Abstract
Study Design
Retrospective cohort study.
Objective
To investigate whether cefazolin can be given to penicillin-allergic patients undergoing spine surgery without significantly increasing rates of allergic reactions. Additionally, we sought to determine if patients receiving cefazolin would be at increased risk of surgical site infection due to allergy-associated skin or wound reactions.
Methods
Patients with a documented penicillin allergy who underwent lumbar fusion from 2017-2022 and received cefazolin for surgical prophylaxis were matched to a cohort of non-penicillin allergic patients. Length of surgery and in-hospital complications related to infections were recorded for each patient. Allergic reactions were recorded, in addition to all-cause mortality, hospital length of stay (LOS), 30 and 90 day readmissions due to infectious etiologies and need for irrigation and debridement (I&D) surgery.
Results
254 penicillin-allergic patients were matched to 762 non-allergic patients. Penicillin- allergic patients had a comparable LOS when compared to non-allergic patients and had comparable incidences of respiratory complaints, sepsis, cellulitis, wound infections and all-cause mortality. While one penicillin-allergic patient was diagnosed with a mild drug-induced skin reaction, overall, cefazolin did not increase rates of allergic reactions compared to the non-allergic cohort. Cefazolin administration to penicillin allergic patients also did not significantly increase 90 day readmissions due to surgical site infections or need for subsequent I&D.
Conclusion
Our findings suggest that cefazolin can be safely administered to penicillin-allergic patients undergoing lumbar spine surgery without risk of cross-reactivity. Additionally, we found that cefazolin did not increase rates of non-surgical site infections or postoperative wound complications.
Introduction
Cefazolin is the most commonly used antibiotic for surgical prophylaxis in the United States. 1 A member of the cephalosporin class, cefazolin’s popularity can be largely attributed to its broad coverage against Gram-positive and Gram-negative bacteria, relatively low cost, and favorable side effect profile.2,3 Though cefazolin is generally well-tolerated, its safety among patients with penicillin allergies has long been questioned due to risk of cross-reactivity. A series of studies published during the 1960s and 1970s reported high rates of hypersensitivity reactions in penicillin-allergic patients treated with cefazolin that included rashes, wheezing, and anaphylaxis.4,5 In response, the United States Food and Drug Administration (FDA) began reporting a 10% incidence of cross-reactivity based on these findings. 6 Though subsequent research from the 1980s and 1990s has questioned the validity of these earlier studies, many providers continue to avoid cefazolin for penicillin-allergic patients due to concerns of cross-reactivity.4,5
Recent data has suggested that only 1-4% of patients with a history of a reported penicillin allergy will have a true cefazolin allergy.2,3 Cefazolin and penicillin are biochemically similar in that they share a core beta lactam ring, though they possess different side chains. 6 Despite their shared beta lactam ring, true allergies to cefazolin are usually specific to its unique side chains, and are therefore not the result of cross-reactivity. 5 High rates of hypersensitivity reactions reported in the 1960s and 1970s have largely been attributed to contamination, as both antibiotics were being produced by the same fungi during this time. 4 As artificial production of beta lactam drugs became standardized in the mid 1980s, incidence rates of hypersensitivity reactions in penicillin-allergic patients treated with cefazolin dropped significantly. 4 Finally, while penicillin allergies are reported by 8-15% of the United States population, only about 5% of these constitute true allergic reactions.5,7 Taken together, these findings suggest a significant overestimation of cross reactivity risk among penicillin-allergic patients receiving cefazolin and that cefazolin is safe to give to most patients despite a documented penicillin allergy.
Though cefazolin is frequently administered for surgical prophylaxis to penicillin-allergic patients, there are, to our knowledge, no large spine surgery studies that investigate its effect on short-term postoperative outcomes. Thus, the purpose of this study is to assess rates of allergic reactions, hospital length of stay (LOS), and 90-day surgical site infections (SSIs) among penicillin-allergic patients and to compare them to a propensity matched cohort of non-allergic patients who also received perioperative cefazolin.
