Abstract
Study Design
Prospective, randomized controlled study.
Objective
Assess early radiological outcomes in transforaminal lumbar interbody fusion (TLIF) with 3D-printed porous titanium (3DPPT) compared to PEEK.
Methods
Single-blinded prospective, randomized controlled trial comparing 1-2 level TLIF with micro- & nano-textured, 3DPPT vs PEEK cages from 11/2021 to 5/2023. Interbody fusion was assessed on CT at 6 months according to Brantigan and Steffee method, modified to describe the Fraser definition of locked pseudoarthrosis [(BSF) scale]. Primary outcome was radiographic fusion at 6 months.
Results
Initial study protocol included 70 total patients but was ended early given the significance on interim analysis. Amongst 17 patients with 25 interbody levels implanted, 10 3DPPT and 15 PEEK cages were implanted. 3DPPT levels had a significantly higher rate of successful fusion (BSF-3) at 6 months compared with PEEK (100% vs 0.0%, P < 0.001). The posterior probability that 3DPPT increased the odds of fusion versus PEEK was > 99.9%, indicating a near-certain beneficial effect. Using a Bayesian mixed-effects model, the predicted probability of 6-month BSF-3 fusion was 9.0% for PEEK and 91.2% for 3DPPT. There were no significant differences in lumbar pathologies, level of fusion, number of fused levels, cage height, length of hospital stay, surgery duration, postoperative complications, subsidence, or reoperations.
Conclusions
The rate of successful lumbar interbody fusion at 6 months was significantly higher in 3DPPT levels compared to PEEK. 3DPPT may accelerate the rate and quality of bony fusion. Additional studies are needed to further delineate the impact of these radiographical findings on long-term clinical outcomes.
Introduction
Lumbar interbody fusion is a common surgical procedure used to treat various degenerative spine pathologies including spinal stenosis, spondylolisthesis, and degenerative disc disease. An interbody cage serves to restore spinal alignment and intervertebral/foraminal height, provide anterior column support with bone grafting matrix facilitating osseointegration between vertebral bodies, and reduce screw loosening or hardware failure. 1 Failure of spinal fusion, pseudarthrosis, remains a challenging complication influenced by factors such as age, smoking status, number of levels operated, and implant material and design. Subsidence, which occurs when the interbody cage sinks into the adjacent endplates, is a distinct complication that may alter segmental alignment and potentially compromise fusion, although successful fusion can still occur despite subsidence. 2
As technology advances, interbody engineering utilized various materials and designs conferring distinct properties that enhance mechanical stability or rigidity, decrease the elastic modulus and stress shielding, and/or facilitate osteointegration.3,4 Titanium implants offer high durability and strength profiles as well as superior biocompatibility demonstrating cellular adhesion and thus increased osseointegration. 5 However, conventional titanium implants have a high elastic modulus which may lead result in higher rate of subsidence. 6 Polyetheretherketone (PEEK) is an alternative to titanium with a lower elastic modulus similar to trabecular bone and has been associated with lower subsidence rates in some studies. 7 PEEK is also radiolucent allowing for accurate assessment of radiographical fusion. 8 However, PEEK is hydrophobic and biologically inert, which poses limitations on osseointegration potential and increasing the risk of pseudoarthrosis. 9
Although macroscale properties including interbody material impact fusion potential, microscale properties including surface topography and porosity may have a significant impact by catalyzing osteoblastic differentiation, osteoid synthesis, and mineralization.10,11 The porous design and nano-textured titanium surface facilitate osteointegration while also decreasing the elastic modulus to that of physiologic bone.12,13 Current literature is relatively limited regarding studies, particularly class 1 evidence, directly comparing a 3D-printed porous titanium (3DPPT) with unique surface topography and PEEK cages for lumbar interbody fusion with several being animal or nonclinical studies. 14 In this prospective, randomized controlled study, we investigate the efficacy of early lumbar interbody fusion using either PEEK or 3DPPT cages in TLIF.
Methods
Study Design
This single-blinded prospective, randomized controlled superiority trial was conducted at the Ohio State University Medical Center evaluating the safety and efficacy of 1-2 level TLIF with a micro- & nano-textured, 3DPPT interbody compared to a representative PEEK cage currently used in routine fashion for lumbar interbody fusion procedures. IRB approval was obtained, and the study was registered with the ClinicalTrials.gov database (registration no. NCT05182489). Demographic, clinical, and radiographic data were prospectively recorded. Randomization was performed in a 1:1 ratio and dichotomized by smoking status given its association with increased rates of pseudoarthrosis. Patients were blinded to their group status for the duration of study assessments and procedures. Investigators were blinded to group status until the time of cage implantation to minimize potential bias.
Study Participants
Screening was performed on consecutive patients eligible for a 1-2 level TLIF to undergo placement of either 3DPPT or lordotic PEEK cages at each level in a 1:1 ratio. All patients who met inclusion criteria and agreed to participate in this study signed informed consent prior to any study specific procedures. Inclusion criteria was defined as adult patients >18 years of age scheduled to undergo combined interbody and posterolateral spinal fusion has been unresponsive to conservative care for a minimum of 6 months. Exclusion criteria included prior lumbar arthrodesis, inadequate tissue coverage over the operative site, open local wound, rapid joint disease, bone absorption, osteoporosis, condition requiring medications that may interfere with bone or soft tissue healing (i.e., glucocorticoids, immunosuppressants), active infection, metal/foreign body sensitivity, body mass index (BMI) > 45, history of substance abuse, worker’s compensation, prisoners, and less than 6 month follow-up or failure to complete 6 month follow-up CT scan. Data collection was performed on baseline demographics, medical and surgical history, physical examination, pre-operative imaging studies, and current medications within 60 days of the surgery date. A current smoker was defined as any smoking within 6 months of the surgery date, while a nonsmoker was defined as never smoking or quitting at least 6 months before surgery. Data collection was also performed on surgical details, complications, and intra-and post-operative imaging.
