Abstract
Study design
Retrospective comparative cohort study.
Objective
To validate FDA IDE–sponsored trial findings on cervical disc replacement (CDR) and anterior cervical discectomy and fusion (ACDF) by comparing reoperation outcomes with real-world data.
Methods
Patients undergoing one- or two-level ACDF or CDR from 2010-2022 were identified in the PearlDiver (PD) database. FDA IDE trials were retrieved through review of databases. Subsequent surgeries and sample sizes were extracted at 2-, 4-, 5-, 7-, and 10-year intervals. Event rates were calculated as reoperations divided by patients at risk, and survival probabilities as S(t) = 1−event rate. Kaplan–Meier–style survival curves were reconstructed using the Guyot algorithm with a monotonicity constraint. Pooled FDA survival probabilities were weighted by number at risk. Hazard ratios (HRs) were derived from log-transformed survival probabilities, and statistical significance was tested using Pearson’s chi-square or Fisher’s exact test. Analyses were stratified by surgical level (1- vs 2-level) and procedure type (CDR vs ACDF).
Results
In total, 277 146 ACDF and 25 957 CDR patients from PD and 46 FDA trials were analyzed. CDR reoperation risks were similar between FDA and PD cohorts, though differences were significant at 2-, 7-, and 10-year follow-up for 1-level procedures. ACDF reoperation risks were consistently and significantly lower in PD (eg, HR 0.4 at 7 years).
Conclusion
Reoperation risks for CDR were broadly comparable, while ACDF outcomes were superior in practice compared with trials. These differences may reflect trial selection bias or stricter adjudication of ACDF outcomes. Validation against real-world data supports long-term safety assessment and personalized surgical decision-making.
Keywords
Introduction
Degenerative cervical spine disease is a common disorder characterized by neck pain, radiculopathy, and myelopathy. 1 Early surgical treatment involved laminectomy and posterior forminotomy, however, due to anatomical limitations in effectively addressing centrally or paracentrally located discs, anterior cervical discectomy and fusion (ACDF) emerged as the favorable method by the mid-20th century. 2 ACDF is now regarded as the gold standard for degenerative cervical disc disease. 3
To address the limitations of ACDF, particularly reduced spinal mobility and adjacent segment disease (ASD), cervical disc replacement (CDR) was developed as a motion-preserving alternative. Since its commercial debut in 2007, 4 CDR use has grown significantly.5-8 Currently, 9 devices are approved for single-level CDR, with 3 also approved for two-level procedures.4,9-17 Clinical trials have shown CDR to be non-inferior to ACDF, with better long-term outcomes in pain relief, function, and lower rates of ASD and revision surgery.
CDR approvals were based on FDA investigational device exemption (IDE) trials, which were non-blinded, multicenter randomized controlled trials. 18 However, concerns have been raised about the rigour of these studies due to limitations in design, bias, and post-approval oversight.19,20 Although post-approval studies (PAS) are required, 18 they often suffer from small sample sizes, poor follow-up, and limited generalizability. 21
Given these limitations, this study aims to validate the findings of FDA IDE-sponsored trials by comparing long-term outcomes of CDR and ACDF using real-world data from a national database. Specifically, this study will assess subsequent surgery rates reported in the FDA trials and compare them to those observed in a large, nationally representative cohort over the last decade.
Methods
Data Source and Study Population
This study utilized the PearlDiver (PD) Mariner Database (PearlDiver Technologies, Colorado Springs, CO, USA) to identify patients who underwent ACDF and CDR between 2010 and 2022. The PD database is a nationwide insurance claims database containing approximately 157 million de-identified patients, that includes demographic, diagnostic, and procedural data from various payer types across inpatient and outpatient settings. As the study relied on anonymized data, already published articles and was retrospective in nature, informed consent was waived, and institutional review board approval was not required.
Pubmed, Scopus and Cochrane databases were used to identify FDA-approved IDE trials on April 14, 2025.
Identification of Study Cohorts in PearlDiver
International Classification of Diseases (ICD), 9th (ICD-9) and 10th (ICD-10) editions, along with Current Procedural Terminology (CPT) codes, were utilized to identify patients (Supplemental Table 1). Specifically, patient records containing codes for one- or two-level ACDF and CDR procedures were queried. To exclude hybrid surgical constructs, patients in the ACDF group were removed if they had undergone CDR within 1 week of the index procedure. Similarly, patients in the CDR group were excluded if they had undergone ACDF within 1 week of the procedure. Patients in both study cohorts were also excluded if they had a history of prior cervical surgery before the index ACDF or CDR procedure. Additional exclusion criteria included patients under 18 years of age, those with less than 1 year of postoperative follow-up, and cases involving surgical indications related to trauma, malignancy, or infection. Patients were further categorized based on subsequent surgeries performed in the cervical region.
To simulate an intention-to-treat analysis similar to FDA IDE trials, only patients with complete follow-up data were included. For example, in the 2-year cohort, patients were included if their index surgery occurred between 2010 and 2020, as PD contains data through 2022, ensuring all patients could have full follow-up. Similarly, patients in the 3-year cohort were included if their index surgery occurred between 2010 and 2019; for the 4-year cohort, between 2010 and 2018; for the 5-year cohort, between 2010 and 2017; for the 7-year cohort, between 2010 and 2015; and for the 10-year cohort, between 2010 and 2012.
