Abstract
Study Design
Prospective Study.
Objective
Spinal cord injury leads to severe limitations in autonomy including locomotor function. Intermittent theta burst stimulation (iTBS) has shown promising therapeutic effects on various neuropsychiatric disorders. We analyzed the efficacy of excitatory iTBS on motor and walking functions in patients with incomplete spinal cord injury (iSCI).
Methods
The prospectively selected iSCI patients were divided into the iTBS (n = 69) and sham-iTBS (n = 69) groups, and underwent iTBS and sham-iTBS treatment, respectively, followed by conventional physiotherapy. Clinical assessments were conducted on the muscle strength of quadriceps, gluteus maximus, hamstring, tibialis anterior and gastrocnemius muscle, lower extremity motor score (LEMS), walking speed (WS), stride length (SL), Berg balance scale (BBS), Holden walking ability scale (HWAS) score, the static eyes-open and eyes-closed center of gravity locus length, and the modified Barthel index (MBI) score.
Results
There were no significant differences among all patients in baseline data. After 9-week treatment, all the patients had notably elevated muscle strength levels of quadriceps, gluteus maximus, hamstring, tibialis anterior and gastrocnemius, WS, SL, and HWAS, BBS, LEMS and MBI scores, as well as decreased static eyes-open and eyes-closed center of gravity locus length, with the changes more remarkable in the patients treated with iTBS. The total effective rate for patients treated with iTBS was 86.96%, which was apparently higher than that of patients treated with sham-iTBS.
Conclusion
Excitatory iTBS significantly improved lower limb strength and walking function in iSCI patients.
Keywords
Introduction
Spinal cord injury (SCI) stands out as a severe disorder of the central nervous system; incomplete spinal cord injury (iSCI) is the primary type of SCI recognized as the sensory and functional impairments of some spinal cord segments indirectly or directly triggered by external forces. 1 Notably, the leading factors resulting in augmented mortality and worsened quality of life are motor control impairment and motor dysfunction in SCI patients. 2 It has been documented that in addition to facilitating the arm or hand function recovery with the highest priority, SCI patients give priority to the restoration of lower limb motor function within 10 years following SCI.3,4 Consequently, how to ameliorate lower limb motor function and restore walking ability in patients with SCI are the predominant challenges of rehabilitation medicine.5,6
SCI represents a quite complex pathological process, where a single treatment modality shows limited efficacy, so its clinical management is focused on comprehensive treatment, and the conventional treatments include exercise therapy, decompression surgery, respiratory training, physical factor therapy, stem cell transplantation, and anti-inflammatory drugs. 7 With the in-depth study of the pathological mechanisms of SCI, new techniques have been gradually introduced into the rehabilitation of SCI.8-10 Transcranial magnetic stimulation (TMS), as a safe, non-invasive and acceptable treatment to patients, is capable of producing a widespread effect at the level of the brain neural network by modifying the excitability of neurons at the stimulation site. 11 Importantly, Leszczy et al have proposed that TMS possesses the ability to attenuate upper limb muscle tone and improve neurotransmission function of the spinal cord in patients with iSCI. 12 TMS generates varying effects based on its different frequency, and the TMS modes for therapy consist of theta burst stimulation (TBS) and traditional repetitive TMS, with the TBS mode further allocated into continuous TBS mode with inhibitory effect and intermittent theta burst stimulation (iTBS) mode with excitatory effect. 13 iTBS has been demonstrated to be a particular type of stimulation modality of TMS, with its action mechanism consisting of altering cortical excitability, triggering long-term depression or long-term potentiation, facilitating the release of neurotransmitters, and accelerating neural remodelling.14-16 In the past few years, iTBS has been extensively applied in the clinical treatment of patients with stroke, acute and chronic pain, and post-ischemia-reperfusion nerve injury.17-19 Furthermore, existing evidence suggests that iTBS can effectively alleviate neuropathic pain associated with SCI. 17 Gharooni et al. have further demonstrated that iTBS may attenuate upper limb spasticity in individuals with SCI. 13 The long-term potentiation-like effects induced by iTBS have been shown to continuously enhance the connectivity between the motor cortex and target muscles, promote corticospinal tract (CST) outgrowth, and facilitate the molecular signaling transduction underlying motor evoked potential (MEP) plasticity, thereby contributing to the restoration of motor function. 20 Moreover, improvements in hand function have been observed following iTBS stimulation, along with enhanced endogenous coupling between the supplementary motor area (SMA) and the primary motor cortex (M1). 21 Furthermore, iTBS demonstrates the merits of shorter duration of stimulation, higher frequency, longer duration of cortical excitability changes, as well as lower intensity22-24 relative to traditional TMS.25,26 Nevertheless, there is a dearth of reports regarding the efficacy of iTBS on motor and walking function in iSCI patients. Accordingly, this study aimed to analyze the efficacy of excitatory iTBS on motor and walking function in patients with iSCI.
Materials and Methods
Ethics Statement
The experiments were authorized by the academic ethics committee of First Affiliated Hospital of Kunming Medical University (approval number: kmmu20241833). All procedures were implemented in strict accordance with Declaration of Helsinki. All subjects involved were fully informed of the study objective and signed the informed consents before sampling.
