considered more beneficial than each one isolated for the treatment of pain associated to OA.
In our point of view, this is particularly important since, to date, no data have been found in the literature investigating this approach. This fact reinforces the novelty of the study as well as the importance of the theme and the beneficial role of the combination of different medical approaches, as previously shown for different associations of complementary therapies. 23,24 The use of integrative health practices in rheumatology has been common with an increase in the interest of the affected patients. Thus, some complementary therapies may represent an opportunity to improve patients’ quality of life and, in the future, be integrated into the treatment of different pain conditions. 25
Besides this, it is still important to mention that it has already been suggested in the literature that the use of complementary therapies should be evaluated as to the possibility of their uses in a combined manner to investigate possible synergism between them. 25 By showing the augmentation of the antihyperagesic effect produced by exercise (HISP) with the previous use of a plant extract (ECE-CS), the present work is also important to strengthen evidence for the use of both therapies in humans, especially if considered that the treatment of the animals for three consecutive days with the ECE-CS did not promote behavioral changes related to locomotor activity, which was evaluated in the test that is in agreement with the absence of toxicity presented by the plant in another study. 26
Regarding the results on mice’s paw edema, the fact that CFA produced an increase in the paw thickness of all the animals in 24 h after its injection demonstrates the validity of the model in causing another symptom attributed to OA. Here again, the recent study demonstrated that both exercise and ECE-CS, alone or in combination, were effective in reducing this outcome. Antiedematogenic effect observed for ECE-CS reinforces data obtained previously with the hydroalcoholic extract of the plant 27 in the same dose used for ECE-CS in the present study. In the mentioned work, antiedematogenic property of the plant was attributed both to an antioxidant action as well as to its capability to inhibit neutrophil migration and myeloperoxidase activity in the injured site. Another possible property for the extract explored in a recent work that could interfere in edema and mechanical hyperalgesia here presented is its potential to promote activation of the pathways of resolution of inflammation, as recently suggested by Piovezan et al., 13 who evaluated the effect of the plant in the inflammatory phase of an animal model of complex regional pain syndrome type I.
Expanding these data, the present work also observed that while not-exercised animals presented a significant augment of IL-1β after CFA stimulation, all other groups that received any of the complementary therapies investigated (exercise or phytotherapy), alone or in combination, demonstrated reduction in the levels of this pro-inflammatory cytokine in paw tissues. IL-1β, together with tumor necrosis factor (TNF), plays a pivotal role in inflammatory cascade of mediators and is found locally or systemically in chronic inflammatory conditions, which was significantly reduced by exercise practice (review 28 ). In the present study, as no synergistic effect was observed in the group exercise/ECE-CS in relation to exercise-vehicle or not-exercise/ECE-CS groups, it is reasonable to hypothesize that TNF could be the other contributor, which played a role for the discrepancy observed in inflammatory response and functional performance.
Based on the literature, it is also speculated that swimming has its antihyperalgesic effect mediated by the activation of endogenous pain control systems, since the HISP used in the present study was adapted from a previous study, 15 that demonstrated, through a study involving an animal model of acute visceral nociception, a reduction in the pain behavior of animals exercised by the activation of opioidergic and serotoninergic receptors. Similarly, Lüdtke 16 used this same protocol of exercise and the same animal model of chronic pain induced by CFA and demonstrated the participation of the endocannabinoid system in the antihyperalgesic effect of HISP. These studies corroborate current data, strengthening evidence in the use of swimming in the treatment of different pain conditions and mimicking the benefits found in the clinic, as demonstrated in the study by other authors, who observed a significant improvement in pain and functional capacity in humans suffering from different disorders. 9
These findings are also in agreement with previous results that demonstrated analgesic and anti-inflammatory activity for CS. De Mattos et al. 14 demonstrated antinociceptive action for the hydroalcoholic crude extract of the plant in different models of nociceptive, inflammatory, and immunologic-based pain. Furthermore, it was later verified that the treatment of mice with this same type of extract promoted the reduction of leukocyte migration and myeloperoxidase enzyme activity in a pleurisy model. 27 In the present work, the reduction of IL-β observed in all groups of treatment (Figure 6) could be the result of its inhibitory effect on infiltration or activation of cells involved in the inflammatory process.
As a limitation of the present study, a gold standard drug that is currently in the market for reducing edema and hyperalgesia in patients could be used in the experiments of mechanical hyperalgesia and edema; this approach could be evaluated in future studies to better compare the efficacy of HISP and ECE-CS in relation to this drug.
In conclusion, the results presented here demonstrate for the first time ever the benefit of simultaneous use of ECE-CS and physical exercise on hyperalgesia observed in the CFA-induced pain in an animal model of OA as well as support the hypothesis that there is an advantageous consequence when associating these two complementary therapies, thus contributing to stimulate the evaluation of protocols using both complementary therapies in humans with the same clinical condition.
Footnotes
Acknowledgements
The authors also thank Ana Paula da Silva (EIU Unisociesc) for the English review.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from UNISUL/Brazil (Universidade do Sul de Santa Catarina, Programas PUIC e PIBIC-CNPq), CNPq/Brazil (Conselho Nacional de Desenvolvimento Científico e Tecnológico), CAPES/Brazil (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, PNPD and Finance Code 001) and FAPESC/Brazil (Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina, Call 09/2015).