Identification of Study Cohorts in FDA IDE Trials
Pubmed, Scopus and Cochrane databases were queried since inception on April 14, 2025, using human participants and the English language as a search filter and (“Anterior Cervical Discectomy and Fusion” OR “ACDF” OR “Cervical Discectomy” OR “Fusion” OR “Cervical Spine Fusion” OR “Cervical Degenerative Disease” OR “Cervical Degeneration”) AND (“Cervical Disc Replacement” OR “CDR” OR “Cervical Total Disc Replacement” OR “CTDR” OR “Total Disc Replacement” OR “TDR” OR “Artificial Disc Replacement” OR “Cervical Arthroplasty” OR “Disc Arthroplasty” OR “Cervical Degenerative Disease” OR “Cervical Degeneration”) AND (“Randomized Controlled Trial” OR “RCT” OR “Randomized Study” OR “Clinical Trial” OR “Interventional Study”) as a search strategy. Our exclusion criteria included trials that compared devices not approved by the FDA for commercial distribution or that reported subsequent surgery rates beyond a 10-year follow-up period due to the small sample size available for comparison in the PD database. Duplicates were removed, and relevant full-text randomized controlled clinical trials (RCT) were screened for data collection following the title and abstract screening (Figure 1). Flow Chart for the Selection of FDA IDE Trials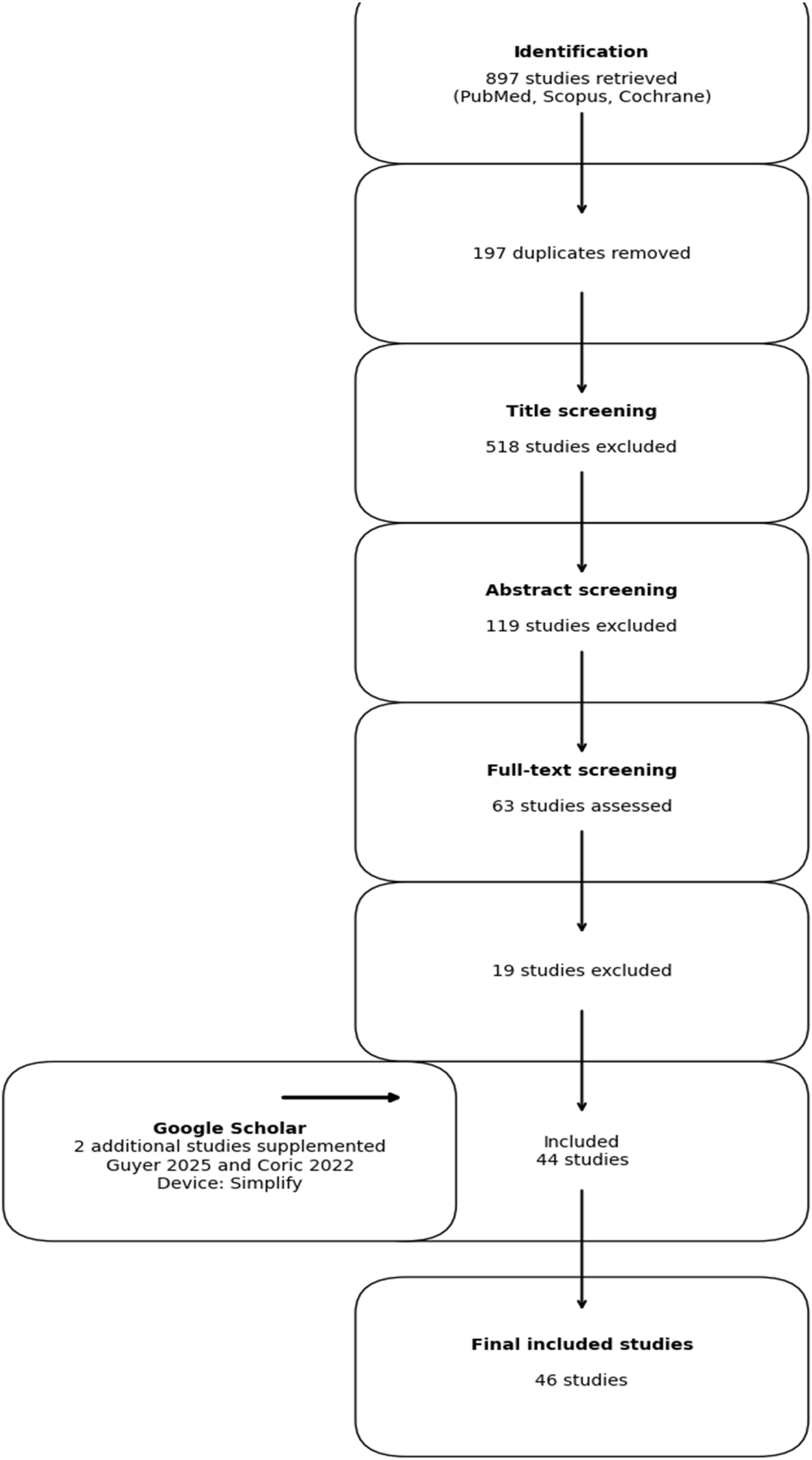
Primary Endpoints
Subsequent Surgery Event In PearlDiver And FDA IDE Trials Cohort.
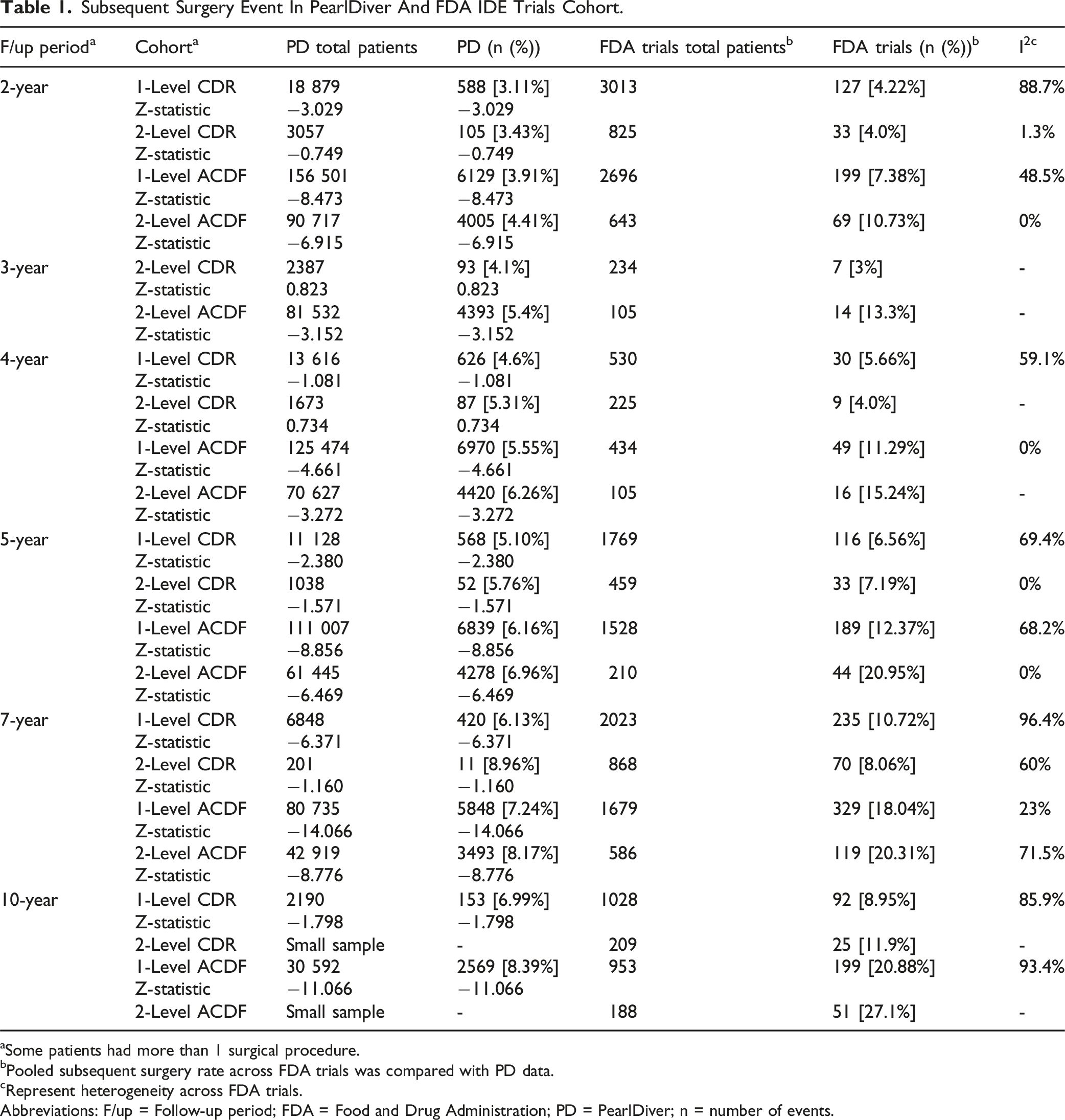
aSome patients had more than 1 surgical procedure.
bPooled subsequent surgery rate across FDA trials was compared with PD data.
cRepresent heterogeneity across FDA trials.