Interbody Characteristics
All participants were randomized to either the Adaptix micro- and nano-textured, 3DPPT interbody or CAPSTONE PEEK cage (Medtronic, Minneapolis, Minnesota, USA) (Figure 1). These implants were chosen based on matched macro properties: shape, modulus of elasticity, and volume for osseous ingrowth. However, the 3DPPT cage incorporates a micro- and nano-textured surface topography and porosity designed to mimic cancellous bone and enhance osseous integration.12,13 All implants were 10 mm in width. All constructs were supplemented with a titanium pedicle screw system. (A) Adaptix micro- and nano-textured, 3D-printed porous titanium interbody; (B) Capstone polyetheretherketone interbody.
Surgical Protocol
An open midline approach was performed followed by laminectomies and bilateral foraminotomies, facetectomies, and posterolateral pedicle screw insertion. Randomization was performed at the time of surgery following endplate preparation and determination of cage height based on tactile feedback and radiographic findings. A 50:50 mixture of autograft and allograft using milled local autograft bone and GRAFTON DBM DBF were packed within the cages and posterolateral bilaterally. Cages were inserted via the more symptomatic side with positioning guided by intraoperative fluoroscopy and confirmed with postoperative radiographs prior to discharge. Titanium rods were then positioned and posterior elements compressed to achieve segmental lordosis.
Radiographic Assessment
A radiographic analysis was performed independently by 2 surgeons evaluating fusion status and potential complications. Each subject underwent AP, lateral and flexion/extension radiographs at the 3, 6, 12, and 24-month visits as well as a CT scan at 6 months to assess device/graft integrity including migration or subsidence, hardware fracture, and fusion consolidation progress. Fusion was assessed on 6-month follow-up CT according to Brantigan and Steffee as modified to describe the Fraser definition of locked pseudoarthrosis (BSF) scale (Figure 2) as below:11,15-17 • BSF-1: radiographic pseudarthrosis potentially demonstrating construct collapse, loss of disc height, vertebral slip, displacement of the cage, or significant bone resorption. • BSF-2: radiographic locked pseudarthrosis with lucency visible within the cage despite solid bone growth from vertebral endplates into the cage periphery. • BSF-3: radiographic fusion with solid bone bridges within at least half of the fusion area. Brantigan and Steffee as modified to describe the Fraser definition of locked pseudoarthrosis (BSF) scale. (A) BSF-1: radiographic pseudarthrosis; (B) BSF-2: radiographic locked pseudarthrosis; (C) BSF-3: radiographic fusion, presenting bone bridges within at least half of the fusion area with at least the density originally achieved at surgery.
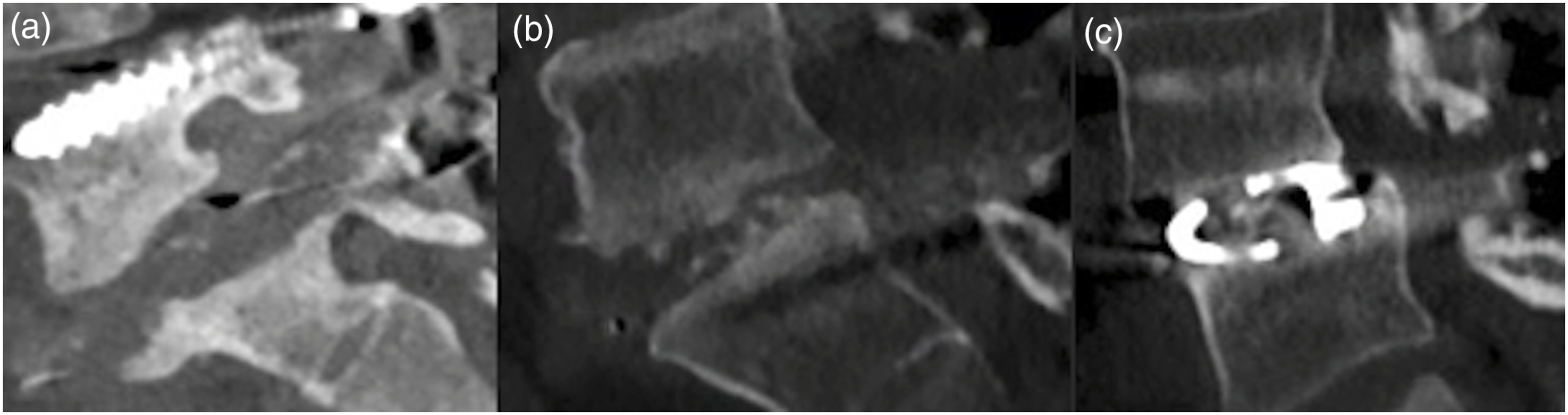
Outcomes
The primary outcome of this study was interbody fusion success at 6-months postoperatively corresponding to early osseointegration and fusion. Successful radiographic fusion was defined BSF-3, while failure was defined by radiographic pseudarthrosis (BSF-1 or -2). Procedural safety was a secondary outcome and analyzed according to clinical and radiographic complications.