Materials and Methods
A retrospective chart review was conducted at a single, large health care system to identify patients who underwent lumbar fusion surgery from 2017-2022. This was an IRB-approved retrospective study with exempt status, for which all patient information was de-identified and patient consent was not required. Approval for this study was granted by Thomas Jefferson University’s Institutional Review Board. All eligible patients were aged 18 years or older. Patients were excluded if they underwent surgery due to a traumatic injury, infection, or malignancy, and if they received an antibiotic for surgical prophylaxis that was not cefazolin. Patients with a documented penicillin allergy in our institutions electronic medical record (EMR) underwent a 3:1 propensity match to a cohort of penicillin non-allergic patients who also received cefazolin. Matching was based on age, race, gender, body mass index (BMI), smoking status, Charlson Comorbidity Index (CCI), surgical approach, number of levels fused, and revision status. Data on operative duration (cut to close time) and hospital length of stay was collected. Documented historical reactions to penicillin were recorded for all penicillin-allergic patients. All patient charts were reviewed for the development of postoperative allergic reactions, in-hospital complications, and mortality rates. We also identified 30- and 90-day readmissions due to infectious etiologies (both surgical site infections and non-spinal infections), as well as need for irrigation and debridement (I&D).
Statistical Analysis
Statistical analysis was performed using t-tests or Mann-Whitney U tests to compare continuous data and Chi-Squared or Fisher’s Exact tests to compare categorical data. All statistical analyses were completed using R studio (version 4.1.2, Vienna, Austria) and a P-value <0.05 was considered statistically significant.
Results
Propensity-Matched Demographic and Surgical Characteristics.
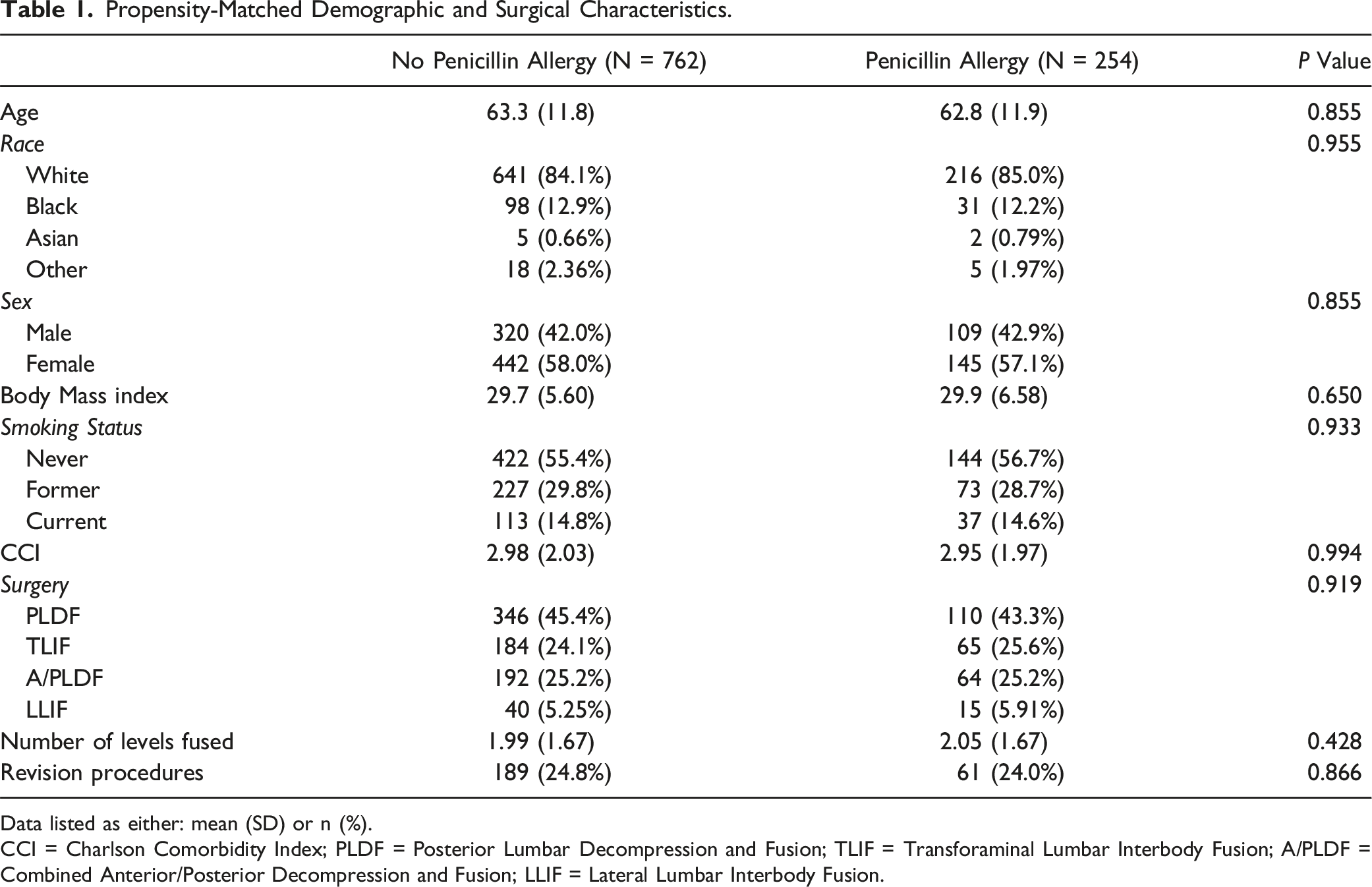
Data listed as either: mean (SD) or n (%).