Abbreviations: F/up = Follow-up period; FDA = Food and Drug Administration; PD = PearlDiver; n = number of events.
Statistical Analysis
Demographics of PearlDiver Patients Cohort.
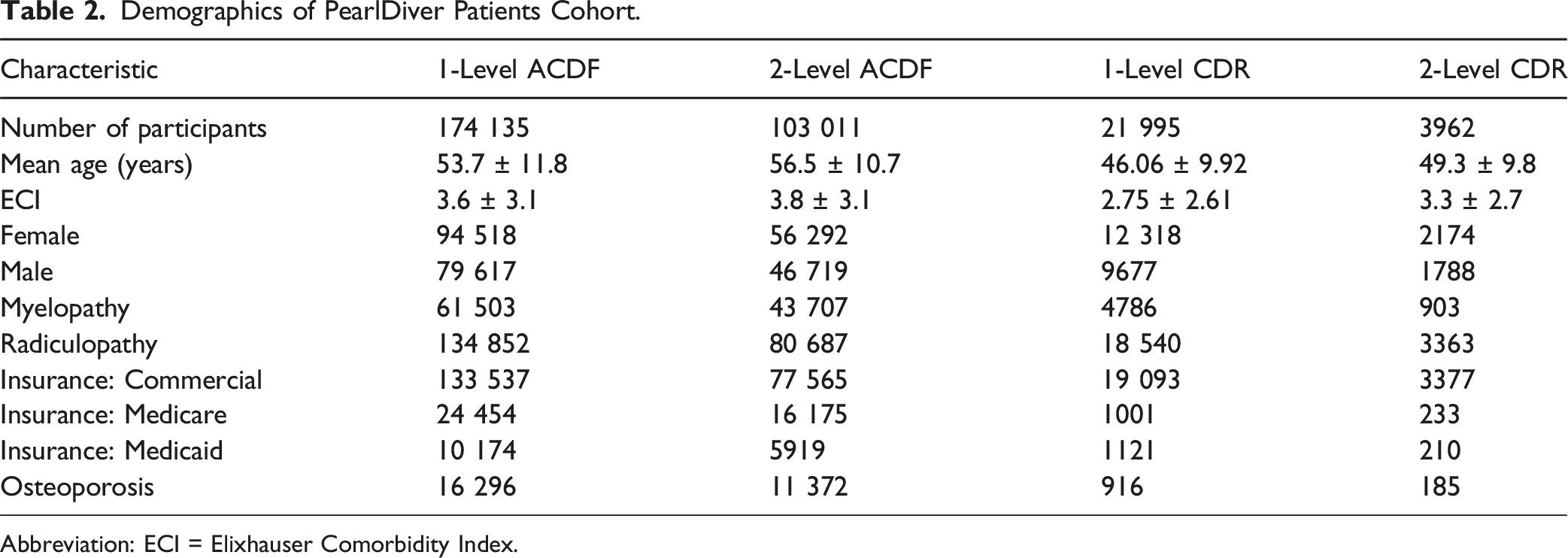
Abbreviation: ECI = Elixhauser Comorbidity Index.
Demographics of FDA Trials Patient Cohort.
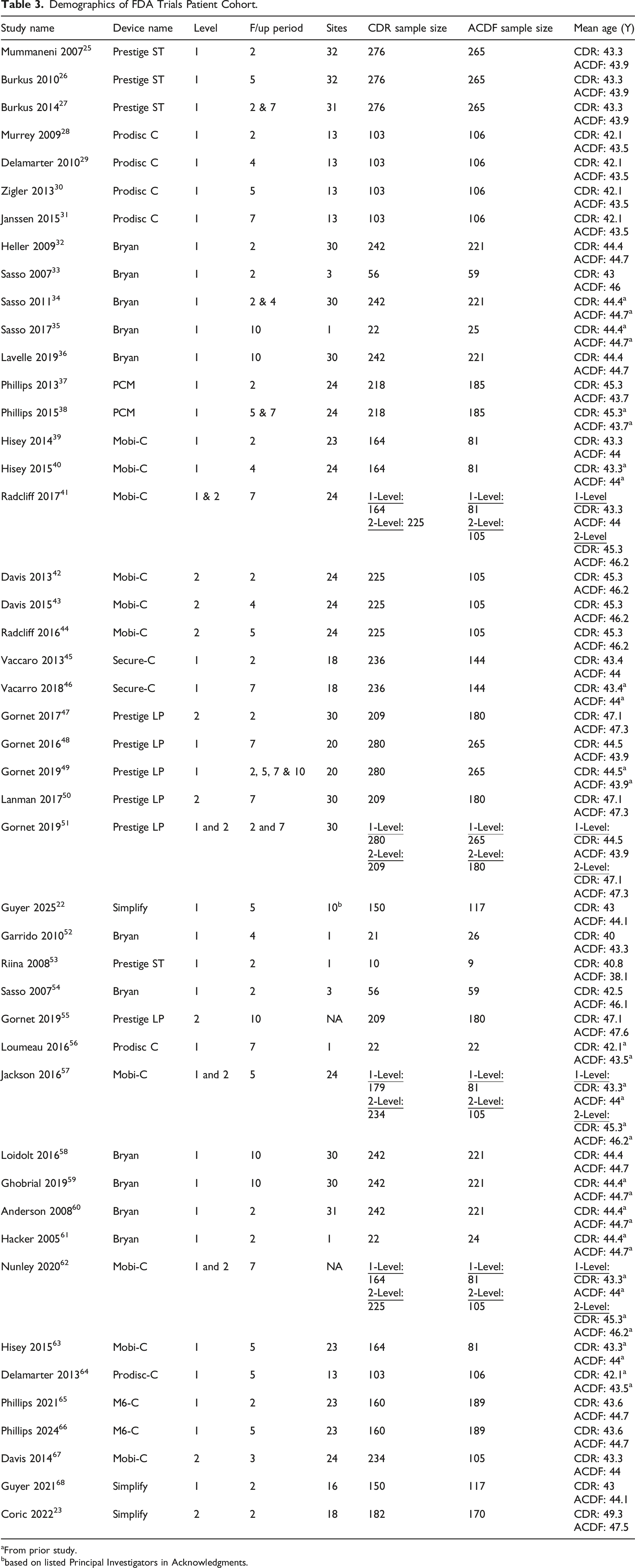
aFrom prior study.
bbased on listed Principal Investigators in Acknowledgments.
To generate Kaplan–Meier–style survival curves from aggregate data, patient-level event times were reconstructed from the published discrete survival probabilities using the algorithm described by Guyot et al (2012) 22 for individual patient data (IPD) reconstruction from published Kaplan–Meier curves. In cases where reported survival probabilities showed non-monotonic trends (ie, survival increasing over time), a monotonicity constraint was applied by replacing each value with the cumulative minimum of all preceding survival probabilities. This ensured biologically plausible curves and addressed potential artefacts arising from small sample sizes or reporting variability. For each surgical level (1-level, 2-level) and procedure type (CDR, ACDF), pooled FDA survival probabilities were calculated as weighted means, with weights proportional to the number at risk in each trial.
