Abstract
Study Design
Retrospective cohort study.
Objective
Frailty is defined as a state of minimal “physiologic reserve.” The modified 5 factor frailty index (mFI-5) is a recently proposed metric for assessing frailty and has been previously studied as a predictor of morbidity and mortality. However, its utility in predicting postoperative patient reported outcomes (PROMs) and return to activities remains understudied.
Methods
This is a retrospective cohort study of patients undergoing 1 or 2 level minimally invasive transforaminal lumbar interbody fusion queried from a prospectively maintained multi-surgeon registry. Comorbidities including hypertension, congestive heart failure, diabetes, chronic obstructive pulmonary disease, and functional status were utilized to calculate the mFI-5. Patients were separated into 3 cohorts, “non-frail,” “moderately frail” or “severely frail” based on their mFI-5. Patient outcomes were compared between the cohorts.
Results
392 patients were included of which 227 patients were non-frail, 134 were moderately frail, and 31 were severely frail. Frail patients had longer operative time (P = 0.002), greater estimated blood loss (P = 0.038). Despite similar preoperative PROMs, at a longer term follow up, frail patients also had worse PROM scores for ODI (P = 0.009), VAS-back (P = 0.028), and VAS-leg (P = 0.004). Frail patients had worse preoperative (P = 0.017) and postoperative (P < 0.001) SF-12 PCS. At 1-year, frail patients had lower rates of fusion (P = 0.002). Frail patients also demonstrated lower rates of returning to work (P = 0.018), returning to driving (P = 0.027), and discontinuation of narcotics (P = 0.004).
Conclusion
Frail patients as measured by the mFI-5 index demonstrated worse postoperative outcomes following 1 or 2 level MI-TLIF. Careful patient selection and preoperative optimization may be especially important in frail patients.
Introduction
As the demand for surgical treatment of degenerative lumbar spinal disease continues to grow, 1 it becomes increasingly important to understand which patients will benefit the most and which patients may experience suboptimal outcomes.
Frailty is one such patient specific factor that has the potential to influence how patients improve after spine surgery. Frailty represents a state of minimal “physiologic reserve” or the decreased ability of a patient to rebound after significant stressors. 2 Specifically, since first described in 2018, the 5-item Modified Frailty Index (mFI-5) has been extensively studied within the orthopedic and spine surgery literature as a prognostic indicator for morbidity and mortality following surgery.3-6 These studies were largely conducted utilizing large national databases such as the American College of Surgeons National Surgical Quality Improvement Program database. While these databases can be highly valuable for discerning overarching trends in morbidity and mortality, especially when these complication rates are low, they do not capture long-term outcomes.
Very few studies have analyzed the impact of frailty on patient reported outcomes measures (PROMs) following minimally invasive transforaminal lumbar interbody fusion (MI-TLIF). Furthermore, prior studies have all utilized the mFI-11 index, while the mFI-5 may be more applicable clinically and for research purposes.2,3 In this context, the relationship between mFI-5 and long-term PROMs and recovery kinetics deserves investigation. The purpose of the present study is to provide more granular data on longer term patient outcomes in frail patients defined with the mFI-5 index following one- or two-level MI-TLIF for the treatment of degenerative lumbar disorders. Specifically, we aim to 1) study the relationship between PROMs and mFI-5 and 2) evaluate whether mFI-5 influences recovery kinetics.
Methods
Study Design and Patient Cohort
This was a retrospective cohort study conducted following approval by the Institutional Review Board. Patients who underwent primary one or two-level MI-TLIF for the treatment of degenerative conditions of the lumbar spine between April 2017 and August 2023 with a minimum follow up of 6 months were included. The data was derived from a single-center multi-surgeon prospectively maintained database. MI-TLIF was performed as previously described in the literature.7,8 Patients less than 18 years old, revision surgeries, or those with a surgical indication of trauma were excluded.
Data Collection
Data was collected and managed using REDCap (Research Electronic Data Capture)9,10 hosted at Weill Cornell Medicine Clinical and Translational Science Center supported by the National Center For Advancing Translational Science of the National Institute of Health under award number: UL1 TR002384. Following data was collected and analyzed: 1. Demographics: age, gender, race, insurance type, body mass index (BMI), age adjusted Charlson Comorbidity Index (CCI), American Society of Anesthesiologists (ASA) class were all recorded. Past medical history collected additionally included hypertension (HTN) requiring medication, diabetes mellitus (DM), chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), renal disease, tumor (both benign or malignant), autoimmune disease, and functional status (independent, partially dependent, or fully dependent. 2. PROMs: Oswestry Disability Index (ODI) Visual Analog Scale, (VAS) back and leg, 12-Item Short Form Survey Physical Component Score (SF-12 PCS), and global rating change (GRC) were collected at 2 weeks, 6 weeks, 12 weeks, 6 months, 1 year, and 2 years after the surgery. GRC is a single question “Compared to preoperative, you feel 1) much better, 2) somewhat better, 3) about the same, 4) somewhat worse, or 5) much worse?).” Responses of “much better” and “somewhat better” were defined as “feeling better” while “much worse” and “somewhat worse” were defined as “feeling worse.” Two postoperative timepoints - early (<6 months) and late (>6 months) - were defined for analyses.11,12 3. Surgical details and outcomes: Estimated blood loss (EBL), operative time, number of levels operated, and specific operated levels were recorded. Postoperative length of stay (LOS) was also collected. Evidence of fusion was assessed on the 1-year CT scan as part of our institution’s patient standard of care. Intraoperative and postoperative complications as well as all reoperations through 2 years postoperatively when available were collected. 4. Return to activities: patients completed a return to activities survey as part of their standard of care. Patients who were driving and working preoperatively were asked if they had begun driving and working again at every follow-up visit until they returned to that activity. Patients who took opioids postoperatively were asked the date of cessation if applicable.
