Abstract
Study Design
Retrospective analysis of randomized controlled trial.
Objectives
Surgical site infection (SSI) after spine surgery has severe negative health and financial consequences. Surgical antibiotic prophylaxis (SAP) is a routinely used method to prevent SSIs in the spine patient population. The most commonly used antibiotic is cefazolin, with vancomycin often being substituted in the case of penicillin or cephalosporin allergy. Vancomycin as SAP has been associated with increased SSI in the joint replacement literature, but this is not yet well defined in the spinal surgery population. The purpose of this study was to determine whether vancomycin SAP compared to cefazolin SAP is associated with increased risk of SSI.
Methods
535 patients, aged 16 years or older, underwent elective multi-level open posterior spinal fusion surgery at the thoracic, thoracolumbar, or lumbar levels. Demographic and operative characteristics as well as post-operative outcomes were compared between the following groups: (1) noninfected-cefazolin, (2) noninfected-vancomycin, (3) infected-cefazolin, and (4) infected-vancomycin. Primary outcomes were superficial and complicated (deep and organ/space) infections.
Results
The following risk factors for SSI were identified in a logistic regression analysis: vancomycin (OR 2.498, 95% CI, 1.085-5.73, P = 0.031), increasing operating time (OR 1.006, 95% CI, 1.001-1.010 P = 0.010), weight (OR 1.020, 95% CI 1.006-1.034, P = 0.005), revision procedure (OR 2.343, 95% CI 1.283-4.277, P = 0.006), and depression (OR 2.366, 95% CI 1.284-4.360, P = 0.006).
Conclusions
In open posterior approach spinal fusion surgery, vancomycin SAP is associated with increased risk of infection compared to cefazolin SAP.
Introduction
Surgical site infection (SSI) following spine surgery not only extends hospital length of stay and necessitates additional surgery and/or prolonged antibiotic treatment, but also increases morbidity, mortality, and negatively impacts quality-of-life outcomes.1,2
One commonly used method to prevent SSIs is with surgical antibiotic prophylaxis (SAP). 3 In general, an appropriate SAP agent should be active against those organisms most likely to cause an infection at the surgical site, and they should be given at an appropriate time and dose to ensure adequate concentration of antibiotic at the surgical site throughout the operation. 4
In the setting of spine surgery, cefazolin is the most commonly used drug for SAP.5,6 It’s typically dosed at 2g within 60 minutes of surgical incision for patients <120 kg, and at 3g for patients >120 kg4. Cefazolin is effective as an agent for SAP for spine surgery, 7 but sometimes alternative antibiotics are indicated as a replacement.
One of the most often administered alternative SAP agents is vancomycin. 8 Typically, vancomycin is used when a patient has a severe allergy to beta-lactams, 9 or when MRSA colonization is present. 10
Recently, we published a randomized controlled trial (RCT) comparing 24 hours of post-operative SAP to 72 hours. 1 This RCT found no difference in infection rates between the 2 different treatment methods. However, the infection rate in this study was relatively high. Given previous literature demonstrating increased risk of SSI with vancomycin SAP in patients undergoing total joint arthroplasty,11,12 we sought to determine whether vancomycin increases the risk of SSI when compared to cefazolin in this population of patients undergoing multilevel spinal fusion. Our hypothesis is that when vancomycin SAP is used instead of cefazolin SAP, risk of SSI increases.
Methods
Study Setting and Design
This is a retrospective analysis of prospectively collected data from a consecutive series of patients enrolled in a previously completed RCT that compared 2 postoperative antibiotic treatment strategies: standard preoperative SAP with cefazolin or vancomycin and (1) 24 hours of postoperative antibiotics, or (2) 72 hours of postoperative antibiotics (to provide an additional 24 hours antibiotic coverage following postoperative drain removal on postoperative day 2). 1
Patient Population
Patients were enrolled between October 2011 and April 2016 at a single centre. The details of the primary trial have been published previously.1,13 Briefly, patients were randomly assigned by 1:1 allocation to duration of antibiotics for 24 hours after the operation or for 24 hours after drain removal (72 hours after the operation). The initial trial randomization was stratified by surgeon and presence or absence of diabetes. Drains were discontinued on the second postoperative day. The duration of antibiotic administration was not blinded. The operative technique used was chosen according to the surgeon’s discretion on a patient-by-patient basis. All patients received pre-operative antibiotic prophylaxis. This included either 2g of cefazolin intravenously 1 hour to no later than 15 minutes prior to incision and a second dose of cefazolin intraoperatively at the 4-hour mark (if necessary), or 1g of vancomycin intravenously within 1 hour of incision if the patient was allergic to penicillin or cephalosporin. 14 All patients had a closed-suction drain used (10-French Jackson Pratt Wound drain, Cardinal Health). Postoperatively, patients received 1g of cefazolin every 8 hours or 1g of vancomycin every 12 hours, according to their randomization. Drain removal was standardized to approximately 48 hours postoperatively.
