Abstract
Tuberculosis is a mycobacterial infection, but it can mimic many diseases including diseases of the musculoskeletal system. The need to consider it as a differential diagnosis of musculoskeletal tumours and tumour-like conditions cannot be overemphasized. The opinions of some, that tuberculosis is a disease of the undeveloped world, can no longer be sustained especially in the presence of immunodeficiency disorders, including HIV/AIDS, which have a worldwide prevalence. Traditionally, literature evidence shows that tuberculosis can mimic quite a number of diseases, including cancers. This has been found to be true, even in some technologically advanced countries. We present a case of hepatocellular cancer patient incidentally found to have concomitant hepatic tuberculosis who complained of bone lesion, with clinico-radiologic features of metastasis, but which turned out to be a tuberculous infection of the bone.
Keywords
Introduction
In all less than 10% of patients with tuberculosis have skeletal involvement. 1 A patient with musculoskeletal tuberculosis may not have any complaints related to the respiratory system at the time of diagnosis. 2 This explains the clinical diagnostic difficulty in differentiating osseous tuberculosis from metastatic bone tumours. Clinically, multifocal skeletal tuberculosis can closely mimic the distribution of multiple metastatic disease to the central skeleton, ribs, vertebrae and pelvis. 1,3 Modern radiological investigations, including Flourodeoxyglucose Positron Emission Tomography/Computerized Tomography (FDG PET/CT), may also not be able to conclusively distinguish between tuberculosis and metastasis or primary malignancy. Both of these diseases can produce areas of abnormally increased FDG activity on PET/CT. 2 Other types of infections and inflammatory conditions are also known to produce significantly increased FDG activity on the PET/CT scan. 4,5 High index of clinical suspicion as well as judicious biopsy procedures for both histopathological and microbiological examinations remains the gold standard for avoiding the embarrassment of diagnostic and treatment errors that otherwise may arise in some of these patients.
Another source of diagnostic concern in these patients is that they may not have any systemic symptoms and signs and may otherwise be fit and well. 1 This was the situation of the patient presented in this article. Evidence exists in the literature that two coexisting distinct non-malignant processes masqueraded as primary malignancy with spinal metastases on PET or FDG PET/CT. 2 These coexisting conditions proved to be a combination of lung sequestration and Pott’s disease. A greater diagnostic dilemma may arise when osseous tuberculosis presents in a patient with a suspected or background history of malignancy. Much greater dilemma may arise as in the index case when hepatic and osseous tuberculosis coexist with a hepatocellular carcinoma.
The aim of this article is to illustrate a case of osseous tuberculosis masquerading as bone secondary in a university hospital in Hong Kong and to demonstrate that a confusing clinical double pathology of tuberculosis and tumour may occur in the same patient.
Case report
A 58-year-old man and a known Hepatitis B carrier on lamivudine and adefovir was referred to the Department of Surgery for a suspected hepatocellular carcinoma in the left lobe of the liver. There was also a medical history of fatty liver and multiple benign liver cysts, detected on regular ultrasonographic liver surveillance for a Hepatitis B virus carrier. The patient was asymptomatic. He neither took alcohol nor cigarette. He had surgery for pectus carinatum at age 21. There was no history of pulmonary tuberculosis in the patient, but there was a history that his colleague at work had pulmonary tuberculosis. Physical examination was unremarkable.
The complete blood picture, renal function tests, liver function tests, bone profile, inflammatory and tumour markers were normal (Table 1). Triphasic contrast CT scan of the liver showed a 3.7 × 4.2 × 3 cm3 arterial enhancing mass with faint washout of enhancement in the portovenous phase, in the left lateral section and segment 2/4a of the liver. There were three benign-looking liver cysts in the right lobe, with the largest measuring 6.2 cm in the segment V of the right lobe. There was normal portal vasculature, no ascites and no obvious extrahepatic disease. A joint medical and radiologic review confirmed a diagnosis of hepatocellular carcinoma, and the patient was referred for surgical opinion, which was favourably inclined towards a left hepatectomy. Meanwhile, dual-tracer PET-CT scan showed C-11 avid liver tumour corresponding to the area outlined in the previous CT scan. No other hypermetabolic liver lesion was seen. There was also a markedly FDG-avid left proximal femoral lytic lesion with standardized uptake value (SUVmax) = 22.3, but mild C-11-acetate uptake at the level of the lesser trochanter. This raised a concern for a possible lytic osseous metastasis, but with a radiologist advise to exclude solitary bone lesion of second pathology such as myeloma. Patient was referred for Haematology and Orthopaedic reviews prior to the planned left hepatectomy. Plain radiography showed a lytic lesion in the left proximal femur at the level of the lesser trochanter (Figure 1). Bone scan showed moderate increased uptake at the left proximal femur. MRI of the left hip with contrast showed a 2.1 × 1.4 × 2.6 cm oval mass located centrally in the medulla of the proximal shaft of the left femur. There were homogenous intermediate signals on T1W1 and high signals on T2W1, a thin enhancing rim and adjacent bone marrow oedema (Figure 2). The radiologist’s opinion suggested a Brodie abscess and stated that the lesion was unlikely to be bone metastasis. Fluoroscopy-guided trephine biopsy of the left proximal femoral lesion showed no evidence of malignancy.