Sample Size Estimation
In this study, sample size estimation was conducted in advance using G*Power version 3.1.9.2 (Heinrich-Heine-Universität Düsseldorf, Germany) (Supplemental Figure 1). A t-test was selected as the testing method, with the significance level (α) set at 0.05, testing power (1-β) set at 0.80, and anticipated effect size (Cohen’s d) as 0.5 (medium effect). The results showed that the minimum sample size was 128 to ensure sufficient statistical power to detect the study outcomes.
Study Subjects
A total of 170 patients with iSCI admitted to First Affiliated Hospital of Kunming Medical University between January 2022 and June 2023 were prospectively selected, with 142 patients sorted out as per the inclusion and exclusion criteria. Among these, 4 cases refusing to participate were excluded, and 138 cases were finally included as the study subjects (105 traumatic iSCI patients and 33 non-traumatic iSCI patients). The subjects were allocated into the iTBS group (n = 69) and the sham-iTBS group (n = 69) utilising the randomized numerical table method, and underwent conventional physiotherapy (5 times/week) for 9 weeks. Prior to conventional physiotherapy, the two groups of patients underwent TMS, with iTBS applied for patients in the iTBS group and sham-iTBS for patients in the sham-iTBS group. No one withdrew through the session.
Inclusion and Exclusion Criteria
Inclusion criteria were as hereunder: 1) conformed to the diagnostic criteria updated by the American Spinal Injury Association in 2015, and diagnosed as iSCI by speciality check-up and imaging means including magnetic resonance imaging (MRI); 2) with American Spinal Cord Injury Association Impairment Scale (ASIA) grade C or D, or motor dysfunction of both lower limbs; 3) iSCI patients received spinal stabilization surgery and ≥ 4 weeks of postoperative stabilization; 4) had stable vital signs, good cognitive function, and could cooperate with specialized physicians for evaluation and treatment; 5) 18-70 years old.
Exclusion criteria were as hereunder: 1) combined with disorders influencing motor function such as joint contracture, lower limb fracture, and lower limb spasticity; 2) had severe complications and grave brain, heart and lung diseases; 3) implanted with intracranial metallic foreign bodies, cochlear implants, and pacemakers; 4) had drugs that might affect cortical excitability; 5) pregnant or gestational women; 6) injuries below the L3 segment and of cauda equina nerve; 7) underwent spinal surgery within the past month, had spinal instability, or presented with progressive neurological deterioration requiring emergency surgery.
Based on the principle of randomization, 138 patients were randomly assigned into the sham-iTBS group and the iTBS group at a ratio of 1:1 utilising the randomized numerical table method. The detailed methodology was as below: 138 iSCI patients (numbers 1-138) were allocated into two groups with the aid of the principal investigator (PI). One group included the subjects that had corresponding 69 numbers, and another group included the rest of the subjects. The randomization schedule was hidden in a locked cabinet that was only accessible to the investigators using iTBS and the PI. Therefore, patients were blinded to the TMS type they underwent (sham or real). Neither the researchers evaluating outcomes nor the subjects were aware of the administered interventions. The participants were blinded to the experimental hypothesis and were not allowed to share their experience with other participants engaging in evaluating the outcomes or the researchers during the intervention.
Standard Physical Therapy Program
All participants were subjected to standard SCI rehabilitation treatment and nursing care, such as nutritional neurologic medication, routine rehabilitation care, electro-acupuncture (paralyzed limbs, twice a day, 20 min/time), electronic biofeedback therapy (paralyzed limb sites, twice a day, 15-20 min/time), lower limb robotics training (once a day, 30 min/time), and motor function training of paralyzed limbs (once a day, 30-40 min/time). Routine rehabilitation care consisted of prevention of decubitus ulcers, good limb position, psychological counseling and guidance, urethral care, and dietary advice. The subjects received treatment for 9 consecutive weeks (5 days/week). The project was carried out by 2 specially trained physiotherapists blinded to the project.
iTBS Intervention
Cerebral stimulation was implemented using a CCY-1 magnetic stimulator (Yiruide, Wuhan, Hubei, China) with figure-8-coil at a diameter of 12.5 cm and with a maximum stimulation intensity of 3T (Yiruide Medical Treatment Equipment New Technology Co., Ltd., Wuhan, Hubei, China). The subjects were kept in a supine position during the treatment. The magnetic stimulation coil was tangent to the surface of the skull of the subjects, whilst the subjects kept their eyes closed. The midpoint of the coil was in alignment with the cortical M1 region (motor area) of the legs of the bilateral brain. 27 The chosen stimulation was iTBS mode, with a total of 600 pulses. The two sides underwent treatment for 6 min 40 s in total, with each side treated for 3 min 20 s. The stimulation intensity was set as 100% of the rest motor threshold of the cerebral hemisphere of the subjects, and the treatment was implemented for 9 weeks (once a day, 5 times/week).
Sham-iTBS Intervention
Subjects in the sham-iTBS group were subjected to a 9-week treatment (once a day, 5 consecutive times/week). Attenuation of the magnetic field was ensured using a dummy coil, which presented the same shape as the active coil and possessed a good approximation of the auditory feedback. 28 The coil also maintained a tactile sensation with the skull. Neither could the sham coil produce an electric field nor induce any neural activation.
Clinical Assessment
Lower limb muscle strength, gait parameters, motor function, ability of daily living activities, balance function, and walking function were assessed before and following 9 weeks of intervention, separately.
