Abstract
Study Design
Prospective Cohort Study.
Objective
To compare the effectiveness and safety of perioperative denosumab, bisphosphonates, and a control group in promoting lumbar fusion after midline lumbar fusion (MIDLF) surgery in patients with osteopenia or osteoporosis.
Methods
This prospective cohort study enrolled 54 patients with osteopenia or osteoporosis undergoing MIDLF surgery. Patients were divided into 3 groups: denosumab (60 mg subcutaneous injection), bisphosphonates (5 mg zoledronic acid intravenous infusion), and control (no anti-osteoporotic drugs, only calcium + vitamin D). Outcomes included fusion rates, clinical scores, bone metabolism markers, and adverse events.
Results
54 patients were included in the study. Among them, 50 patients completed the 6-month postoperative follow-up. Due to the impact of the COVID-19 pandemic, only 29 patients completed the final follow-up. At 6 months, fusion rates were significantly higher in the denosumab (25% complete, 68.6% partial) and bisphosphonate (25% complete, 75% partial) groups compared to the control group (10.5% complete, 57.9% partial, P < 0.001). No significant difference was found between the 2 drug groups. Postoperative fever was more common in the bisphosphonate group (71.4%) than in the denosumab group (11.8%, P =0.004) and control (23.5%, P = 0.011) groups.
Conclusions
In the short-term follow-up, both denosumab and bisphosphonates significantly improved the early bone fusion rate after MIDLF surgery, with no significant difference observed between the two. Denosumab was associated with a lower incidence of postoperative fever, making it a recommended choice for perioperative anti-osteoporosis therapy.
Introduction
Lumbar degenerative diseases (LDD), including lumbar spinal stenosis, lumbar spondylolisthesis, and lumbar degenerative scoliosis, have a high incidence rate in the elderly population. Pedicle screw instrumentation and fusion surgery have become one of the effective treatments for these conditions. However, with the progression of population aging, osteoporosis has become an increasingly severe issue, which significantly impacts the outcomes of spinal fusion surgeries. 1 Studies have shown that the axial pullout strength of pedicle screws is positively correlated with bone density.2,3 In patients with osteoporosis, decreased vertebral bone density leads to insufficient screw purchase, resulting in an increased risk of screw loosening, displacement, and even pullout, which are associated with internal fixation failure. 4 To reduce the risk of postoperative internal fixation loosening in patients with osteoporosis, current strategies include screw structural modifications,5,6 optimized screw placement techniques or special methods,7,8 cement augmentation of pedicle screws,9-11 and the use of anti-osteoporosis drugs.12,13
The cortical bone trajectory (CBT) screw fixation technique, first proposed by Santoni et al in 2009, 14 can maximize the contact between the screw and cortical bone, thereby enhancing the stability of fixation in osteoporotic bone. This technique is known to reduce soft tissue dissection and postoperative complications,15-17 and was integrated into the midline lumbar fusion (MIDLF) procedure performed in this study.
Spinal fusion is often considered as an important indicator to evaluate the efficacy of lumbar fusion surgery, and is also closely related to the long-term prognosis of patients. 18 Osteoporosis is an important risk factor for postoperative fusion failure, pseudoarthrosis, and revision surgeries. 19 Therefore, perioperative anti-osteoporosis treatment is gaining increasing attention. Common anti-osteoporosis drugs include calcium + vitamin D, teriparatide, and bisphosphonates. 20 Studies have shown that patients with osteoporosis who received teriparatide after lumbar fusion surgery had higher fusion rates, better clinical outcomes, and lower adverse event rates compared with placebo and bisphosphonate groups. 21 However, teriparatide has a relatively long treatment cycle and is expensive, limiting its widespread clinical use. Bisphosphonates can specifically bind to hydroxyapatite on the bone surface, especially in areas with active bone resorption, thereby inhibiting osteoclast function, inducing apoptosis of mature osteoclasts, and reducing bone remodeling. Although bisphosphonates’ efficacy is not as high as that of teriparatide, they have achieved certain therapeutic effects in treating osteoporosis.12,21 However, bisphosphonates have more contraindications, and patients may experience more noticeable adverse reactions such as fever and renal impairment during the acute phase of treatment. 22 Denosumab is a monoclonal antibody anti-osteoporosis drug that specifically binds to the receptor activator of nuclear factor-κB ligand (RANKL) in humans, reducing the formation of the RANKL-RANK complex and directly inhibiting the formation, function, and survival of osteoclasts. Studies have shown that denosumab can increase bone density in various skeletal sites. 23 Additionally, it has been demonstrated that the combination of denosumab and teriparatide is more effective in increasing bone density than teriparatide alone and can significantly accelerate spinal fusion.21,24 Although denosumab is widely used in the treatment of osteoporosis, there are few studies on its use in the perioperative period, especially regarding its effects on promoting intervertebral fusion when used alone. Therefore, we conducted this study to explore the impact of denosumab on spinal fusion after MIDLF surgery and to compare the effects of denosumab and bisphosphonates on MIDLF outcomes.
Objects and Methods
Eligibility Criteria
This study is a prospective cohort study (registered with Chinese Clinical Trial Registry ChiCTR2100054769) and has been approved by the Biomedical Ethics Committee of our center (Approval No. 2022-744). The study followed the STROBE guidelines and adhered to the principles of the Declaration of Helsinki, and informed consent was obtained from all patients. 25
Subjects
Clinical and radiological data of patients diagnosed with osteopenia or osteoporosis who underwent MIDLF surgery from May 2021 to October 2022 at our center were prospectively collected.
