Abstract
Study Design
Narrative review.
Objective
The objective of this review was to discuss the various surgical techniques and developments in cervical laminoplasty and outline any reported differences in outcomes between the different techniques used.
Methods
A PubMed literature search was performed using the terms “expansive”, “open door laminoplasty”, “cervical”, “double door laminoplasty”, “French door laminoplasty”, and “endoscopic”. All articles written or translated into English were considered and synthesized to provide a narrative overview of cervical laminoplasty techniques.
Results
Cervical laminoplasty techniques can be categorized either into “open door” or “double door” (“French door”) laminoplasty. Several iterations of each technique to address long-term closure of the laminoplasty, muscle preservation, and migration of bone grafts have been developed. There are no consistent reported differences in outcomes in the literature between the 2 techniques for individuals with degenerative cervical myelopathy.
Conclusions
Cervical laminoplasty is an effective surgical technique to expand canal size and achieve spinal cord decompression in the context of degenerative cervical myelopathy secondary to spondylosis or OPLL. Both the “open door” and “double door” laminoplasty techniques generally provide equivalent outcomes. Minimally invasive techniques including endoscopic approaches also continue to evolve, and future research comparing all approaches is warranted.
Introduction
Cervical laminoplasty is a posterior cervical surgical procedure that aims to decompress the spinal cord while preserving motion in the cervical spine, and it has been reported to be an effective technique for patients with degenerative cervical myelopathy (DCM) secondary to multi-level degenerative cervical spondylosis and/or ossification of the posterior longitudinal ligament (OPLL). Although laminectomy and instrumented fusion is a common procedure that has also been reported to be effective for these patients, concerns about adjacent segment degeneration, implant problems, and pseudoarthrosis warrant exploration of other surgical techniques. 1
Cervical laminoplasty was first developed in the 1970s, and its rationale was based on the principle of preserving the posterior tension band and facet joints while avoiding the complications of fusion, thereby maintaining an arguably more physiologic replication of native cervical biomechanics.2,3 Cervical laminoplasty has developed into a reasonable option to consider when aiming to perform a multilevel cervical decompression from a posterior approach. A variety of techniques have been described, each with its own potential advantages and disadvantages, including not only open techniques but the evolution of minimally invasive techniques including endoscopic approaches. 4 The purpose of this narrative review is to summarize the literature on the main surgical techniques that have been developed and used to perform cervical laminoplasty, and to examine any differences in outcomes between them.
Methods
A literature search of PubMed was performed using the following search terms: “expansive”, “open door laminoplasty”, “cervical”, “double door laminoplasty”, “French door laminoplasty”, and “endoscopic”. Articles published in or translated into English were used. There were no restrictions on date of publication. All study types were included. There were no systematic assessments of article quality. Articles were deemed relevant if they provided information on the technical aspects of cervical laminoplasty and comparisons of different laminoplasty techniques employed. All included articles were then synthesized to perform a narrative summary of the laminoplasty techniques in their original form and their associated variations.
Results
Anesthesia
Depending on the level of spinal cord compression, awake fiberoptic intubation or video laryngoscopy can be considered to avoid extensive cervical extension and possible worsening of cord injury. 5 Avoidance of hypotension which can exacerbate cord injury is important. More recent evidence suggests there may be a role for augmenting mean arterial pressure (MAP) during surgery for DCM. A clinical trial of 60 DCM patients randomly divided individuals into a “normotensive” group (30 patients) and “hypertensive” group (30 patients; intraoperative MAP 20 mm Hg higher than preoperative average MAP ±5 mm Hg). 6 There were no statistically significant differences in modified Japanese Orthopaedic Association (mJOA) scores between groups up to 1 year of follow-up although the VAS scores were lower in the “hypertensive” group at follow-up. 6 Larger trials are required to fully elucidate the effects of MAP parameters on outcomes after surgery for DCM.
Positioning
Individuals undergoing cervical laminoplasty are positioned in the prone position under general anesthesia. Use of the K-line (a line connecting the midpoints of the spinal canal at C2 and at C7) pre-operatively especially in the setting of OPLL can help determine if a posterior approach will result in adequate neurologic recovery, more specifically if the OPLL does not extend beyond the K-line on radiographs. 7 Stabilization of the neck can be performed in a variety of ways, including use of a Mayfield three-pin head holder or a Sugita head frame.5,8,9 In-line cervical traction with Gardner-Wells tongs has also been described. 10 The spine is generally positioned parallel to the floor and neutral. Patients can also be positioned with up to 30° of reverse Trendelenberg and surgeons can ensure that the abdomen is free to avoid venous congestion and increased bleeding risk during surgery. Care is taken to avoid hyperextension of the cervical spine to prevent further neurologic decline from cord compression. It is also important to ensure enough padding of all bony prominences and pressure points with arms secured parallel to the body. The shoulders can be taped caudally to allow more visualization of the cervical spine with intraoperative x-rays to facilitate localizing the levels of interest. Care should be taken to avoid excessive shoulder retraction in order to avoid brachial plexus injury and decrease risk of C5 palsy due to nerve traction. 11
Surgical Adjuncts
The use of neuromonitoring is not necessarily required although it has been recommended as an adjunct to monitor neurologic status of the individual during the procedure, especially in cases of OPLL with severe cord compression and moderate to severe myelopathy. 12 Transcranial motor-evoked potentials and somatosensory-evoked potentials may be used to monitor throughout the case. 5 Baseline recordings prior to positioning can be obtained while supine.