For lower limb muscle strength appraisement, the manual muscle testing (MMT) method 29 was used to evaluate the lower limb muscle strength of all subjects prior to and after the intervention, including gluteus maximus, quadriceps, hamstring, tibialis anterior, and gastrocnemius muscle.
Gait parameters and walking function estimation were performed as follows: the data of walking function before and following treatment was gathered from the two groups of participants, with the stride length (SL) and walking speed (WS) of the lower extremities assessed with the help of the digital treadmill Tecno Body Walker-view (Bergamo, BG, Italy). The gathered data were processed by Tecno Body Medical Fitness Software (version number: 2.7.8.0), and SL and WS were acquired, with 3 repeated determinations taken and the mean determined. 30 Holden Walking Ability Scale (HWAS) was adopted for evaluating the walking ability of the patients, with grade 0 standing for no walking ability, grade 1 for the need of help with walking, grade 2 for the need of substantial assistance with walking, grade 3 for the need of a little help with walking, grade 4 for the ability to independently walk on the flat ground, and grade 5 for normal walking. 15
Balance function assessment was carried out as below: the Berg Balance Scale (BBS) was employed for assessing the balance ability of the two groups before and following therapy, with a total score of 56. A higher score represented a better balance ability of patients. 31 Meanwhile, balance tests were conducted using a balance tester (Active Balancer ABC-20, AYIMA, Japan), and the static eyes-open center of gravity locus length and static eyes-closed center of gravity locus length were recorded. 32
The evaluation of lower extremity motor function was carried out using ASIA lower extremity motor score (LEMS) before and after the treatment. The total score of the scale was 50. A higher score indicated a better lower extremity motor function of the patients. 33
The modified Barthel index (MBI) scale was adopted for assessing the pre- and post-treatment ability of daily living activities of patients, including 10 items such as defecation, urination, toileting, eating, grooming, transferring, dressing, and going upstairs, with a maximum score of 100. The higher the score, the better the ability to perform daily living activities. 34
Clinical efficacy was evaluated as per the following standards: significantly effective: after treatment, the functional disorders of muscle motor function below the injury plane, superficial sensory function and bowel control function were basically restored; effective: the functional disorders of muscle motor function below the injury plane, superficial sensory function and bowel control function were partially restored after treatment; ineffective: the functional disorders of muscle motor function below the injury plane, superficial sensory function and bowel control function did not get any improvement after treatment. 35 Total effective rate = [(significantly effective + effective)/total number of cases] × 100%.
Statistical Analysis
SPSS 27.0 statistical software (IBM Corp. Armonk, NY, USA) and GraphPad Prism 9.5.0 software (GraphPad Software, San Diego, CA, USA) were applied for statistical analysis and plotting. Normal distribution was tested by the Kolmogorov-Smirnov test, with measurement data conforming to normal distribution denoted as mean ± standard deviation. Independent sample t-test was employed for data comparisons between two groups, with paired sample t-tests used for pre- and post-intervention comparisons within groups. Non-normally distributed measurement data were expressed as quartiles [median (minimum, maximum)], with the Mann-Whitney U test adopted for inter-group comparisons. This method was to combine two sets of independent sample data and calculate the rank (sort position), and compare the rank and difference between the two groups, so as to determine whether there is a statistically significant difference in medians. Wilcoxon signed-rank test was used for pre- and post-treatment comparisons within groups. It was based on the absolute value of the paired difference to calculate the rank and analyze the systematic differences of the same group of samples under different conditions. The counting data were represented as cases and percentages, with intergroup comparisons conducted by the Chi-square test. P was a two-sided test, with the differences considered statistically significant at P < 0.05.
Results
Comparisons of Baseline Data Between the Two Groups
Comparisons of Clinical Baseline Characteristics.