For comparative analyses, hazard ratios at each follow-up interval were calculated from survival probabilities using the formula: HR (t) = −log SPD (t)/−log SFDA(t). While these values do not represent Cox regression–derived hazard ratios, they provide a consistent relative measure of survival differences between FDA and PD cohorts at discrete time points. Statistical significance of differences in event rates between FDA and PD cohorts at each follow-up interval was assessed using Pearson’s chi-square test when all expected cell counts were ≥5, and Fisher’s exact test otherwise. A two-sided P value <0.05 was considered statistically significant. All analyses were conducted separately for each surgical level and procedure type. Survival curves were plotted for visual comparison, and numerical results including HR values and corresponding P values, were tabulated.
Result
Index And Adjacent Segment Surgery Event in FDA Trials Patient Cohort.
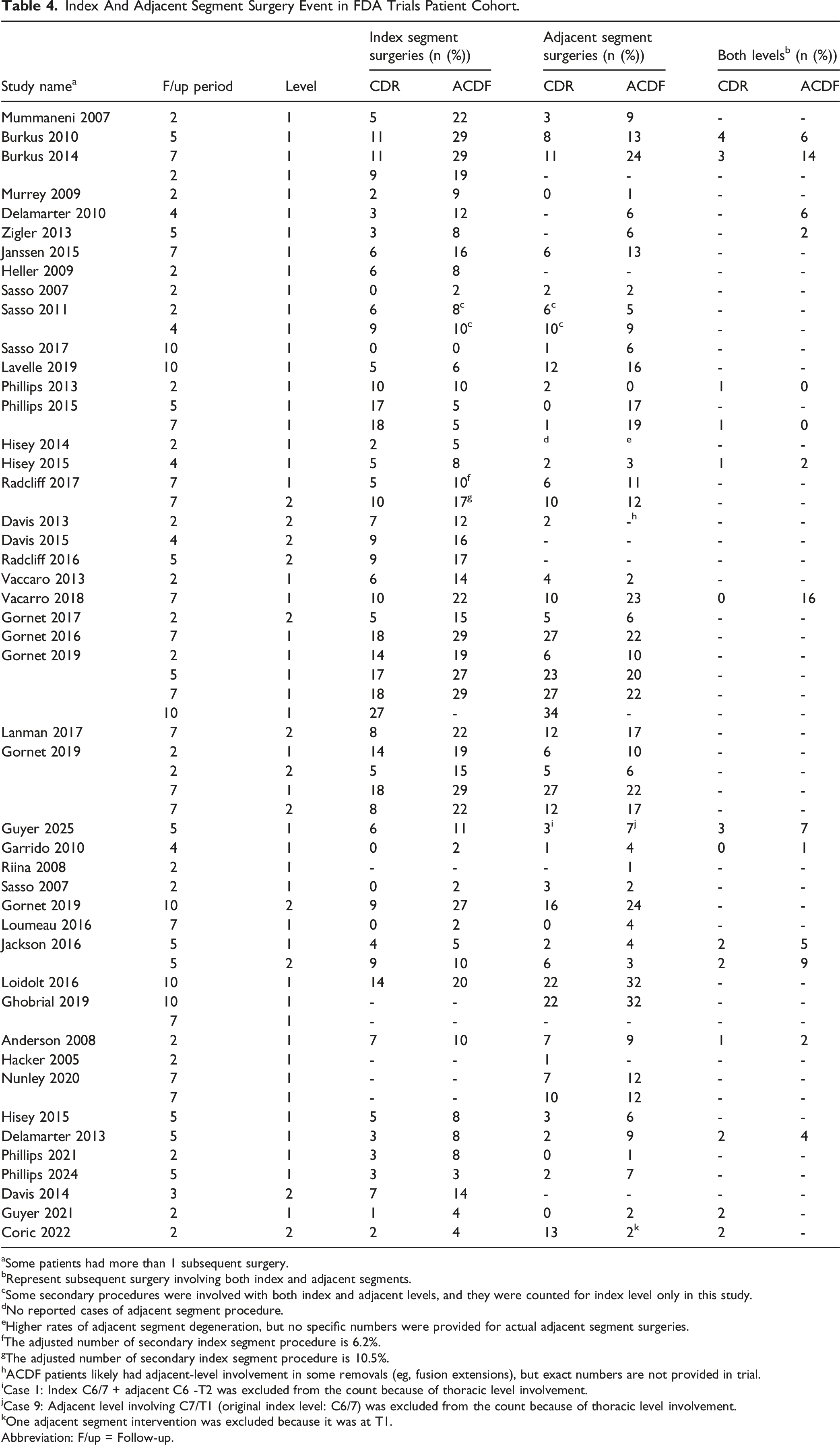
aSome patients had more than 1 subsequent surgery.
bRepresent subsequent surgery involving both index and adjacent segments.
cSome secondary procedures were involved with both index and adjacent levels, and they were counted for index level only in this study.
dNo reported cases of adjacent segment procedure.
eHigher rates of adjacent segment degeneration, but no specific numbers were provided for actual adjacent segment surgeries.
fThe adjusted number of secondary index segment procedure is 6.2%.
gThe adjusted number of secondary index segment procedure is 10.5%.
hACDF patients likely had adjacent-level involvement in some removals (eg, fusion extensions), but exact numbers are not provided in trial.
iCase 1: Index C6/7 + adjacent C6 -T2 was excluded from the count because of thoracic level involvement.
jCase 9: Adjacent level involving C7/T1 (original index level: C6/7) was excluded from the count because of thoracic level involvement.
kOne adjacent segment intervention was excluded because it was at T1.
Abbreviation: F/up = Follow-up.
Survival Probability And Hazard Ratio.
