Abstract
Objective
To undertake an updated meta-analysis to obtain more evidence from randomized controlled trials (RCTs) to assess the effect of repetitive transcranial magnetic stimulation (rTMS) for the treatment of tinnitus.
Methods
PubMed®, Embase®, Web of Science, Cochrane Database of Systematic Reviews, CBM, CNKI and Wanfang were searched for RCTs from inception up to March 2020. Studies meeting the eligibility criteria were included in the meta-analysis. The mean difference was calculated and the effect size was evaluated using a
Results
The analysis included 12 randomized sham-controlled clinical trials with a total of 717 participants. Active rTMS was superior to sham rTMS in terms of the short-term and long-term effects (6 months) on the tinnitus handicap inventory scores, but an immediate effect was not significant. There was no significant immediate effect on the tinnitus questionnaire (TQ) and Beck depression inventory (BDI) scores.
Conclusions
This meta-analysis demonstrated that rTMS improved tinnitus-related symptoms, but the TQ and BDI scores demonstrated little immediate benefit. Future research should be undertaken on large samples in multi-centre settings with longer follow-up durations.
Introduction
Tinnitus is considered as the perception of sound without an external source and different brain areas may be involved in this different perception.1,2 Chronic tinnitus is a disabling, almost incurable disease that can cause attention or sleep disturbances, even severe anxiety and depression.3,4 Furthermore, epidemiological studies show that the incidence of tinnitus in the adult population is 10–15%; 5 and most patients suffer from a certain degree of hearing loss, which seriously affects quality of life.6,7
In early research, tinnitus was thought to be due to the hyperactivation and neural synchronization of the auditory cortex.8,9 Some recent studies have found that the changes in neuronal activity and synaptic plasticity of the auditory cortex and non-auditory cortex play an important role in improving tinnitus.10–14 Although not fully elucidated, various attempts have been implemented to modulate changes in neuronal activity, particularly repetitive transcranial magnetic stimulation (rTMS), which has emerged as a potential option and has become the focus of clinical research in tinnitus.15,16 As a proven safe and non-invasive brain stimulation technique, rTMS regulates the activities of related cerebral cortex by using the rhythmic application of short magnetic pulses delivered by a coil placed on the scalp. The positive effects of rTMS are probably caused by the complex interactions between the associated brain network and the stimulation target of tinnitus in terms of mood, attention and perception.14,17–19 A previous study found that the application of rTMS significantly improved the severity of tinnitus and was well tolerated. 20 In addition, a second application for patients whose tinnitus worsened during the treatment interval was particularly promising. 20 Compared with sham rTMS, real stimulation had a significant suppressive effect on tinnitus and the magnetoencephalogram showed that the oscillation power of the alpha band increased after rTMS treatment. 21 However, due to differences in the optimal stimulation target, parameters, range and time of rTMS in the treatment of tinnitus, the definitive therapeutic effect of rTMS on tinnitus remains controversial.22–24
In 2016, a meta-analysis on tinnitus was published and concluded a moderate efficacy of low-frequency rTMS for chronic tinnitus. 25 In recent years, some studies of rTMS in patients with tinnitus that included relatively large sample sizes were published,26–28 but several different conclusions were reported. Therefore, an updated meta-analysis was conducted based on the latest research with the aim of finding more evidence to evaluate the efficacy of rTMS for the treatment of tinnitus.
Materials and methods
Study methods
This meta-analysis was conducted following the recommendations of the PRISMA guidelines. Two investigators (L.Y. & X.C.) performed independent selections of the articles and data extraction. Any discrepancies were resolved by consensus. The present meta-analysis was registered at the International Prospective Register of Systematic Reviews (PROSPERO: CRD42019145538.).