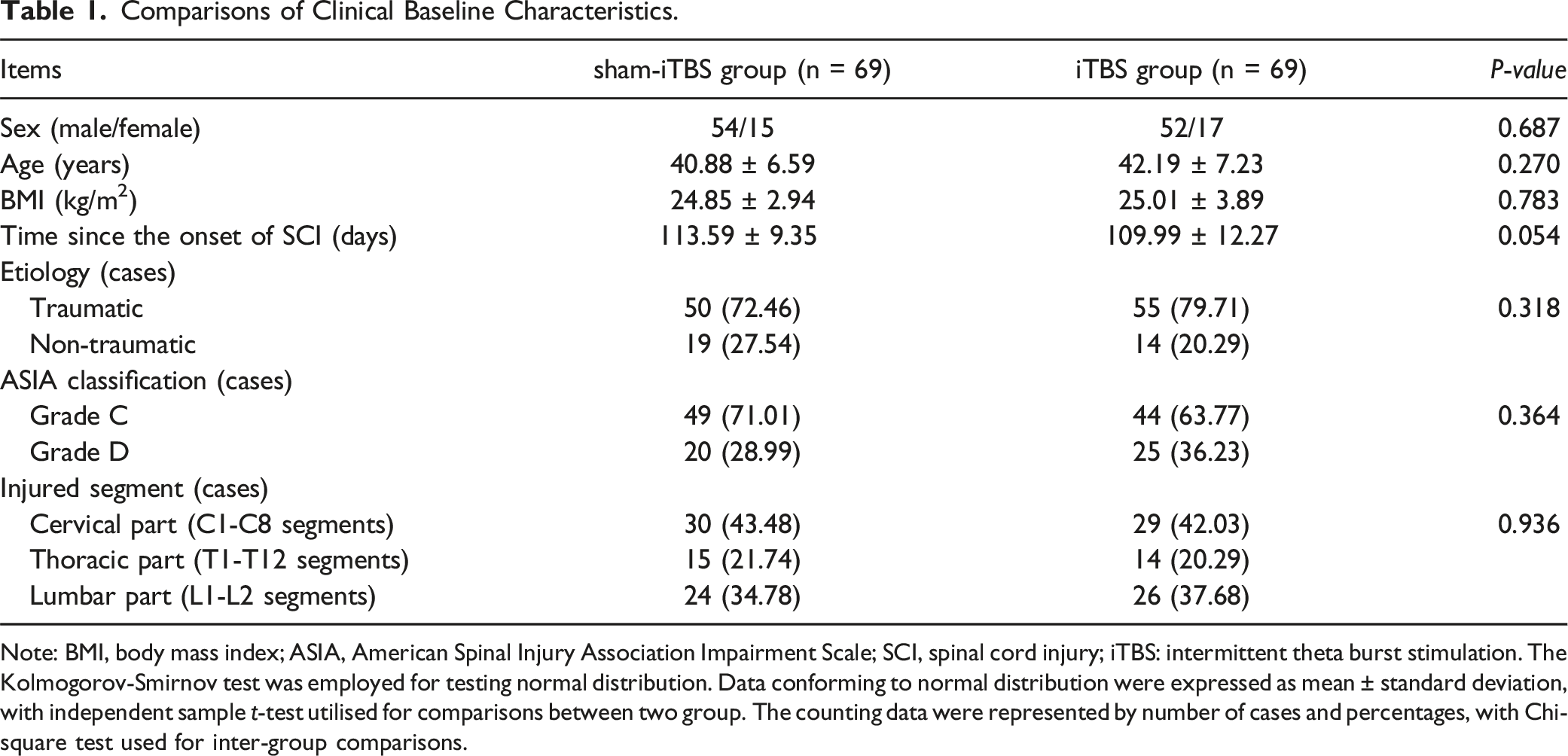
Note: BMI, body mass index; ASIA, American Spinal Injury Association Impairment Scale; SCI, spinal cord injury; iTBS: intermittent theta burst stimulation. The Kolmogorov-Smirnov test was employed for testing normal distribution. Data conforming to normal distribution were expressed as mean ± standard deviation, with independent sample t-test utilised for comparisons between two group. The counting data were represented by number of cases and percentages, with Chi-square test used for inter-group comparisons.
iTBS Prominently Attenuated Lower Limb Muscle Strength in iSCI Patients
Comparisons of Lower Extremity Muscle Strength.

Note: Normal distribution test was performed by the Kolmogorov-Smirnov test, with non-normally distributed measurement data expressed as quartiles [median (maximum, minimum)]. Inter-group comparisons were performed using the Mann-Whitney U test, and the intragroup comparisons pre- and post-treatment were performed using the Wilcoxon signed-rank test.
Comparisons of Gait Parameters and Walking Ability of iSCI Patients before and after Treatment
We evaluated the WS and SL of the lower extremities of iSCI patients before and after treatment and the HWAS score. The findings disclosed that the differences in WS, SL and HWAS scores of the two groups before treatment were not statistically significant (all P > 0.05). Following treatment, the WS, SL and HWAS scores of all patients were conspicuously higher than those before treatment (all P < 0.001), and those in the iTBS group were noteworthily higher than the sham-iTBS group (all P < 0.001), as shown in Figure 1. Comparisons of gait parameters and walking ability of iSCI patients pre- and post-treatment. A: Comparison of WS between the two groups; B: Left SL was compared