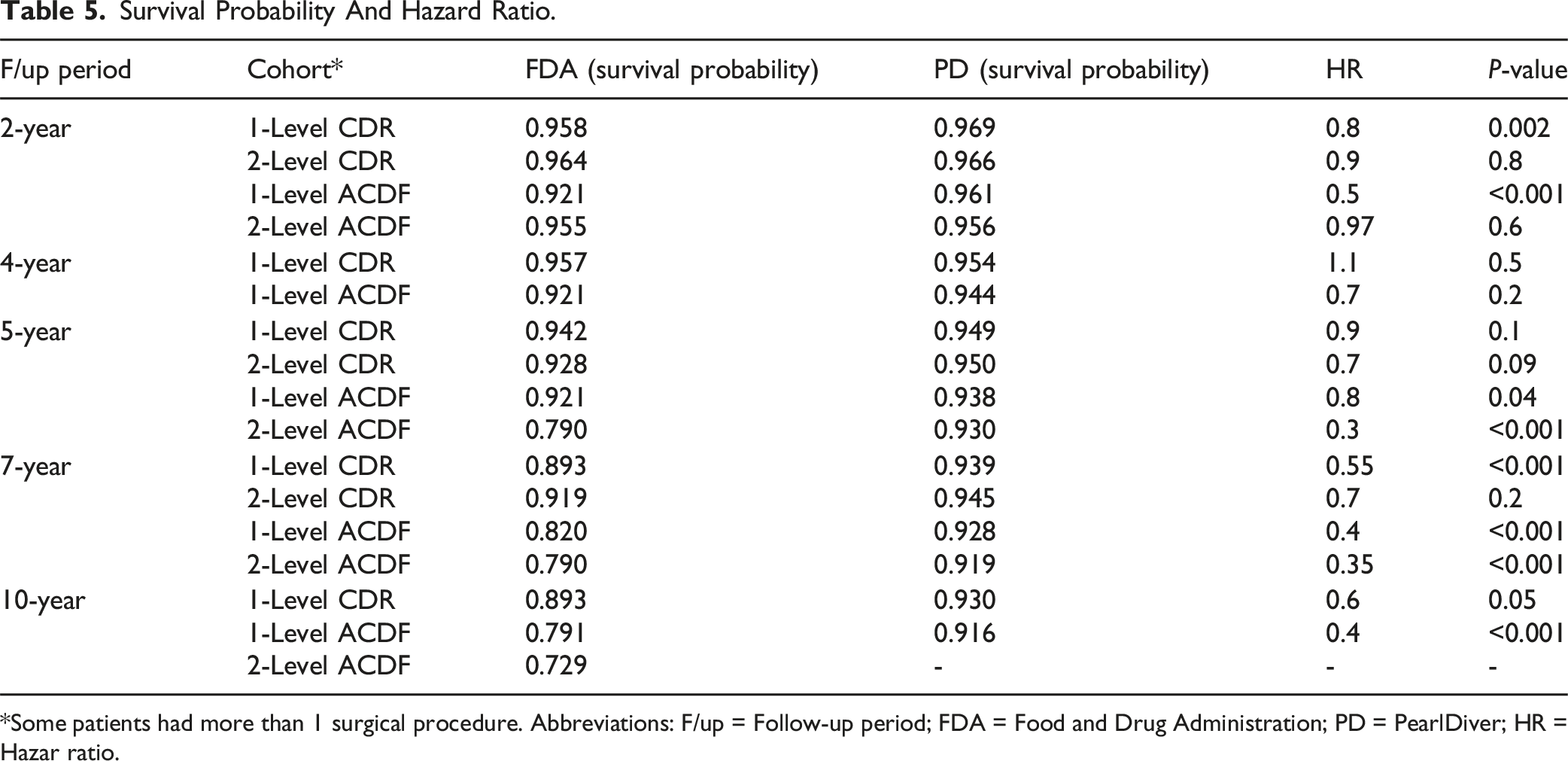
*Some patients had more than 1 surgical procedure. Abbreviations: F/up = Follow-up period; FDA = Food and Drug Administration; PD = PearlDiver; HR = Hazar ratio.
Survival Analysis
Level 1 ACDF
In the Level 1 ACDF cohort, the PD group consistently demonstrated higher survival probabilities compared to the FDA IDE cohort across most follow-up intervals. At year 2, survival was 96% in the PD group vs 92% in the FDA group (P < 0.001). This difference persisted at year 5 (PD: 94% vs FDA: 92%, P = 0.038) and became more pronounced at years 7 and 10, where PD maintained survival above 92%, while the FDA group declined to 82% at year 10 (P < 0.001). The separation between curves widened substantially after year 7, highlighting a sustained survival advantage for the PD cohort at longer follow-up durations. (Figure 2). Kaplan–Meier Survival Curve (ACDF, Level 1)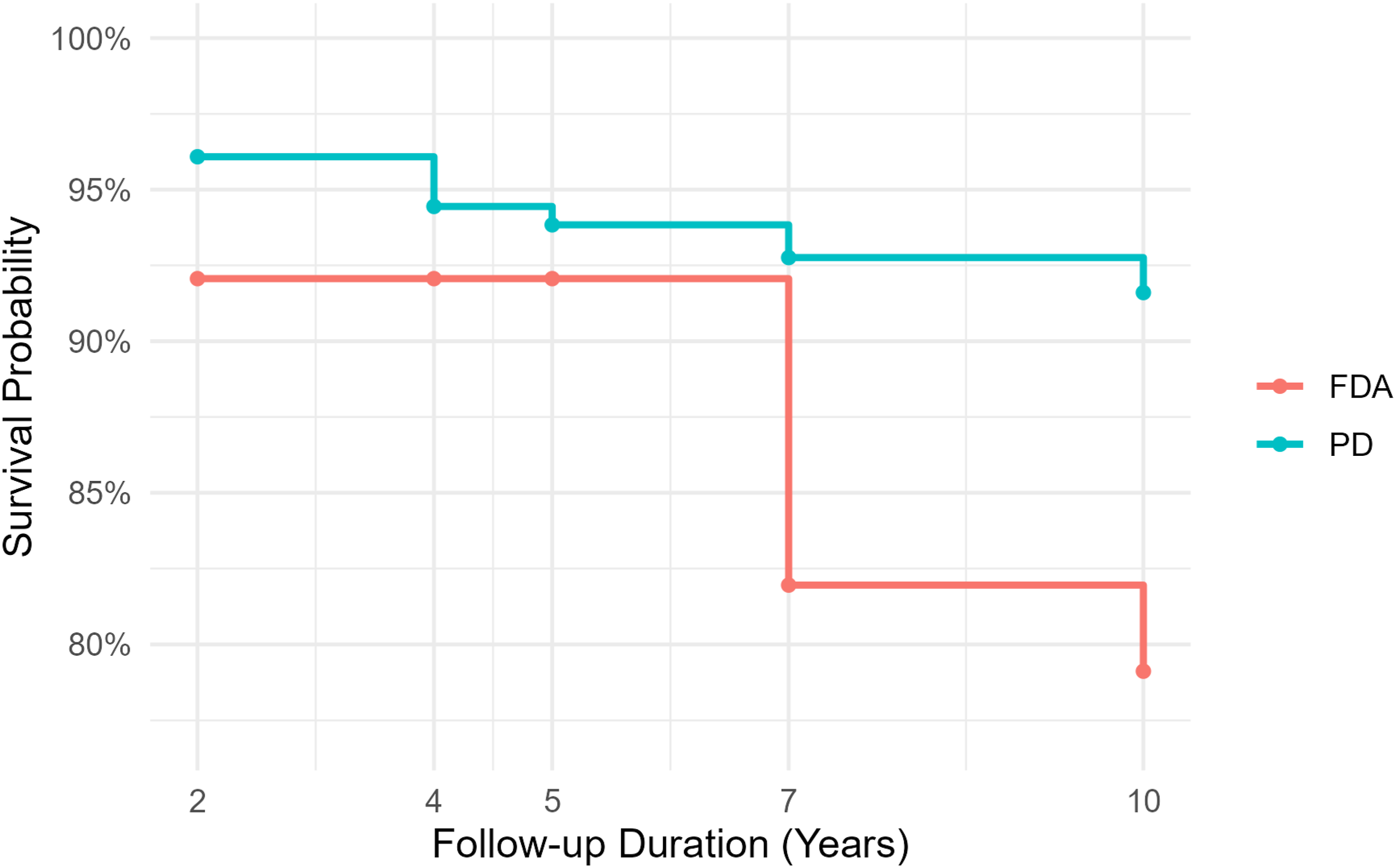
Level 2 ACDF
In the Level 2 ACDF cohort, the PD group showed consistently higher survival probabilities than the FDA IDE cohort at mid- and long-term follow-up. At year 2, both groups had nearly identical survival (PD: 95.6% vs FDA: 95.5%, P = 0.645). However, by year 5, the difference became striking, with PD maintaining a survival probability of 93% compared to only 79% in the FDA cohort (P < 0.001). This trend continued at year 7, where survival remained higher in the PD group (92% vs 79% in FDA, P < 0.001). (Figure 3). Kaplan–Meier Survival Curve (ACDF, Level 2)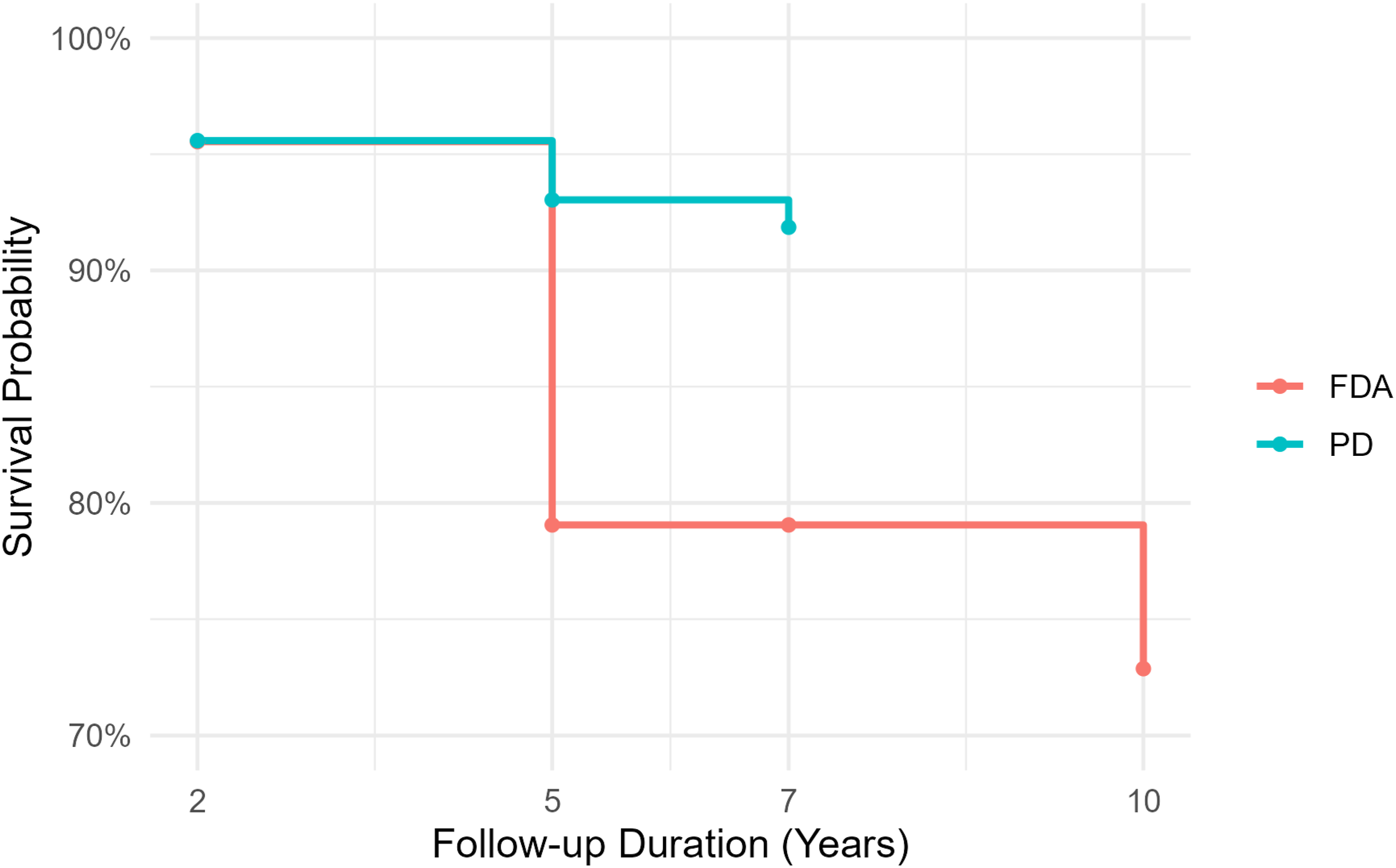
Level 1 CDR