Laboratory indices.
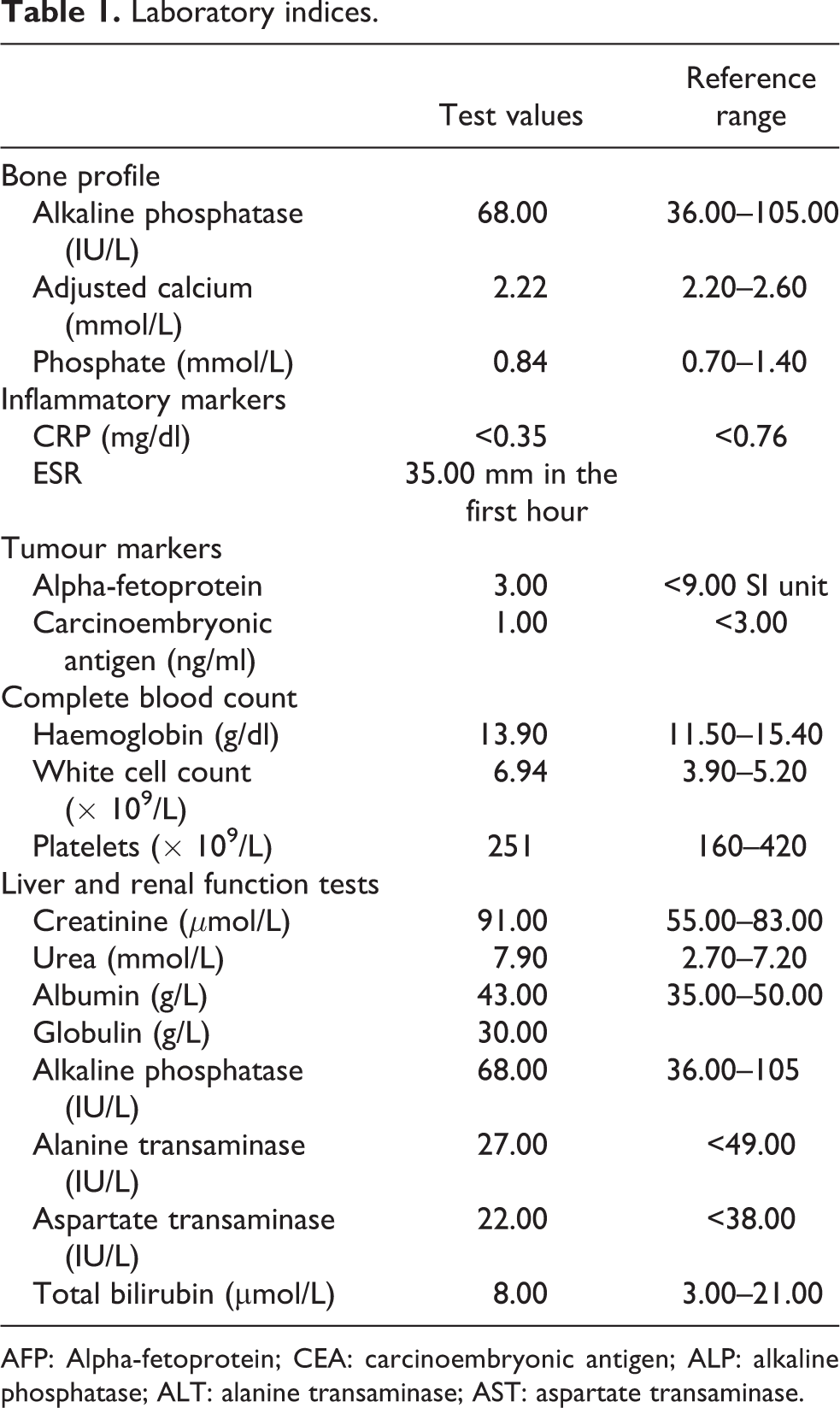
AFP: Alpha-fetoprotein; CEA: carcinoembryonic antigen; ALP: alkaline phosphatase; ALT: alanine transaminase; AST: aspartate transaminase.