between the two groups; C: Comparison of right SL between the two groups; D: Comparison of HWAS score between the two groups. Data were expressed as mean ± standard deviation, n = 69. Independent sample t-test was employed for inter-group comparisons, followed by intragroup comparisons pre- and post-treatment using paired t-test. ***P < 0.001.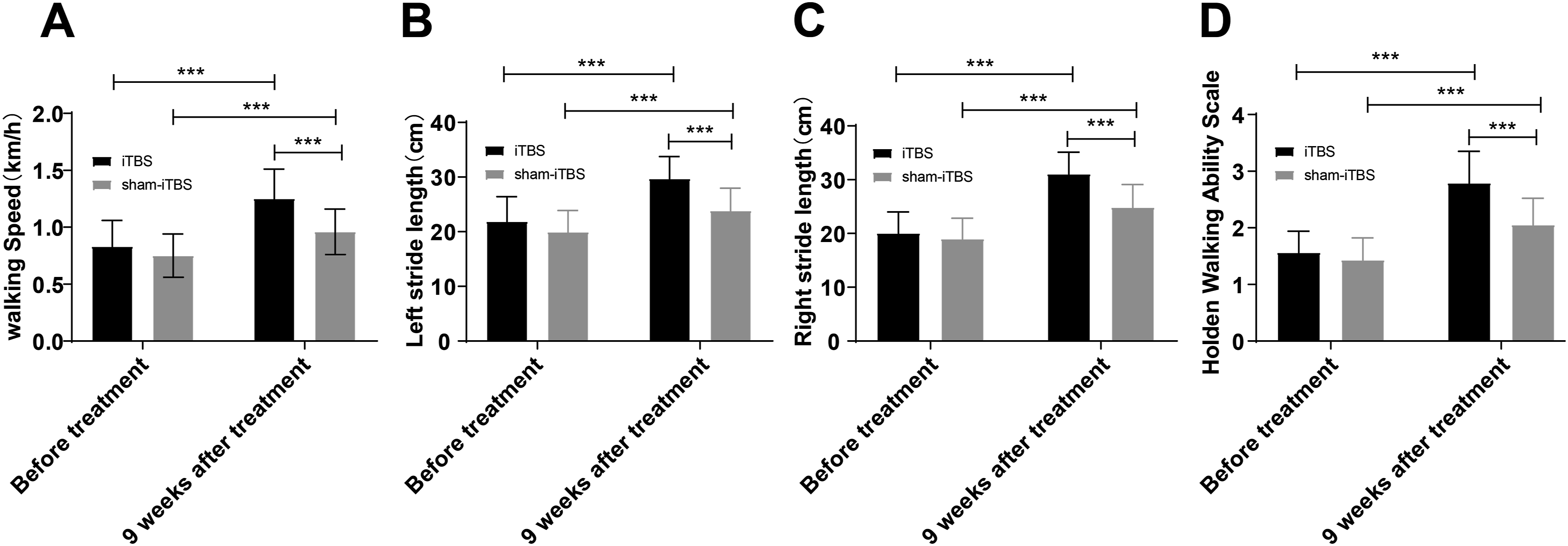
Comparison of Balance Function of iSCI Patients before and after Treatment
We further evaluated the balance function of iSCI patients before and after treatment, including the static eyes-open center of gravity locus length and static eyes-closed center of gravity locus length, as well as the BBS score. No statistically significant differences were found regarding the static eyes-open center of gravity locus length and static eyes-closed center of gravity locus length, as well as BBS scores between the two groups before treatment (all P > 0.05). After treatment, the static eyes-open center of gravity locus length and static eyes-closed center of gravity locus length of all patients were prominently lower than those before treatment, and BBS scores were elevated (all P < 0.001, Figure 2). Meanwhile, the static eyes-open center of gravity locus length and static eyes-closed center of gravity locus length were apparently lower (all P < 0.05, Figure 2A-B), while the BBS score was higher (P < 0.05, Figure 2C) in the iTBS group than the sham-iTBS group. Comparison of balance function in iSCI patients before and after treatment. Data were denoted as mean ± standard deviation, n = 69. Inter-group comparisons were carried out using an independent sample t-test, followed by intragroup comparisons pre- and post-treatment using a paired t-test. *P < 0.05, ***P < 0.001.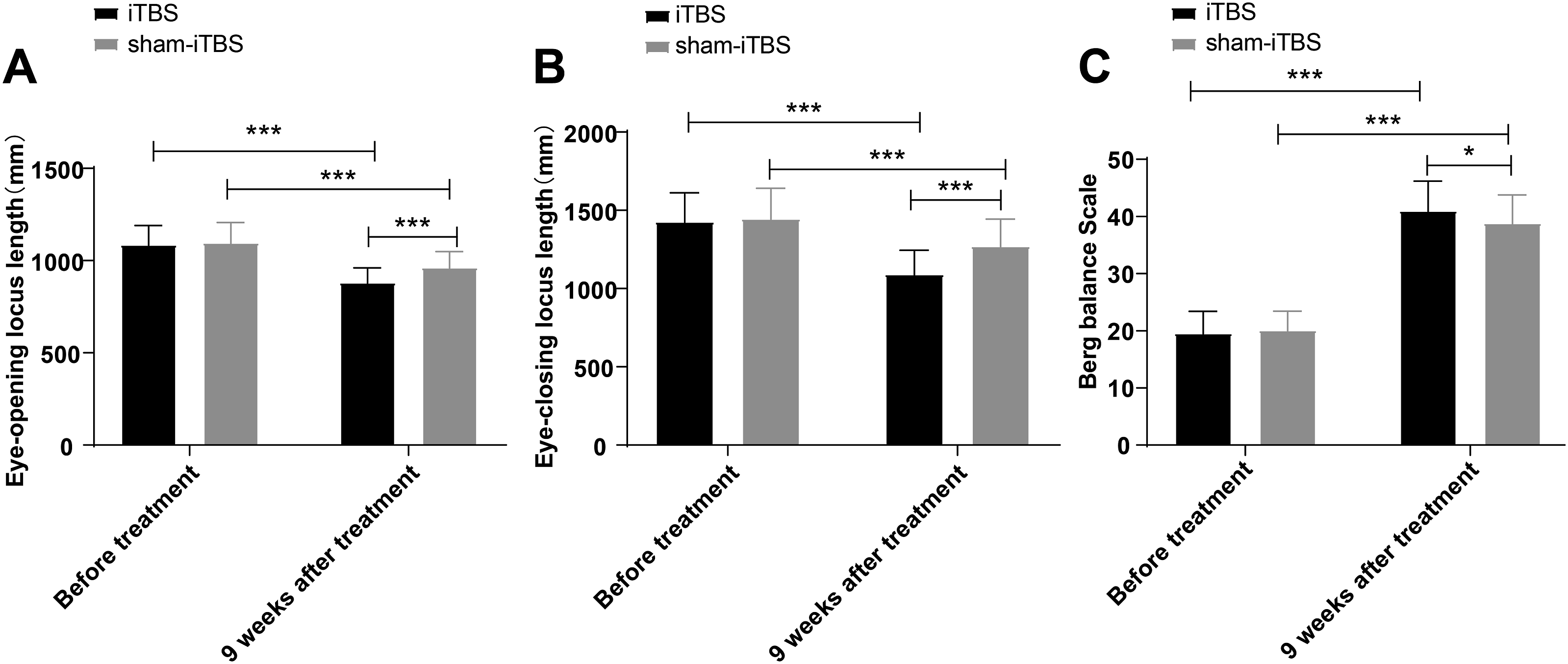