5-item Modified Frailty Index (mFI-5)
The 5-item modified frailty index is a validated measure of frailty and has been shown to be a reliable predictor of complications and mortality in the spine literature and across various surgical subspecialties. The index accounts for 5 comorbidities in patient medical history - hypertension requiring medication, history of chronic obstructive pulmonary disease, history of congestive heart failure, diabetes mellitus, and functional health status. For the calculation of frailty index, patients received 1 point on the mFI-5 index for each comorbidity present preoperatively (HTN, DM, COPD, CHF) and 1 point if their functional status was either partially or fully dependent. For the purpose of this analysis, patients were then separated into 3 cohorts based on their mFI-5 index: 1) 0 comorbidities (non-frail), 2) 1 comorbidity (moderate frailty), or 3)
PROMs and Statistical Analysis
VAS-back, VAS-leg, ODI, and SF-12 PCS were assessed for improvement. Changes in PROMs from their preoperative baseline were assessed at both the early and late timepoints. Additionally, the percentage of patients achieving patient acceptable symptom state (PASS) at the late time point and minimal clinically important difference (MCID) at both the early and late time points were calculated. PASS achievement was defined as an absolute postoperative ODI <25.2 as previously defined by Shahi et al. 13 MCID achievement was defined as a decrease in ODI by 12.8, VAS-back by 1.2, VAS-leg by 1.6, and SF-12 PCS by 4.1 as previously defined by Copay et al. 14 Since preoperative baseline affects the utility of PASS and MCID in predicting patient improvement,15-17 patients with preoperative ODI<25 were appropriately excluded from PASS achievement calculations. Patients with ODI<15, VAS-back/leg<2.5, and SF-12 PCS>92.1 were similarly excluded from MCID achievement calculations.
Statistical analyses were conducted to understand differences between the 3 defined groups. Normally distributed continuous variables between groups were compared with a one-way Analysis of Variance (ANOVA) or 2-tailed independent sample t test when appropriate. Non-normally distributed variables were compared between groups with a Kruskal-Wallis test or a Mann-Whitney U test as appropriate. Continuous variables were reported as mean ± standard deviation (SD) or as median [interquartile range (IQR)] when non-normally distributed. Categorical variables were compared between groups with a χ2 test or a fisher exact test when the number of subjects was less than 10. Categorical variables are reported as sample size (%). Statistical significance was taken at P value r <0.05. For comparisons between 3 groups that were significant, a post hoc analysis between each group was conducted to determine specific intergroup differences. Analyses were performed using the IBM Statistical Package for the Social Sciences (SPSS) version 25 (IBM Corp., Armonk, NY).
Results
Patient Demographics. Superscripts a, b Indicates Post-hoc Significance Between Groups.