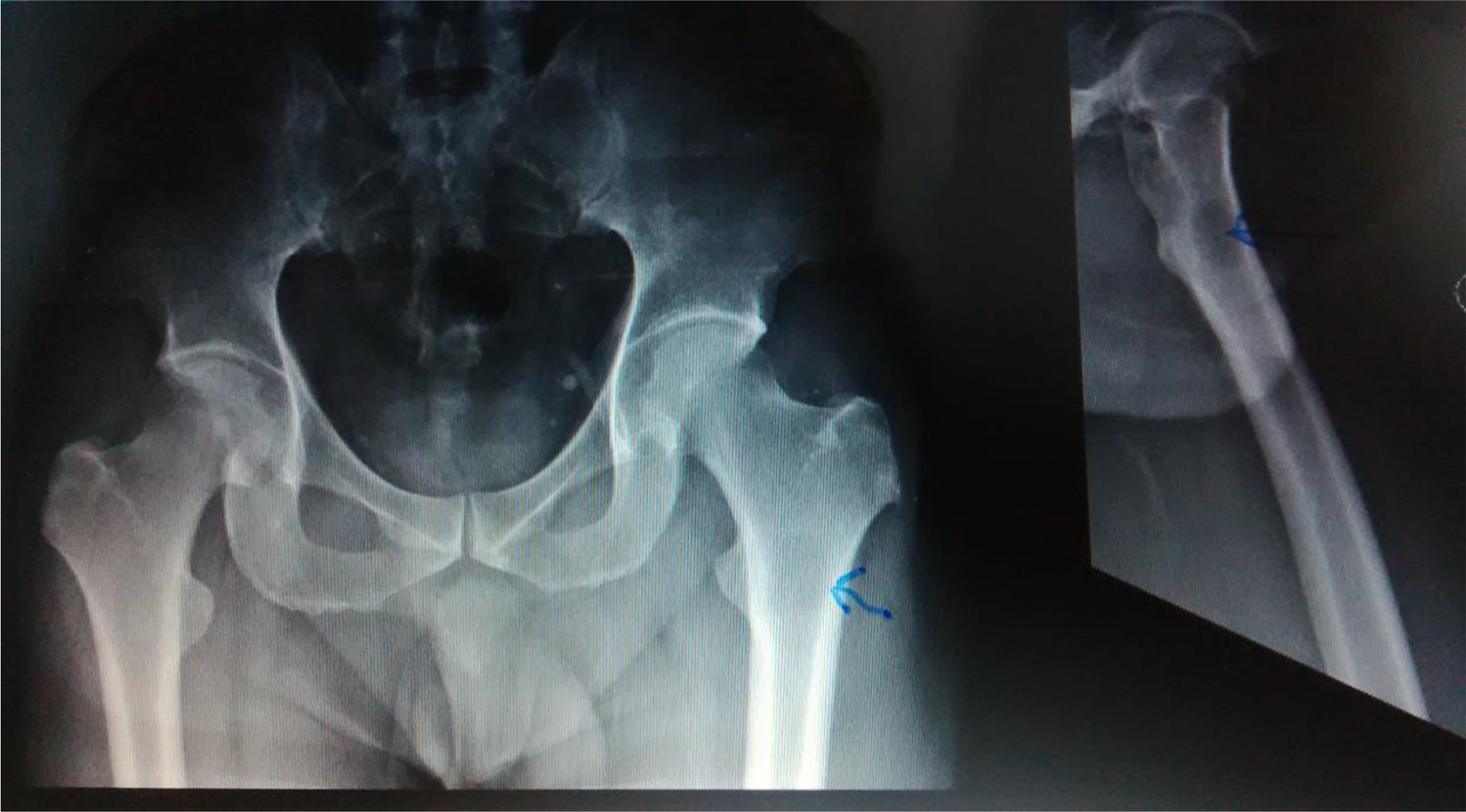
X-Ray showing circumscribed lytic lesion in the proximal left femur.