Comparisons of Lower Extremity Motor Function and Daily Living Activity Ability in iSCI Patients Pre- and Post-Treatment
In addition, we used ASIA-LEMS to evaluate the lower extremity motor function of patients before and after treatment, with the MBI scale conducted to evaluate the ability of daily living activities of patients before and after treatment. The findings elicited no statistically significant differences in the LEMS and MBI scores between the two groups before treatment (all P > 0.05), but elevations in the LEMS and MBI scores in all patients following treatment vs before treatment (all P < 0.001), with obvious enhancements in the LEMS and MBI scores in the iTBS group relative to the sham-iTBS group (all P < 0.001, Figure 3). LEMS and MBI scores of iSCI patients were compared before and after treatment. A: LEMS score was compared between the two groups; B: Comparison of MBI score between the two groups. Data were presented as mean ± standard deviation, n = 69, with independent sample t-test utilised for inter-group comparisons, and paired t-test used for intragroup comparisons between pre- and post-treatment. ***P < 0.001.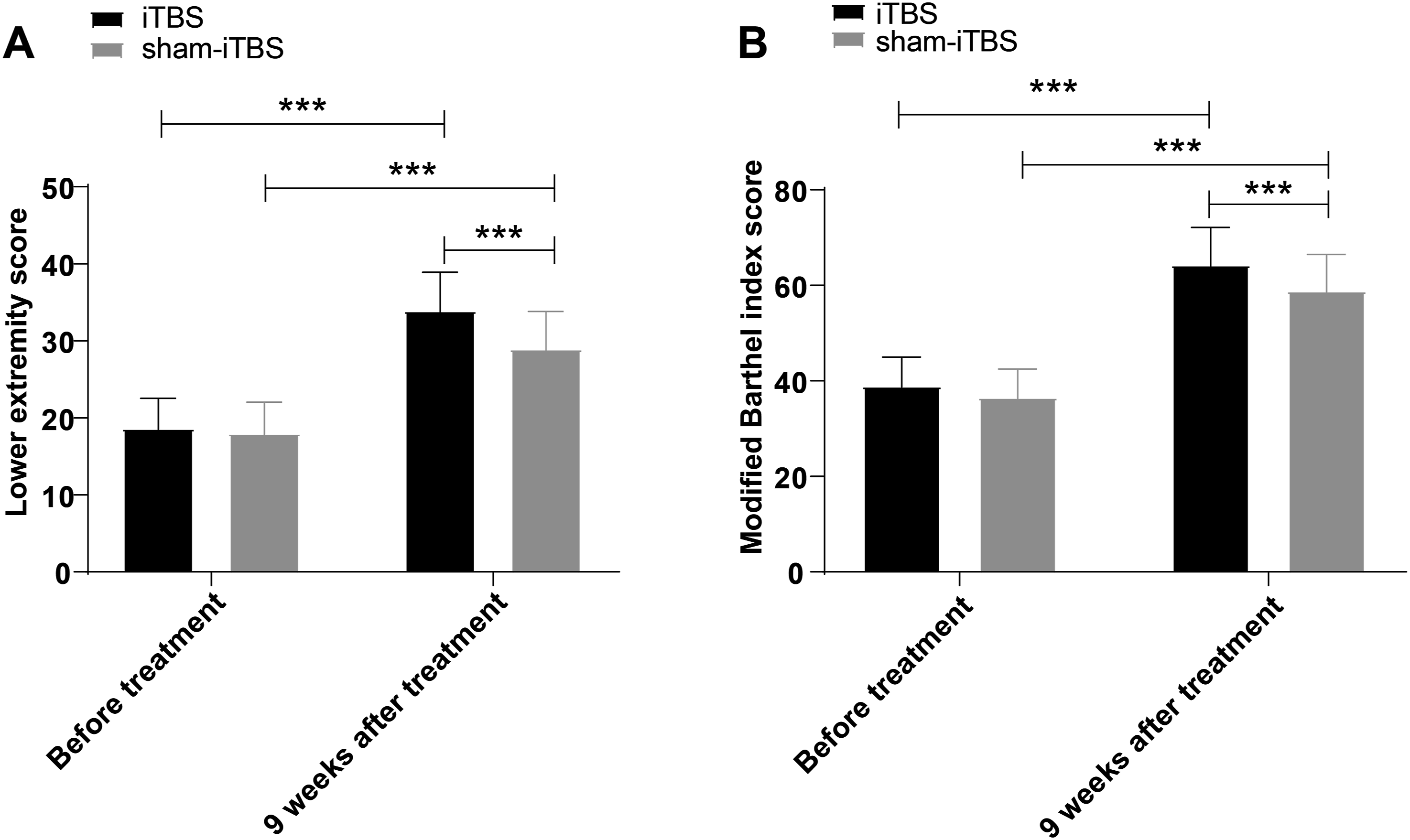
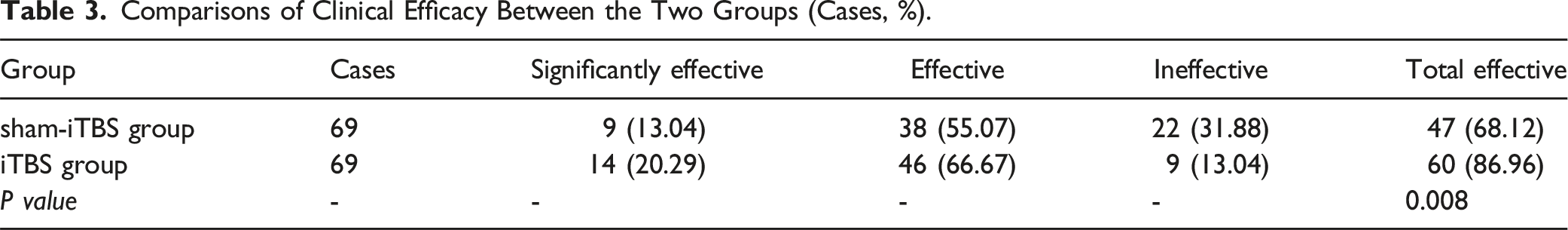
Comparison of Clinical Efficacy Between the Two Groups
Comparisons of Clinical Efficacy Between the Two Groups (Cases, %).
Discussion
SCI leads to the impairment of autonomic, motor, and sensory functions, which almost invariably induces permanent physical disability, with a dynamic range of global prevalence of 11 to 16 cases per 100000 people. 36 It is reported that 81% of SCI patients undergo chronic pain and 86% of patients have neuropathic pain, posing a heavy burden on their quality of life and daily living. 