Inclusion and Exclusion Criteria
For the present study, patients were included if they were 16 years of age and older and underwent elective, multi-level open posterior spinal fusion surgery at the thoracic, thoracolumbar, or lumbar levels with placement of a closed-suction drain. Exclusion criteria were known hypersensitivity to both cefazolin and vancomycin; renal function impairment (creatinine level of >100 μmol/L); antibiotic therapy for concomitant infection; surgery for spinal tumor or spinal trauma; surgery for spinal infection; pregnancy, concomitant corticosteroid therapy, and permanent residence >5 hours driving distance from surgical geographic location. In the original trial, 552 patients were enrolled. However, in the present study, patients that had deformity (n = 12) or multilevel decompression with no fusion (n = 5) were excluded.1,13 Therefore, 535 patients were included. Approval was obtained from the institutional ethics board (#100561). Written informed consent was obtained from all participants.
Outcome Measures
Primary outcome was a complicated infection (deep or organ/space) or a superficial infection within 1 year of the surgical procedure. Superficial, deep, and organ/space surgical site infections were defined according to the U.S. Centres for Disease Control and Prevention criteria. 15 Demographic variables evaluated as risk factors for surgical site infection were age, gender, body mass index, diabetes, anemia or other blood disease, hypertension, cerebrovascular disease, osteoarthritis and inflammatory arthritis, heart disease, lung disease, gastrointestinal disease, liver disease, history of cancer, smoking history, depression, pre-operative Medical Outcomes Study-12 item Short-Form General Health Survey (SF12) mental component summary score (MCS) and physical component summary score (PCS) and Oswestry Disability Index (ODI), and diagnosis. 16 BMI was dichotomized to ≥30 kg/m2 as this is a cut-point for obesity. Operative variables evaluated as risk factors included duration of postoperative antibiotics (randomized to 24 hours or 72 hours of postoperative antibiotics in the original RCT), type of procedure (interbody fusion or instrumented fusion), ASA classification, antibiotic type (vancomycin vs cefazolin), revision procedure, number of operated levels, operating time, and estimated blood loss. Other factors associated with infection that were identified for collection a priori included the season that the procedure was performed, number of surgical assistants, number of scrub events, and postoperative day 1 hemoglobin level.
Statistical Analysis
Data analysis was performed using SPSS Statistics version 27 (SPSS Inc., Chicago, IL, USA). The demographic and operative characteristics as well as post-operative outcomes were compared between the following groups: (1) noninfected-cefazolin, (2) noninfected-vancomycin, (3) infected-cefazolin, and (4) infected-vancomycin. Between group comparisons were made using a one-way ANOVA for continuous parametric variables or Kruskal-Wallis test for continuous nonparametric variables. Comparisons for categorical variables were made using the Chi-square test or the Fisher’s exact test. Multiple comparisons were conducted using a Bonferroni post hoc test or an independent samples test for proportions.
To identify the risk factors associated with surgical site infection, type of antibiotic as well as variables that yielded a P value of ≤0.05 in univariate analysis were included in the multivariate analysis. Variables were also included if they were clinically important by the authors’ consensus. Collinearity was evaluated by calculation of tolerance. Multivariate backward stepwise, conditional, logistic regression was used to identify the best combination of variables that predict having a surgical site infection vs not having an infection. Individual predictor variables were eliminated in a backward fashion if the corresponding P value was ≥0.10. Cases with missing data were excluded from the model. The discriminative ability was assessed using ROC curves and by comparing the predicted vs the observed surgical site infections by area under the curve.
Results
Patient Demographics
Patient-Specific Risk Factors Stratified by Antibiotic Type and the Presence of Infection.
