Abstract
Study Design
Prospective Cohort Study.
Objectives
The aim of this study was to assess how sound pressure levels (SPL) change throughout spine procedures and which phases may place surgeons at higher risk of noise induced hearing loss.
Methods
SPL was recorded by a microphone attached to the surgeon’s collar beneath the sterile gown for 6 of each: MI-TLIF, microdiscectomy, decompression. dBA was recorded every second. An overall average (ADL) and maximum (MDL) for each phase of the procedure were calculated and compared across procedure type. The ADL and MDL of two electric burrs and one pneumatic burr were collected and compared.
Results
ADLs were significantly different for all three procedure types. There was no difference in the mean MDL (mMDL) between phases within each procedure type. The irrigation/closure phase had the lowest ADL and mMDL for all three procedure types. In MI-TLIF, facetectomy had a significantly greater ADL than all other phases; the highest mMDL was during the exposure phase. In microdiscectomy, laminotomy had the greatest mMDL, and ADL and was significantly higher than all other phases except exposure. In decompression, the laminectomy phase had the highest ADL and mMDL. The brand A-e burr had a significantly lower ADL and the lowest MDL.
Conclusion
During common spine surgery procedures, phases involving powered instruments such as burrs, electrocautery machines or suction had higher ADL and mMDL. Sound levels in the OR may peak above 85 dBA for short periods despite the average being below dangerous levels. Therefore, while surgeons are not usually at an elevated risk of NIHL, periods of elevated dBA warrant attention for elevated risk.
Introduction
Noise-induced hearing loss (NIHL) is a bilateral, sensorineural condition caused by prolonged exposure to hazardous sound levels and affects over 22 million U.S. workers annually.1,2 It is the most common occupational illness and the second leading cause of hearing loss worldwide, after presbycusis. 3 NIHL is associated with impaired speech perception, reduced quality of life, and increased risk of social isolation, depression, and cognitive decline. 3 Occupational sound exposure is measured as a sound pressure level (SPL) which is typically recorded in A-weighted decibels (dBA), with the National Institute for Occupational Safety and Health (NIOSH) recommending a maximum of 85 dBA over an 8-hour time-weighted average (TWA).1,4 However, noise levels below 85 dBA may still contribute to hearing loss, particularly in susceptible individuals. 4
Although NIHL has been well characterized in industrial and military settings, its impact in healthcare environments has only recently gained attention. Longitudinal data shows that hospital staff exposed to noise levels above 65 dBA over 6 months experience significant shifts in hearing thresholds, underscoring the cumulative risk of NIHL in clinical settings. 5 Operating rooms (ORs) are dynamic and high-noise environments due to powered instruments, alarms, and team communication, as commonly used tools such as suction and dermatomes have been shown to produce average sound levels exceeding the 85 dBA threshold. 6 In orthopedic surgery —where high-speed burrs, drills, saws, and mallets are routinely used—noise exposure is particularly pronounced. One review found that 76% of orthopedic procedures and 70% of instruments exceeded acceptable A-weighted noise levels, with NIHL affecting more than half of staff in some cohorts. 7 Subspecialty analysis has further shown that 84% of spine procedures produce peak SPLs above 85 dBA, with spine surgeries carrying the second-highest projected daily noise dose. 8
However, these findings primarily reflect maximum SPLs recorded during largely open spine procedures, which—while emphasizing the presence of intense acoustic peaks—do not characterize temporal fluctuations or average exposure across distinct procedural steps.8,9 Moreover, prior literature has largely excluded minimally invasive spine (MIS) surgery, a growing segment of spine care with unique workflows and tool usage patterns that may yield different auditory profiles.
The present study addresses these gaps by providing a detailed, phase-specific analysis of average intraoperative SPLs during MIS spine procedures. Using second-by-second sound monitoring throughout microdiscectomy, decompression, and minimally invasive transforaminal lumbar interbody fusion (MI-TLIF), this study identifies operative phases associated with elevated noise exposure. By characterizing the intraoperative noise profile of MIS spine surgery, the findings aim to inform targeted hearing protection strategies and refine risk assessment for MIS spine surgeons.