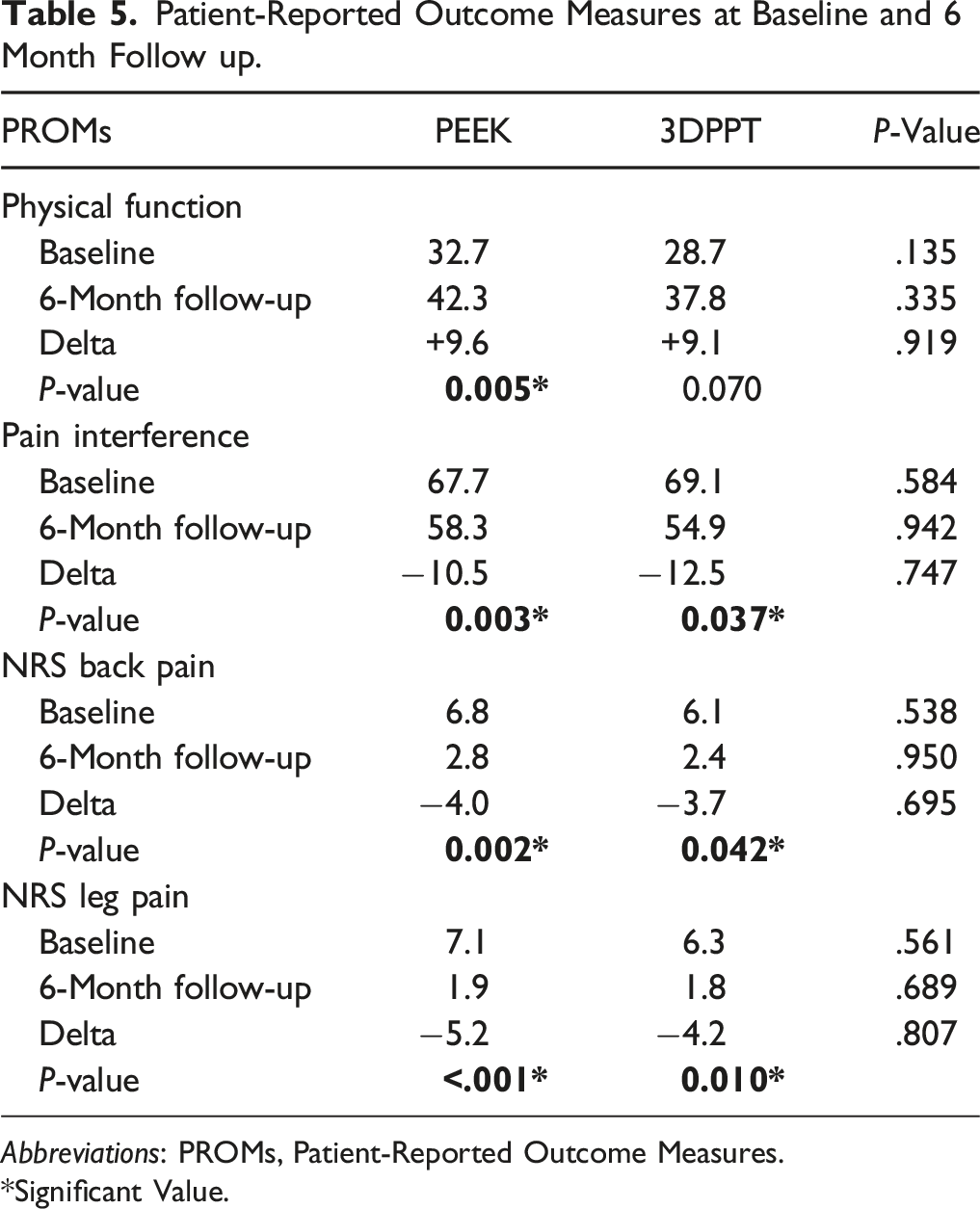
Several parameters were assessed postoperatively as secondary outcome measures. Procedural safety was assessed via complication rates including subsidence, hardware failure, and new neurological deficits, as well as rate of reoperation. Additional radiographic parameters including lumbar lordosis, segmental lordosis, disc height (anterior, middle, and posterior), and foraminal height were also evaluated. Patient reported outcome measures (PROMs) were collected at baseline and 6-month follow-up. This included PROMIS questionnaire physical function and pain interference as well as Numeric Rating Scale (NRS) back and leg pain.
Statistical Analysis
A total sample size of 70 cages was calculated with a power of 0.8 and alpha of 0.05 based on the assumption of 90% and 60% fusion rates in the 3DPPT and PEEK levels, respectively. These assumptions were based on retrospective review of radiographic results. Interim analysis yielded statistically significant results, therefore enrollment ceased as it was unlikely for investigators to maintain clinical equipoise in continuing patient randomization. Analysis was performed using unpaired t test, χ2, Fisher’s exact tests, as appropriate. Data normality was determined using a Shapiro-Wilk test. Non-normal data was analyzed using a Mann-Whitney U test. A Bayesian mixed-effects logistic regression model was used to estimate the association between implant type and probability of successful fusion given the data separation. Fixed effects included patient implant type, age, and BMI. Random effects model was used to account for within-patient variability and multiple observations in 1 patient. The model was fit using Hamiltonian Monte Carlo Sampling and and an R Bayesian regression model package (version 4.3.3). Convergence was assessed using
Results
Our prospective randomized controlled study included 17 patients with 25 interbody cages implanted in 1-2 level transforaminal lumbar interbody fusion for degenerative disease from 11/2021 to 5/2023: 10 patients with 15 PEEK cages and 7 patients with 10 micro- & nano-textured, 3DPPT cages. Initial study protocol included 70 total patients; however, the study was ended early given the significance of the interim results. A comparative analysis was performed between the 3DPPT and PEEK implant levels.
Baseline Characteristics
Baseline Demographics.