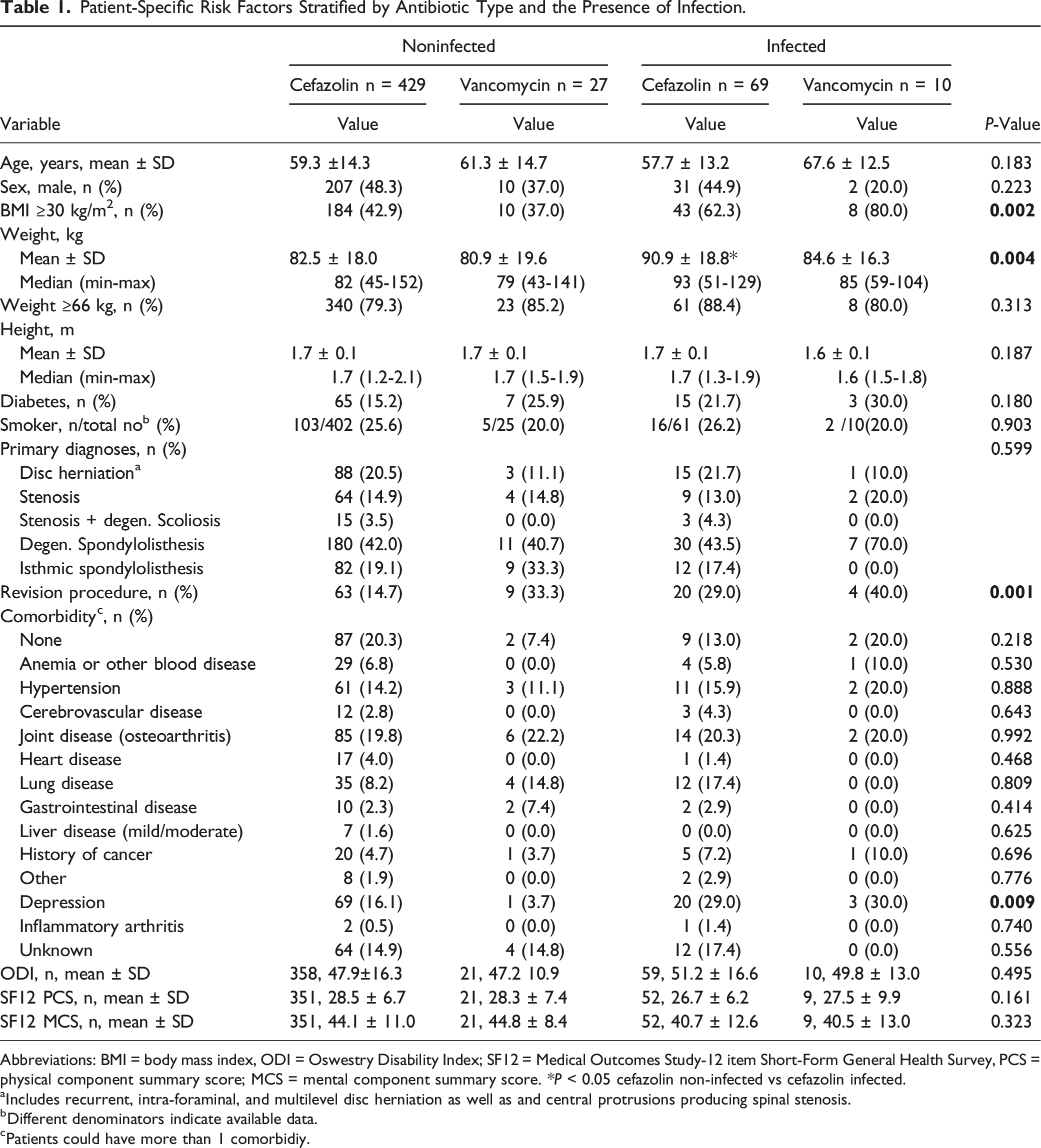
Abbreviations: BMI = body mass index, ODI = Oswestry Disability Index; SF12 = Medical Outcomes Study-12 item Short-Form General Health Survey, PCS = physical component summary score; MCS = mental component summary score. *P < 0.05 cefazolin non-infected vs cefazolin infected.
aIncludes recurrent, intra-foraminal, and multilevel disc herniation as well as and central protrusions producing spinal stenosis.
bDifferent denominators indicate available data.
cPatients could have more than 1 comorbidiy.
Intra- and Peri-Operative Factors
Comparison of Intra-Operative and Peri-Operative Risk Factors Stratified by Antibiotic Type and the Presence of Infection.