Inclusion Criteria
1. Patients with lumbar degenerative diseases (lumbar disc herniation, lumbar spinal stenosis, or degenerative lumbar spondylolisthesis) with lumbar or leg pain or impaired walking function affecting the quality of life; 2. Patients who had undergone at least 3 months of conservative treatment with poor outcomes and required surgical intervention; 3. Patients aged ≥50 years; 4. Patients with osteopenia (−2.5 < T-score < −1.0) or osteoporosis (T-score ≤ −2.5) confirmed by dual-energy X-ray absorptiometry (DXA).
Exclusion Criteria
1. Patients with severe cardiovascular or pulmonary diseases contraindicating surgery; 2. Patients who had used anti-osteoporosis drugs for ≥3 months preoperatively; 3. Patients with glucocorticoid-induced osteoporosis due to long-term use of corticosteroids; 4. Patients with a history of previous lumbar surgery; 5. Patients with lumbar trauma, tumor, or infection; 6. Non-menopausal women (Menopausal status was recorded for all women to ensure that all women in the study population were menopausal); 7. Patients with contraindications to anti-osteoporosis drugs, such as hypocalcemia or renal insufficiency.
Grouping Design
This study employed a computer-generated random sequence to ensure randomness. Participants were randomly assigned to the denosumab group, the bisphosphonate group, and the control group using the Sequentially Numbered, Opaque, Sealed Envelopes (SNOSE) method.
All patients provided informed consent for the observational study. The same experienced spinal surgeon performed MIDLF surgeries for all patients in the study population. Postoperatively, all patients were allowed to use analgesics, muscle relaxants, and neurotrophic drugs. The specific treatments according to our center’s perioperative anti-osteoporosis protocol are as follows: 1. Denosumab Group: A single subcutaneous injection of 60 mg denosumab on postoperative day 5 or before discharge, along with daily oral administration of 600 mg calcium and 800 IU vitamin D. 2. Bisphosphonate Group: A single intravenous infusion of 5 mg zoledronic acid on postoperative day 5 or before discharge, along with daily oral administration of 600 mg calcium and 800 IU vitamin D. 3. Control Group: Daily oral administration of 600 mg calcium and 800 IU vitamin D.