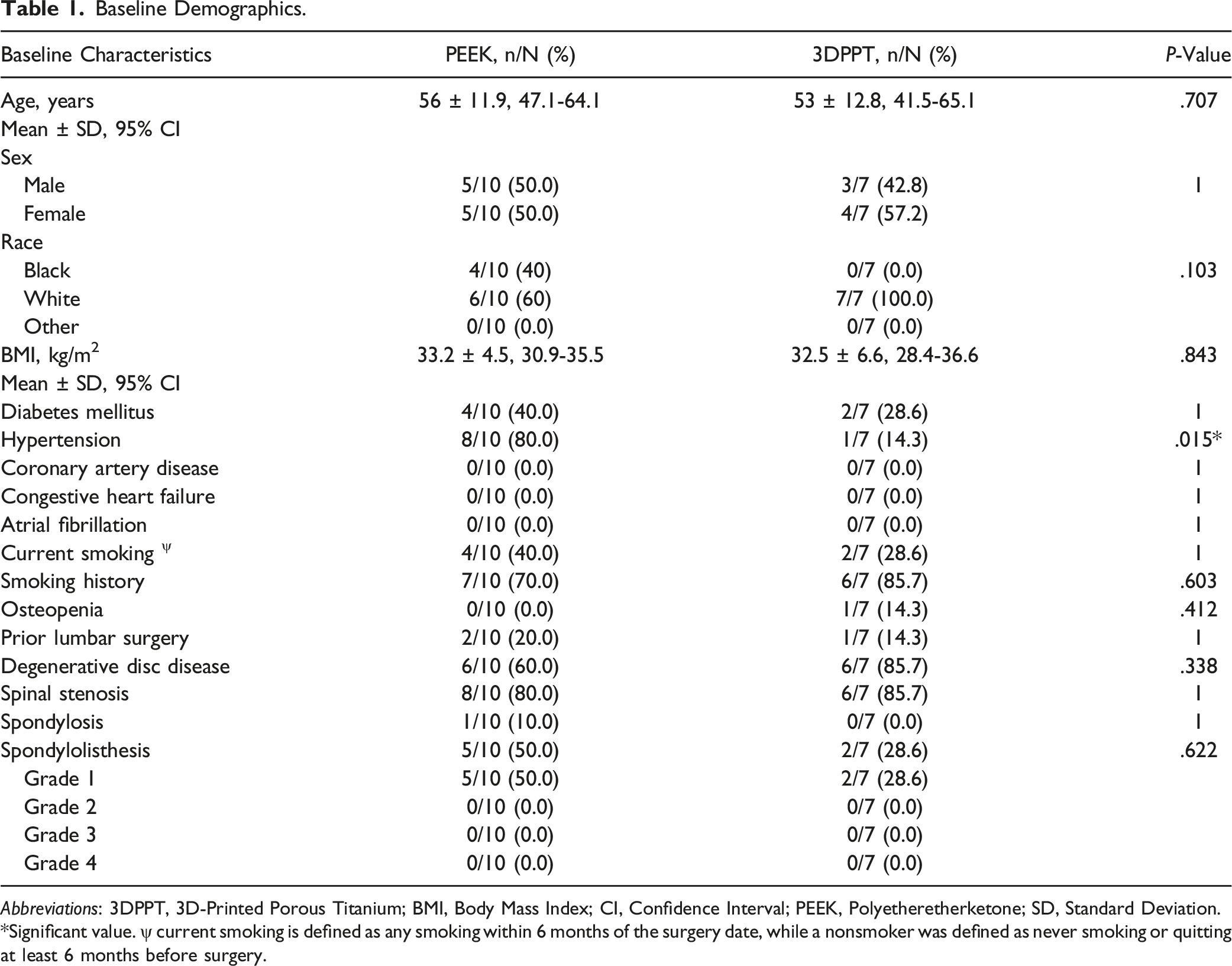
Abbreviations: 3DPPT, 3D-Printed Porous Titanium; BMI, Body Mass Index; CI, Confidence Interval; PEEK, Polyetheretherketone; SD, Standard Deviation.
*Significant value. ψ current smoking is defined as any smoking within 6 months of the surgery date, while a nonsmoker was defined as never smoking or quitting at least 6 months before surgery.
Surgical Details
Surgical Details Presented Based on Number of Interbody Levels.
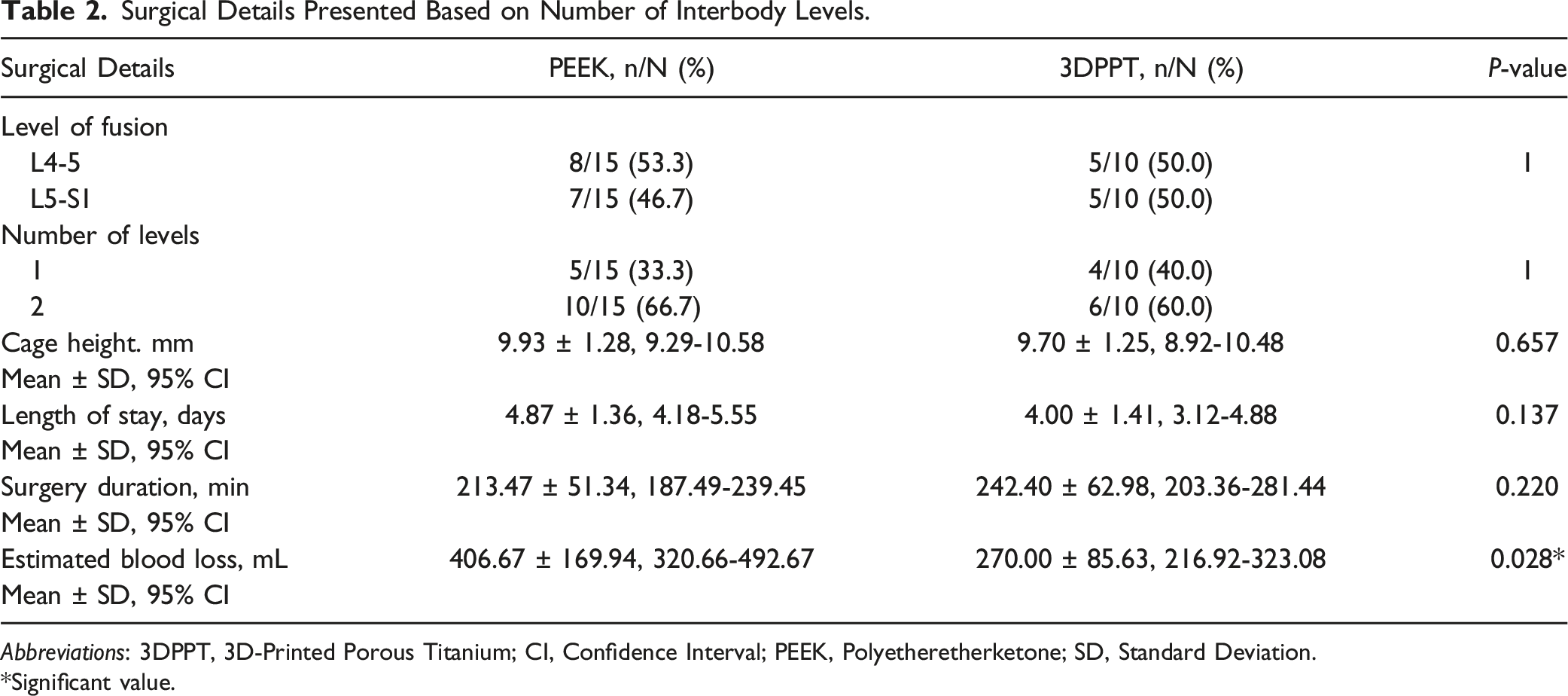
Abbreviations: 3DPPT, 3D-Printed Porous Titanium; CI, Confidence Interval; PEEK, Polyetheretherketone; SD, Standard Deviation.