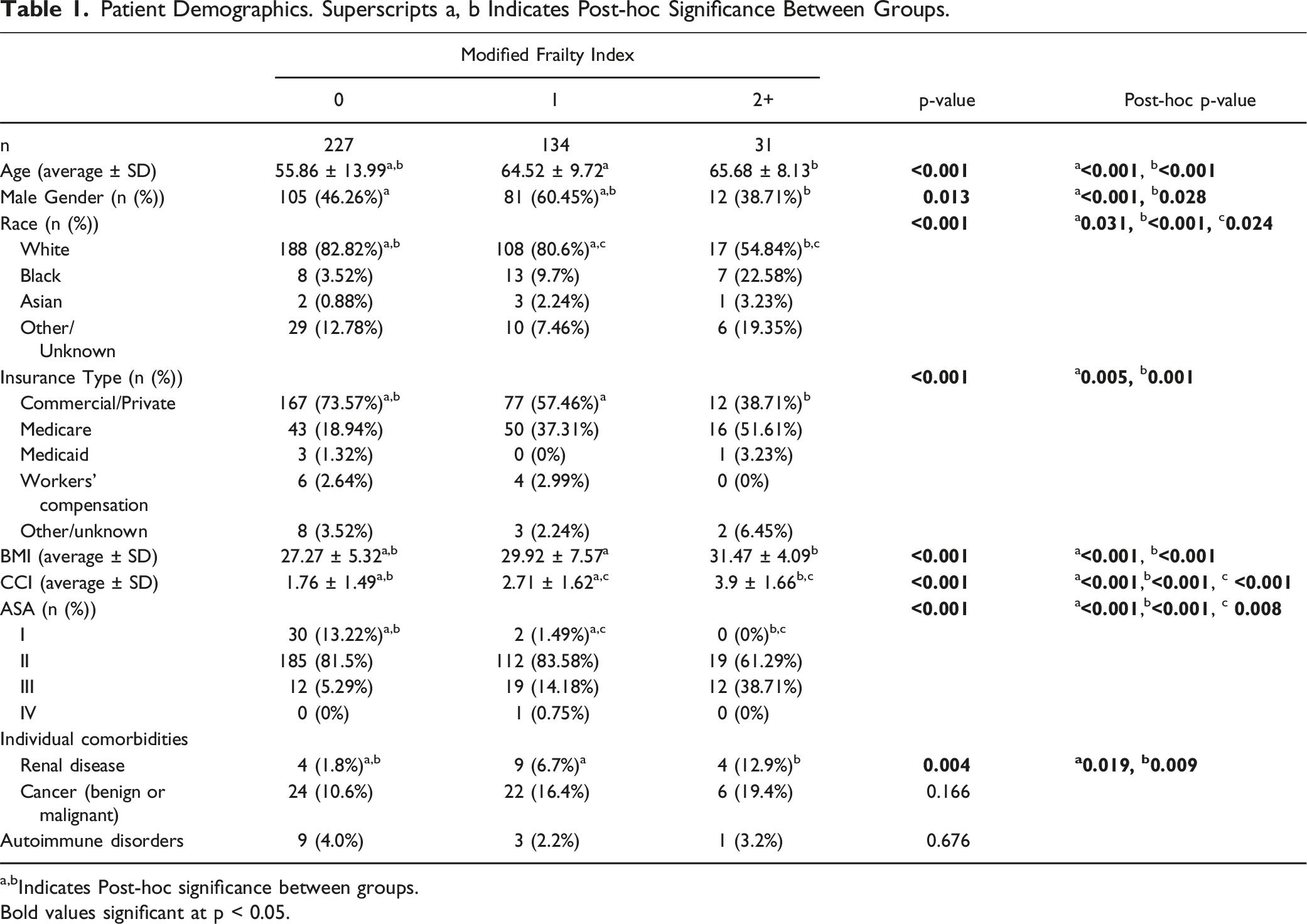
a,bIndicates Post‐hoc significance between groups.
Bold values significant at p < 0.05.
Surgical Details. Superscripts a, b Indicates Post-hoc Significance Between Groups.
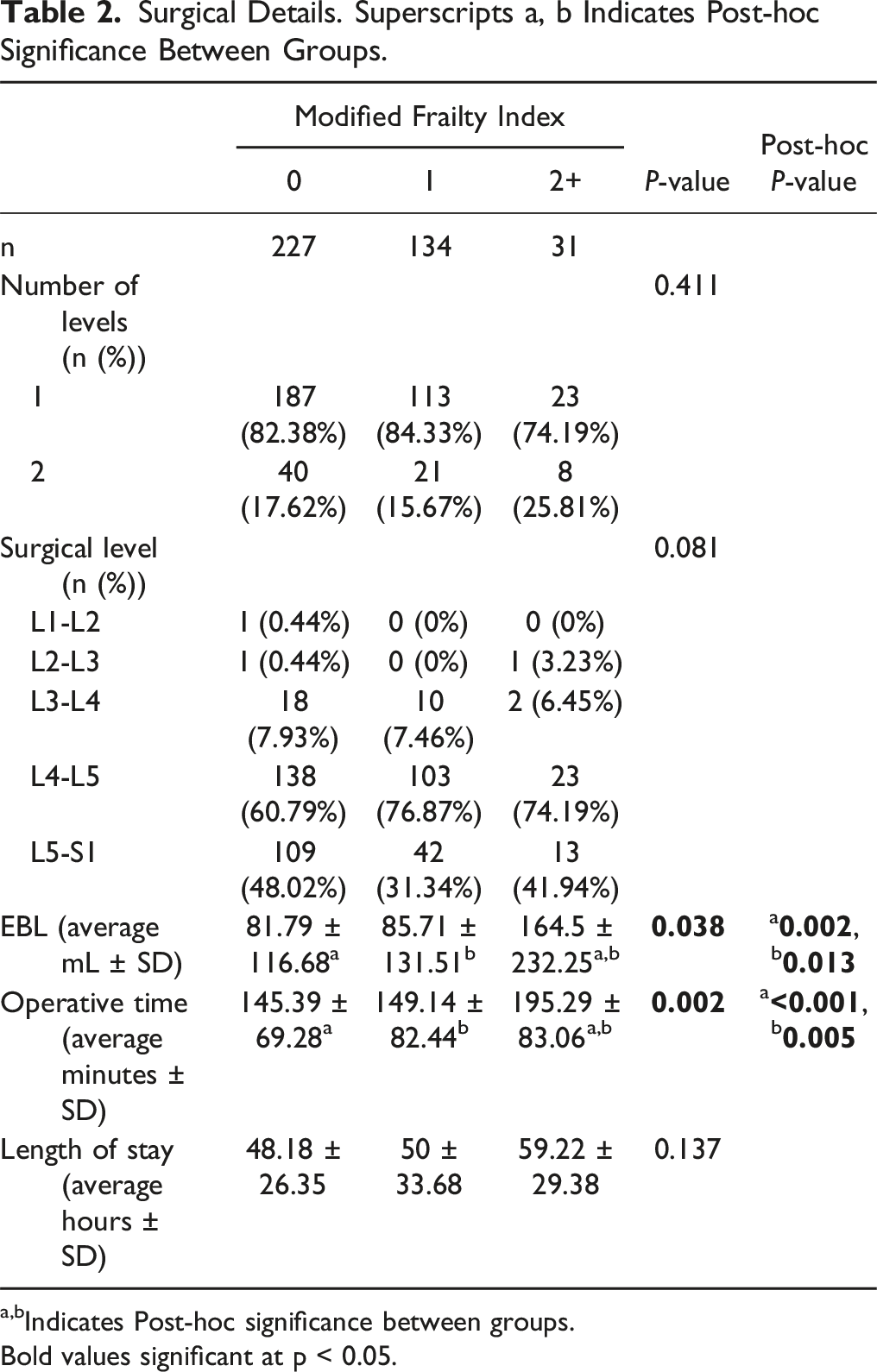