37 Existing evidence suggests that iTBS effectively decreases neuropathic pain associated with SCI. 17 However, there are few reports on the efficacy of iTBS on motor and walking functions in patients with iSCI. Our findings highlighted that iTBS had remarkable therapeutic effects on iSCI patients, which could dramatically ameliorate the lower limb muscle strength and lower limb motor function of iSCI patients, thereby improving their walking function and their ability of daily life activities.
Reportedly, three months after injury, the musculature of iSCI patients atrophy by 10%, and after four to 5 months progress to a significant muscle loss, highlighting that restoring the volume and strength of lower limb muscle is of vital importance for iSCI patients as it ameliorates daily living activity performance and over-ground walking to a higher quality. 38 At present, the effects of TBS in SCI individuals have been disclosed on the mitigation of muscle tension in upper extremity muscles; repetitive TMS ameliorates the function of upper extremity muscle motor units, relieves the muscle tension in upper extremity muscles, and marginally strengthens the transmission of efferent neural impulses within the spinal pathways, and moreover, restrains unavoidable neural pathological changes. 12 However, the role of iTBS in protecting against iSCI is not fully understood. Our findings revealed that the patients treated with iTBS manifested more significant elevation in the levels of muscle strength after treatment than patients treated with sham-iTBS. In a similar light, iTBS plays a role in raising the corticomotor excitability of hand muscles in SCI patients, and also heightens the excitability of biceps corticomotor in patients with tetraplegia. 39 Conjointly, iTBS demonstrates efficacy in improving the lower limb muscle strength of iSCI individuals.