*Significant value.
Radiographic Outcomes
Radiographic Outcomes at 6 Months.
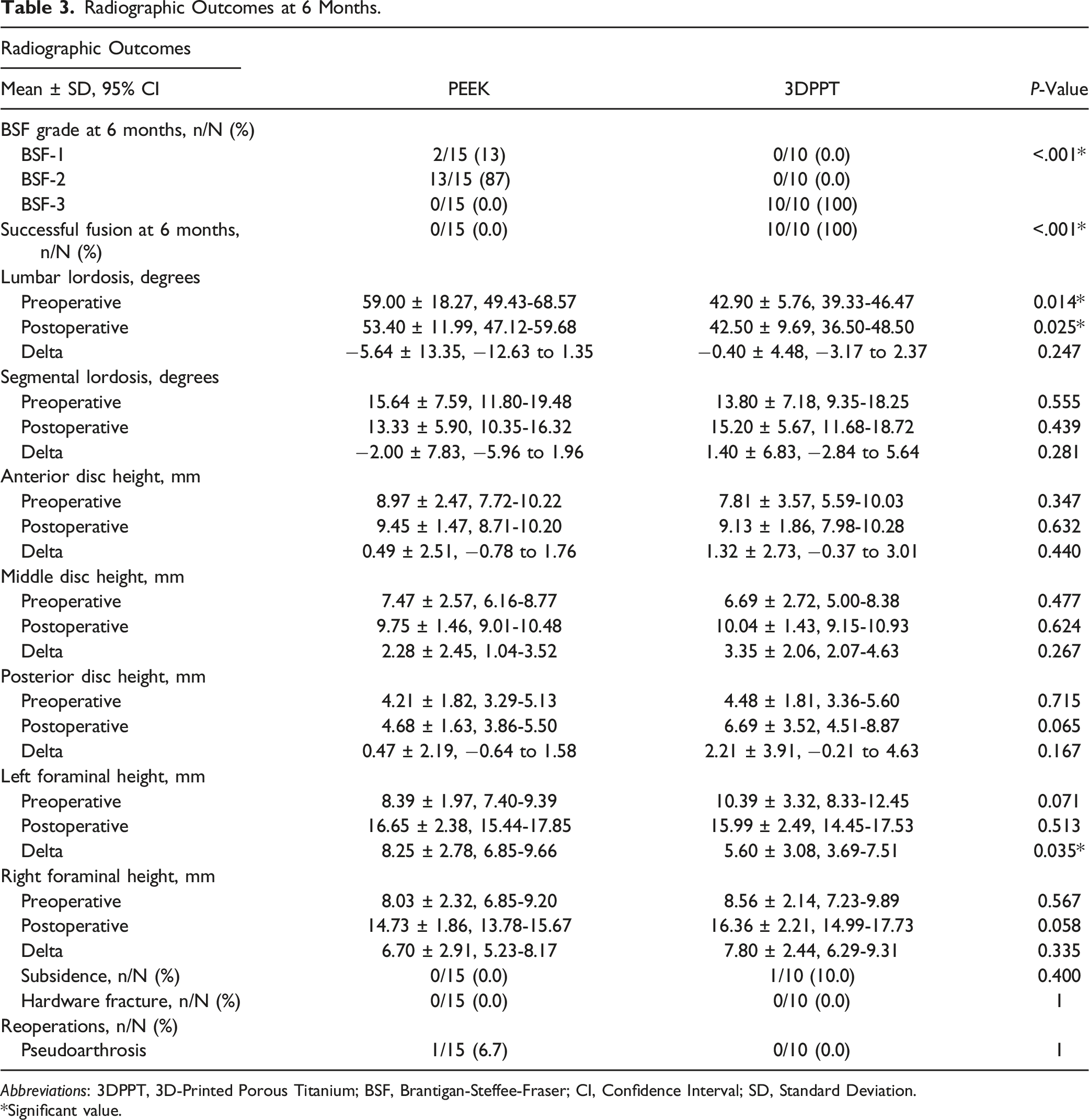
Abbreviations: 3DPPT, 3D-Printed Porous Titanium; BSF, Brantigan-Steffee-Fraser; CI, Confidence Interval; SD, Standard Deviation.
*Significant value.
Bayesian Mixed-Effects Model
Bayesian Regression Model for Predictors of Successful Fusion.

Abbreviations: SD, Standard Deviation of Random Patient Intercept.
*Significant value.