Methods
Study Design
List of Phases by Procedure Type

Intraoperative Recording Procedure and SPL Assessment
Intraoperative SPL recordings were collected using the AudioTools iOS (Apple) application using the SPL Graphing feature (Studio Six Digital) on an iPhone X or iPad Mini 4 (Apple) connected to an external iMM-6 calibrated measurement microphone (Dayton Audio), which was attached to the surgeon’s collar beneath the sterile gown. The SPL was recorded every second using an A-weighted decibel (dB) scale, which is most commonly used since it discriminates against lower frequencies, more closely reflecting the sensitivity of the human ear. 9 A separate observer noted the start and stop time of each phase, adding additional timepoints if the surgeon returned to a prior one. Intraoperative communication between the observer and surgeon was encouraged to ensure accurate recording of sound levels specific to each procedural phase. Following each recording, the raw data values were exported and separated by phase.
Burr Recording Procedure and SPL Assessment
To assess the SPL of the burr independently, three available burr types at our institution were compared: two electric burrs (brand A-e and brand B-e) and one pneumatic burr (brand A-p). All burrs were tested using the corresponding 3 mm matchstick burr tip. Using a cadaveric lumbar spine sample, each burr was used to perform as much of an open laminectomy as possible in 45 seconds without turning off the burr. SPL was collected every second using the same equipment and A-weighting as previously described and similarly with the microphone on the surgeon’s collar beneath a sterile gown.
Statistical Analysis
For the intraoperative phase assessment, for each procedure type, the data from each phase of each recording was combined and the average decibel level (ADL) was obtained for each phase. The maximum decibel level (MDL) of each phase of each recording was collected. The mean MDL (mMDL) for each phase was calculated by averaging the phase-specific MDL values across all 6 cases within each procedure type. For the burr assessment, the ADL and MDL were obtained for each burr tested. Data is represented as mean ± standard deviation in dBA unless otherwise noted. ANOVA with post-hoc tests using Bonferroni correction was performed for all analyses. P values of P < .05 were considered significant. Statistical analysis was performed using SPSS v29 (IBM, Inc., Armonk, NY, USA).
Results
Intraoperative SPL Assessment
Mean and Mean Maximum SPL by Procedure Phase
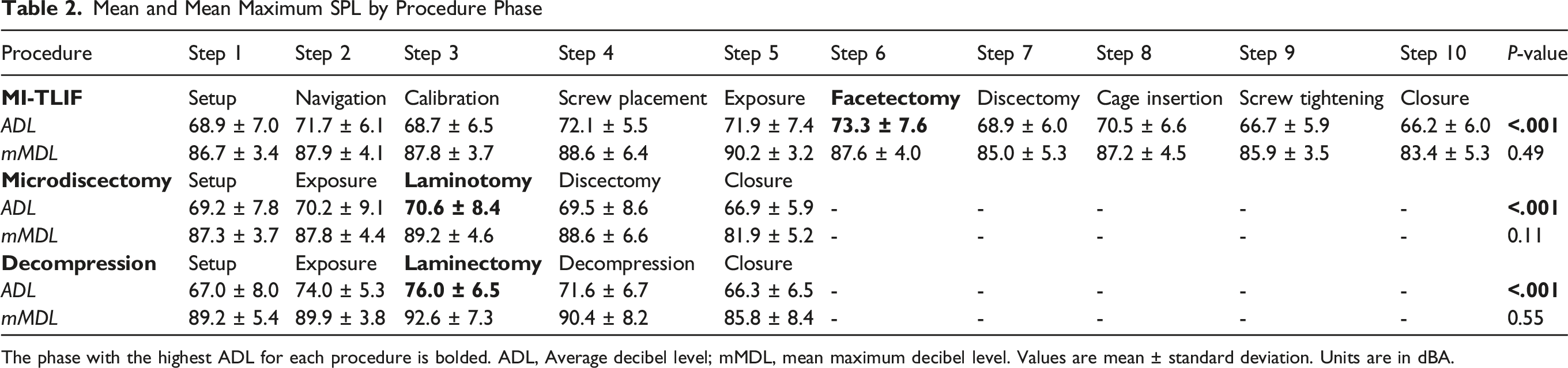
The phase with the highest ADL for each procedure is bolded. ADL, Average decibel level; mMDL, mean maximum decibel level. Values are mean ± standard deviation. Units are in dBA.