Laminoplasty Approach
Historically, there has been the development of several key techniques for cervical laminoplasty. While there have been many different variations, they have generally been classified into “expansive open-door” laminoplasty and “French door” or “double door” laminoplasty.
“Expansive Open-Door” Laminoplasty
In the original description of the “expansive open-door” laminoplasty by Hirabayashi in 1983, the muscles are dissected from the involved spinal levels bilaterally to expose the laminae, and care is taken to avoid violating the facet joints bilaterally.2,13 The tips of the spinous processes are first removed in order to prevent re-closure of the laminoplasty when suturing the muscles back together. This is followed by burring completely through the interface of the lateral mass and laminae on 1 side only, just medial to the pedicles (Figure 1). The side with greater canal or foraminal stenosis or clinical evidence of radiculopathy is the side of the “open door”.
14
On the opposite side, the bone of the laminae-lateral mass interface is only partially burred, leaving the ventral cortex intact. This side acts as the hinge of the door. Ligamentum flavum is removed in the most rostral and caudal interspaces to allow then the laminae to be opened. The interspinous ligaments are preserved. The ligamentum flavum underlying the open laminae is removed with Kerrison rongeurs. Steps to Completing the “Open Door” Laminoplasty. (A) The Lamina is Burred Fully at the Interface of the Lateral Mass and Laminae on One Side Only, Just Medial to the Pedicles. This Forms the “Open Door”. Partial Burring on the Opposite Side is Performed to Create the “Hinge” of the Door. (B) The Lamina is Then Elevated Towards the “Hinge”. (C) Miniplates are Then Secured to Keep the “Door” open. Reproduced With Permission From Grochulla F., Laminoplasty. In: Vieweg U and Grochulla F. Editors. Manual of Spine Surgery. Second Edition. Springer-Verlag GmbH Germany 2023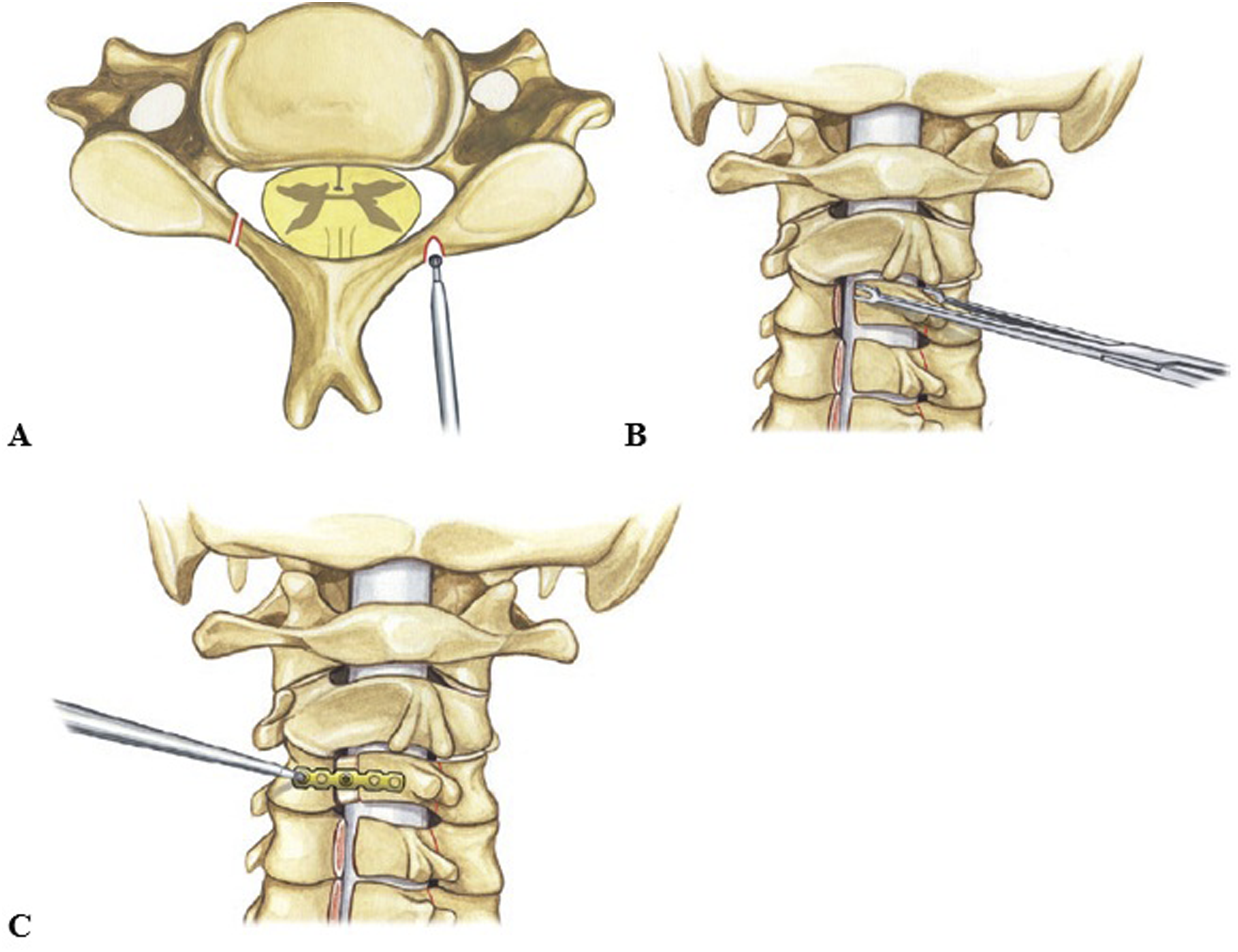
Various techniques have been described to keep the “door” open. Originally, stay sutures were stitched through the deep muscles and facet capsules on the hinge side, which were then sutured around the base of the spinous processes to tether the “door” open. 13 However, the use of sutures resulted in questionable long-term stability and undesirable “spring back” closure of the hinge, resulting in restenosis due to breakage or stretching of the sutures over time.15,16 Various adaptations of the suture technique were then proposed, including the use of suture anchors to add stability.17-19 To address this problem, bone grafts and synthetic implants including hydroxyapatite implants placed between the open lamina and lateral mass, secured with suture, have also been described, with some evidence of less laminar closure compared to suture-only techniques.20-22
Proponents of these techniques argue that reconstruction of the laminae is required post-laminoplasty in order to ensure recovery from cervical myelopathy and prevent post-operative kyphosis that can result from sinking of the laminae. Thus, promoting the fusion of the “open-door” hinge and spacer can allow for a rigid construct. However, the use of grafts and spacers has been complicated by migration and even thecal sac compression.