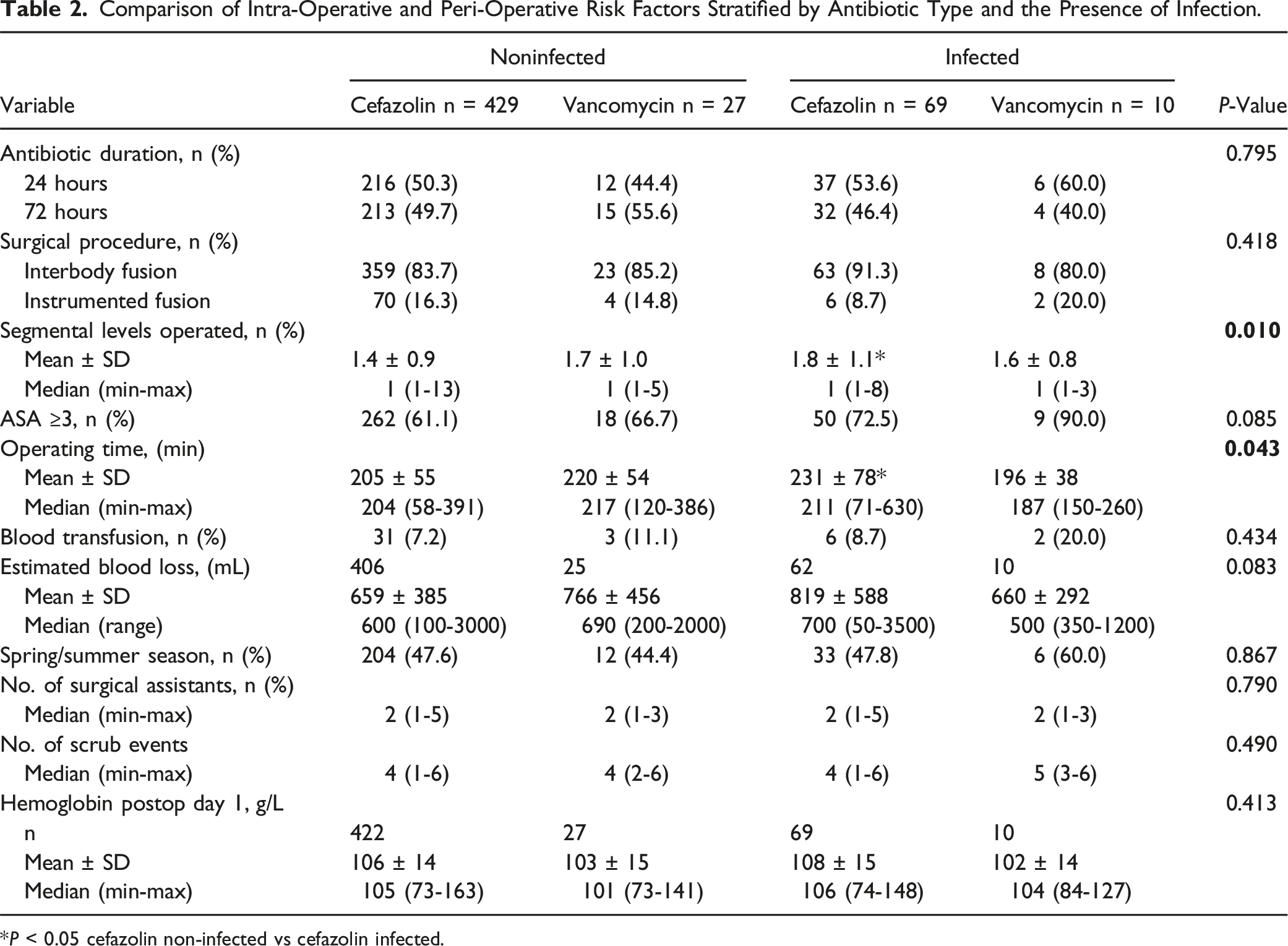
*P < 0.05 cefazolin non-infected vs cefazolin infected.
Infection Details
Infection Details.
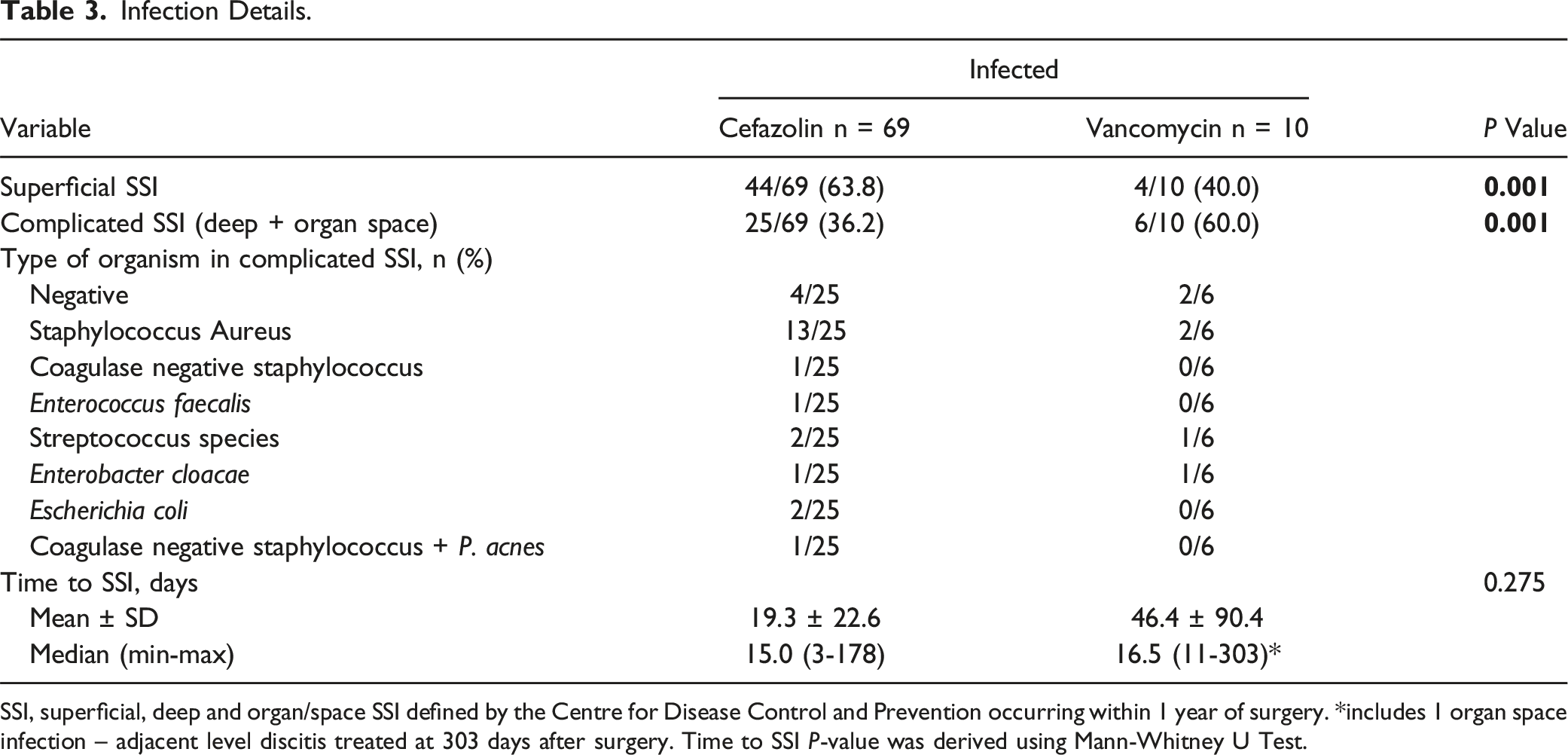
SSI, superficial, deep and organ/space SSI defined by the Centre for Disease Control and Prevention occurring within 1 year of surgery. *includes 1 organ space infection – adjacent level discitis treated at 303 days after