For MI-TLIF, facetectomy had a significantly greater ADL than all other steps (73.3 ± 7.6 dBA, P < .001) (Supplemental Table 1). Although differences were not significant on pairwise comparison, posterior screw instrumentation (72.1 ± 5.5 dBA) had the second highest ADL, followed by foraminal exposure (71.9 ± 7.4 dBA) and navigation array placement (71.7 ± 6.1 dBA). The next loudest ADL were cage insertion (70.5 ± 6.6 dBA), discectomy (68.9 ± 6.0 dBA), setup (68.9 ± 7.0 dBA), calibration and surgical planning (68.7 ± 6.5 dBA), final screw tightening (66.7 ± 5.9 dBA), and lastly, closure (66.2 ± 6.0 dBA) (Figure 1, Table 2). The highest mMDL was for foraminal exposure (90.2 ± 3.2 dBA) (Table 2), although differences were not significant between phases. Mean differences and pairwise significance of ADLs is illustrated in Supplemental Table 1. Mean SPL by MI-TLIF Phase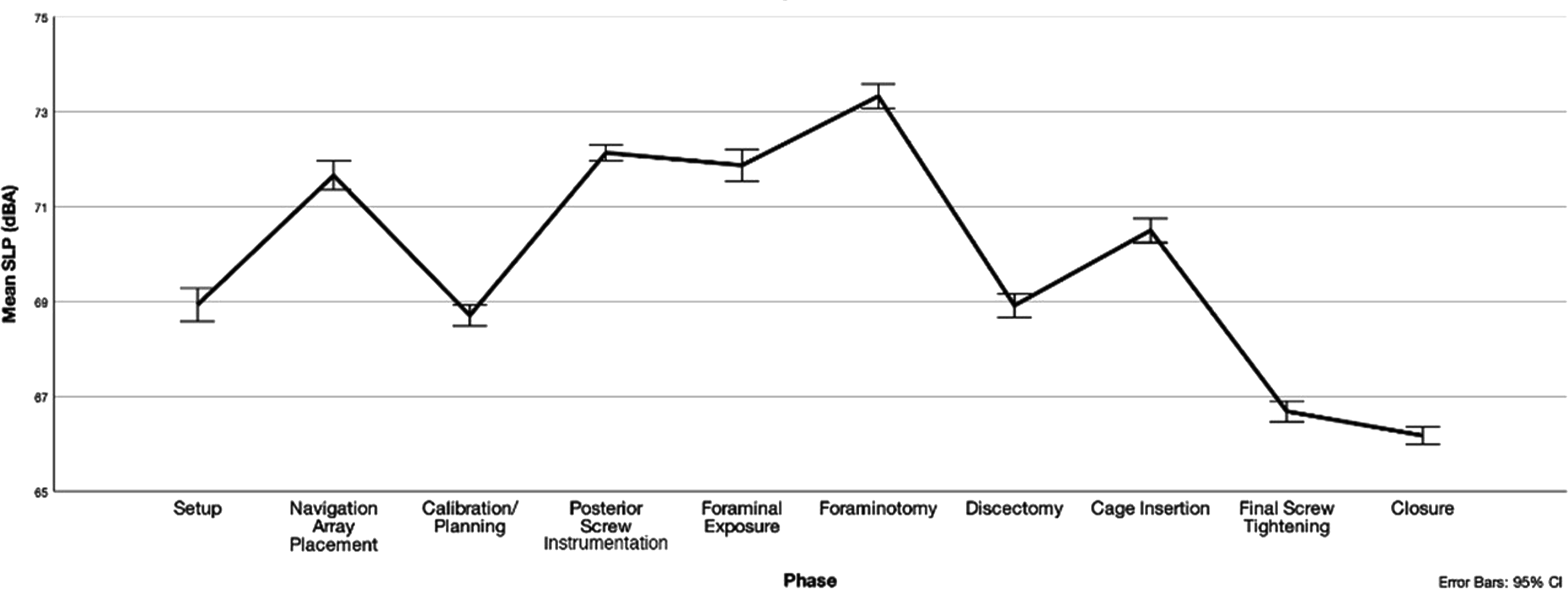