23
As such, use of titanium miniplates to stabilize the laminoplasty was eventually proposed and is now considered a key modern technique to keep the “door” open (Figure 2). Advantages include relative cost-effectiveness compared to using combo plate and bone graft constructs, relative ease of technique, and immediate stability of the construct leading to better fusion on the hinge side of the laminoplasty, especially in comparison with suture-only techniques.14,15,24-26 Post-operative CT Scan Showing Use of a Titanium Miniplate to Maintain Expansion in an “Open Door” Laminoplasty. In This Case, the “Hinge” on the Patient’s Right Side has Healed, Such that the Drilled Incomplete Trough is No Longer Visible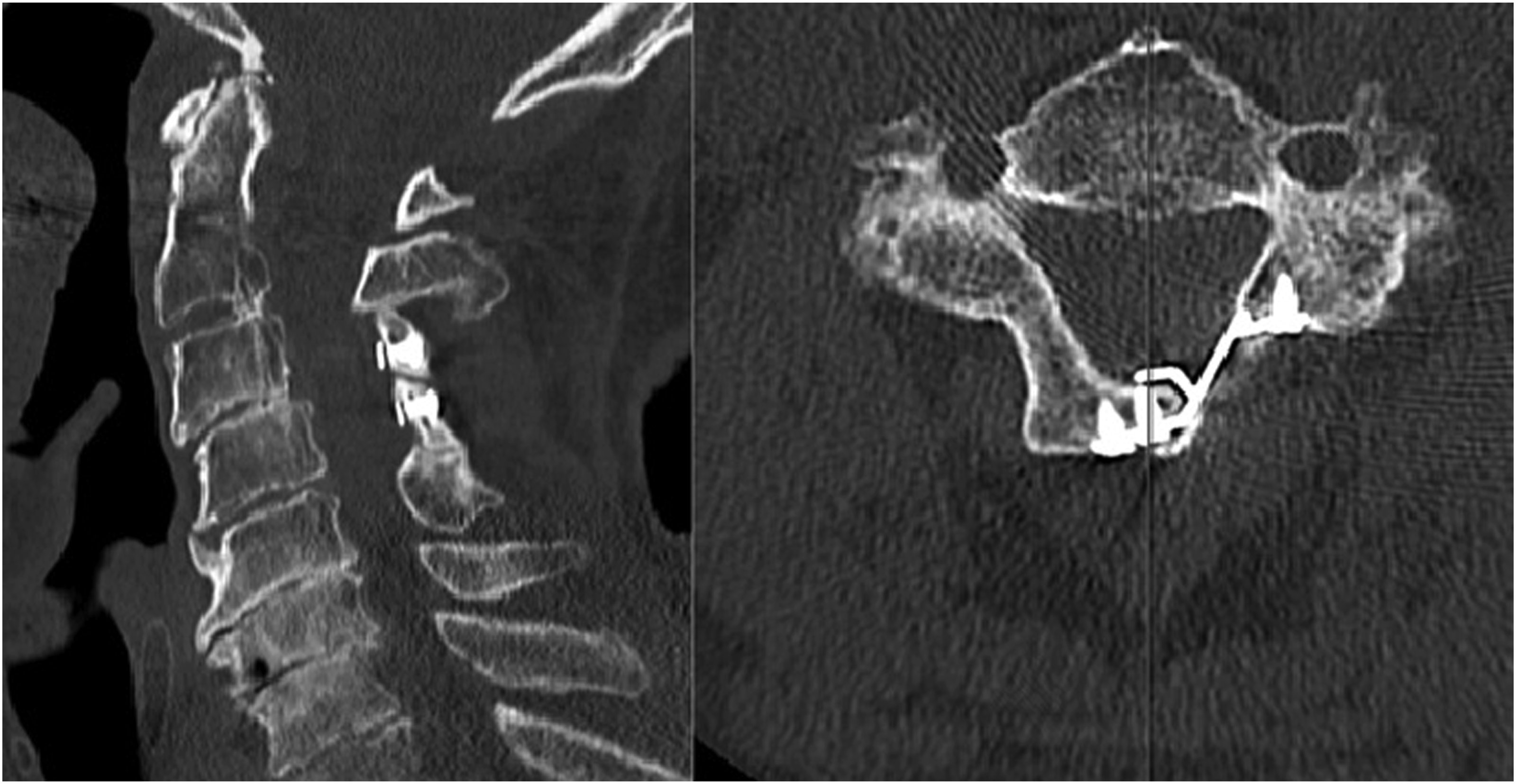
Generally, the miniplates are configured into an “open Z” configuration with 1 hole secured to the lateral mass and another hole secured to the open lamina. Plates available include those that are specifically designed for cervical spine laminoplasty and non-specific plates for maxillofacial reconstruction that are adapted for laminoplasty. The plates are secured with screws approximately 2 mm in diameter and 5 to 9 mm in length, with longer screws in the lateral mass and shorter screws in the lamina. Typically, 1 plate is placed per level of the laminoplasty or 1 plate at every other level, with potentially better clinical outcomes using plates at every level but at higher cost, although other literature reports equivalent clinical outcomes.14,15,27-29
“French Door” or “Double Door” Laminoplasty
The “French door” or “double door” laminoplasty was originally described by Kurokawa in 1982.30,31 In this technique, muscle is dissected to expose the cervical laminae to the medial aspect of the facet joints with removal of the interspinous ligaments. In contrast to the “expansive open-door” laminoplasty, a split opening through the midline of the lamina is made in addition to bilateral hinges at the lamina-facet joint mass border on each side. The opening in the midline and the hinges can be performed with a high-speed burr. The spinal canal is then enlarged by opening the laminae in the middle. Classically, iliac crest autograft was then inserted in between the split laminae and secured with sutures or wires to promote fusion (Figure 3). Steps to Completing the “Double Door” or “French Door” Laminoplasty. A Split Opening Through the Midline of the Lamina is Made in Addition to Bilateral Hinges at the Lamina-Facet Joint Mass Border on Each side. The Spinal Canal is Then Enlarged by Opening the Laminae in the Middle, and the Opening is Maintained With a Graft, Spacer, or Miniplates. Reproduced With Permission From Kaito T and Yonenobu K. Cervical Laminoplasty. In: Fessler RG and Sekhar LN. Editors. Atlas of Neurosurgical Techniques: Spine and Peripheral Nerves. Second Edition. Thieme Medical Publishers, Inc 2016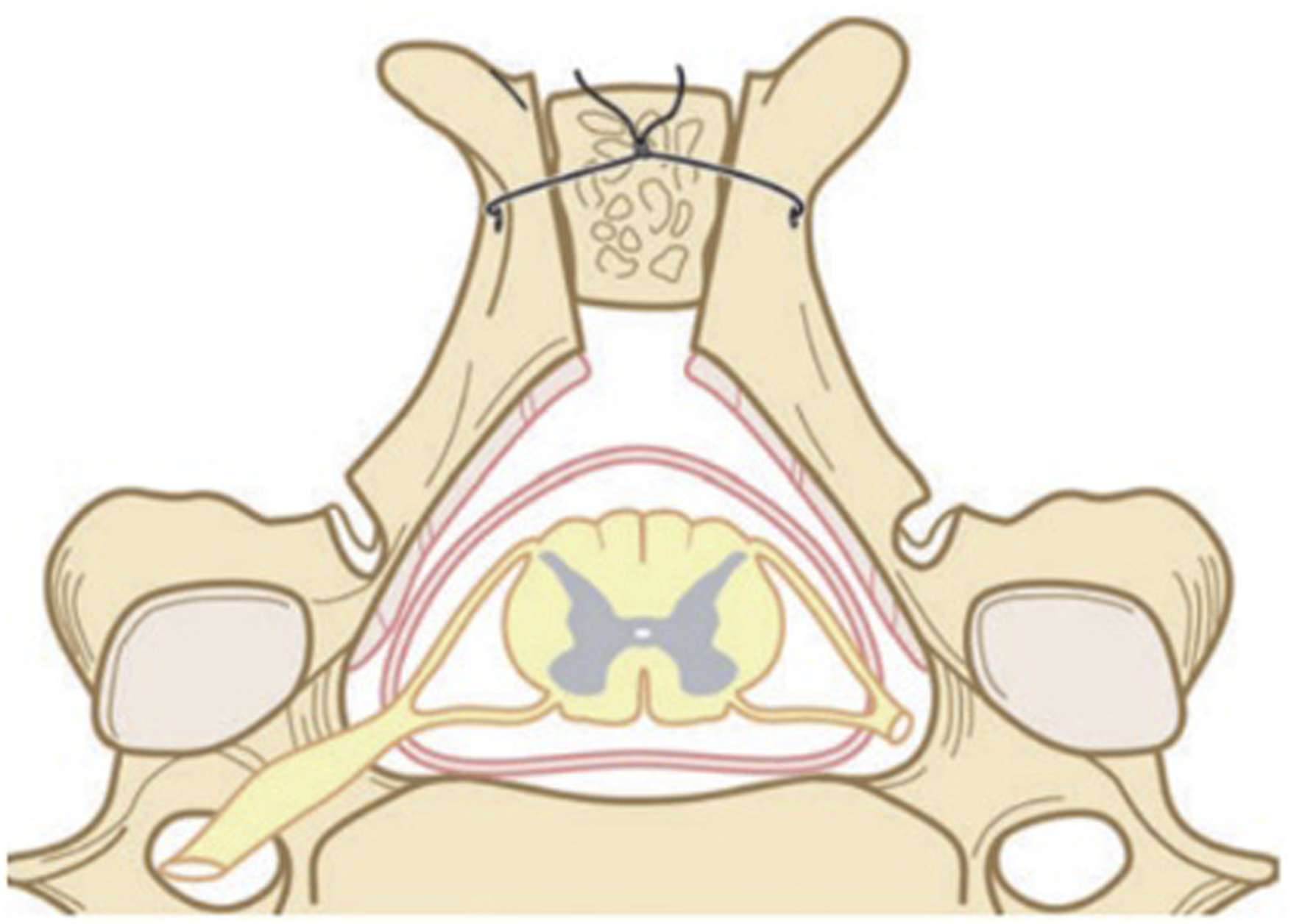