Both denosumab and bisphosphonate treatments were administered only once, while calcium and vitamin D were continued until the end of the follow-up period. Drug safety was assessed based on the occurrence of adverse events, and all patients, regardless of whether they experienced adverse reactions or not, were included in the final analysis, and relevant adverse events were recorded.
Operating Procedure
All the patients in this study underwent midline lumbar fusion (MIDLF) performed by the same experienced spinal surgeon, using the cortical bone trajectory (CBT) screw fixation technique & a PEEK cage filled with bone graft.
Data Collection
Basic Characteristic Data
Age, gender, height, weight, body mass index (BMI), comorbidities (hypertension, diabetes, coronary heart disease), ASA classification, preoperative bone density, hospital stay duration, number of surgical segments, surgical time, bone metabolism indicators (serum PINP and CTX), Oswestry Disability Index (ODI), and Visual Analog Scale (VAS) were collected through the electronic medical record system.
Imaging Data
Bridwell Four-Point Scale for Intervertebral Assessment

Note: For multi-planar/multi-level evaluations, the lowest grade was used to determine the final classification.
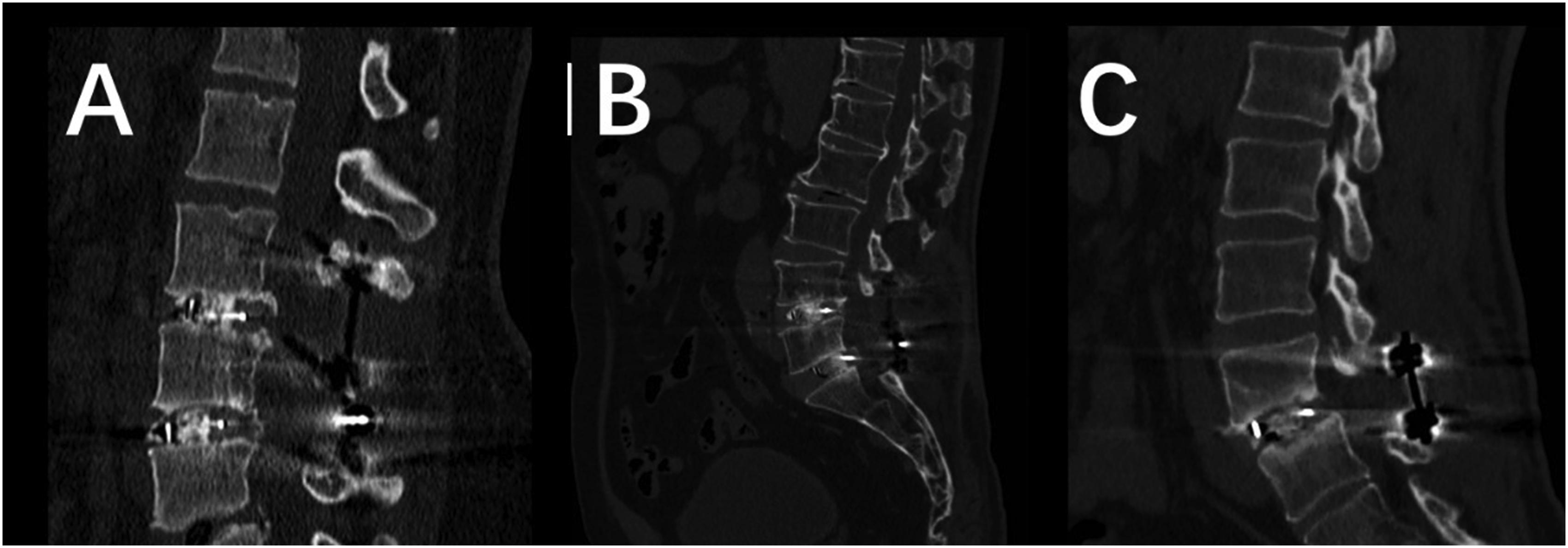
CT Imaging Results of Some patients: Figure (A) shows the CT Image at 1-Year Follow-Up of a 45-Year-old Female Who Underwent MIDLIF Surgery at the L3-5 Levels, With a Grade 1 Fusion at the L3-4 Segment and a Grade 2 Fusion at the L4-5 Segment. Figure (B) Shows the CT Image at 6-Month Follow-Up of a 68-Year-old Male Who Underwent MIDLIF Surgery at the L4-S1 Levels, With Grade 3 Fusion at Both the L4-S1 Segments. Figure (C) Shows the CT Image at 6-Month Follow-Up of a 55-Year-old Male Who Underwent MIDLIF Surgery at the L4-5 Level, With No Evidence of Fusion, Indicating a Grade 4 Fusion
Statistical Methods