In microdiscectomy, laminotomy had the greatest ADL (70.6 ± 8.4 dBA) and was significantly higher than all other steps except exposure (70.2 ± 9.1 dBA, P = 1.0) on pairwise comparison (Supplemental Table 2). Discectomy (69.5 ± 8.6 dBA) was third loudest, followed by setup (69.2 ± 7.8 dBA) and closure (66.9 ± 5.9 dBA) (Figure 2, Table 2). The highest mMDL was for laminotomy (89.2 ± 4.6 dBA), although differences were not significant between phases (Table 2). Mean differences and pairwise significance of ADLs is illustrated in Supplemental Table 2. Mean SPL by Microdiscectomy Phase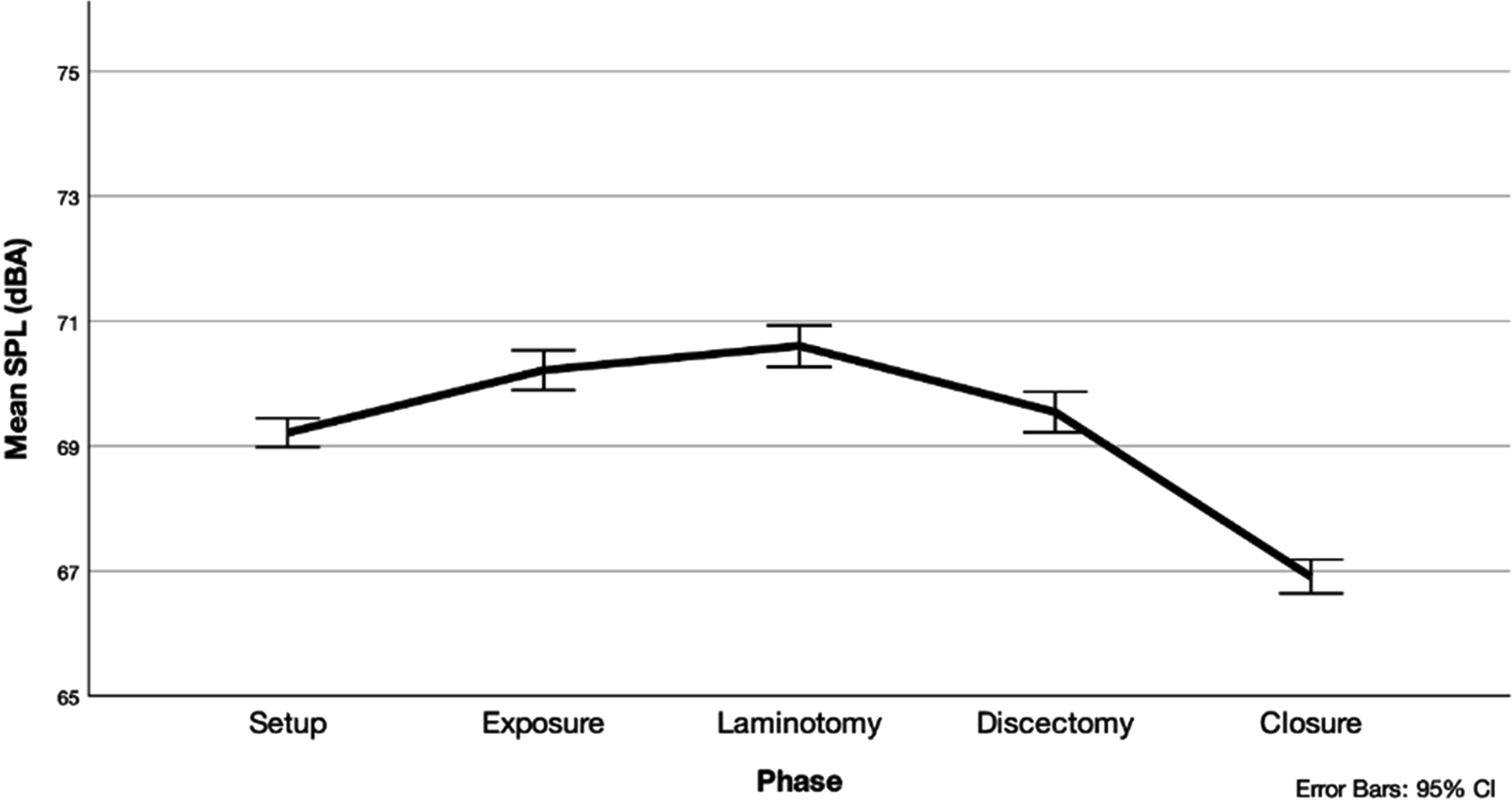
For decompression procedures, all phases had significantly different noise levels from one another (P < .001) with the highest ADL being the laminectomy phase (76.0 ± 6.5 dBA). Although differences were not significant between phases, laminectomy phase also had the highest mMDL (92.6 ± 7.3 dBA). Following laminectomy, the next loudest phases were exposure (74.0 ± 5.3 dBA), decompression of ligamentum (71.6 ± 6.7 dBA), setup (67.0 ± 8.0 dBA), and closure (66.3 ± 6.5 dBA) (Figure 3, Table 2). Mean differences and pairwise significance of ADLs is illustrated in Supplemental Table 3. Mean SPL by Decompression Phase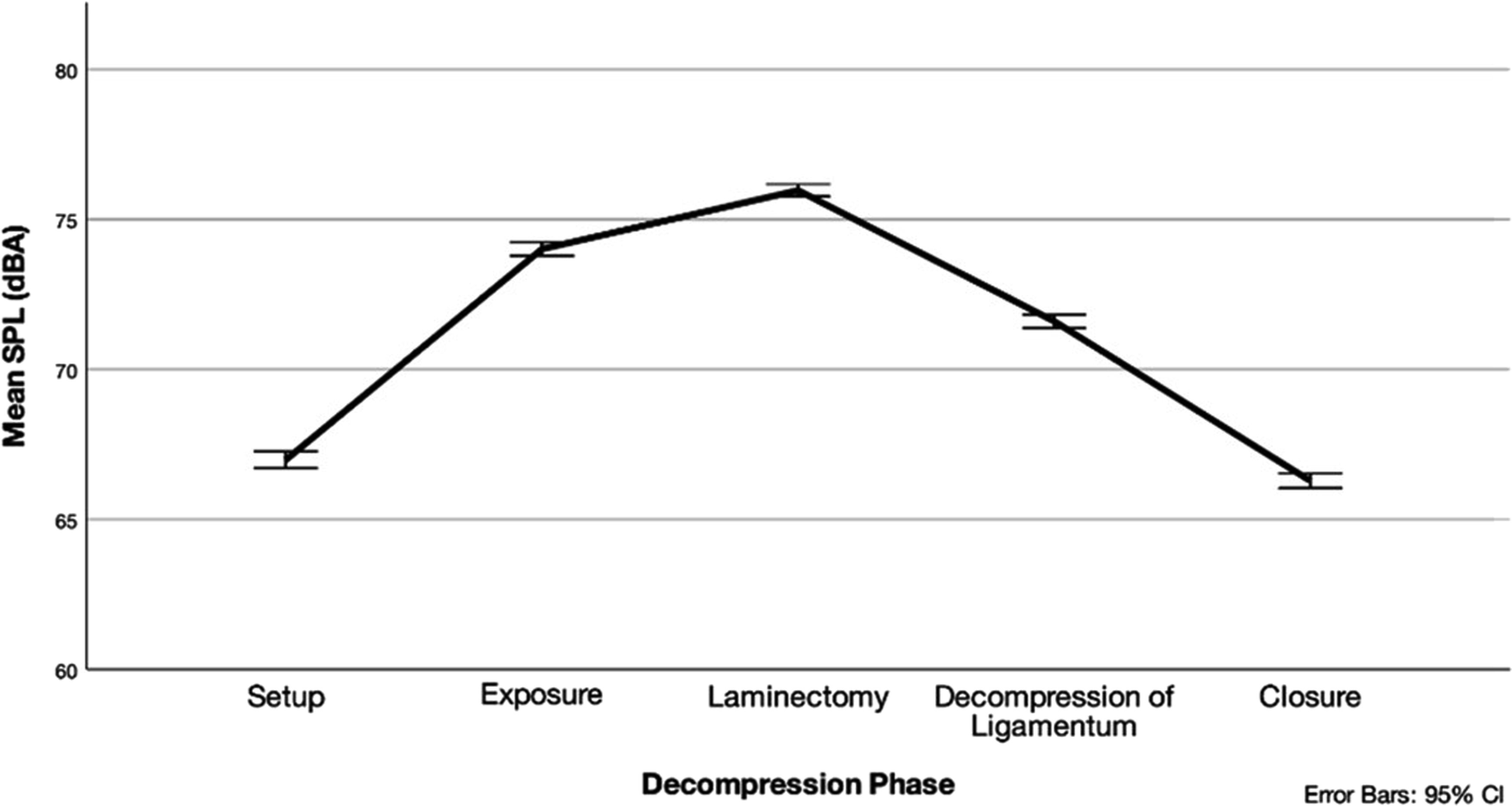