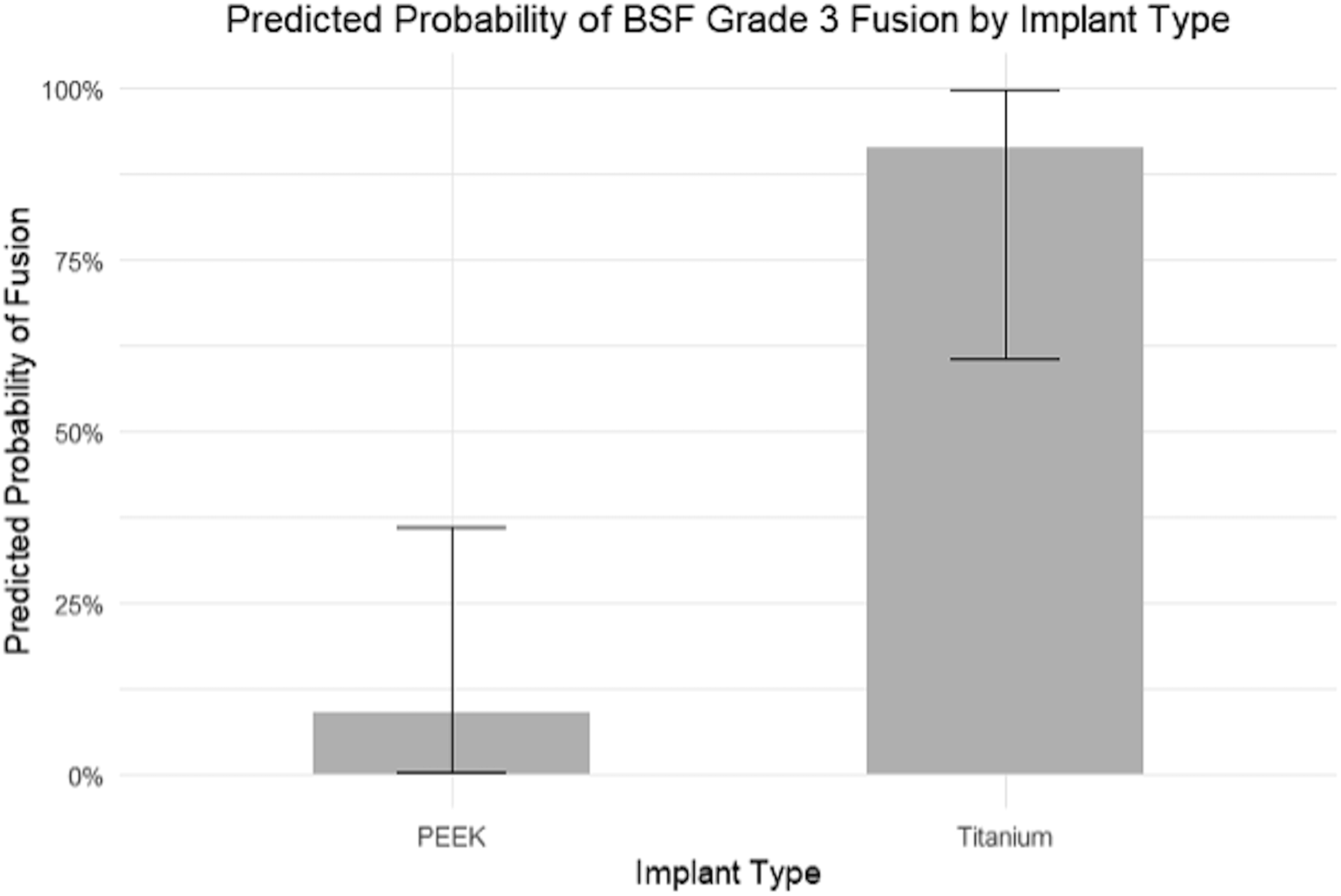
3DPPT implants were associated with significantly higher odds of fusion compared to PEEK (OR = 384.6, 17.5-12,680). The posterior probability that titanium increased the odds of fusion relative to PEEK was >99.9%, indicating near-certain evidence of a beneficial effect. Neither age (OR = 0.93, 0.76-1.16) nor BMI (OR = 0.93, 0.58-1.45) showed a significant association with fusion in this model (Figure 3). The predicted probability of 6-month BSF-3 fusion was 9.0% (0.3-36.1%) for PEEK implants and 91.2% (60.4-99.8%) for 3DPPT implants, based on the Bayesian mixed-effects model (Figure 4). The estimated standard deviation of the patient-level random effects 1.11, reflecting moderate heterogeneity in baseline fusion probability across individuals. Posterior odds ratios with 95% credible intervals for predictors of successful fusion from a Bayesian mixed-effects model. Odds ratios are shown on a log scale. *Predictor with a posterior probability >0.999 for the observed effect direction. Predicted probability of BSF grade 3 fusion at 6 months by implant type, based on a Bayesian mixed-effects logistic regression model. Error bars represent 95% credible intervals.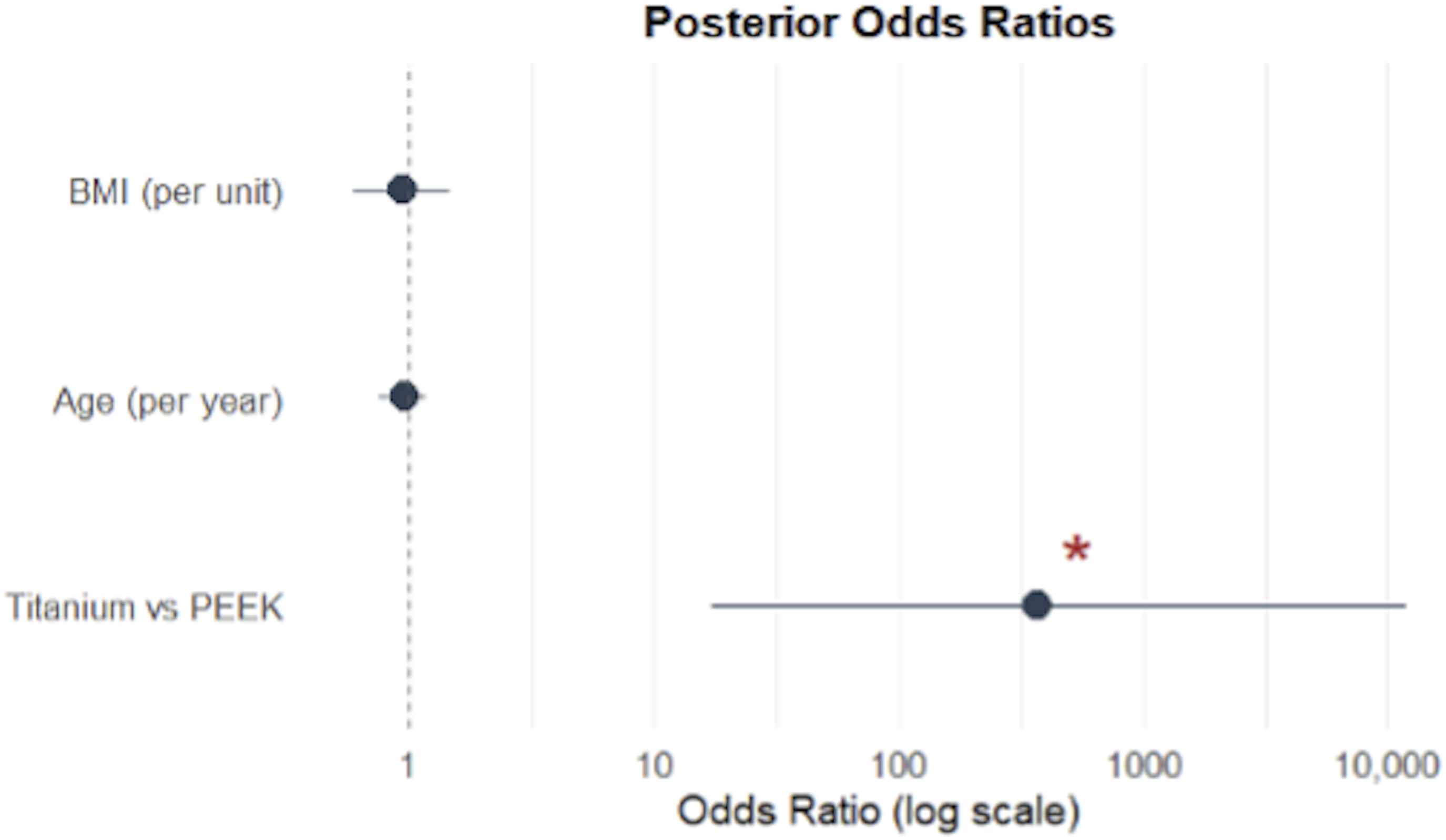
Patient Reported Outcome Measures
Patient-Reported Outcome Measures at Baseline and 6 Month Follow up.