a,bIndicates Post-hoc significance between groups.
Bold values significant at p < 0.05.
PROMs Comparison. Superscripts a, b, c Indicates Post-hoc Significance Between Groups.
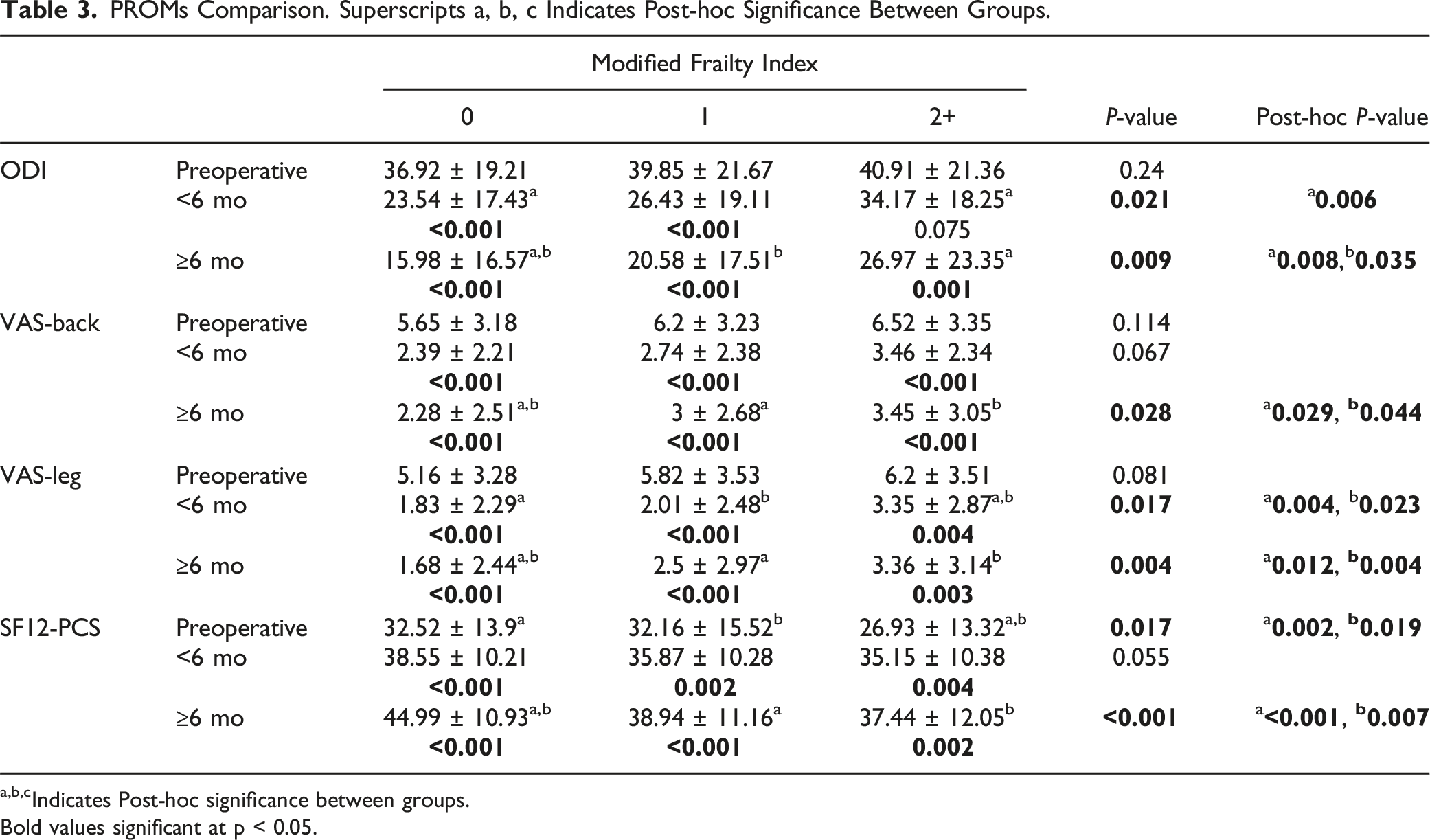
a,b,cIndicates Post-hoc significance between groups.