1. All data were processed and statistically analyzed using SPSS 27.0 (IBM Corporation, Armonk, NY, USA). Two-sided tests were used, and P < 0.05 was considered as the level of significance. 2. Shapiro-Wilk test was used to test the normal distribution of continuous variables. The continuous variables with normal distribution were expressed as mean ± standard deviation, and ANOVA was used to test the comparison between multiple groups. The data not conforming to normality and homogeneity of variance were tested by non-parametric test (Kruskal-Wallis test). 3. The ordinal variables were tested by non-parametric test (Kruskal-Wallis test). When the non-parametric test showed statistical difference, the Bonferroni method was used to correct the multiple comparisons. 4. The categorical variables were expressed as numbers or proportions, and the chi-square test or Fisher’s exact test was used.
Results
Enrollment and Baseline Characteristics of the Enrolled Population
A total of 54 patients (29 males and 25 females, mean age 63.7 ± 8.4 years) were included in this study. According to the postoperative medication, the patients were divided into 3 groups: there were 17 cases in denosumab group, aged 50-74 years; there were 17 patients in the bisphosphonate group, aged 52-80 years; there were 20 cases in the control group, aged 50-78 years. During the follow-up, a total of 4 patients were lost to follow-up, including 3 patients in the bisphosphonate group due to personal reasons and 1 patient in the control group due to severe postoperative heart failure. Thus, 17 patients (mean age, 64.9 ± 8.9 years) in the denosumab group, 14 patients (mean age, 66.6 ± 7.1 years) in the bisphosphonate group, and 19 patients (mean age, 62.5 ± 8.2 years) in the control group completed 6 months of follow-up and had a complete data set for analysis. The last follow-up time was from 1 year to 3 years after surgery due to COVID-19, and 12 patients in denosumab group, 8 patients in bisphosphonate group and 9 patients in control group completed the last follow-up. The flow chart of patient enrollment is shown in Figure 2. Flow Chart of Study Enrollment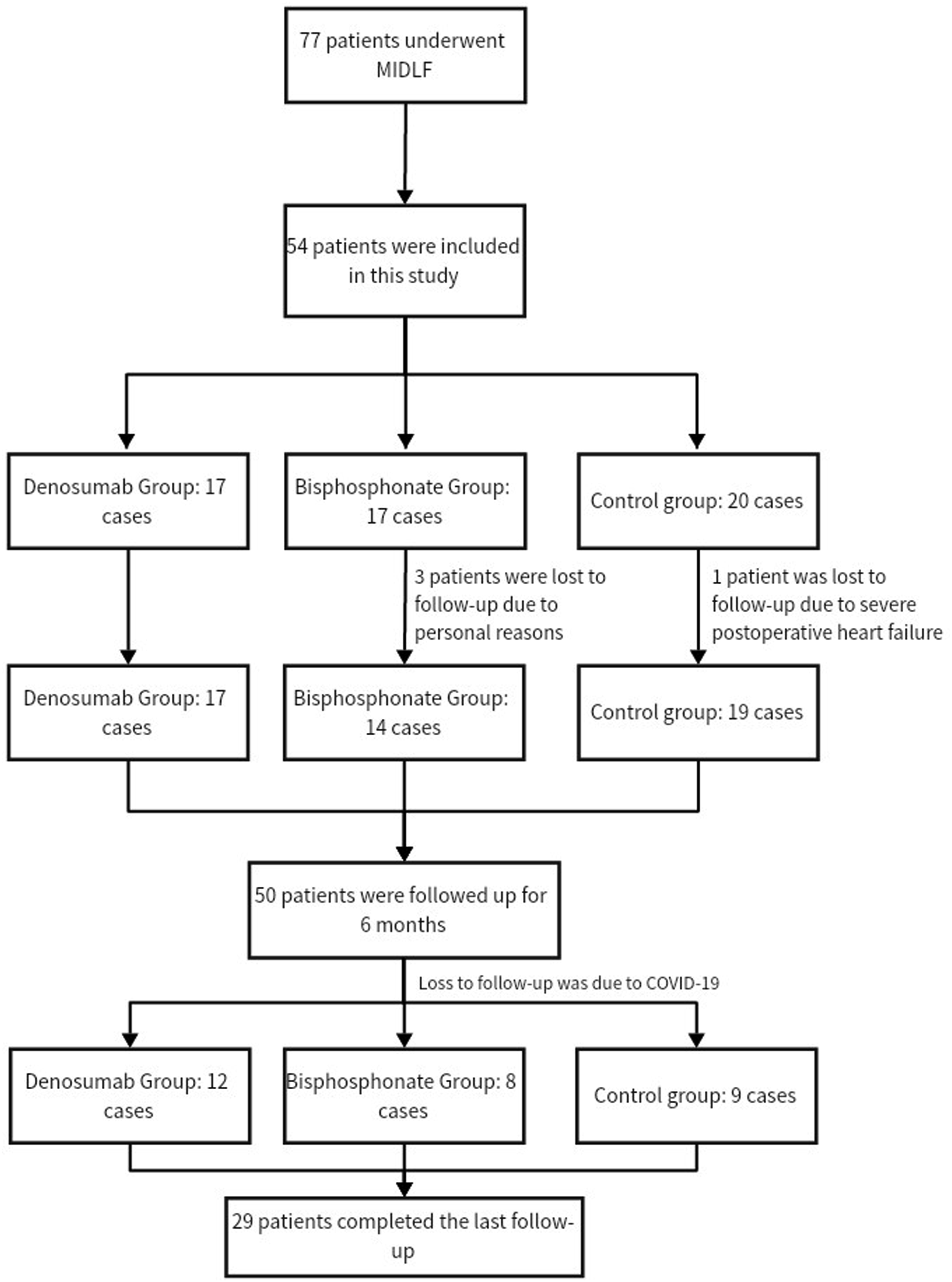
Baseline Data of Enrolled Patients
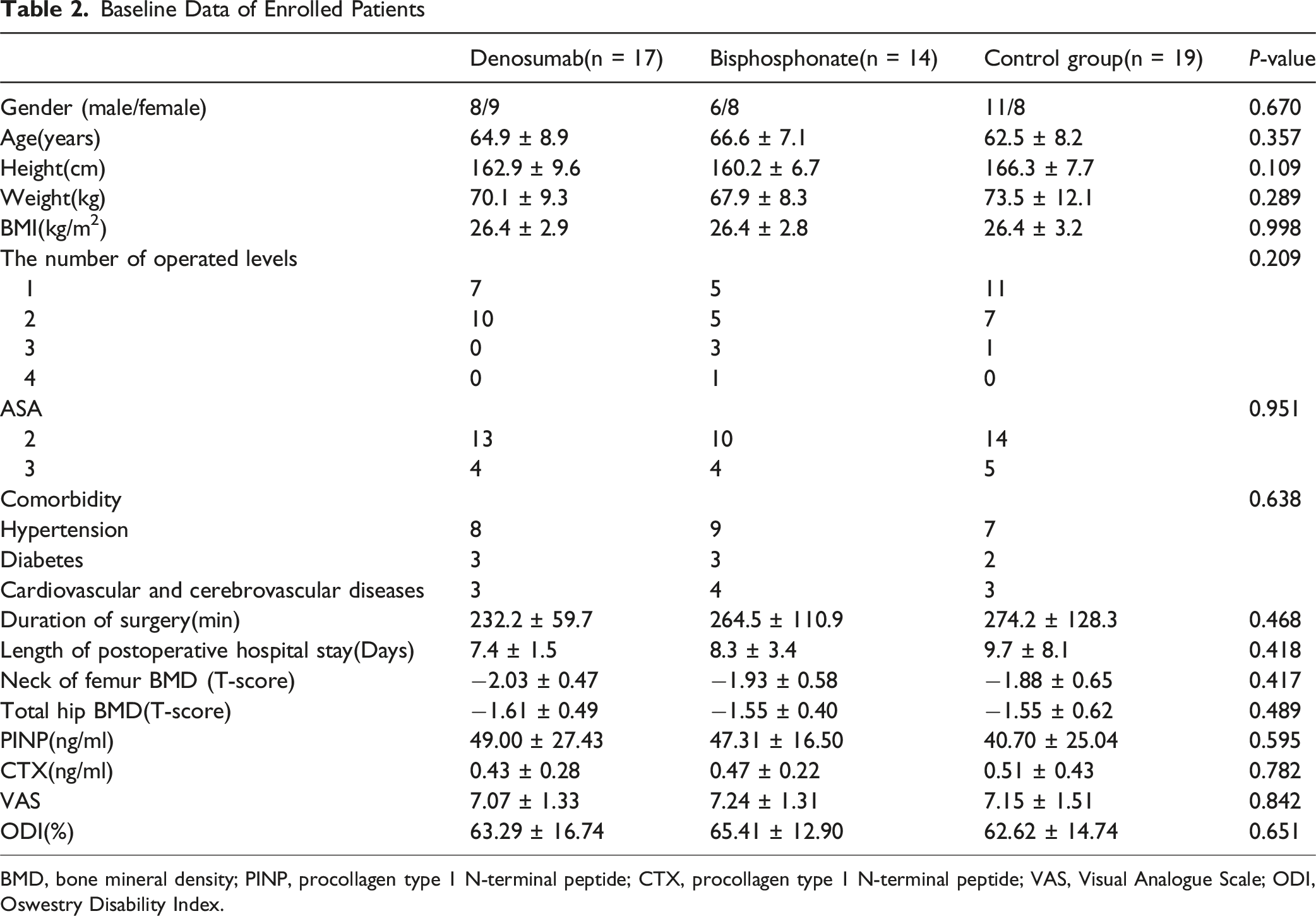
BMD, bone mineral density; PINP, procollagen type 1 N-terminal peptide; CTX, procollagen type 1 N-terminal peptide; VAS, Visual Analogue Scale; ODI, Oswestry Disability Index.