Abbreviations: PROMs, Patient-Reported Outcome Measures.
*Significant Value.
Discussion
Findings Summary
In the present prospective randomized controlled study, we aimed to assess the efficacy and safety of 1-2 level TLIF using a micro- & nano-textured, 3DPPT interbody compared to PEEK. The rate of lumbar interbody fusion at 6 months was significantly higher in 3DPPT levels (100% vs 0.0%, P < .001). Given the statistically significant difference in fusion rates at 6 months on early evaluation, the study was stopped prematurely. Otherwise, there was no significant difference in baseline demographics or surgical details including complications. Although the estimated blood loss was higher in the PEEK group, this difference may be attributable to random variation given the small sample size and standardization of the procedure. Therefore, the observed EBL difference should be interpreted with caution. TLIF using the 3DPPT implants displayed a significant improvement in patient reported outcomes at 6-month follow-up. However, PROMs at 6-months and the extent of improvement did not significantly differ from the PEEK group. Our results suggest the efficacy of lumbar interbody fusion, assessed by successful interbody fusion at 6 months, with a 3DPPT implant is superior to PEEK. Procedural safety, assessed according to complication and reoperation rates, was similar. However, failure of spine fusion can contribute to complications and disability requiring future revision surgeries,18,19 which suggests a superior safety profile with 3DPPT. To our knowledge, this is one of the few studies providing class 1 evidence for improved early fusion with 3DPPT implants during lumbar interbody fusions.
Interbody Fusion & Implant Design Considerations
Failure of spinal fusion is associated with increased postoperative complication risk including pseudoarthrosis and instrumentation failure such as screw loosening or subsidence, which are associated with poor clinical outcomes and may necessitate revision surgeries.18,19 Subsidence is often the result of stress shielding on interbody adjacent vertebral bodies and associated with loss of sagittal plane correction as well as neuroforaminal/intervertebral height loss.14,20 Spinal fusion is dependent on several variables such as patient comorbidities and healing capacity, spine mechanical stability, and material properties associated with intervertebral fusion devices. 21 The addition of lumbar interbody cages to posterolateral instrumentation improved fusion rates as opposed to posterolateral fusion alone,22,23 yet there are still several variables to consider, in addition to biocompatibility and load carrying capacity, when engineering an interbody to optimize fusion rates. 24 The implant’s elastic modulus should be similar to cortical bone (3-30 GPa) yet allow for radiographical assessment of fusion (ideally radiolucent). 25 It should also have a high level of osteogenic tissue support with surface modifications that optimize bone on- and in-growth.26-31
Titanium vs PEEK
Titanium and PEEK are common interbody materials; however current evidence supports superior fusion rates with titanium implants. Biomaterial studies have demonstrated deficient osteoblast integration with PEEK implants secondary to inhibited osteoblast differentiation and increased inflammatory reactions induced by macrophages and lymphocytes as well as poorly vascularized fibrous connective tissue (fibrous union).32-34 The hydrophobic nature of PEEK also contributes poor bone-implant bonding and osteointegration. 35
As opposed to biologically inert PEEK, 36 titanium promotes osteointegration and osteoblast osteogenic factor secretion (BMP2, alkaline phosphatase, osteocalcin) as well as improved bone-implant interface cell adhesion, therefore potentially inducing a more substantial and faster rate of bony fusion.30,33,37 However, the high elastic modulus (50-110 GPa) of conventional titanium cages may result in stress shielding at the bone-implant interface and pseudoarthrosis or subsidence in accordance with Wolff’s law.20,30,31,35 Studies have reported subsidence rates associated with conventional titanium cages up to 24.9-75% due to the significant elastic modulus mismatch.38,39
3D-Printed Porous Titanium
Advances in 3D printing technology enable the development 3DPPT cages improving osteointegration and decreasing complications by varying porosity (directly correlates with elastic modulus), strut widths, and surface modifications.34,40,41 The titratable porous lattice enables greater load sharing and confers an elastic modulus (2.-4.6 GPa) similar to that of cancellous/trabecular bone (0.02-2 GPa) or PEEK,24,42 thereby significantly reducing the stress shielding effect and resultant subsidence to an incidence lower than that of PEEK. 43 Additionally, porosity enhances osteocyte adhesion and ingrowth as well as implant-bone interface osteointegration, 44 therefore increasing bone-implant mechanical stability and improving fusion capacity.21,34 McGilvray et al 34 assessed bony ingrowth on histologic sections in an ovine lumbar fusion model comparing PEEK, plasma sprayed porous titanium-coated PEEK, and 3DPPT. 3DPPT demonstrated superior osteointegration with increased osteocyte remodeling and complete bony filling without the fibrous union seen with PEEK and titanium-coated PEEK implants. Laratta et al 45 even demonstrated superior osseointegration in 3DPPT without bone graft compared to PEEK with autologous iliac crest bone graft at 4-week and 8-week follow-up. A biomechanical analysis also demonstrated the potential for 3DPPT to increase osteointegration and decrease subsidence risk. 46 Additionally, increased porosity also increases the degree of radiolucency enabling a more accurate assessment of bony fusion. 47
In a 2024 meta-analysis including 460 patients comparing 3DPPT vs PEEK, 3DPPT was associated with a significantly higher fusion rate (RR: 1.24, 95% CI [1.02, 1.50], P = .03) and grade 1 fusion (RR: 2.51, 95% CI [1.30, 4.87], P = .006) at 6-month follow-up. Subsidence rate in the 3DPPT group was also significantly lower (RR: 0.61, 95% CI [0.42, 0.88], P = .009). There was also a trend towards lower complication and reoperation rates in the 3DPPT group, however, did not reach statistical significance. 48 In a 2023 randomized controlled study, Toop et al 11 provided the first class 1 evidence supporting enhanced early fusion with activated titanium cages in TLIF surgeries at 6-months. Our results further support the findings in the aforementioned meta-analysis as all patients in our 3DPPT group demonstrated successful fusion at 6 months including the single patient that demonstrated cage subsidence. No patient in our PEEK group had successful fusion at 6 months and one required revision for pseudoarthrosis. Our study is limited by long-term radiographic and clinical follow-up as not enough patients had 12-month follow-up to perform any meaningful statistical analyses. However, in a retrospective study, Kim et al 49 reported no difference in fusion rate at 12 months yet 3DPPT demonstrated a significantly higher quality or grade 1 fusion rate at 12 months (77.5% vs 51.2%, P = .013). In patients completing 12-month follow-up, Khan et al 50 also found that 3DPPT cages were associated with significantly increased fusion rates (90% vs 73%, P = .003), decreased subsidence (23.5% vs 40.2 %, P = .010), better ODI scores at both 3 and 12 months (P = .013 and 0.048, respectively), and decreased indirect costs associated with surgery (P = .006) compared to a propensity-matched PEEK cohort.