Burr SPL Assessment
Burr Mean and Maximum SPL Values

ADL, average decibel level; MDL, maximum decibel level.
Discussion
This study provides a granular, phase-specific analysis of intraoperative noise exposure during minimally invasive spine (MIS) procedures and demonstrates that average SPLs (ADLs) fluctuate significantly across surgical steps. We found that the highest ADLs were observed during instrument-intensive phases such as exposure, laminectomy, and pedicle screw insertion, all of which involve powered tools like electrocautery, burrs, or suction. While not significant, the highest average mMDLs were also observed during these phases. Importantly, we identified the exposure phase as a period of elevated ADL, most likely due to the sound emitted by the activation feedback tone of the electrocautery machine. The purpose of this tone is to allow the surgeon to know when the device is active, and the volume of the tone is controlled independently from the amount of power delivered to the device. This should be adjusted to a minimal level to protect against NIHL.
While the ADLs recorded in this study did not exceed the 85 dBA threshold recommended by the National Institute for Occupational Safety and Health (NIOSH), 4 they remain clinically relevant given the cumulative risks associated with long-term, repeated exposure.8-11 Prior studies have shown that even chronic exposure above 65 dBA can lead to progressive threshold shifts in the 3-6 kHz range—frequencies essential for speech perception and intraoperative communication5,12,13
Unlike prior studies that predominantly examined open spine procedures, our analysis focused exclusively on minimally invasive spine surgeries.8,9 MIS techniques typically involve smaller incisions, limited soft tissue disruption, and more controlled instrumentation, all of which may contribute to distinct noise exposure profiles not previously described in the literature.