Imaging Evaluation
Radiological assessment of bone fusion was performed using postoperative X-ray and CT slices in the sagittal and coronal planes, with results shown in Figure 3. At 6 months postoperatively, the complete fusion rate was 25% and the partial fusion rate was 68.6% in the denosumab group; the complete fusion rate was 25% and the partial fusion rate was 75% in the bisphosphonate group; while in the control group, the complete fusion rate was 10.5%, the partial fusion rate was 57.9%, and the non-fusion rate was 31.6%. At 6 months postoperatively, a Kruskal-Wallis test was performed to compare the fusion status distribution among denosumab, bisphosphonate, and control groups. The results showed a significant difference in fusion status across the 3 groups (H = 14.54, df = 2, P < 0.001). Subsequently, Dunn’s test with Bonferroni correction was conducted for pairwise comparisons. All pairwise comparisons demonstrated statistically significant differences (P < 0.001, corrected α = 0.0167): denosumab group vs control group (Z = −15.72), denosumab group vs bisphosphonate group (Z = −26.34), and control group vs bisphosphonate group (Z = −13.31). The bisphosphonate group had the highest rank sum, followed by the denosumab group, and then the control group, indicating that both denosumab and bisphosphonate significantly improved fusion status compared with the control group, but the difference between the 2 drug groups was not obvious. Distribution of Bone Fusion Grades Among the 3 Groups: (A) Distribution of Fusion Grades at Six Months Postoperatively. (B) Distribution of Fusion Grades at the Last Follow-Up. Panels a and B Show that Patients Treated With Denosumab and Bisphosphonates had Significantly Higher Bone Fusion Rates than the Control Group at Six Months and the Last Follow-Up, while There was No Significant Difference Between the Two Drug Groups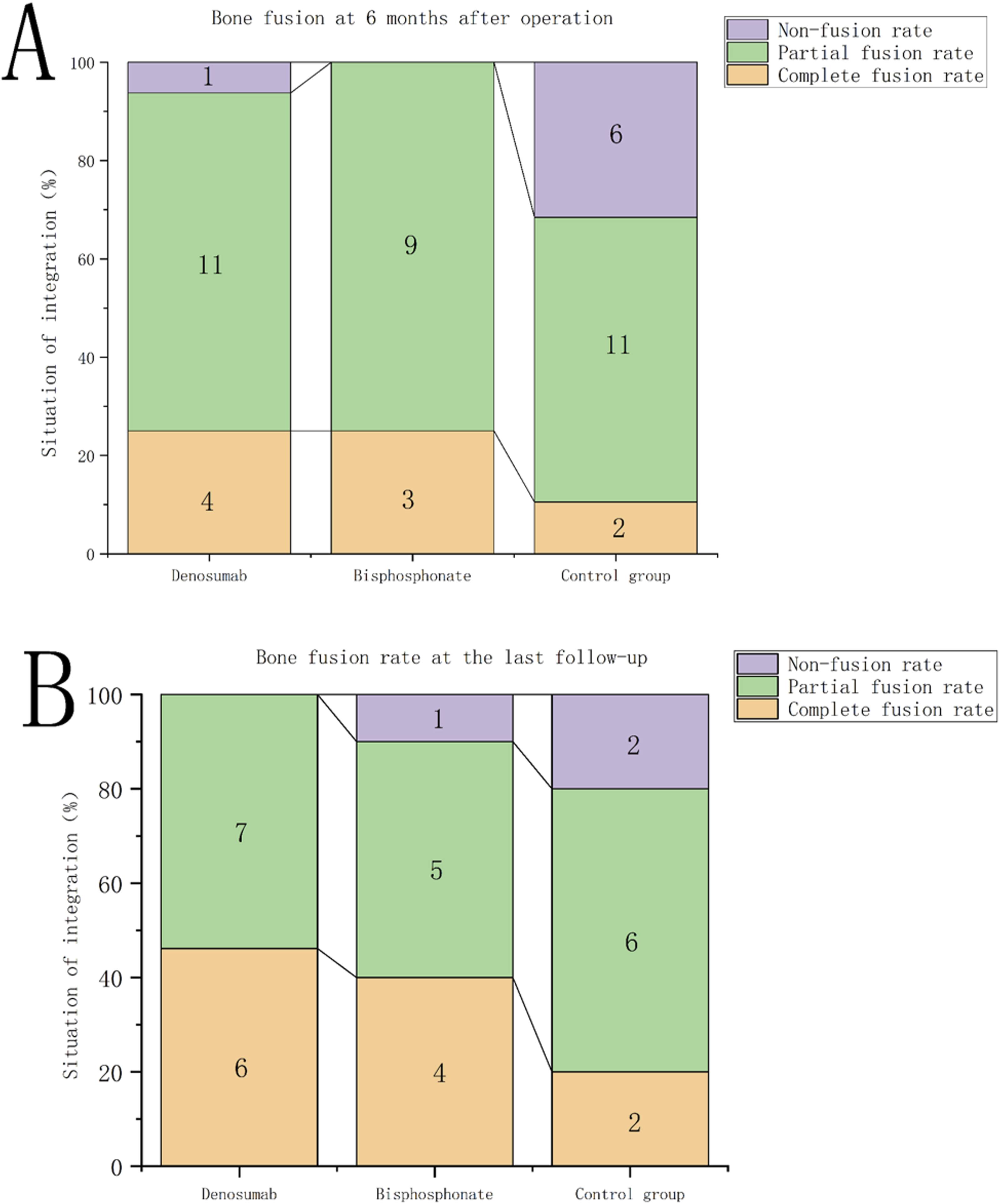
At the last follow-up (12-36 months postoperatively), complete fusion was observed in 5 of 12 patients (42%) in the denosumab group, 3 of 8 (38%) in the bisphosphonate group, and 2 of 9 (22%) in the control group. Partial fusion was noted in 7/12 (58%), 4/8 (50%), and 5/9 (56%) respectively.