surgery. Time to SSI P-value was derived using Mann-Whitney U Test.
Post-Operative Outcomes
Comparison of Post-operative Outcomes Stratified by Antibiotic Type and the Presence of Infection.
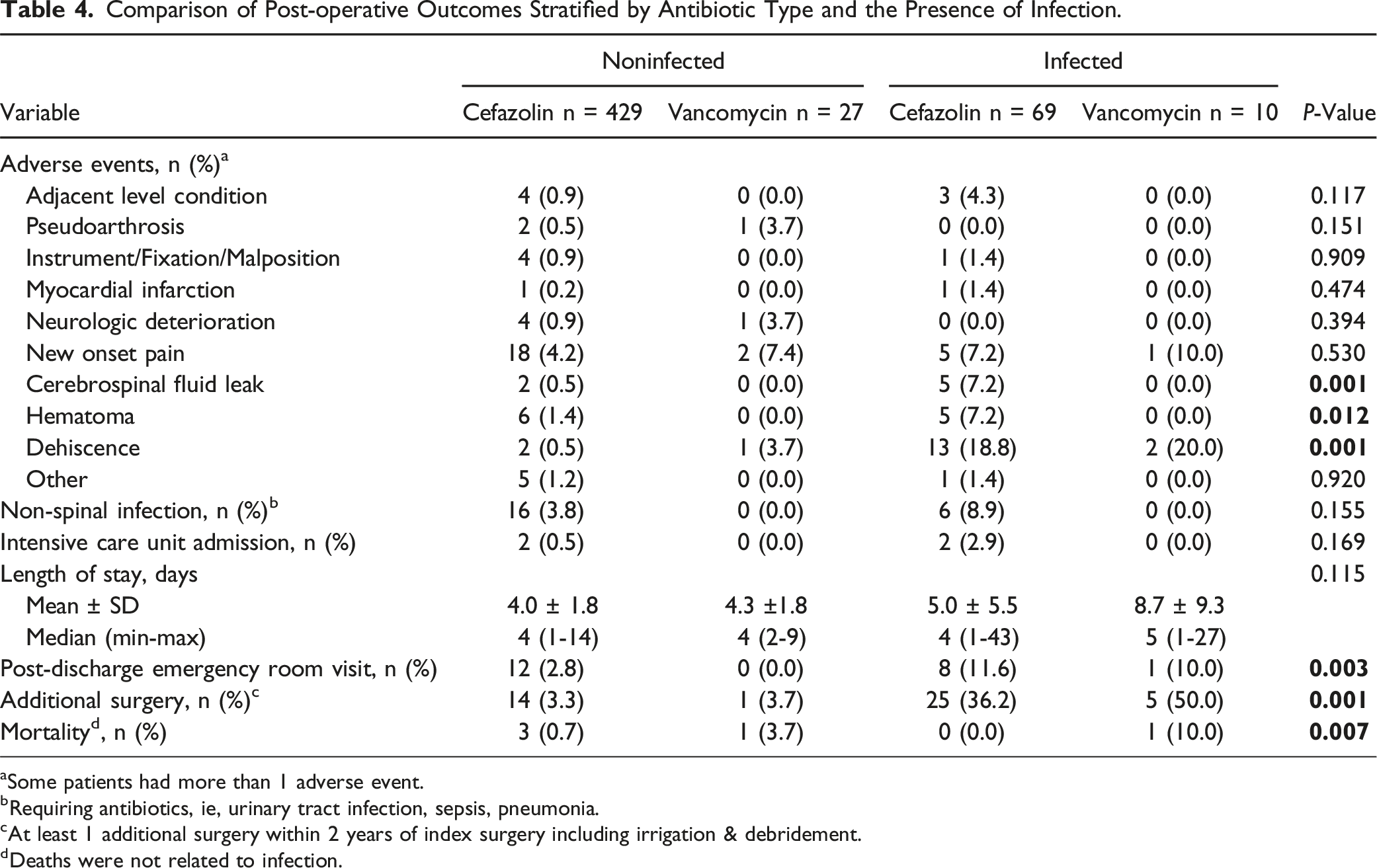
aSome patients had more than 1 adverse event.
bRequiring antibiotics, ie, urinary tract infection, sepsis, pneumonia.
cAt least 1 additional surgery within 2 years of index surgery including irrigation & debridement.