Our findings contrast with earlier studies that reported maximum SPLs (MDL) exceeding 100 dBA during spine procedures as our results show the majority of our MDLs were below this value, both overall and by phase.8,9 MDL values reflect peak intensities at a singular instant rather than sustained exposure. There is an inherent variability in peak noise events, which, for example, could be a result of an anomalous event, such as equipment falling to the floor, which could have limited the ability of mMDL to represent the MDL of a phase. Future studies should utilize a larger sample size when comparing mMDLs across phases. When comparing ADLs, our results align more closely with prior literature: reported ADL values range from 73.6 ± 4.4 dBA 8 to 78.6 ± 6.2 dBA, 9 consistent with the average ADLs in our study, which ranged from 70.6 to 76.0 dBA depending on procedure type. This suggests that mean exposure levels during minimally invasive spine procedures are comparable to those reported across studies of predominantly open spine surgeries, despite differences in surgical approach and instrumentation.
Additionally, the surgeons in our study consistently used an electric burr (electric brand A), rather than pneumatic systems. Independent testing conducted as an adjunct to this study confirmed that the electric burr utilized by surgeons during the procedures in our study generated lower ADLs during typical usage (79.9 ± 2.4 dBA) compared to the pneumatic version of brand A (83.4 ± 1.7 dBA) and the electric brand B (82.5 ± 3.8 dBA). These objective measurements support the notion that both surgical approach and tool selection meaningfully influence intraoperative sound exposure and likely contributed to the lower SPLs observed in our cohort. Therefore, to decrease the risk of NIHL, surgeons should consider instrument brand and drive mechanism.
Risk is further compounded by inconsistencies in regulatory guidelines: while the Occupational Safety and Health Administration (OSHA) uses a 5 dB exchange rate, 14 NIOSH recommends a more conservative 3 dB exchange rate, effectively halving allowable exposure time with each 3 dB increase. 1 As such, even modest elevations in SPL can rapidly push cumulative exposure beyond safe limits. In high-volume surgical environments, where providers may perform multiple instrument-heavy procedures per day, adhering to NIOSH guidelines may offer a more stringent safeguard against long-term hearing loss. Though the peak SPLs (MDL) in this study were brief, their repeated occurrence across multiple cases highlights the potential need for effective, proactive hearing conservation strategies in MIS spine surgeons. Future studies should characterize the risk of these brief periods of elevated exposure such as their length and frequency.
Additionally, high-frequency sounds emitted by surgical burrs and suction devices, often falling within the 2-8 kHz range, can significantly contribute to auditory masking.12,15,16 This phenomenon occurs when loud background noise interferes with the perception of other sounds, especially those in similar frequency ranges. Given that 3-6 kHz is essential for speech discrimination, sustained exposure in this range can hinder intraoperative communication.3,13 In the OR environment, such communication breakdowns may compromise team coordination and patient safety.
Finally, the implications of OR noise extend beyond the surgical team. Anesthesia suppresses the stapedius reflex, a key defense against acoustic trauma, rendering patients more vulnerable to loud intraoperative sounds.17,18 Prior studies have associated elevated intraoperative noise with increased postoperative pain, anxiety, and analgesic use.19-21 As powered instruments become increasingly central to minimally invasive spine procedures, targeted mitigation strategies—such as quieter surgical tools, noise-dampening protocols, and improved awareness of intraoperative noise patterns—will be critical for protecting both providers and patients.
This study provides a phase-specific analysis of intraoperative noise exposure during MIS spine surgery, yet several limitations warrant discussion. First, although SPL was measured continuously, the study did not capture frequency spectra, limiting the ability to assess how specific frequency bands contribute to auditory masking or potential hearing loss. Auditory masking is a phenomenon where a loud sound makes a quieter one inaudible. This process is frequency dependent, such that a masking sound at the same frequency as a signal exhibits a greater amount of masking compared to ones at different frequencies. In the context of an OR, intraoperative communication is a signal while ambient and machine noise is a masker. A prior study on OR noise assessed spectral analyses of OR sounds and found machine noise to obscure surgeon instruction. 22 Second, while all cases used an electric burr, surgical instruments and room acoustics were not otherwise standardized, and variations in tool condition, OR layout, background music, and personnel positioning may have influenced SPL readings. Third, although the microphone was placed at the surgeon’s collar to approximate ear-level exposure, head orientation and use of PPE may have affected actual perceived noise. The adjunctive burr testing strengthens our interpretation of tool-specific acoustic differences, but further research is needed to evaluate how instrument selection interacts with surgical technique and OR acoustics. Future studies should include frequency-based measurements, include a larger sample size, and investigate the impact of intraoperative noise on team communication and cognitive load, and explore long-term hearing outcomes among spine surgeons to guide evidence-based protection strategies.