Clinical Outcomes
The VAS and ODI scores of the denosumab group, the control group and the bisphosphonate group decreased significantly at 1, 3 and 6 months after operation compared with those before operation, and there was no significant difference in VAS and ODI scores among the 3 groups (P > 0.05). Postoperative clinical pain and dysfunction were improved in all 3 groups, but there was no difference in the improvement of pain and dysfunction among the 3 groups, proving that denosumab and bisphosphonates did not significantly promote the improvement of postoperative pain and dysfunction. (Figure 4) Trend Plot of VAS and ODI Scores: (A) VAS Scores in Denosumab Group, Control Group and Bisphosphonate Group at 1, 3 and 6 months after Surgery. (B) ODI Scores of Denosumab Group, Control Group and Bisphosphonate Group at 1, 3 and 6 Months after Surgery. (A, B) Show that the Postoperative Clinical Pain and Dysfunction of the Three Groups Were Improved Compared With Those before Surgery, but There was No Significant Difference Between the Three Groups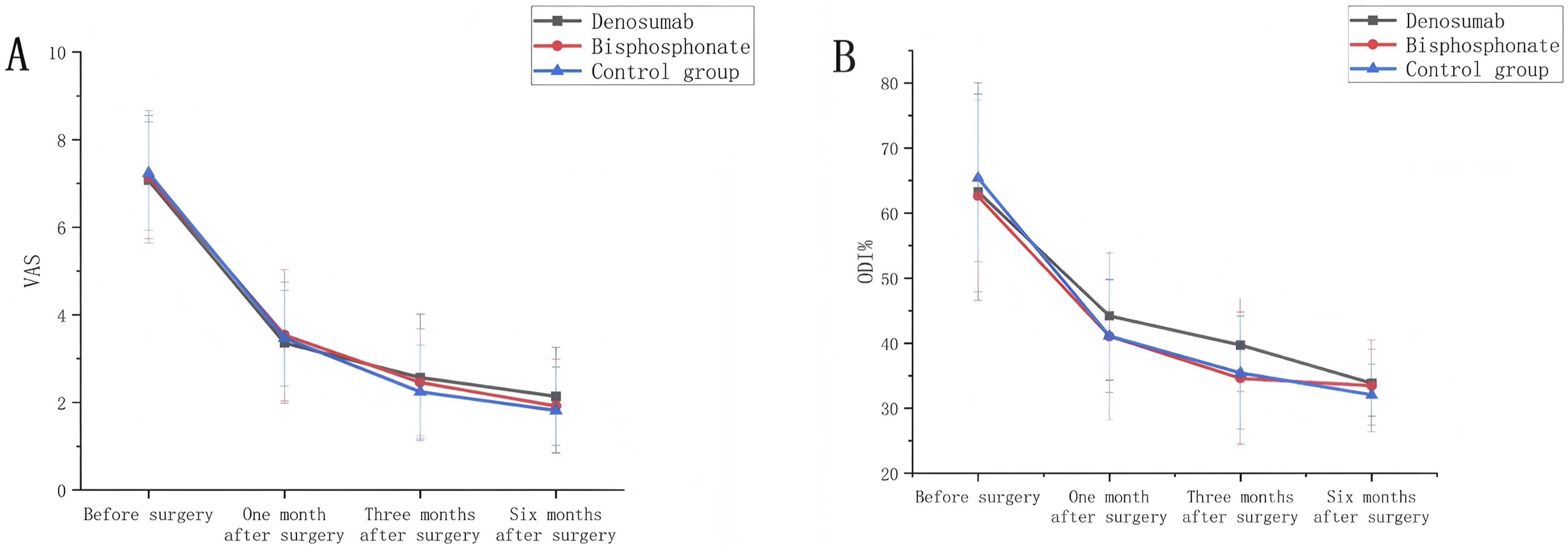
Bone Metabolism Indexes
Preoperative serum levels of PINP and CTX showed no significant differences among the 3 groups (P = 0.595 and 0.782, respectively). As shown in Figure 5, the control group exhibited significantly higher serum PINP and CTX levels at all time points except baseline (P < 0.05). Trend of Bone Metabolic Indicators: (A) Trend of Serum PINP. (B) The Change Trend of Serum CTX. Except before Surgery, the Serum PINP and CTX in the Control Group Were Higher than Those in the Denosumab and Bisphosphonate Groups at all Time Points. Excluding the Effect of Surgery on Bone Metabolism Indexes, the Serum PINP and CTX in the Bisphosphonate Group Showed a Trend of Decreasing after Surgery and Maintaining a Balance, while the Serum PINP and CTX in the Denoumab Group Showed a Rebound Trend after Decreasing and Maintaining a Balance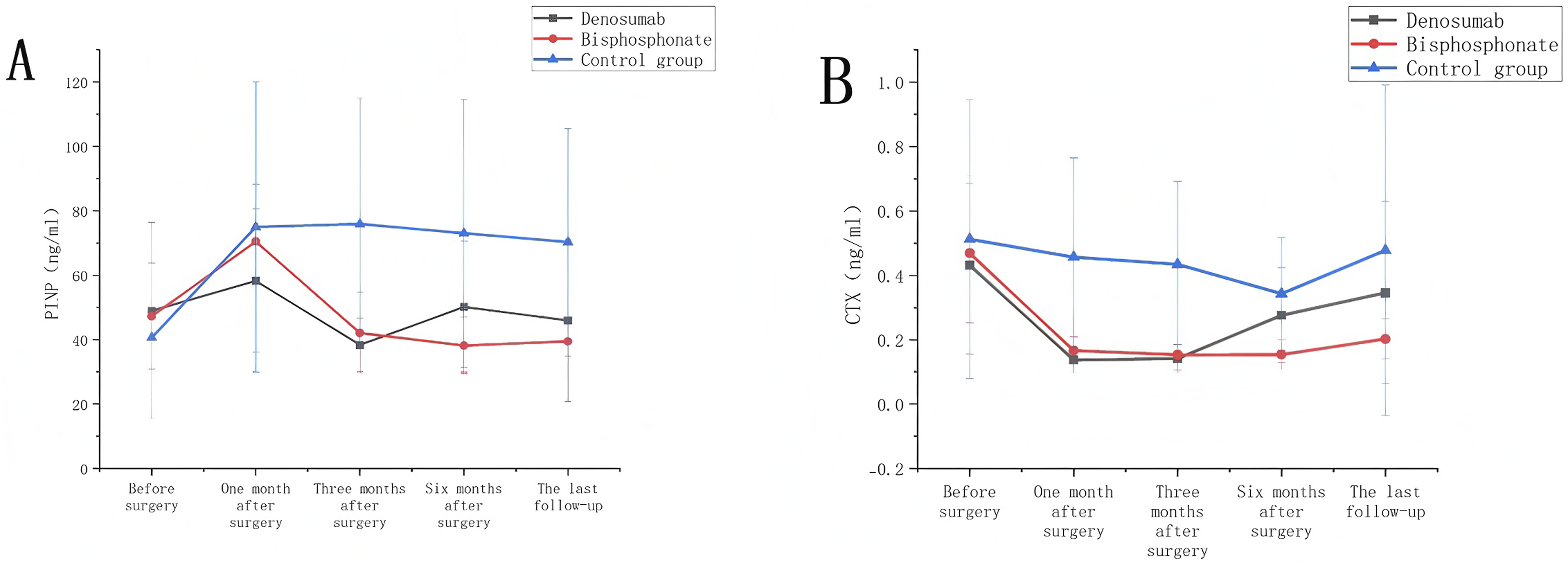
At 1 month postoperatively, although the reduction in serum PINP did not differ significantly between the denosumab group, bisphosphonate group, and control group (P > 0.05), both active treatment groups showed significantly greater reductions in serum CTX than the control group (P < 0.05), with no intergroup difference in CTX reduction (P > 0.05). By 3 months postoperatively, the denosumab and bisphosphonate groups demonstrated significantly greater reductions in serum PINP compared with the control group (P < 0.05), while serum CTX levels remained stable (the fluctuation range is less than 10%).
During the subsequent 6-month follow-up, serum PINP and CTX levels remained stable in the bisphosphonate group, whereas a rebound increase trend was observed in the denosumab group.