In the Level 1 CDR cohort, both the FDA IDE and PD groups demonstrated high survival probabilities throughout follow-up, but the PD group consistently maintained a modest survival advantage. At year 2, survival was 97% in the PD cohort compared to 96% in the FDA group (P = 0.002). Differences narrowed at years 4 and 5, where survival remained above 94% in both groups and no statistically significant differences were observed. However, by year 7, the PD cohort again demonstrated significantly higher survival (94% vs 89% in FDA, P < 0.001). At year 10, survival declined further in the FDA group (89%) compared with 93% in the PD cohort (P = 0.050). (Figure 4). Kaplan–Meier Survival Curve (CDR, Level 1)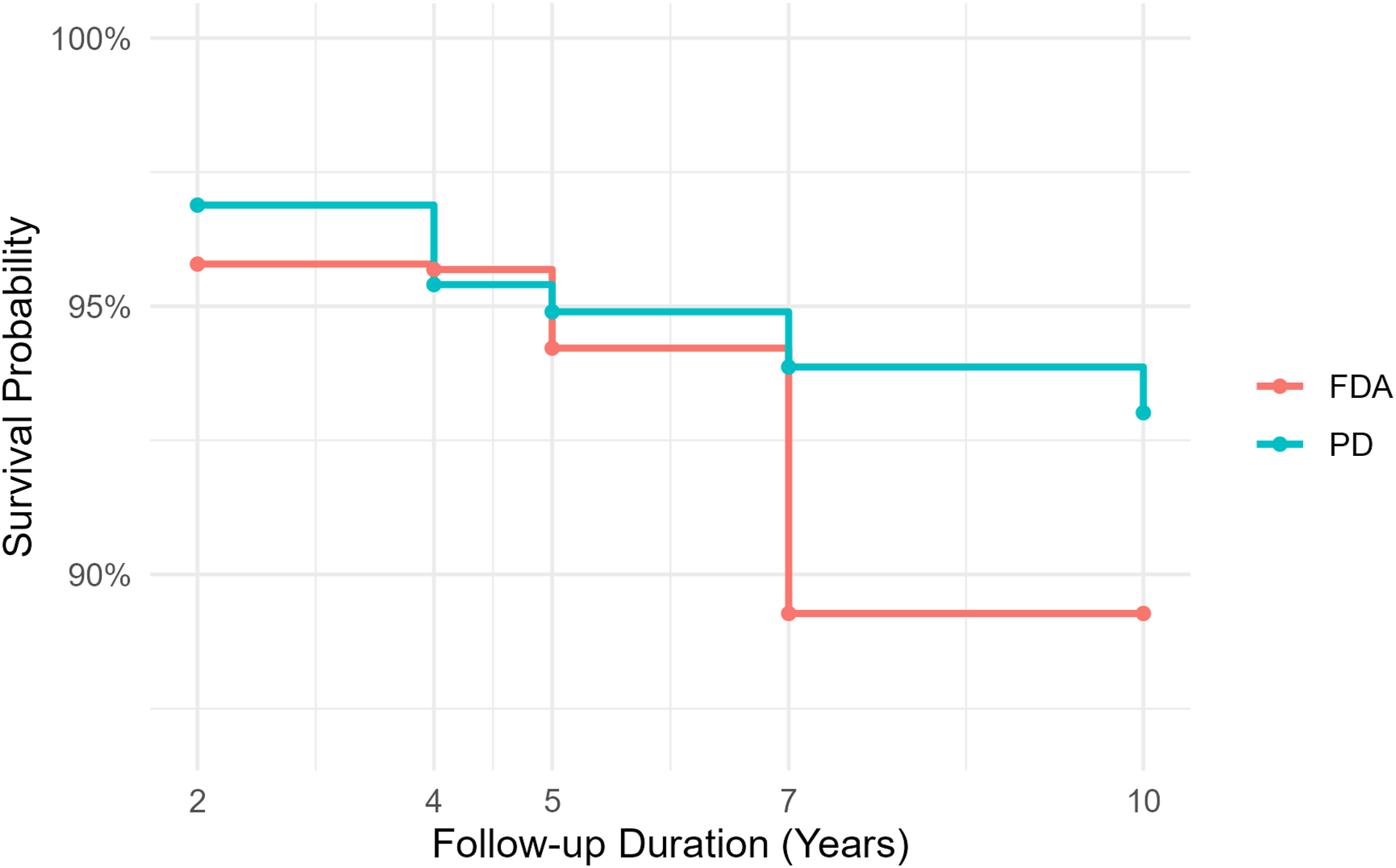
Level 2 CDR
In the Level 2 CDR cohort, survival probabilities remained high in both FDA IDE and PD groups throughout the follow-up period, with no statistically significant differences at any time point. At year 2, both groups demonstrated nearly identical survival (97%). By year 5, PD maintained survival at 95% compared to 93% in the FDA group (P = 0.093), and this modest difference persisted at year 7 (95% vs 92%, P = 0.211). At year 10, survival further declined in the FDA group (88%), while PD remained at 95%, though statistical testing was not available due to missing PD data. (Figure 5). Kaplan–Meier Survival Curve (CDR, Level 2)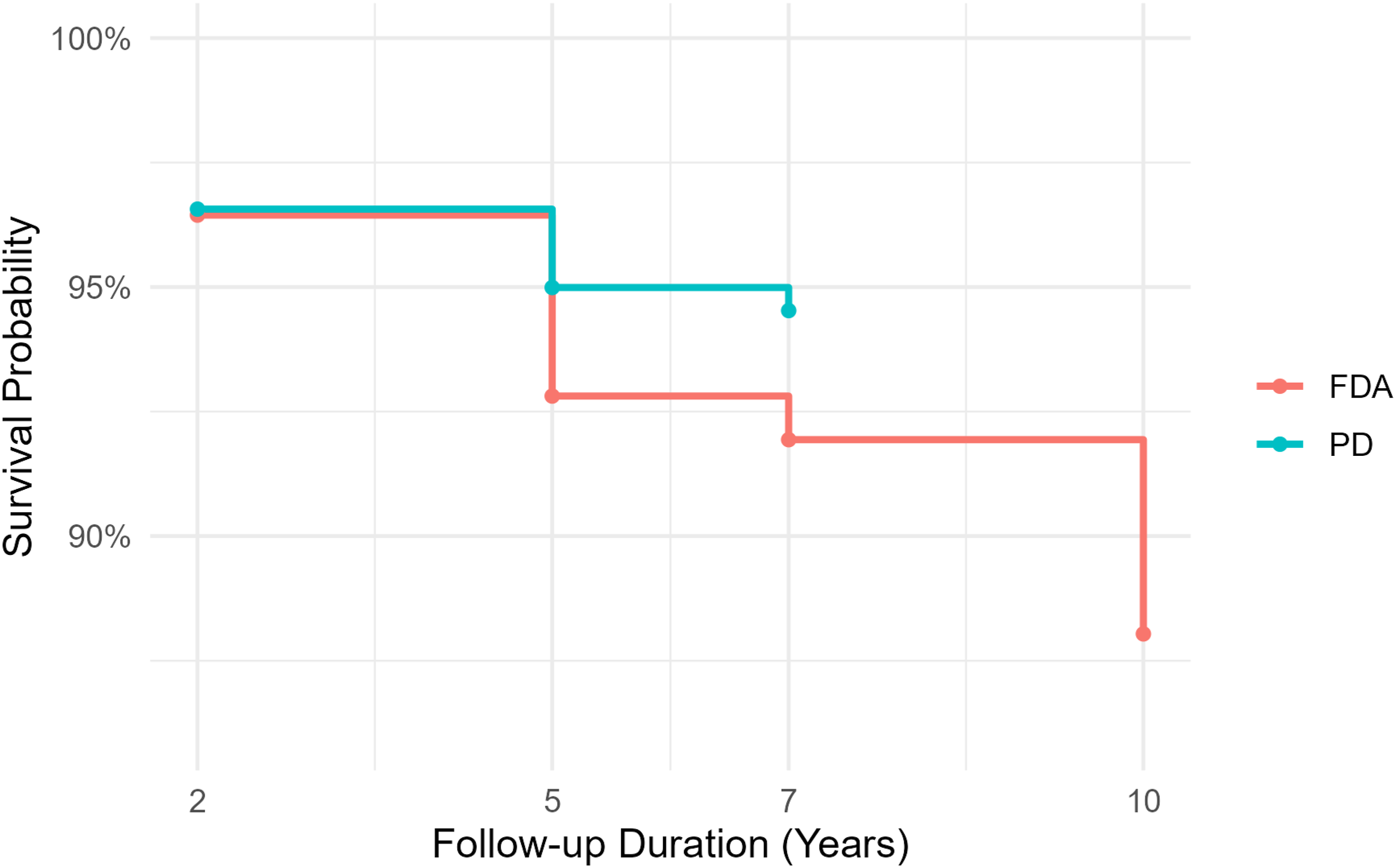
Discussion
Our study aimed to compare the long-term outcomes of CDR and ACDF using real-world data from the PD database and FDA IDE trials. The results highlight notable differences in the risk of subsequent surgeries between these 2 datasets for both procedures. In our analysis