Conclusion
This study demonstrates that average intraoperative noise levels during minimally invasive spine procedures generally remain below harmful thresholds, with elevations above 85 dBA occurring only in short, transient bursts. Across all procedure types, operative phases involving powered instruments—such as burrs, electrocautery devices, and suction—produced the highest ADL and mMDLs. In contrast, setup and closure phases had the lowest ADL and mMDLs, consistent with limited use of powered instruments. These findings contrast with prior reports suggesting more sustained hazardous exposure, likely reflecting differences in surgical approach, instrumentation, and technique. Notably, exposure steps involving electrocautery produced some of the highest SPLs; therefore, modifying its volume in real time may offer a practical strategy to reduce intraoperative noise levels. Additionally, there was a difference in ADL between burr brands and drive mechanisms upon independent testing. Accordingly, targeted noise-reduction strategies—such as disabling suction when idle, minimizing sustained use of powered instruments, selection of quieter burr brands, and incorporating hearing protection—should be considered to mitigate long-term auditory risk. By identifying specific, controllable contributors to peak noise, these results support the adoption of simple, targeted interventions to further reduce intraoperative auditory burden.
Supplemental Material
Supplemental Material - How Loud is Minimally Invasive Spine Surgery? Identifying Intraoperative Risk of Hearing Loss
Supplemental Material for How Loud is Minimally Invasive Spine Surgery? Identifying Intraoperative Risk of Hearing Loss by Adin M. Ehrlich, Sophie Kush, Chad Z. Simon, Eric R. Zhao, Tomoyuki Asada, Maximilian K. Korsun, Stephane Owusu-Sarpong, Luis Felipe Colon, Farah Mushbarash, Quante Singleton, Gregory S. Kazarian, Sheeraz A. Qureshi, Sravisht Iyer in Global Spine Journal
Footnotes
ORCID iDs
Ethical Considerations
This research was approved by the HSS Institutional Review Board (IRB# 2018-0552).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sheeraz Qureshi has the following disclosures: AMOpportunities: Other financial or material support; Annals of Translational Medicine: Editorial or governing board; Association of Bone and Joint Surgeons: Board or committee member; Cervical Spine Research Society: Board or committee member; Contemporary Spine Surgery: Editorial or governing board; Globus Medical: IP royalties; Paid consultant; Paid presenter or speaker; Hospital Special Surgery Journal: Editorial or governing board; HS2, LLC: Stock or stock Options; International Society for the Advancement of Spine Surgery (ISASS) - Program Committee member: Board or committee member; Lifelink.com: Other financial or material support; Lumbar Spine Research Society: Board or committee member; Minimally Invasive Spine Study Group: Board or committee member; North American Spine Society: Board or committee member; Simplify Medical, Inc.: Other financial or material support; Society of Minimally Invasive Spine Surgery (SMISS) - Program Committee member: Board or committee member; Spinal Simplicity: Other financial or material support; SpineGuard, Inc.: Paid consultant; Stryker: IP royalties; Paid consultant; Surgalign: Paid consultant; Tissue Differentiation Intelligence: Stock or stock Options; Viseon, Inc.: Paid consultant; Research support; Sravisht Iyer has the following disclosures: Globus Medical: Paid presenter or speaker; Stryker: Paid presenter or speaker; Vertebral Columns/International Society for the Advancement of Spine Surgery (ISASS): Editorial or governing board.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