There were previously documented concerns around the technical difficulty of this approach because of the risk of cord injury with the midline opening of the laminae with the burr. 32 A modification of this approach with an alternative midline splitting technique using a threadwire saw (T-saw) was proposed by Tomita. 32 In this approach, a subperiosteal exposure to the medial aspect of the lateral mass is performed. Then removal of the ligamentum flavum is performed in the interspace of the level above and below the intended levels for the laminoplasty. The T-saw is then passed first in its polyethylene sleeve in the midline epidural space between the levels of interest through the openings in the ligamentum flavum created previously. The sleeve is then removed and the T-saw is then used to perform the midline split in the ventral to dorsal direction. Lateral gutters are then burred in the medial one-third of the lateral mass with preservation of the ventral cortical bone. These gutters act as the hinges similar to the classic “double door” laminoplasty, and the split laminae are opened with a laminar spreader. The hemi-laminae are held open with the use of a graft (ie, iliac crest autograft, allograft rib or fibulae, ceramic/hydroxyapatite spacers) held in place with sutures or wires.
Concerns around the use of the bone graft or other spacer to keep the hemi-laminae open have again led to alternative methods. Cases of delayed dural injury and spinal cord injury by migration of the hydroxyapatite spacers were reported. 33 One study examined 86 individuals who underwent “double-door” laminoplasty with 278 hydroxyapatite spacers used and found dorsal migration >2 mm in 42% of cases, spacers rotation >10° in 38%, and bony fusion in only 8.7%. 34 Loosening of hydroxyapatite spacers due to movement of the cervical spine or breakage of the suture holding the spacers before bony fusion have been posited as potential reasons for neurological injury. 33 To avoid this complication, 1 technique variation uses a 2.0 mm drill to make holes on the medial edge of each hemi-laminae to fit a plate that keeps the hemi-laminae in an open position, avoiding the use of a bony spacer. Another technique removes the spinous processes of the levels of interest and splits open the hemi-laminae as previously described but uses mini-plates secured to each hemi-lamina with an 8 mm screw, resulting in a “box-shape” laminoplasty.35,36 In a similar response to the concerns around the stability of sutures in the “expansive open-door” laminoplasty, another group proposed the use of suture anchors to maintain the split hemi-laminae open.37,38 Suture anchors are placed in the lateral masses and holes are made in the medial sides of the laminae using a 1.7 to 2 mm burr. Sutures are then placed through the holes and tied to prop each set of hemi-laminae open. Combinations of mini-plates and bone grafts have also been described. 10