Bold values significant at p < 0.05.
Comparison of Magnitude of Improvement. Superscript a Indicates Post-hoc Significance Between Groups.
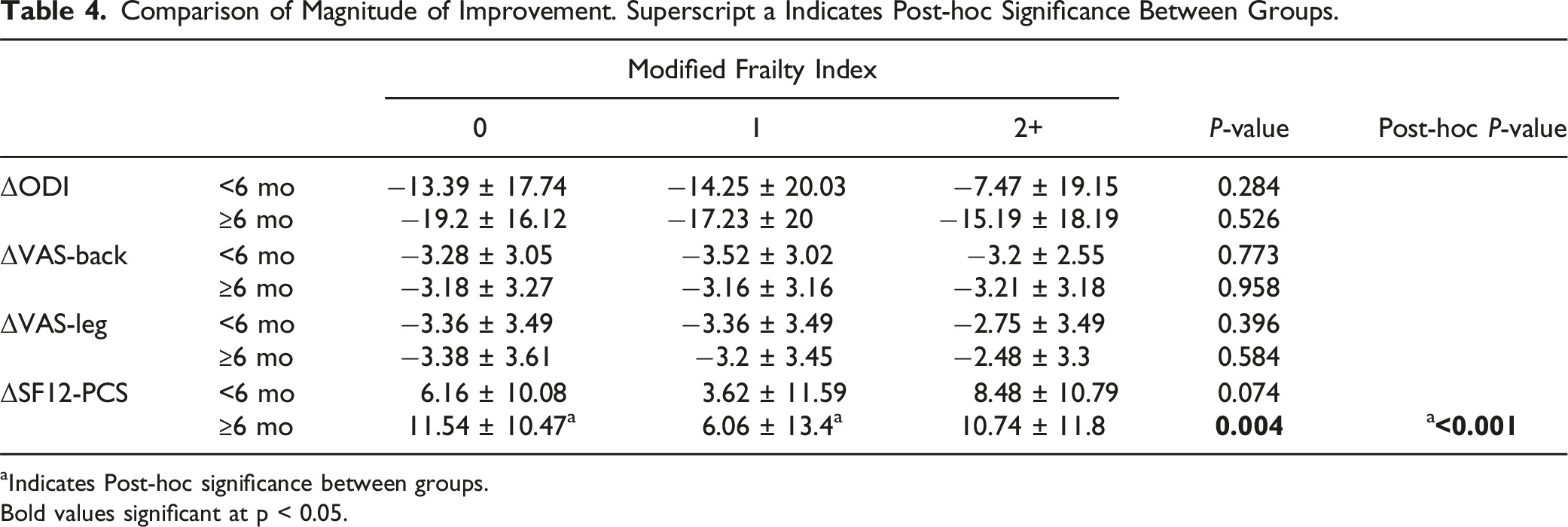
aIndicates Post-hoc significance between groups.
Bold values significant at p < 0.05.
MCID, PASS, GRC Achievement. Superscript a Indicates Post-hoc Significance Between Groups.