dDeaths were not related to infection.
Multivariable Results
Multivariate Logistic Regression Analysis: Significant Risk-Factors for Infection.
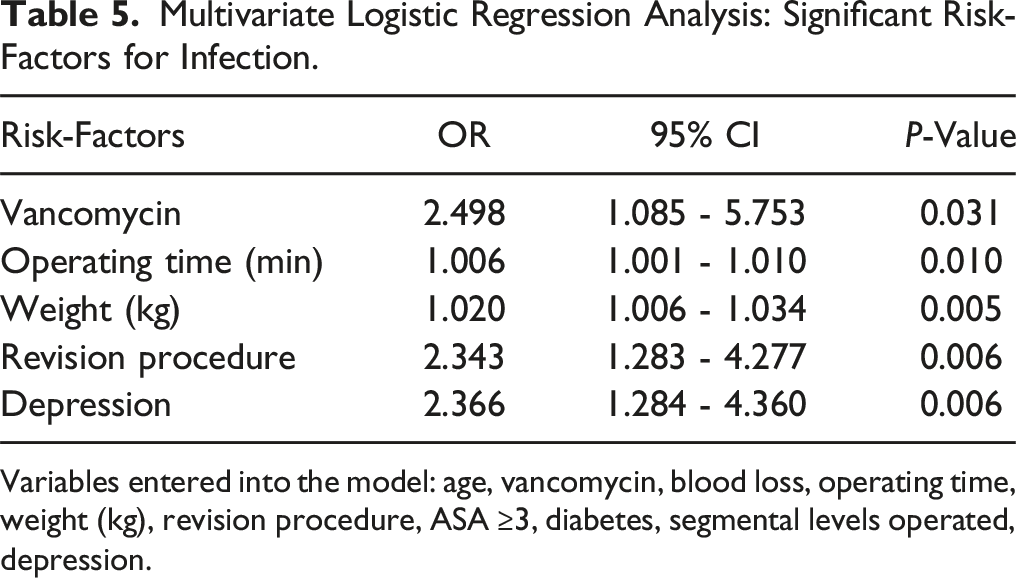
Variables entered into the model: age, vancomycin, blood loss, operating time, weight (kg), revision procedure, ASA ≥3, diabetes, segmental levels operated, depression.