CCI = Charlson Comorbidity Index; PLDF = Posterior Lumbar Decompression and Fusion; TLIF = Transforaminal Lumbar Interbody Fusion; A/PLDF = Combined Anterior/Posterior Decompression and Fusion; LLIF = Lateral Lumbar Interbody Fusion.
Historical Reaction to Penicillin Allergies.
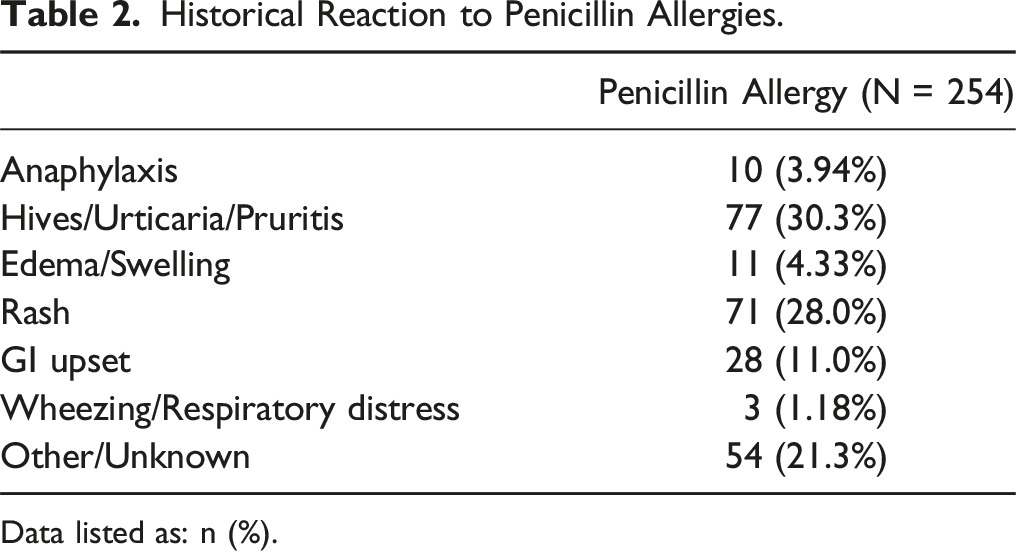
Data listed as: n (%).
In-Hospital Complications.