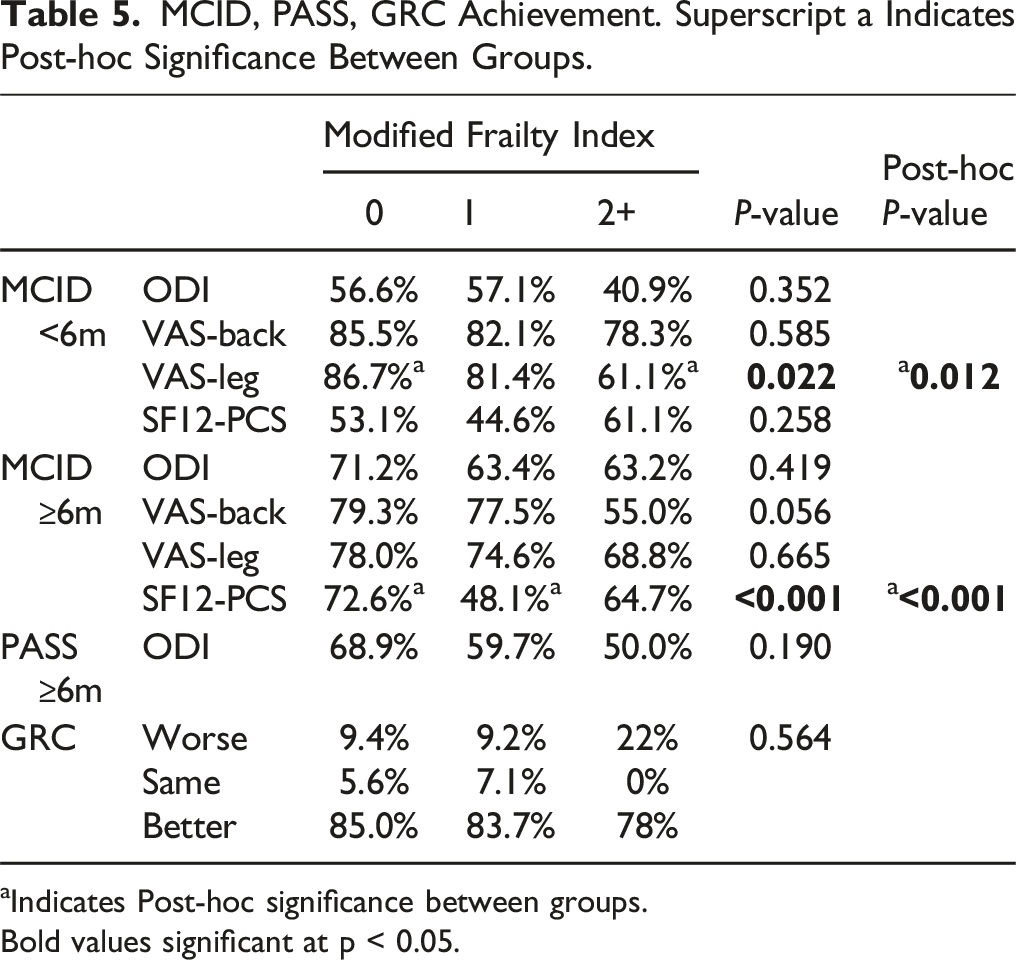
aIndicates Post-hoc significance between groups.
Bold values significant at p < 0.05.
Return to Activities. Superscripts a, b Indicates Post-hoc Significance Between Groups.
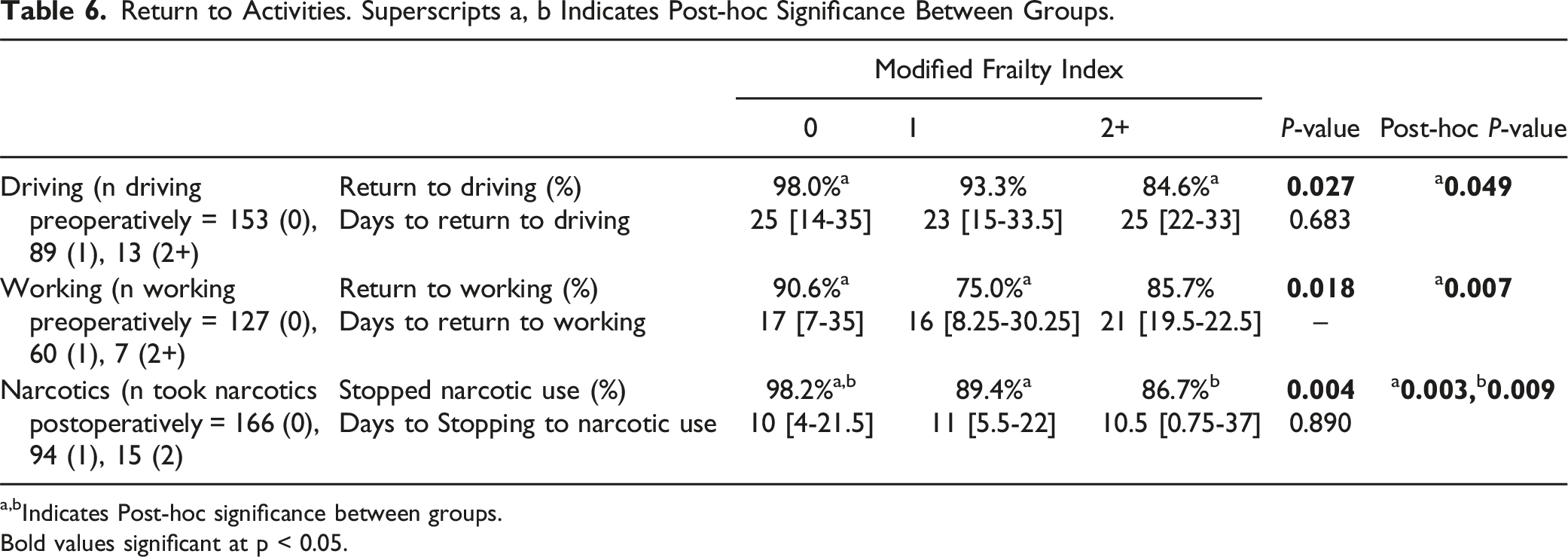
a,bIndicates Post-hoc significance between groups.