To address concerns around extensor muscle disruption and in order to prevent post-operative kyphosis, another variation was proposed by Yoshida in which the nuchal ligament is left intact. 30 In this technique, the paravertebral muscles on the left side are removed from their spinous process attachments and from the laminae in a subperiosteal fashion. The spinous processes of the levels of interest are removed including 1 level above and below. The right sided muscles are then removed from the laminae in a subperiosteal fashion with the spinous processes still attached in addition to the supraspinous and interspinous ligaments. The resulting muscle-ligamentous complex is then retracted laterally. The remaining bases of the spinous processes are split sagittally and lateral gutters are created with a high-speed burr as for a classic “double door” laminoplasty. The hemi-laminae are then opened, and bone graft is then placed into the opening to prop the hemi-laminae open and secured with wire to the spinous processes. The previously cut spinous processes are then reattached and the dissected left-sided muscles are re-sutured to those spinous processes.
Endoscopic Laminoplasty
More recent advances in minimally invasive surgical techniques have also been applied to cervical laminoplasty. Endoscopic approaches can be used when there is a single level laminoplasty. 4 Biportal endoscopic approaches involve making small incisions at the midpoint between the pedicle and midline. As with classic open approaches, the soft tissue is dissected, exposing first the ipsilateral laminae and interlaminar spaces of the levels of interest. Then, the spinous process is drilled out while preserving the interspinous ligaments in order to create a contralateral working space. The contralateral gutter in the bone is then created, which acts as a hinge. An ipsilateral gutter is then created along the medial border of the ipsilateral facets. Hooks and dissectors are then used to release the ligamentum flavum from the drilled laminar borders. The laminar flap is then elevated using a scope retractor and a dissector. Metal plates are then applied through the portals and secured with screws, first on the lateral mass then on the lamina. 4 Another variation of the technique has used suture anchors on the hinge side of the laminoplasty between the lateral mass and spinous process to keep the laminoplasty open. 39 While an advanced technique with advantages related to a reduction in invasiveness, individuals with thin laminae and osteoporosis are not good candidates for this approach given the risk of hinge failure. Individuals with multi-level pathology may not be good candidates for this approach. 4 There is also a significant learning curve requiring first expertise in endoscopic spine approaches before attempting endoscopic cervical laminoplasty.