Postoperative Complications
Regarding postoperative complications, the number of patients experiencing fever after treatment with bisphosphonates was significantly higher than in the denosumab group (10 vs 2, P = 0.004) and the control group (10 vs 4, P = 0.011), with an average fever duration of 2.3 days, all presenting as low-grade fever. Notably, no cases of tachycardia, nausea, myalgia, vertebral fracture, cage subsidence, or pseudoarthrosis were observed in any of the 3 groups during the follow-up period. Only one case of screw breakage and displacement was observed in the bisphosphonate group at the last follow-up, which required a secondary surgery. In the control group, 1 patient experienced postoperative wound issues, requiring readmission for treatment, diagnosed with wound effusion without evidence of infection on microbiological examination.
Discussion
This study employed a prospective cohort design to compare the efficacy of perioperative denosumab, bisphosphonates, and conventional calcium and vitamin D treatment in patients with osteopenia or osteoporosis undergoing midline lumbar fusion (MIDLF) surgery. The results showed that at 6 months postoperatively, the fusion rates in the denosumab and bisphosphonate groups were significantly higher than those in the control group (P < 0.01), while there was no significant difference between the 2 drug groups (P = 0.999). Additionally, the postoperative fever rate in the denosumab group was significantly lower than that in the bisphosphonate group (11.8% vs 71.4%, P = 0.004). The results are consistent with our hypothesis, indicating that compared with conventional calcium and vitamin D treatment, both denosumab and bisphosphonates can significantly increase the early bone fusion rate in patients after lumbar surgery, and the 2 treatment regimens are equally effective in promoting bone fusion.
Perioperative anti-osteoporosis treatment is of crucial importance for patients with osteopenia or osteoporosis undergoing spinal surgery. Previous studies have shown that patients with osteoporosis face a significantly increased risk of screw loosening, displacement, and pseudoarthrosis after lumbar fusion surgery, which directly affects the long-term outcomes and recovery process. 4 Enhancing screw purchase or increasing vertebral bone density can significantly reduce these risks. Moreover, long-term use of anti-osteoporosis drugs has been proven to effectively prevent or slow bone loss. However, the efficacy and safety of perioperative anti-osteoporosis drugs still require further investigation. Therefore, identifying a safer and more effective anti-osteoporosis drug for the perioperative period of lumbar fusion surgery is an important direction for current research.