Search strategy
Electronic databases, including PubMed®, Embase®, Web of Science, Cochrane Database of Systematic Reviews, CBM, CNKI and Wanfang, were searched from inception to March 2020, to identify relevant studies. The keywords used in literature search were as follows: (i) “Transcranial Magnetic Stimulation*”; (ii) “Magnetic Stimulation*, Transcranial”; (iii) “Stimulation*, Transcranial Magnetic”; (iv) “TMS”; (v) “rTMS”; (vi) “Theta-burst”; (vii) “Tinnitus”; (viii) “Ringing”; (ix) “Buzzing”; (x)“Phantom Sound*”. The detailed retrieval strategy was performed as follows: ((((((((Transcranial Magnetic Stimulation*[MeSH Terms]) OR Transcranial Magnetic Stimulation*[Title/Abstract]) OR Magnetic Stimulation*, Transcranial[Title/Abstract]) OR Stimulation*, Transcranial Magnetic[Title/Abstract]) OR TMS[Title/Abstract]) OR rTMS[Title/Abstract]) OR theta-burst[Title/Abstract])) AND (((((tinnitus[MeSH Terms]) OR tinnitus[Title/Abstract]) OR Ringing[Title/Abstract]) OR buzzing[Title/Abstract]) OR phantom sound*[Title/Abstract]). An English language restriction was imposed. Study selection was based on an initial screening of identified abstracts or titles and a second screening of full-text articles. The reference lists of relevant review articles and meta-analyses were examined to identify other potentially eligible studies.
Eligibility criteria
The eligibility criteria were as follows: (i) participants were adults diagnosed with tinnitus; (ii) the intervention was rTMS; (iii) comparison between sham rTMS or normal care or blank; (iv) the outcomes tinnitus handicap inventory (THI), tinnitus questionnaire (TQ) and Beck depression inventory (BDI) were used to evaluate the severity of tinnitus and other physical or psychological symptoms; (v) studies with a randomized controlled design; (vi) provided data (on the manuscript or upon request) for the estimation of the outcomes, i.e. mean (±SD) values. Case reports, non-efficacy assessment and trials assessing conditions other than tinnitus or interventions other than rTMS were excluded.
Data extraction
Two investigators (L.Y. & X.C.) extracted the following variables from the studies: (i) basic information (i.e. authorship, publication date); (ii) participant characteristics (i.e. sample size, mean age, mean duration of tinnitus); (iii) parameters of rTMS (i.e. stimulation frequency, targets, pulses, days of treatment, motor threshold); (iv) primary outcomes that were assessed before and after treatment by changes in overall tinnitus severity and other physical or psychological symptoms. Patient questionnaires included the THI, TQ and BDI;29–31 (v) research methods (i.e. randomization protocol, sham technique, blinding assessment). Authors were contacted and invited to send additional data if publications involved insufficient or incomplete results.