Post-operative Care
Reported use of a cervical collar after cervical laminoplasty and length of time in a collar vary in the literature. In general, cervical collars are not required post-operatively, and 1 randomized clinical trial demonstrated no differences in neck pain, JOA scores, range of motion, and complications up to 1 year after “double door” laminoplasty surgery between groups that did not have collar compared to use of a Philadelphia collar for 2 weeks post-operatively. 40 Post-operative upright x-rays of the cervical spine to assess alignment and plate placement are important. Individuals can usually be discharged within 2 to 3 days after the operation. Routine follow-up 1 month post-operatively and at regular intervals thereafter is important to monitor for implant failure, worsening kyphosis, or evidence of restenosis or worsening myelopathy.
Comparisons of Laminoplasty Techniques
Several studies have been performed to compare the outcomes of “open door” vs “double door” techniques for cervical laminoplasty. Proposed advantages for “double door” laminoplasty include: symmetrical reconstruction of the posterior arch, maintenance of canal enlargement by the midline bone graft, and lack of epidural bleeding because of the midline bony exposure.32,41
One of the largest randomized controlled trials comparing “open door” with “double door” laminoplasty was performed by Nakashima et al. 42 In this study, a total of 90 patients with cervical myelopathy were analyzed after randomization to undergo either “open door” (44 patients) or “double door” laminoplasty (46 patients). They found no differences in pre-operative or post-operative JOA scores, surgical duration, and estimated blood loss with an average follow up of approximately 30 months in both groups. There were no perioperative complications in either group. While clinical outcomes were not different between groups, they did find differences in radiographic parameters including greater decrease in cervical lordosis and significantly lower post-operative cervical range of motion in the “open door” group. However, the “open door” group also demonstrated a significantly greater percent increase in spinal canal enlargement. It is difficult to make definitive conclusions with respect to the superiority of 1 technique compared to the other given the relatively small sample size and overall similarity in clinical outcomes.
Okada et al 43 performed a randomized controlled trial comparing 20 patients who underwent “open door” laminoplasty vs 20 individuals who underwent “double door” laminoplasty. 43 They found reduced operative time with the “open door” laminoplasty but greater mean blood loss. Axial pain was worse after “open door” laminoplasty, but JOA scores demonstrated no differences for neurological function at final follow-up (at least 1 year after surgery). 43
In a retrospective review of 51 patients who underwent cervical laminoplasty (23 “open door” and 28 patients “double door” laminoplasty), with a mean follow-up of approximately 26 months, Lee et al. found that mean operative time was longer in the “double door” group although not statistically significant. 44 This study also noted significantly greater mean blood loss but no significant differences for radicular pain, axial neck pain, and JOA or Nurick scores at last follow-up. There were more complications after “double door” laminoplasty including more wound infections (0 in “open door” group, 3 in “double door” group) and C5 palsies (1 in “open door” group, 2 in “double door” group).
Wang et al performed a systematic review and identified 4 studies, 2 of which were randomized controlled trials. They found that “open door” laminoplasty had greater JOA scores post-operatively compared to “double door” laminoplasty, with no significant differences in operative time, intraoperative blood loss, and peri-operative complications including C5 palsy. 45 Luo et al reported a meta-analysis of 6 studies (including 3 randomized controlled trials) and found no significant differences for operative time, blood loss, complication rates, or length of hospital stay. 46 There were also no significant differences in post-operative JOA scores between the group, but the magnitude of canal expansion was significantly larger in the “open door” group compared to the “double door” group. More recently, Chen et al 47 reviewed 14 studies (including 6 randomized controlled trials) with a total of 1010 patients (519 cases of “open door” laminoplasty, 491 cases of “double door” laminoplasty) and found no significant differences for operative time, JOA scores, cervical lordosis, post-operative cervical range of motion, mean sagittal canal diameter, C5 palsy, or CSF leak. 47 There was significantly more intraoperative blood loss in the “open door” group, as well more axial pain.
In contrast, Ma et al also performed a systematic review and demonstrated better JOA scores, no difference in operative time, lower length of hospital stay, and less adverse events in the “double door” laminoplasty group. 48 Limitations of this study included absence of follow-up data and lack of granularity regarding adverse events. Only 7 studies were included, for a total of 99 individuals in the “double door” group and 125 individuals in the “open door” group.
Nagoshi et al. performed a prospective study that included 205 patients with cervical myelopathy secondary to OPLL who were followed across multiple centers for up to 2 years after undergoing laminoplasty. 49 The choice of technique was at the discretion of the surgeon, and 41 individuals underwent “open door” laminoplasty while 164 individuals underwent “double door” laminoplasty. There were no statistically significant differences in the number of decompressed levels or frequency of complications (including surgical site infection, dural tears, and C5 palsy). There were no differences in the changes in JOA score between the 2 groups. Pain scores for neck and shoulder were significantly reduced in the “open door” group but not the “double door” group at 2 years after surgery, but the differences were small and not likely to be clinically important.
Overall, there do not appear to be any reliable or consistent reported differences in outcomes in the literature between the 2 techniques for individuals with cervical myelopathy. Therefore, both techniques may be reasonable options for posterior decompression of the spinal canal for appropriately selected individuals with degenerative cervical myelopathy.