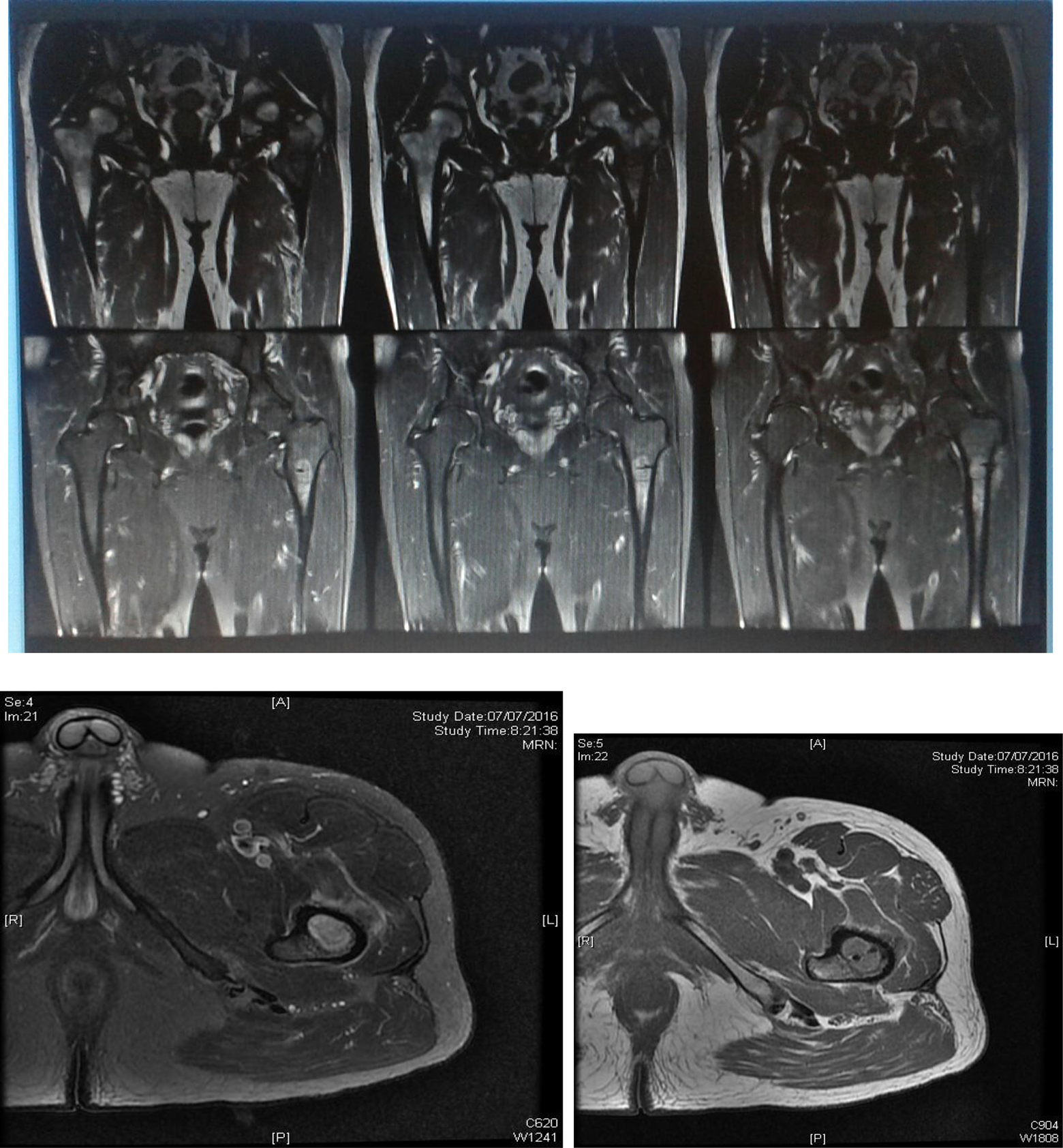
Coronal and axial MRI views of the left hip, showing enhancing lesion with thin enhancing rim and moderate surrounding marrow oedema.