Bold values significant at p < 0.05.
Surgical Outcomes and Complications. Superscripts a, b Indicates Post-hoc Significance Between Groups.

a,bIndicates Post-hoc significance between groups.
Bold values significant at p < 0.05.
While the severe frailty cohort experienced higher rates of intraoperative, in-hospital medical, or postoperative complications, these differences were not statistically significant. Reoperation rate between the 3 cohorts was similar as well (Table 7).
Discussion
As the aging population continues to expand, a corresponding rise in lumbar spondylosis and frailty is anticipated. Recent evidence suggests that the assessment of perioperative risks and outcomes following spine surgery may be better informed by biological age rather than chronological age. 18 Biological age encompasses the accumulation of physiological challenges over time, alongside an individual’s physiological reserve, which reflects their capacity to withstand stressors such as surgery. 19 Frailty indices have been established to enhance the predictive accuracy in identifying patients at risk of experiencing unfavorable perioperative and postoperative outcomes. Specifically, the mFI-5 is a newer index, simplified from the previous mFI-11 index focusing on 5 key comorbidities—functional dependence, diabetes, hypertension, COPD, and congestive heart failure—chosen for their strong predictive association with frailty. We found that frail patients as defined by the mFI-5 had longer operative time, greater EBL, worse PROMs postoperatively, lower rates of return to activities of daily living, lower rate of narcotic discontinuation, and lower fusion rates compared to non-frail patients. Taken together, frailty status likely impacts the patient’s “overall health state” and may make it more difficult to recover from spine surgery.
Frail patients had significantly greater operative time and EBL with similar LOS. While it is not what we found in this cohort, longer LOS has been a well described outcome of frail patients following lumbar fusion.20,21 Moses et al. previously found a 3 day longer LOS among severely frail patients compared with non-frail patients after single level MI-TLIF. 20 Comparing these may be difficult however as the baseline patients with no frailty had a LOS of 3.1 days, a day longer than non-frail patients in our cohort. It is possible differences in institutional postoperative protocols clouds the LOS finding. Nonetheless, frail patients likely do have longer LOS and it is important to consider this within the context of postoperative recovery. Contrastingly, the literature regarding operative time and EBL in frail patients is less clear. Previous studies that have reported operative time and EBL in frail patients following TLIF have not found significant differences.20,21 In our cohort, severely frail patients experienced almost a 100% and 35% increase in operative time and EBL respectively compared to non and moderately frail patients. It is possible patients with significant frailty had more degeneration and therefore complex anatomy, predisposing to challenging surgeries and difficult recovery. Importantly however, frail patients did not experience greater rates of intraoperative complications. While these patients may undergo surgery safely, surgeons should be wary of the potential risks associated with prolonged anesthesia time and increased blood loss in frail patients.
Although the magnitude of improvement was similar between the cohorts, our findings demonstrated that frail patients had significantly worse PROMs at both the early and late time points despite similar preoperative symptom states. To our knowledge, 2 previous studies have reported PROMs in frail patients after TLIF, both utilizing the mFI-11 index.20,21 Postoperatively, in an analysis of 488 one or 2 level TLIF, not necessarily conducted using minimally invasive techniques, Tran et al. found that frail patients are associated with worse one-year postoperative PROMs including ODI, SF-12 PCS, VAS-back, and VAS-leg. 22 Frail patients demonstrated worse MCID achievement for SF-12 PCS and VAS-back. In their cohort the magnitude of recovery was significantly worse in frail patients as well for ODI, SF-12 PCS, and VAS-back. The second study, Moses et al. included a uniform cohort of 198 only single level MI-TLIF procedures and demonstrated frail patients had significantly worse one-year ODI, SF12-PCS, and SRS function score with no significant differences in delta change or MCID achievement. 20 We found similarly worse PROMs for ODI, VAS-back, VAS-leg, and SF12-PCS in frail patients and significantly worse MCID achievement in VAS-leg and SF12-PCS. For predicting postoperative PROMs, both mFI-5 and mFI-11 may serve as beneficial indices for providing adequate preoperative counseling concerning expectations for symptom improvement among frail patients.