Discussion
In the present retrospective analysis of an RCT of patients that underwent multilevel thoracolumbar surgery, vancomycin SAP was associated with almost a 2.5-fold higher risk of SSI than cefazolin SAP. These results echo previous findings within the orthopaedic literature but also have important distinctions. For instance, in a retrospective review of SAP agents in 622 patients who underwent elbow arthroplasty, vancomycin was associated with a 3.3 times greater risk of SSI when compared to cefazolin. 17 Similarly, in a retrospective review of 7713 shoulder arthroplasties, when compared to cefazolin, a 2.3-fold greater risk of SSI was identified in patients who received vancomycin SAP. 18 Large dataset reviews have found similar increased risk of SSI with vancomycin.19,20 However, other studies have shown similar rates of SSI between those receiving vancomycin or cefazolin SAP. 21 Some have even demonstrated vancomycin to be advantageous in reducing SSI with drug resistant organisms. 22 These conflicting results highlight 2 significant issues with vancomycin administration—dosing and timing.
In regards to timing of administration, Marigi et al re-examined a cohort of shoulder arthroplasty patients who had previously demonstrated higher rates of SSI with vancomycin SAP than with cefazolin. 18 They showed that when patients received a complete dose of vancomycin—defined as at least 30 minutes of vancomycin infusion prior to incision—no difference was observed in rates of SSI compared to cefazolin. 23 Hawn et al looked at a large dataset of patients who received various forms of antibiotic prophylaxis. In general, timing of antibiotic delivery was not a significant risk factor for SSI, but vancomycin compared to cefazolin was associated with higher risk of SSI, and it was administered 12 minutes earlier than cefazolin on average (40 vs 28 minutes). 20 Since vancomycin is usually administered at a rate of 1 g/hour to avoid vancomycin flushing syndrome, 24 this can potentially cause issues where the medication is not fully delivered prior to skin incision, and SAP delivery after skin incision has been linked to higher rates of SSI. 25 In our analysis, a well-established protocol during the initial RCT ensured patients had completed their administration of vancomycin within 1 hour of skin incision. As such, in the present analysis, timing of vancomycin administration was not the cause of the increased risk of SSI with vancomycin SAP.
In this cohort of patients who underwent multilevel thoracolumbar fusion surgery, prophylactic vancomycin dosing was standardized to 1g for all patients.10,26 This “standardized” dose has been called into question, with more recent guidelines recommending weight-based dosing of 15 mg/kg4. Using these recommendations, Catanzano et al found that 69% of patients who received 1g vancomycin SAP prior to elective joint or spine surgery were underdosed. 27 Similarly, when Kheir et al examined arthroplasty patients receiving vancomycin SAP, they showed that 94% of those who received 1g were underdosed, and that no patients who received the recommended 15 mg/kg dose developed a SSI. 28 In the present cohort, the proportion of patients with BMI ≥30 kg/m2 was significantly different in the infected-vancomycin group compared to the noninfected-vancomycin group (80% vs 37%), which points to obesity as a significant risk factor for infection and potentially dosing issues as well. However, after examining patients with body weight ≥66 kg (the weight-based cutoff point for adequate therapeutic dosing using 1g vancomycin), the proportion of patients with weight ≥66 kg in the noninfected-vancomycin was actually higher than in the infected-vancomycin group (85% vs 80%), but this was not statistically significant. Although most patients in each group were underdosed according to newer weight-based dosing guidelines, weight ≥66 kg and weight on a continuous scale were not independent predictors of increased SSI when comparing the noninfected-vancomycin and infected-vancomycin groups.