The surgical team opted for a left hepatectomy with an open biopsy of the bony lesion by the orthopaedic team at the same operative session. The intraoperative findings include 4.5 × 2.5 cm well-encapsulated tumour located at the segment 2 region, extending to segment 4b. There was also a 0.8 × 0.8 cm malignant looking tumour located at the subcapsular region of segment 3 and segment 6. Frozen section confirmed this as a granuloma. There was a 0.5 × 0.5 cm whitish nodule at segment 5 near the gall bladder fossa whose frozen section confirmed to be biliary adenoma. There were also multiple liver cysts with the largest located at the tip of the segment 6. Frozen section for bone biopsy tissue obtained at the same operative session showed infective lesion with no viable tumour cells seen. The final pathology report on the bone biopsy tissue showed fragmented pieces of bone with necrosis and granulomatous inflammation. Ziehl-Neelsen stain showed a few acid-fast bacilli. Subsequent culture yielded mycobacterium tuberculosis.
The final pathological report of the liver biopsy showed a well-differentiated hepatocellular carcinoma, chronic hepatitis, as well as mixed lymphocytes and multinucleated giant cells in the section nodules of the liver lesion. Caseous necrosis was observed, and Ziehl-Neelsen stain showed occasional acid-fast bacilli.
The Patient was reviewed by microbiologists. The femoral lesion was polymerase chain reaction (PCR) positive. He was commenced on anti-tuberculous chemotherapy. Subsequent follow-up in the orthopaedic clinic showed that the femoral lesion has been reducing in size and the blood parameters were all within normal limits. At 1-year after commencement of anti-tuberculous drugs, patient had no pain, erythrocyte sedimentation rate and C-reactive protein were normal, and X-ray of the proximal femur was normal (Figure 3).
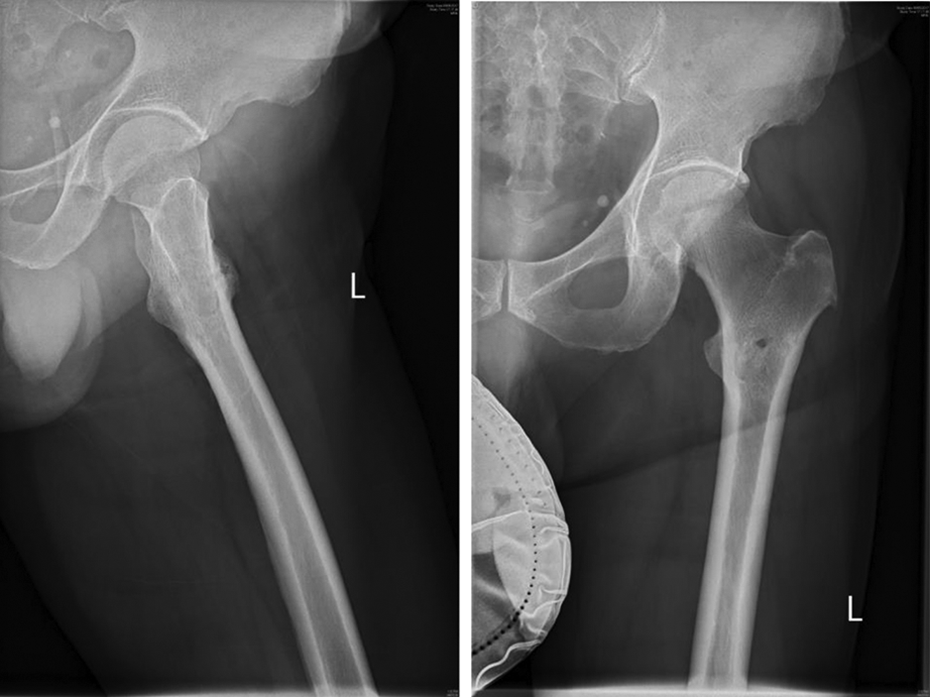
Anteroposterior and lateral X-ray views of the proximal femur at 1-year following anti-tuberculous treatment.