iSCI patients regain or retain the ability to walk with limited gait parameters. 40 iSCI often leads to impaired balance resulting in functional impairments, and restoration of standing balance function is a vital goal of the rehabilitative programs. 41 Recovery of mobility, including recovery of walking function, is expected to ameliorate the quality of life and the ability to perform daily living activities.42,43 From these aspects, gait is a latent non-intrusive biomarker that mirrors an individual’s movement features, and standard gait parameters consist of cadence, WL and SL. 44 ASIA-LEMS is a well-known measure to concern the MMT of 5 vital muscles in the lower extremity that have correspondence to the L2-S1 spinal cord segment. 4 Besides, MBI has shown good effectiveness, sensitivity, as well as reliability in the evaluation of activities of daily living of Chinese stroke patients. 45 Thus, we explored the impacts of iTBS on the gait parameters, walking ability, balance function, lower extremity motor function and ability of daily living activities of iSCI patients, and our results displayed that after treatment, the WS, SL, HWAS, BBS, LEMS, and MBI scores of all patients prominently rose, and the static eyes-open and eyes-closed center of gravity locus length declined, all of which were more noticeable in patients treated with iTBS. Consistently, the intervention of 9-week iTBS produces positive impacts on the HWAS in iSCI patients, as well as MBI. 15 iTBS stimulation may influence the endogenous coupling between the SMA and the primary motor cortex (M1), 21 and increase the functional connectivity of bilateral frontal lobe-striatum. 46 Additionally, iTBS stimulation may improve motor and walking functions in patients with iSCI by enhancing the excitability of the posterior parietal cortex to promote cerebellar cortical reorganization, 47 as well as by facilitating the molecular signaling transduction involved in CST growth and MEP plasticity. 20 Furthermore, the cerebellar iTBS can facilitate and accelerate recovery, particularly the balance function and gait, which can be an efficient and facilitative treatment for patients with stroke. 48 Koch et al have declared that cerebellar iTBS ameliorates balance and gait function, and advances the excitability of the posterior parietal cortex in individuals with chronic stroke. 47 Precise root stimulation combined with cortical iTBS contributes to ameliorating lower limb motor function recovery and nerve conduction of the corticospinal tract in chronic SCI individuals. 8 Feng et al. have obtained that physiotherapy combined with cerebral iTBS encourages the recovery of lower extremity motor in individuals with iSCI. 15 Evidence also shows that iTBS possesses the ability to advance the quality of life and overall function in individuals with Parkinson’s disease by physiotherapy, with this efficacy related to modified neuroplasticity in exercise-associated brain regions. 49 Furthermore, Liao et al. have found that iTBS is capable of facilitating post-stroke patients’ balance and motor recovery and improving the rehabilitation of poststroke. 25 Finally, we confirmed that the total effective rate for patients with iTBS treatment was 86.96%, distinctly higher than sham-iTBS treatment. To support this, iTBS demonstrates its feasibility and preliminary efficacy for posttraumatic stress disorder on clinical symptoms. 22 In this study, we adopted a randomized, double-blind, and sham-controlled design to investigate the effect of iTBS on the lower limb motor function in patients with SCI, which was consistent with previous research.13,15,50 However, our study included a larger sample size (n = 138), a longer intervention period (9 weeks), and a more representative patient population, thereby enhancing the reliability of both data statistics and research results. Additionally, our study only focused on incomplete SCI patients, with an emphasis on the lower limb function and a higher overall effectiveness rate.
In summary, this study affirms that iTBS is effective for the treatment of iSCI, and it results in ameliorated muscle strength and motor function of lower limbs in iSCI patients, along with improved ability to perform daily life activities and walking function. Nevertheless, this study does not measure corticospinal excitability in iSCI patients and lacks an investigation of the potential mechanisms of iTBS. Although the total sample size (n = 138) met the comparison requirement of two groups, due to the small sample size, there was no in-depth study on the differences in key subgroups such as age, sex, race, and iSCI category (traumatic or non-traumatic iSCI), which might limit the evaluation of the specific efficacy of iTBS on spinal cord injury and the selection of clinical individualized treatment options. Furthermore, our study only focused on the short-term effects of 9 weeks after treatment and lacked long-term stability verification of functional recovery, such as future efficacy attenuation or dynamic changes of neuroplasticity in some patients. Future studies with larger sample sizes and extended follow-up durations are warranted to further validate the long-term efficacy of iTBS. In addition, we adopted a unified stimulation intensity and parameters in this study, without dynamical adjustment based on individual neural excitability thresholds, which might reduce the response rate of some patients. These are the limitations of this study. In the future, multi-center, large-sample randomized, double-blind controlled clinical trials are needed for further validation. Future research should integrate multimodal assessments (e.g., functional MRI, TMS-EEG, fNIRS) to quantitatively analyze the characteristics of the sensory-cortical-motor loop, providing a basis for the sustainability of individualized iTBS scheme optimization and functional recovery. In general, although iTBS may be preferred for iSCI patients with partial preserved neural conduction, it is necessary to exclude patients who are sensitive to magnetic stimulation (e.g., those with a history of epilepsy) or those with implanted electronic devices (e.g., metal foreign objects, pacemakers, or cochlear implants) from clinical applications. In addition, iTBS can provide clinical non-invasive, and efficient rehabilitation interventions for iSCI patients. Compared with traditional care, the iTBS stimulation mode can apply the same amount of pulse stimulation to the target area in a shorter time. An efficient stimulation mode is more helpful in improving patient compliance so that patients can better tolerate the treatment process and receive enhanced rehabilitation effect. Meanwhile, the use of iTBS has greatly improved the patients’ functional independence, reduced their dependence on drugs and physical aids, reduced home care needs and clinical care costs, and thus reduces family and social burdens. Consequently, iTBS holds promise for playing a pivotal role in future rehabilitation therapies.
Supplemental Material
Supplemental Material - Efficacy of Excitatory iTBS on Lower Limb Recovery in iSCI Patients
Supplemental Material for Efficacy of Excitatory iTBS on Lower Limb Recovery in iSCI Patients by Shengliu Shi, Jingyu Yao, and Jing Cai in Global Spine Journal
Footnotes
Authors’ Contributions
Guarantor of integrity of the entire study: Shengliu Shi; study concepts: Shengliu Shi; study design: Shengliu Shi; definition of intellectual content: Shengliu Shi; literature research: Shengliu Shi; clinical studies: Jingyu Yao;experimental studies: Jingyu Yao; data acquisition: Shengliu Shi; data analysis: Shengliu Shi; statistical analysis: Jing Cai; manuscript preparation: Jing Cai; manuscript editing: Jing Cai; manuscript review: Jing Cai.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