Endoscopic laminoplasty techniques offer a new minimally invasive strategy for which techniques are continuing to evolve. One single centre prospective series examined 45 DCM patients who underwent cervical endoscopic laminoplasty. 50 The mean mJOA scores improved from 8.0 ± 1.72 to 13.29 ± 1.85 after operative intervention with an associated increase in the sagittal diameter of the canal by 2.6 ± 0.8 mm (18 to 48 months follow-up). 50 The study did not report any serious complications aside from transient upper limb dysfunction and a loose mini-screw without further malposition on flexion-extension radiographs. 50 While potentially safe and effective with the right expertise, at present, there are no large case series or trials comparing endoscopic techniques with other open techniques. Future research comparing more minimally invasive techniques with open approaches to laminoplasty as they develop are warranted.
Discussion
We have provided descriptions of the key foundational techniques used to perform cervical laminoplasty. Various iterations of the techniques have been proposed since the procedure was originally described in order to address various concerns that have developed, including: keeping the laminoplasty open over time, maintaining muscle integrity, and preventing post-operative kyphosis and restenosis. Limited studies have considered the variations of techniques beyond “open door” vs “double door” laminoplasties, such that interpretations of outcomes for each nuanced iteration of technique are not strictly feasible. However, it generally appears that there has been a movement away from the use of sutures to keep the “door” of the laminoplasty open in favor of plating, and the use of bony grafts or artificial spacers may no longer be required to achieve satisfactory outcomes.
Laminectomy and fusion still remains an option to consider, and although there has been substantial geographical variability in use of cervical laminoplasty vs laminectomy and fusion, studies comparing the 2 procedures have failed to demonstrate clear superiority of 1 technique compared to the other. For example, a recent meta-analysis of 22 studies and 1025 individuals undergoing laminoplasty compared with 1103 individuals undergoing laminectomy and fusion did not find differences in rates of reoperation, cervical lordosis, neurological function, or neck pain scores. 51 The study did demonstrate statistically significant shorter operative times and less blood loss during surgery with laminoplasty, as well as reduced rates of C5 palsy, although C5 palsy risk remains controversial. 51 C5 palsy remains 1 of the more debilitating morbidities after cervical laminoplasty with rates between 5% to 17%. 52 Another meta-analysis comparing incidence of C5 palsy between cervical laminoplasty and laminectomy and fusion consisting of 9 studies with 360 individuals in the laminoplasty group and 340 individuals in the laminectomy and fusion group also demonstrated statistically significant higher incidence of C5 palsy in the laminectomy and fusion group. 53 However, risk factors for C5 palsy in cervical myelopathy remain an ongoing area of study and may not entirely be attributable to choice of surgical technique. Pre-operatively alignment and effects on C5 palsy have been investigated with a recent study retrospectively examining 60 individuals with DCM who underwent either laminoplasty (30 individuals) or laminectomy and fusion (30 individuals). 54 Subgroup analysis did not reveal statistically significant differences between the pre-operatively lordotic subgroups (Cobb angle >10° from C2 to C7) compared to straight alignment subgroups (Cobb angle 0 to 10° from C2 to C7) for either procedure although more research into pre-operative alignment and effects on C5 palsy rates are warranted. Foraminal diameter has been highlighted as a factor impacting risk of C5 palsy, which may be independent of surgical approach. Another systematic review including all cervical surgical approaches for degenerative cervical spine pathology found increased risk of C5 palsy per millimeter decrease in pre-operative foraminal diameter at C4/5 based on multivariate analysis. 55 In this regard, prophylactic foraminotomy has been proposed to reduce risk of C5 palsy. However, a study examining the role of prophylactic foraminotomy at C4/5 in 35 individuals with cervical myelopathy who underwent laminoplasty (19 individuals) or posterior decompression and fusion (16 individuals) did not find statistically significant differences in incidence of C5 palsy within each group between individuals who underwent foraminotomy compared to those who did not. 56 In contrast, another study of 141 individuals who underwent cervical laminoplasty with prophylactic C4/5 bilateral foraminotomies was compared with a prior group of 141 individuals who underwent laminoplasty without foraminotomies. 57 Two-year data for 121 individuals who underwent prophylactic foraminotomies compared to 115 individuals who did not demonstrated a statistically significant reduction in incidence of C5 palsy (1.7% vs 7.0%, respectively). 57 Further research into factors contributing to C5 palsy and comparisons between cervical laminoplasty and laminectomy and fusion are warranted.