Discussion
It often comes as a surprise to the surgeon whenever histological examinations of biopsy specimens from suspected malignant lesions reveal pathology other than malignancy. The finding of granulomatous inflammation without evidence of malignancy in a biopsy specimen from a suspected malignant lesion might be the first evidence that the lesion is tuberculous. Less than 10% of patients with tuberculosis have skeletal involvement, 1,6 and in more than 50% of cases, there is no evidence of concurrent active intrathoracic tuberculosis. 6 Extrapulmonary tuberculosis is difficult to diagnose, because of its tendency to mimic any condition, with non-specific clinical and radiological features suggestive of other disease conditions including malignancy. 1,7 The most common site of osseous involvement is the spine, followed by the femur, tibia and the small bones of the hands and feet. Indeed, any bone can potentially be affected. 6,8 Therefore, in suspected cases of primary bone tumour or metastatic bone disease, it is safer that the differential diagnosis of skeletal tuberculosis be considered, particularly in patients from tuberculosis-endemic regions. 1 The emergence of multidrug-resistant strains of mycobacterium tuberculosis, increase in the number of immune-compromised individuals and increase in the number of immigrants from developing world have been thought to be responsible for a significant rise in the prevalence of extrapulmonary tuberculosis in the developed world in recent times. 7
In general, it is pertinent to maintain a high level of clinical suspicion of the possibility of tuberculosis when evaluating suspicious osseous lesions, including malignancy. This is because musculoskeletal imaging studies, including modern radiotracer techniques such as FDG PET-CT, may still not be able to conclusively discriminate between tuberculous lesions in one extreme and malignant lesions in another. 2,3,7 It is this suspicion that underscores the need to also include microbiological studies among the investigations that should be carried out on tissue biopsy specimens from suspicious lesions. The investigations such as Mantoux test on the patient, Ziehl-Neelsen test, PCR test and tissue culture on biopsy specimen are the relevant tools for identifying tuberculous lesions from very close differential diagnoses such as metastatic bone disease or primary bone sarcoma. 1
There is a wide variation in the specific diagnostic yield of the various imaging studies available for evaluating and distinguishing tuberculosis from more sinister bone lesions. It is believed that certain key radiological patterns can help in clinching the diagnosis of extrapulmonary tuberculosis, but these patterns in themselves are not pathognomonic. 7 The whole-body isotope scanning is a non-specific investigation. It can reveal increased tracer uptake in areas of increased osteoblastic activity or increased blood flow. 1,7 On the other hand, MRI scan is thought to be more sensitive and specific and capable of detecting tuberculosis lesions, especially in the spine. The characteristic features on T2-weighted images show increased signal within the affected bone. 9 Notwithstanding, there can still be diagnostic dilemma with these imaging techniques, since these features, as pointed out earlier, are not pathognomonic. Plain radiography is even much inferior to either of the foregoing modalities in the evaluation of suspicious bone lesions. 1,9 At best, plain radiography shows findings that are non-specific, variable, inconstant and can be found in a range of pathological processes, including neoplasia. 7 Therefore, a multimodal approach comprising high index of suspicion, imaging techniques, with histopathological and microbiological confirmations, is the key to resolving the diagnostic dilemma of differentiating osseous tuberculosis from metastatic bone tumours or primary bone sarcomas. To this end, surgical biopsy tissue can now be safely and reliably obtained with minimally invasive techniques under image guidance. 7 Like the imaging techniques, the diagnostic yield of the laboratory tests also varies. It has been reported that acid-fast bacilli was not visible on Ziehl-Neelsen staining of the biopsy specimen of a particular case, but yet the PCR was positive for mycobacterium tuberculosis DNA, and the tissue culture yielded mycobacterium tuberculosis after 6 weeks. 7 This serves to emphasize the need to carry out a whole spectrum of all possible investigations on the biopsy specimens. Positive Mantoux test and chest imaging findings further strengthen the diagnosis of skeletal tuberculosis, but their absence does not exclude it. 10
Conclusion
Osseous tuberculosis can mimic any disease. The presenting complaints of pain and swelling are largely non-specific, with a risk of incorrect initial diagnosis and delay in relevant treatment. There is a need for high level of clinical suspicion on the part of the clinicians. Virtually, any bone can be affected, and this makes the diagnosis even more challenging. Judicious use of multiple imaging techniques augmented by microbiological examinations of biopsy specimens, in addition to histopathological examinations, is the key step in avoiding the diagnostic dilemma that osseous tuberculosis can elicit.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