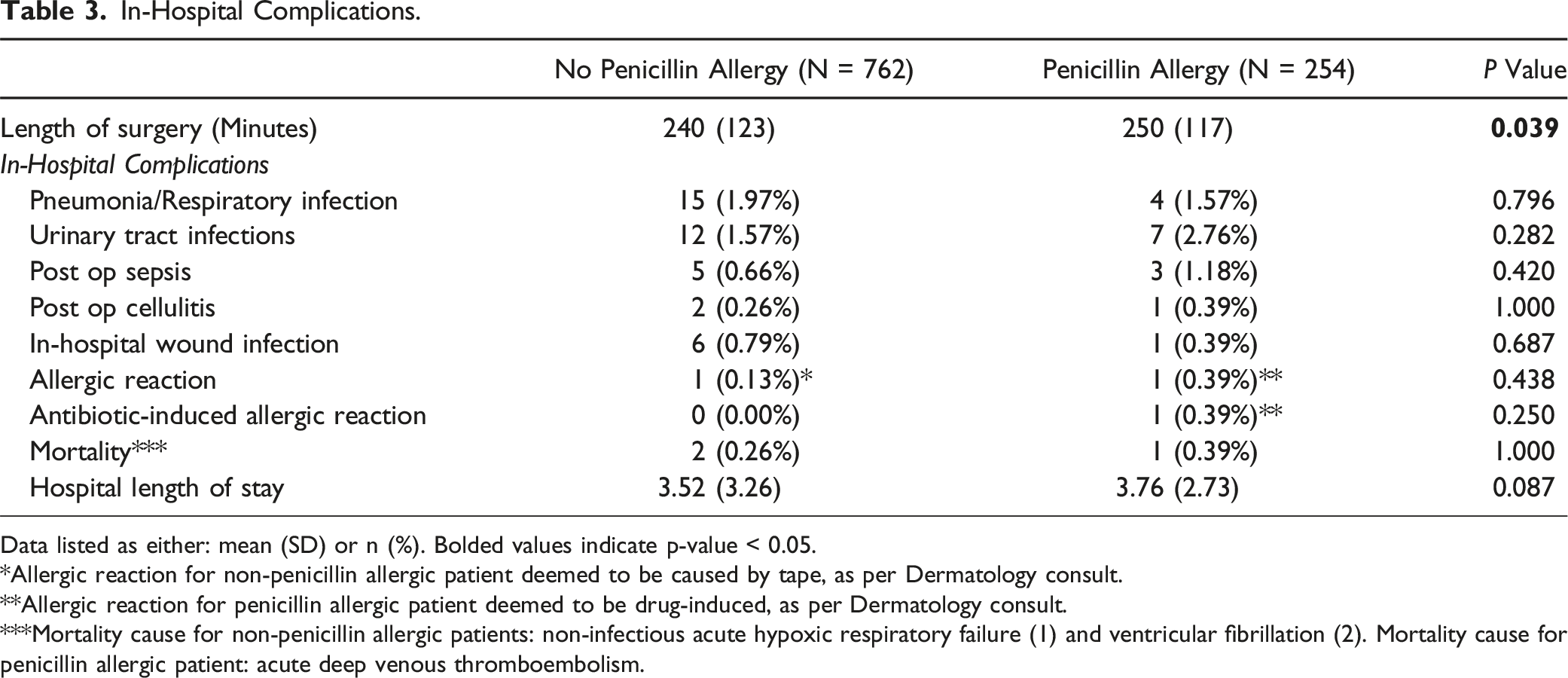
Data listed as either: mean (SD) or n (%). Bolded values indicate p-value < 0.05.
*Allergic reaction for non-penicillin allergic patient deemed to be caused by tape, as per Dermatology consult.
**Allergic reaction for penicillin allergic patient deemed to be drug-induced, as per Dermatology consult.
***Mortality cause for non-penicillin allergic patients: non-infectious acute hypoxic respiratory failure (1) and ventricular fibrillation (2). Mortality cause for penicillin allergic patient: acute deep venous thromboembolism.
Readmissions and Reoperations.

Data listed as: n (%).
I&D: Irrigation and Debridement.
Discussion
Cefazolin is the mainstay of surgical antibiotic prophylaxis, particularly in orthopedic and spine surgery. As a result, research into optimizing its dosing and timing based on various patient factors is extensive, particularly when compared to its alternative antibiotic agents. 8 Thus, it is preferable to use cefazolin when possible, rather than alternatives such as vancomycin or clindamycin. Norvell et al found that patients undergoing knee and hip arthroplasty who received cefazolin rather than clindamycin or vancomycin reported lower rates of postoperative SSIs, as well as periprosthetic joint infections, without any increases in hypersensitivity reactions. 9 Furthermore, in a study of 8385 patients with documented penicillin allergies, Blumenthal et al observed that these patients were much less likely to receive cefazolin perioperatively and subsequently experienced higher rates of surgical site infection. 3
In our analysis of allergic reactions between cohorts, we identified only one penicillin-allergic patient who experienced a drug-induced cutaneous reaction after administration of cefazolin (0.39% of the penicillin-allergic cohort). This percentage is within the expected incidence of all-cause cutaneous reactions among hospitalized patients (0.36-3%), as reported by prior studies.10,11 Additionally, the incidence of allergic reactions was not significantly different when compared to our cohort of non-allergic patients. While it was unclear whether the patient’s skin reaction was directly related to cefazolin administration or another medication, it was nevertheless considered a very mild reaction and managed with topical steroids only. The severity of the patient’s cutaneous reaction is also consistent with prior studies that indicate that the majority of cutaneous reactions to systemic drugs are mild and do not require systemic treatment. 11 Taken together, our findings suggest that penicillin-allergic patients are not at higher risk of developing cutaneous reactions in response to cefazolin exposure when compared to the general population, and that cefazolin is overall safe to administer in these patients.