With increased concern around infection with methicillin resistant organisms, 29 some authors have demonstrated lower SSI rates by using both vancomycin and cefazolin together for SAP. 30 This question was addressed by a recent large multicenter randomized controlled trial in a group of patients undergoing joint replacement surgery. The authors unfortunately did not find any reduction in SSI rates when patients received vancomycin and cefazolin SAP when compared to SAP with cefazolin and placebo. 31 In addition, the combination of the 2 antibiotics has also been associated with higher incidence of acute kidney injury than either 1 of them alone. 32
Although dosing and timing are the most cited reasons for differences in SSI between vancomycin and cefazolin SAP, 2 other less obvious characteristics may play a significant role in this discussion—molecular weight and charge.33-35 In a cohort of 54 patients who received prophylactic administration of either cefazolin, clindamycin, or vancomycin, and subsequently had their disc material analyzed following microdiscectomy, concentration of vancomycin in the disc material was significantly lower than that of cefazolin. 36 The molecular weight of vancomycin is much greater than cefazolin (1449 g/mol vs 455 g/mol), and the authors postulated that this may have impacted the ability of vancomycin to penetrate the intervertebral disc. This is supported by other in vitro work demonstrating slower diffusion within an intervertebral disc with vancomycin compared to oxacillin (molecular weight = 401 g/mol)—despite the positive charge of vancomycin theoretically giving it an advantage over the negatively charged oxacillin (as well as cefazolin) to penetrate the negatively charged extracellular matrix of the disc. 37
Recently, using gene expression analysis in a 2024 study, Hayles et al identified a cellular mechanism of s. aureus that allows it to modify its cell surface charge following attachment to medical grade titanium. 38 In this novel study, prior to exposing s. aureus to titanium, both antibiotics had equal effectiveness. However, after a titanium surface was immersed in a s. aureus solution for 3 hours, only 20% of s. aureus survived upon treatment with cefazolin whereas 60% survived after treatment with vancomycin. When they extended this immersion to 24 hours, 50% of organisms survived with cefazolin while 90% survived after treatment of vancomycin. This study elucidates that not only are organisms more resistant to antibiotics quickly after exposure to titanium, but also that cefazolin is significantly more effective against titanium surface-associated s. aureus than vancomycin. This is particularly relevant to the fields of orthopaedic and spine surgery, as many of the instrumented materials are titanium-based. 39 Of note, similar patterns of resistance have been shown with the positively charged antibiotic daptomycin.40-42
In the present analysis, patients only received vancomycin SAP when a documented beta-lactam allergy was identified. In a 2021 systematic review and meta-analysis of penicillin and cefazolin allergies, a true cefazolin allergy was identified in only 0.6% of patients with an unconfirmed penicillin allergy, and in only 3% of patients with a confirmed penicillin allergy. Furthermore, only 4.7% of patients allergic to cefazolin were also allergic to penicillin, suggesting low cross-reactivity. 43 Given the increased risk of SSI associated with vancomycin, clinicians should ensure patients with suspected beta-lactam allergy are further investigated by an appropriate professional, as many of these individuals could still safely receive cefazolin. Indeed, in 1 2019 arthroplasty study, 97% of 2576 patients referred to an allergist for penicillin allergy prior to surgery were ultimately cleared for the use of cefazolin. 44
Aside from vancomycin use, obesity, revision surgery, and depression were also found to be risk factors for SSI in our cohort. Obesity and revision surgery are well documented elsewhere as risk factors for SSI,45,46 and more evidence continues to emerge regarding risk of SSI with anxiety and depression.47-49 It’s thought that with depression, the increased risk of infection may be secondary to elevated cortisol levels suppressing the immune system and subsequently affecting wound healing rates, higher rates of smoking, or higher rates of nonadherence to medical recommendations in this population.50,51 In the present study, however, smoking was not an independent predictor of SSI.
Limitations
A limitation of this study is selection bias. Due to patients receiving vancomycin only if they had an allergy to beta lactams, and more patients undergoing reoperation (which also carries a higher risk of infection) in the vancomycin group, the sample may not be fully representative of the broader population. Although a number of patient and operative risk factors including reoperation were controlled for in our multivariate analysis, we cannot control for any unknown factors that may have contributed to the increased risk of infection in patients receiving vancomycin. This is a limitation of the retrospective nature of the study. However, this data was gathered from a previous RCT where patients were recruited consecutively, which does strengthen the findings of the study and ultimately decrease the risk of that bias.
Our study population was limited to those who underwent instrumented fusion surgery only. This impacts our ability to generalize the results of the study to the population of spine patients undergoing surgery without instrumentation. Additionally, our study cohort consisted of patients who underwent open posterior approach surgery only. It’s possible that infection rates between those who received vancomycin and cefazolin SAP may be different in those who had more minimally invasive or non-posterior approaches. However, the majority of elective surgeries continue to be done via an open posterior approach, and continue to include instrumentation, which lends to the overall generalizability of this analysis. 52
Conclusions
In our cohort of patients who underwent posterior instrumented spine surgery, we identified a 2.5-fold increased risk of SSI in those who received vancomycin SAP compared to those who received cefazolin SAP, independent of dose and timing of administration. Clinicians should ensure patients with a suspected beta-lactam allergy receive appropriate pre-operative investigation into the nature of this allergy to prevent unnecessary administration of vancomycin in those who can ultimately tolerate cefazolin antibiotic prophylaxis.
Footnotes
Acknowledgments
Thank you to Lori Nutt and Linda Kuska for all their help in the recruitment of patients during the initial trial.
Author Contributions
Conception and Design: BJ Herrington, JC Urquhart Administrative Support: JC Urquhart, Provision of Study Materials: P Rasoulinejad, F Siddiqi, K Gurr, CS Bailey, Collection and Assembly of Data: BJ Herrington, JC Urquhart Data Analysis and Interpretation: BJ Herrington, JC Urquhart Manuscript Writing: All authors Final Approval of Manuscript: All authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