Our study found that frail patients had lower rates of return to activities including driving and working compared to non-frail patients. While to the best of our knowledge, no prior analysis has reported recovery kinetics in frail patients, these findings are in line with the current literature on predictors of delayed or non-return to activities after lumbar spine surgery. Shinn et al. and Singh et al. have both identified that patients with worse physical function and higher comorbidity burden were associated with slower return to activities and non-returning to work.23-25 Similarly, prior studies have identified frailty as an independent risk factor for predicting driving cessation26,27 and have been shown to predict early retirement from both manual and non-manual labor28,29 among elderly individuals. To our knowledge, no prior studies have investigated the rate of return to driving and working rates after spine surgery specifically among frail patients. These findings further confirm that frailty represents a state of minimal “physiologic reserve” or the decreased ability for a patient to rebound after significant stressors. 2 Hence, particularly among frail patients who prioritize returning to work and/or driving postoperatively, it may be important to offer thorough counseling regarding the higher risk of not achieving these goals. The rate of narcotic discontinuation was also lower among frail patients within our cohort. The current data regarding frailty and postoperative narcotic use is limited and mixed. Among patients undergoing elective non-spine-related surgeries, Auckley et al. found that frailty is associated with increased opioid use whereas no associations were found by Admiraal et al.30,31 Shinn et al. demonstrated that elevated pain scores were independent predictor non-cessation of narcotics after elective lumbar surgery. 23 While frailty may not be a direct predictor for increased postoperative narcotic use, frail patients had higher early and late postoperative pain scores, which likely explains the correlation with the higher rate of narcotic use among these patients.
Successful fusion rates were also lower among frail individuals within our study cohort. Frail patients had evidence of fusion on 1 year CT at ∼80% compared with >95% in non-frail patients. This is consistent with a prior multicenter study of adult spinal deformity (ASD) patients by Miller et al. who utilized the adult spinal deformity frailty index (ASD-FI) and found a 13-fold higher likelihood of pseudarthrosis among frail patients undergoing ASD surgery. 32 The mFI-5 evaluates the presence of one or more baseline comorbidities including HTN, DM, COPD, and CHF, as well as the presence of partially or fully dependent functional status. Narain et al. conducted an analysis on predictors of surgical complications after single level MI-TLIF. In their cohort DM was found as an independent predictor of surgical complications of which pseudarthrosis represented 64% of. 33 In a cohort of MI-TLIF with rhBMP-2 or mesenchymal stem cells, Overley et al. found hypertension was predictive of radiographic nonunion. 34 Although no prior studies have identified a robust association between each of these individual variables with pseudarthrosis after MI-TLIF, our studies demonstrate that the evaluation of frailty that comprehensively accounts for the aggregate of all 5 variables is potentially a reliable and clinically useful predictor.
Postoperative complications and reoperation demonstrated a non-significant “dose response” trend towards greater rates in frail patients. It is possible we were not powered to demonstrate significant differences. Much of the literature does support the notion that frail patients have increased rates of medical and surgical complications after spine fusion.20,21,35 A systematic review of the literature by Chan et al. including 32 studies with a total of 127,813 patients concluded that regardless of how frailty was defined it was associated with increased serious adverse events, mortality, increased LOS, readmission, and non-home discharge. 36 Interestingly Burke et al. reported that MIS TLIF is protective against revision surgery and non-home discharge compared to open surgery in frail patients. 37 The exact relationship between MIS and open surgery for these patients remains unclear and the present study is not designed to answer that question.
Several limitations constrain the scope of this study. First, given the retrospective design of the study, the accuracy of the data hinges on the precision of charting and could also be prone to selection bias. Additionally, this study examines data solely from one institution, which may not accurately represent outcomes from other centers where surgical techniques or management approaches could differ. Moreover, the study encompasses a relatively small patient cohort of individuals with mFI-5 of 2+, possibly resulting in insufficient statistical power to identify certain associations such as differences magnitude of PROMs improvement, in LOS, as well as peri- and postoperative complications. Similarly, we are limited by our follow up. It is possible frail patients take longer to improve but will eventually plateau at the same state as non-frail patients. This question remains unanswered by our analysis and may be a subject of future research. Further studies investigating the external validity of this study may be important prior to the application in practice. We additionally did not analyze potential cofounder variables such as spinopelvic parameters, degree of degeneration, muscle health, and mental fitness that can certainly impact a patient’s ability to recover postoperatively, some of which are included in other frailty indices such as the Edmonton Frailty score. Despite these limitations, to our knowledge, this is the largest comprehensive study to date that investigates the association of frailty defined by mFI-5 with outcomes after one- or two-level MI-TLIF.
Conclusion
Increased mFI-5 is associated with inferior postoperative outcomes after one- or two-level MI-TLIF surgery including worse postoperative PROMs, lower rates of return to activities, lower rate of narcotic discontinuation, and higher pseudarthrosis rates. Whether these differences are modulated by overall poor health state or a decreased ability to recover requires further investigation. Additionally, more research is necessary to fully understand the correct patient selection and how best to optimize highly frail patients before surgical intervention.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HS2, LLC Innovasis Globus Medical, Inc. Intrinsic Therapeutics Inc. Bioventus HSS AND ASC Development Network.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