In accordance with the latest guidelines for optimizing perioperative skeletal health, it is recommended that anti-resorptive or anabolic medications be administered for at least 3-6 months preoperatively to optimize bone quality and enhance surgical success rates. However, considering that such a treatment regimen may delay the surgical date and potentially exacerbate patient symptoms, this study opted to administer denosumab and bisphosphonates within 1 week postoperatively. This approach meets the actual clinical treatment needs and makes the study protocol easy to implement and generalize in clinical practice.
In this study, both denosumab and bisphosphonates significantly increased bone fusion rates through their anti-osteoporosis effects, which is consistent with previous finding.13,28,29 Previous studies have also shown that perioperative use of anti-osteoporosis drugs can significantly increase bone fusion rates and postoperative stability, reducing the incidence of complications.12,21 For example, Oba et al 28 found that bisphosphonates can significantly increase bone fusion rates after lumbar fusion surgery and reduce the incidence of postoperative complications. However, the use of bisphosphonates in the perioperative period is often associated with adverse reactions such as postoperative fever and renal impairment.22,30
In this study, the postoperative fever rate in the bisphosphonate group was significantly higher than that in the denosumab and control groups (71.4% vs 11.8% vs 23.5%, P = 0.004 and P = 0.011), which may be related to the acute-phase reaction caused by bisphosphonates. Denosumab directly inhibits the differentiation and maturation of osteoclasts by reducing the formation of the RANKL-RANK complex, thereby reducing bone resorption. Its mechanism of action enables it to exert significant anti-osteoporosis effects in the early postoperative period, reducing the risk of screw loosening in lumbar surgery, increasing bone fusion rates, and having fewer adverse reactions. 23 In contrast, bisphosphonates have a slow onset of action and may also be associated with more adverse reactions. 22 Although studies have indicated that the anti-resorptive effects of bisphosphonates and denosumab on bone remodeling may delay fracture healing in the early stages, fracture healing primarily relies on the natural bone repair process. In contrast, postoperative spinal fusion depends on the mechanical stability provided by the surgery and osteoconductive materials. Therefore, by reducing bone resorption and enhancing the stability of the implants (eg, increasing the pullout strength of screws, thereby improving the fixation effect of the implants), bisphosphonates and denosumab can indirectly promote bone fusion after spinal fusion surgery.
Additionally, bone metabolism indicators showed that both denosumab and bisphosphonates significantly inhibited bone resorption, but the inhibitory effect of denosumab peaked at 3 months postoperatively and then gradually weakened, while the inhibitory effect of bisphosphonates was more sustained but may lead to long-term adverse reactions.31,32 Notably, previous studies have shown that denosumab treatment can significantly increase lumbar bone density, with effects superior to those of bisphosphonates.33,34 In this study, the CTX levels in the denosumab group significantly decreased at 1 and 3 months postoperatively, indicating a significant inhibitory effect on bone resorption (P < 0.001), while the CTX levels in the bisphosphonate group also significantly decreased at all postoperative time points, albeit with relatively smaller changes. This characteristic gives denosumab a significant advantage in perioperative management, especially in patients requiring rapid improvement in bone quality, such as those undergoing lumbar surgery. Moreover, the anti-resorptive effect of denosumab can be rapidly reversed after discontinuation, leading to a rebound phenomenon. Therefore, it is recommended to use denosumab postoperatively on a long-term or even lifelong basis to maintain its anti-osteoporosis effects. 33 Early increases in lumbar bone density, higher bone fusion rates, increased screw pullout strength, and reduced risks of screw loosening and displacement are all benefits associated with denosumab use.
The significance of this study lies in providing a safer and more effective perioperative anti-osteoporosis treatment option, especially for patients undergoing MIDLF surgery, where denosumab may be a better choice. By increasing bone fusion rates and reducing postoperative complications, denosumab has the potential to improve patients’ postoperative recovery and long-term outcomes. This finding is of great clinical value for optimizing perioperative management, increasing surgical success rates, and enhancing patient satisfaction. However, this study also has limitations: the small sample size and single-center may introduce bias; the short follow-up period does not allow for the assessment of long-term complications and drug effects; and the impact of the pandemic meant that only one drug injection was administered, precluding the evaluation of the full treatment cycle’s effects; few patients completed the last follow-up and the follow-up time was inconsistent, resulting in limited statistical power of the last follow-up data, the long-term efficacy needs to be verified by further research. Some of the fusions in this study (Bridwell grades 2-3) may progress to complete fusion or degenerate into pseudo-joints over time. Due to the insufficient follow-up period, it is impossible to rule out the possibility of incomplete healing in the long term or to overestimate the long-term efficacy of the drugs. Future studies should employ large-sample, multicenter, randomized controlled designs with extended follow-up periods to explore efficacy differences among various patient groups and surgical methods while also considering patient adherence and quality of life to comprehensively assess the clinical value of anti-osteoporosis drugs.
Conclusion
During the perioperative period of MIDLF in patients with reduced bone mass or osteoporosis, denosumab and bisphosphonates both significantly increase the early bone fusion rate and the effects are comparable. However, denosumab has fewer adverse reactions such as postoperative fever and is safer. Given the limitations of the study, its long-term efficacy needs to be further verified, but the short-term results support denosumab as an optimized treatment option.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is granted by National High Level Hospital Clinical Research Funding (Scientific Research Seed Fund of Peking University First Hospital) (No. 2023HQ05) and Clinical Special Research Fund of the Chinese Association of Medical Education (No. 2022-006).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