Roebke et al looked at 286 942 patients undergoing elective posterior lumbar fusion utilizing the 100% Medicare Standard Analytical Files (SAF100) database and found that the 7497 patients with a self-reported penicillin allergy had a higher rate of surgical site infections, urinary tract infections, sepsis, acute kidney injury, and readmission rate compared to those without a self-reported penicillin allergy. 12 Due to the database nature of the study, the authors were unable to obtain granular data on the antibiotics administered in place of cefazolin, but they nevertheless concluded that the elevated incidence postoperative complications observed in penicillin-allergic was likely the result of administration of less effective antibiotics. These findings underscore our own findings which demonstrate that cefazolin is not only safe to use in penicillin-allergic patients, but should be used whenever possible for all patients to minimize risk of postoperative complications. A similar finding was observed by Niu et al, who performed a retrospective study of 20 022 patients after a variety of orthopedic procedures including spine, trauma, joint arthroplasty and found that penicillin allergic patients were associated with greater rates of SSI. 13 In their non-matched cohort, greater SSI rates were attributed to the use of alternative antibiotics such as vancomycin and clindamycin.
With the prevalence of penicillin allergy reported in the literature varying from 5-25%,14,15 many patients and surgeons are still faced with the dilemma of how to appropriately choose surgical prophylaxis and manage potential reaction symptoms. As Kuruvilla et al discuss, published antibiotic recommendations continue to advise against using beta-lactam antibiotics in surgical patients with moderate to severe penicillin allergies without either graded provocation testing or referral to allergy clinic for skin testing and delabeling of their “penicillin allergy”. 16 These recommendations may be outdated based on more recent safety data and are also impractical in the immediate perioperative setting. The process of delabeling an allergy can lead to additional outpatient encounters for patients and delay the time to surgery. To address this issue, a working group of physicians from Anesthesiology, Surgery, Infectious Diseases, and Allergy/Immunology at a large academic medical center developed a targeted algorithm to recommend cefazolin except in patients with a history of severe delayed hypersensitivity reactions (skin/mucosal blistering, drug fever/serum sickness, organ involvement, and drug induced anemia). They were able to increase institution administration of cefazolin from 22% to 80%. In addition, they had no patients with allergic reactions requiring epinephrine administration and only one patient with a minor rash that did not require cephalosporin discontinuation as part of management. 16 Their findings provide evidence for increased history-based screening that may be practical for surgeons and anesthesia teams to adopt.
There are several limitations to this study. The majority of historical reactions for the 254 patients with documented penicillin allergies in our cohort were mild (rash, GI upset, etc.), while 54 were unspecified or unknown. Our cohort also only had ten patients (4%) with self-reported anaphylaxis to penicillin, which may suggest that only a small percentage of our total cohort had a true allergy. Interestingly, we also did not know the proportion of patients with a confirmed penicillin allergy obtained through allergic testing, as this is not a part of our institution’s standard preoperative workup. Furthermore, we were not able to collect information on administration of perioperative steroids that may have reduced severity of allergic reactions or stress-dosing of cefazolin by the anesthesia team prior to surgery. Finally, we utilized a 3:1 matched cohort in this study to optimize our sample size and maintain an unbiased and balanced cohort. However, we acknowledge that this may not fully address the potential concerns of power. Despite these limitations, we have one of the largest retrospective cohorts of self-reported penicillin allergy patients who received perioperative cefazolin. Furthermore, in an effort to reduce confounding we performed a propensity match of patients based on various factors that may impact surgical outcomes and infection rates, however there may still be some confounders that we were not able to account for.
Conclusion
Our findings suggest that cefazolin can be administered safely to penicillin-allergic patients undergoing lumbar fusion without increasing rates of allergic reactions or compromising surgical site infection (SSI) risk. This data is consistent with a growing body of literature from other surgical specialties and more research is needed to determine its potential for cross reactivity in patients with even severe reported allergic reactions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