of the CDR group, real-world data from PD showed that the risks of subsequent surgeries were generally similar to those observed in controlled trials, but with some variations across different follow-up periods. For instance, at the 7-year follow-up, the subsequent surgery rate for CDR was 6.13% in PD data compared to 10.72% in FDA trials. However, only 1-level intervention at 2-, 7-, and 10-year timepoints was able to reach statistical significance on survival analysis and observed findings for 2-level intervention could be by chance. This suggests that while CDR performs similarly in real-world settings as in controlled trials, there may be minor differences in outcomes due to variations in patient populations or clinical practices. In contrast, the ACDF group demonstrated a significant discrepancy between real-world and controlled trial settings. The risks of subsequent surgeries were substantially lower in PD data compared to FDA trials across all follow-up periods on survival analysis. For example, at the 7-year follow-up, the subsequent surgery rate for ACDF was 7.24% in PD data vs 18.04% in FDA trials (HR: 0.4, P < 0.001). This indicates that ACDF outcomes in real-world settings may be more favorable than those reported in controlled trials, possibly due to better patient selection or improved surgical techniques not captured in older IDE trials. Additionally, postoperative fusion assessment in the FDA trials was performed by radiologists and included patient-reported outcome measures, reflecting greater scrutiny than typical clinical practice and likely contributing to the observed disparity in findings.