Additional Adaptix Interbody-Specific Design
The Adaptix footprint is similar to that of the well-known PEEK Capstone Spinal System. Both systems utilize lateral windows which enable radiographic visualization of bony fusion, and the open volume design enable continuous graft bridging from endplate to endplate. Unlike open cages, Adaptix has a central honeycomb structure which may minimize stress load on adjacent endplates and decrease subsidence as well as increase bone-implant interface surface area acting as an osteoconductive scaffold. The surface topography of Adaptix is a key distinguishing feature compared to other 3D-printed titanium cages. Adaptix created via a proprietary acid etching process to form nano-, micro-, and macro-textures onto a roughened titanium surface. This surface topography has been shown to facilitate early attachment, migration, and differentiation of human mesenchymal stem cells and therefore more rapid osteointegration than prior technologies. 51 However, further clinical studies are needed to validate whether these design characteristics result in improved long-term clinical outcomes.
Preliminary Results & Future Direction
A micro- & nano-textured, 3DPPT interbody may be a potential solution combating the limitations of PEEK (biocompatibility, osteointegration, pseudoarthrosis) and conventional titanium (subsidence, stress shielding). The rate of successful lumbar interbody fusion at 6 months was significantly higher in the 3DPPT group compared to PEEK (100% vs 0.0%, P < .001). Safety profile in this study was similar, however the only patient requiring reoperation was a result of pseudoarthrosis in the setting of a PEEK cage. A 2024 meta-analysis also showed significantly lower subsidence rates (P = .009) and a trend towards lower complication and reoperation rates in the 3DPPT group. 48 Additional studies are needed to further delineate these observed radiographical findings and determine long-term clinical implications including cost-modeling are needed. Continued advances in 3D printing engineering and computational modeling have the potential to enable patient-specific implants customized to bone density and surface morphology. Although such technology exists, it has not yet been widely implemented likely due to the time- and cost-effectiveness profile of such an endeavor. 14 Additionally, although increasing porosity increases load sharing capacity, 46 structural integrity may be compromised beyond a porosity threshold of 70%. 41 In the current state of material science and 3D-printing technology, surpassing this porosity threshold (implant mass-to-volume ratio) results in thinner struts that are more susceptible to buckling and degradation. 41
Limitations
The limitations of this study include the small sample size and lack of longer-term patient follow-up, which limits conclusions regarding long-term clinical and radiographic outcomes. Clinical and patient-reported outcomes were recorded. However, meaningful statistical analyses were limited given the sample size. This study was not powered to detect a statistically significant difference in complication or reoperation rates, particularly given their relatively low incidence. 95% confidence intervals are reported for descriptive purposes to illustrate variability in group differences and should be interpreted with caution given the small sample size. Although 3DPPT interbodies may be associated with higher initial costs, increased fusion rates have the potential to reduce rates of pseudoarthrosis and subsequent reoperation. However, a cost-effectiveness analysis was beyond the scope of this study. Additionally, fusion assessment cannot be blinded to implant type, which may introduce potential bias in the evaluation of fusion outcomes. Future studies with larger sample sizes and extended follow-up periods will be essential to corroborate these preliminary results and explore potential differences in long-term complication, reoperation rates, and functional benefit. This was a single-center study of lumbar interbody fusions performed at a high-volume academic center; therefore, our data may lack generalization.
Conclusion
The rate of successful lumbar interbody fusion at 6 months was significantly higher in the 3DPPT group compared to PEEK. 3DPPT may accelerate the rate and quality of bony fusion and is associated with a low subsidence and reoperation rates. Additional studies are needed to further delineate the impact of these radiographical findings on long-term clinical outcomes.
Footnotes
Authors Contributions
Conception or design of the work: JHW, AG. Data acquisition and analysis: JHW, SK, WK, NR. Interpretation of data: JHW, AG, SV, DX, SK. Drafting the work: JHW, SK, NR. Revising the work for valuable intellectual content: AG, SV, DX, SK, BL. Final approval of the version: JHW, AG.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Xu receives consulting support from NuVasive, Providence Medical Technologies. Dr Grossbach and Dr Viljoen receive consulting and research support from Medtronic and Nuvasive. The other authors have no personal, financial, or institutional interest in any of the drugs, materials, or devices described in this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medtronic provided funding support for the costs to conduct the study only. Medtronic otherwise did not participate in this study.
Clinical Trial Registration
Ethical Statement
Data Availability Statement
The relevant anonymized patient level data are available on reasonable request from the authors.