There are pre-operative factors that also need to be considered when deciding between cervical laminoplasty compared to laminectomy and fusion. Individuals with significant radiculopathy in addition to myelopathy due to foraminal stenosis may be better treated with anterior/posterior cervical fusion as foraminal stenosis is more difficult to address with a laminoplasty. 51 Cervical laminoplasty can be considered in individuals with multi-level cervical stenosis without kyphotic deformity or instability on x-rays. 51 Individuals with a significant neck pain component have been thought to better be treated with a fusion although there is data to suggest that there are no differences in this regard between laminoplasty and fusion.51,58 The examination of pre-operative radiographic features that can predict outcomes after cervical laminoplasty remains ongoing and has more directly operationalized the factors mentioned above. K-line has been shown to be helpful to determine if an anterior approach or posterior approach is ideal for spinal cord decompression, especially in the context of OPLL. 7 Individuals with OPLL that was K-line (+) and did not go beyond the K-line demonstrated greater rates of recovery of neurologic function compared to individuals with OPLL that extended beyond the K-line after posterior decompression, providing a method to determine if posterior decompression is warranted (ie, laminoplasty or laminectomy and fusion) based on radiographic parameters. 7 More recently, the use of the K-line tilt (angle between a line connecting the midpoints of the spinal canal at C2 and C7 and a line vertical relative to the horizon) has been proposed as a predictor of outcomes. 59 Individuals with a higher K-line tilt were found to be at higher risk of developing kyphotic deformity after cervical laminoplasty based on multivariate logistic regression modelling with a cutoff of approximately 10°. 59 Cervical parameters other than K-line tilt including C2-7 Cobb angle, T1 slope, and C2-7 sagittal vertical axis have also been examined. Research is still ongoing as to which parameters accurately predict outcomes after cervical laminoplasty and the relation of these parameters to clinical outcomes. Previous work examining individuals with cervical myelopathy who underwent laminoplasty and compared pre-operative and post-operative alignment parameters at 1 year follow-up found a higher T1 slope and greater C2-7 sagittal vertical axis were associated with higher loss of lordosis based on a multiple linear regression model. 60 A study of 60 individuals with cervical myelopathy with local kyphosis (defined as ≥ 5°) who underwent either laminoplasty or fusion with reduction of kyphosis found statistically significant improvements in C2-7 Cobb angle in the fusion group, without differences in area of the spinal cord at follow-up, with associated better rate of recovery of mJOA. 61 Another recent study retrospectively examining 167 individuals who underwent anterior cervical discectomy and fusion (45 individuals), laminectomy and fusion (55 individuals), and laminoplasty (67 individuals) demonstrated no differences in pre-operative and post-operative neck disability index for a mean post-operative follow-up period of approximately 19 months. 62 There were no statistically significant differences between the laminectomy and fusion and laminoplasty groups pre- or post-operatively in terms of C2-7 Cobb angle, T1 slope, and C2-7 sagittal vertical axis. 62 In contrast, another recent study examined 67 individuals with OPLL who underwent lateral mass screw fixation and decompression (36 individuals) compared to cervical laminoplasty (31 individuals). 63 Pre-operative C0-2 Cobb angle, C2-7 Cobb angle, C2-7 sagittal vertical axis, C7 slope, T1 slope, and spino-cranial angle (angle between the C7 slope and a straight line joining the middle of the C7 endplate and middle of the sella turcica) demonstrated no statistically significant differences between groups. 63 At last follow-up, there was a statistically significant lower C2-7 sagittal vertical axis, C7 slope, and T1 slope in the laminoplasty group compared to the laminectomy and fusion group. Furthermore, there were no statistically significant differences pre-operatively for neck disability index and VAS neck score. However, at last follow-up, there was a statistically significant lower neck disability index and VAS neck score in the laminoplasty group compared to the laminectomy and fusion group. 63 In this regard, there are mixed results with respect to the ability to preserve cervical alignment and associated pain outcomes comparing laminectomy and fusion with laminoplasty. Taken together, it does appear that loss of preservation of cervical alignment may result in worsening pain and neurological outcomes in general and, for cervical laminoplasty specifically, higher risk of cervical malignment and poor outcomes are associated with local kyphosis, higher T1 slope, greater C2-7 sagittal vertical axis, and higher K-line tilt.
Another common theme that has emerged is meticulous attention to muscle dissection. Care is generally taken to avoid injury to the semispinalis cervicis connecting to the C2 spinous process, the trapezius connecting to the C7 spinous process, and the multifidus to the inferior laminar border of C7 in order to maintain muscle stabilization of the cervical spine and mitigate worsening post-operative kyphosis. 37 Care should also be taken to avoid violation of the facet joint capsules as much as possible to avoid introducing instability during the exposure.
Cervical laminoplasty has been most commonly described for the subaxial spine from C3 to C6. There is recent literature describing C1 and C2 laminoplasty, but this is not commonplace because degenerative compression occurs less frequently at these levels. The atlanto-axial cervical spine presents a unique challenge for the use of laminoplasty because of the significant muscle attachments, especially to C2 including the obliquus capitis inferior, rectus capitis posterior major, semispinalis cervicis, spinalis cervicis, interspinalis, and multifidus muscles, which play a key role in cervical extension and increase risk of pain and post-operative kyphosis if detached. 64 Dissection strategies to preserve as much as possible these muscle attachments have been proposed. “Extensive dome-like laminoplasty” of C2 has been proposed as an alternative solution, which involves performing a C3 laminectomy followed by high-speed burr drilling of the inner lamina of C2, creating a groove towards the inner cortex of the lamina. 65 Eventually, the inner lamina of C2 can be removed “en bloc” with an osteotome, creating more room for the spinal cord. Future research is required to fully develop and evaluate reliable techniques to perform laminoplasty at these upper levels and even if the use of laminoplasty in comparison to laminectomy and fusion is appropriate at these levels. Finally, minimally invasive techniques including endoscopic laminoplasty are continuing to evolve and will provide alternatives with minimal tissue dissection that could also play an important role in preservation of muscle stabilization to improve cervical alignment outcomes and reduction of complications.
Conclusion
Cervical laminoplasty is an effective surgical technique to expand canal size and achieve spinal cord decompression in the context of degenerative cervical myelopathy secondary to spondylosis or OPLL. In general, laminoplasty can be divided into an “open door” or “double door” approach depending on where the opening of the laminoplasty is located. While there have been various iterations of these techniques, the basic principles of the techniques have not largely changed. In the literature, there are no clear differences between the 2 techniques in terms of surgical outcomes or complications although newer minimally invasive endoscopic laminoplasty techniques also continue to evolve, and future research is required to continue to compare and investigate outcomes for all these approaches.4,39,50
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international spine experts, and AO Spine North America. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization based in Davos, Switzerland.