A systematic review by Toci et al underscored the advantages of CDR in reducing ASD and enhancing clinical outcomes, findings further supported by large-scale epidemiological evidence from Singh et al.8,69 In their cohort analysis of 433 660 patients, CDR was associated with significantly reduced rates of anterior revision (3.35% vs 4.96%, P < .001) and posterior fusion (0.79% vs 2.14%, P < .001) relative to ACDF. However, our study uniquely extends these insights by demonstrating that such benefits are also evident in real-world settings, albeit with more modest effect sizes when compared to those observed in controlled trials. This discrepancy may stem from differences in patient demographics, surgical indications, or institutional practices inherent to real-world data as opposed to the tightly controlled environments of IDE trials. Notably, it is plausible that patients presenting with more complex pathology were preferentially selected for ACDF, potentially introducing baseline disparities that attenuate the observable benefit of CDR over ACDF in controlled trials.
Tuchman et al, in a retrospective comparison of single-level CDR and ACDF, reported no significant difference in reoperation rates. 70 Similarly, Kelly et al. noted that while CDR reduced immediate postoperative readmissions and the need for early revision surgeries, it did not offer significant advantages over ACDF in terms of long-term complication rates. 71 However, their study was limited by its reliance on a state-specific nonfederal database that excluded revisions occurring in federal institutions, potentially underestimating the true complication burden.
Further reinforcing this point, Paek et al, in a 10-year retrospective analysis using the Statewide Planning and Research Cooperative System (SPARCS) database, evaluated outcomes for patients undergoing single-level CDR (n = 835) and ACDF (n = 6615). 72 They found no significant difference in revision surgery risk between the 2 cohorts. Nevertheless, SPARCS is limited to data from New York State and may not capture patients who underwent revisions outside the state, thereby restricting the generalizability of the findings.
Importantly, our study addresses several of these limitations through the use of a nationally representative dataset with a broader capture of subsequent procedures, enhancing the external validity of our conclusions. By leveraging real-world evidence on a large scale, we provide a comprehensive and clinically relevant assessment of CDR and ACDF outcomes that complements and contextualizes existing trial data. Our study contributes to the existing literature by demonstrating that while the effectiveness of CDR observed in controlled trials is largely supported in real-world settings, ACDF outcomes appear even more favourable in practice than previously reported in trials. The FDA’s post-approval studies (PAS) program highlights the need for continued long-term follow-up to ensure the safety and efficacy of these devices. 73 This validation is essential for guiding clinical decision-making, ensuring the long-term safety and efficacy of both procedures and helping clinicians select the most appropriate treatment based on individual patient needs and anatomical considerations.
Strength and Limitations
Although this study provides meaningful findings, several limitations should be acknowledged. First, the use of insurance claims data may introduce bias due to potential inaccuracies in coding and inconsistencies in patient follow-up. Additionally, the database only offers aggregate-level information, which limits the ability to assess detailed clinical factors such as spinal alignment before and after surgery. Furthermore, the constraints of CPT coding and the structure of the database restrict the ability to fully understand the indications for surgery, making it challenging to identify whether follow-up procedures were related to adjacent or index-level issues. Another important limitation is the inability to differentiate between devices on PD, as the database lacks specific CPT or ICD-9/10 codes for individual devices, which may obscure device-specific outcomes and trends. Finally, while this study compares FDA IDE trial results with real-world outcomes, we did not adjust for baseline population differences between the more narrowly selected trial participants and the broader, more heterogeneous real-world cohort. Nevertheless, the study highlights the value of real-world evidence in assessing surgical outcomes. Importantly, the use of the large PD dataset enhances the statistical strength of the analysis, allowing for meaningful comparisons between CDR and ACDF. By examining real-world data alongside results from IDE trials, this research offers a broader perspective on the performance of these procedures in both clinical and everyday practice settings.
Conclusion
In conclusion, our study highlights that while CDR performs similarly in real-world settings as in controlled trials for 1-level intervention, ACDF outcomes are more favourable in real-world settings compared to controlled trials. The subsequent surgery risks for ACDF were significantly lower in real-world data compared to FDA trials, suggesting improved outcomes in diverse patient populations. This validation is essential for guiding clinical decisions and ensuring the long-term safety and efficacy of these surgical procedures. Our findings support the continued use of CDR as a viable alternative to ACDF for select patients, however, further research exploring the difference in rate of index and adjacent segment surgeries in the real world is needed to understand the factors contributing to these differences and to optimize patient selection for each procedure. Ultimately, this study underscores the importance of real-world data in complementing controlled trials to provide a comprehensive understanding of surgical outcomes.
Supplemental Material
Supplemental Material - Long-Term Outcomes of Cervical Disc Replacement and Anterior Cervical Discectomy and Fusion: A Review of FDA IDE Trials With Real-World Comparison
Supplemental Material for Long-Term Outcomes of Cervical Disc Replacement and Anterior Cervical Discectomy and Fusion: A Review of FDA IDE Trials With Real-World Comparison by Ali Issani, William J. Karakash, Henry Avetisian, Zeeshan Ahsan, Dil Patel, David Riopelle, Kevin Kashanchi, Matthew Gallo, Raymond J. Hah, Amyn Malik, Ram K. Alluri, John C. Liu, Jeffrey C. Wang in Global Spine Journal
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ali Issani, William J. Karakash, Henry Avetisian, Zeeshan Ahsan, Dil Patel, David Riopelle, Kevin Kashanchi and Matthew Gallo have nothing to disclose. Jeffrey C. Wang has received intellectual property royalties from Zimmer Biomet, NovApproach, SeaSpine, and DePuy Synthes, and stock options from Bone Biologics, Electrocore, PearlDiver, and Surgitech. Raymond J. Hah has received grant funding from SI bone, consulting fees from NuVasive, and support from the North American Spine Society to attend meetings. Amyn Malik was employed by Analysis Group, Inc as a consultant between 2022 and 2024. Ram K. Alluri has received grant funding from NIH, consulting fees and stock options from HIA Technologies, and payment from Eccential Robotics for lectures and presentations. John C. Liu has received consulting fees from vision, illuminant surgical, Nov approach and Spark spine, and support for attending meetings and/or travel from Global spine congress and NASS summer meeting.
Data Availability Statement
Data is not publicly available but can be available upon request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