Quality assessment
The methodological quality of each trial was assessed according to the Cochrane Bias Tool: (i) methods of randomization; (ii) the use of allocation concealment; (iii) blinding of participants and outcome assessment; (iv) integrity of outcome data; (v) selective reporting; (vi) other bias.
Statistical analyses
All analyses were performed using RevMan software (version 5.3; Cochrane Collaboration, Oxford, UK). For continuous data, the mean difference (MD) was calculated for between pre- and post-treatment in the separate conditions and the effect size of the MD was evaluated using a
Heterogeneity was assessed using
Results
A flow chart showing the study selection process is presented in Figure 1. The initial database search identified 1120 studies, of which 859 remained after duplicates were removed. Of these, 12 studies with 717 participants were eligible for inclusion in the meta-analysis.21,26–28,35–42
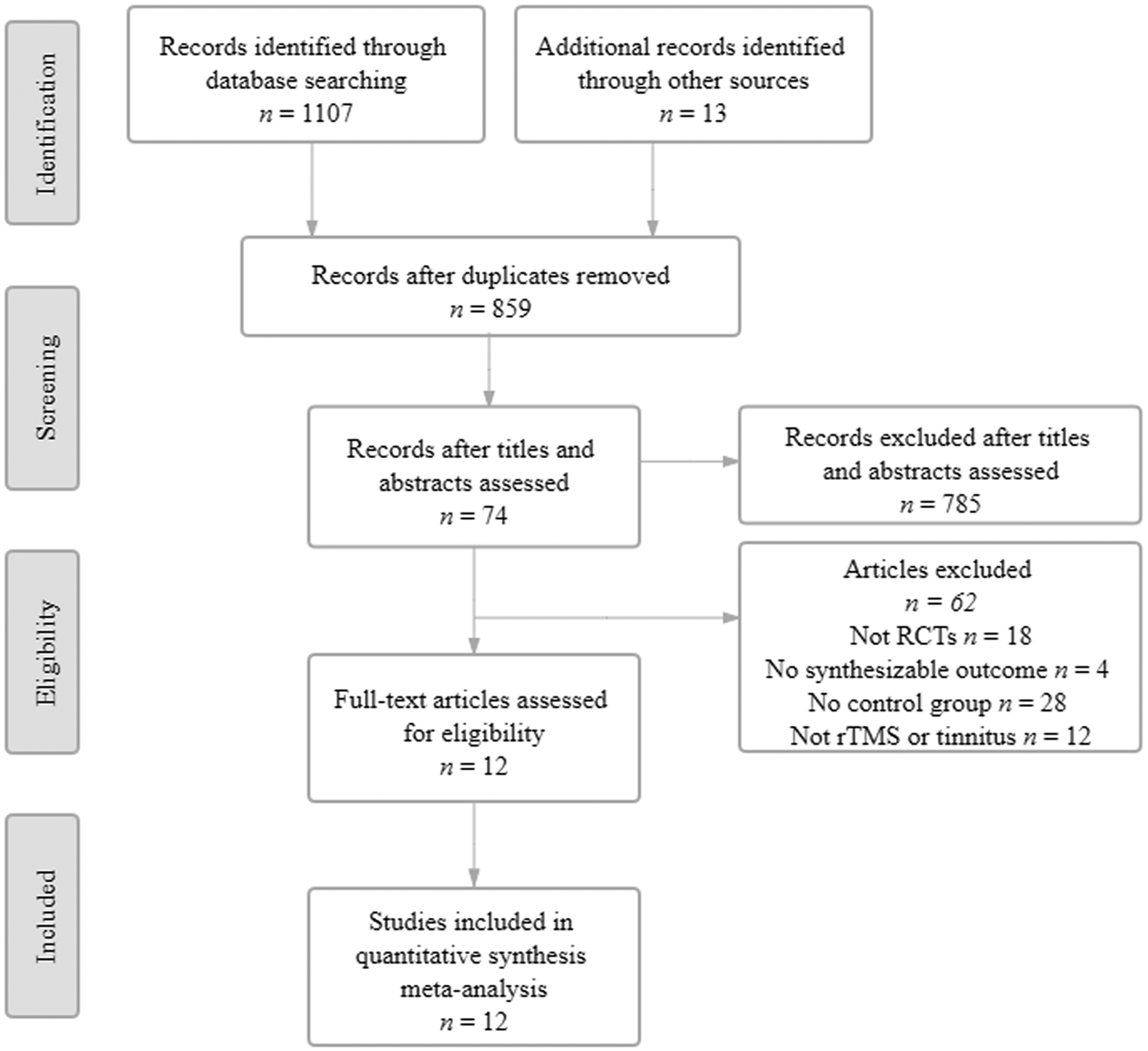
Flow diagram of eligible studies showing the number of citations identified, retrieved and included in the final meta-analysis. RCT, randomized controlled trial; rTMS, repetitive transcranial magnetic stimulation.
The bias risk assessment is presented in Figure 2. In the 12 included studies,21,26–28,35–42 the randomization method was described specifically in eight (67%), allocation concealment was performed in three (25%) and the blinding was reported in eight (67%). Incomplete outcome data had an overall low risk of bias, but the unclear risk of bias mainly occurred in selective reporting and other bias.

In this meta-analysis, an overall short-term effect on THI was calculated post-rTMS. This result showed the MD of active rTMS was –7.05 (95% confidence interval [CI] –11.65, –2.44), which was superior to sham rTMS (

For both TQ27,41,42 and BDI,27,28,39 three trials were included in the meta-analysis to evaluate the immediate effect of rTMS compared with sham interventions. The MD was –2.50 (95% CI –5.68, 0.69) for the TQ score and –0.21 (95% CI –1.85, 1.43) for the BDI score, which were insignificant effect sizes for both outcomes following active rTMS (Figure 4).
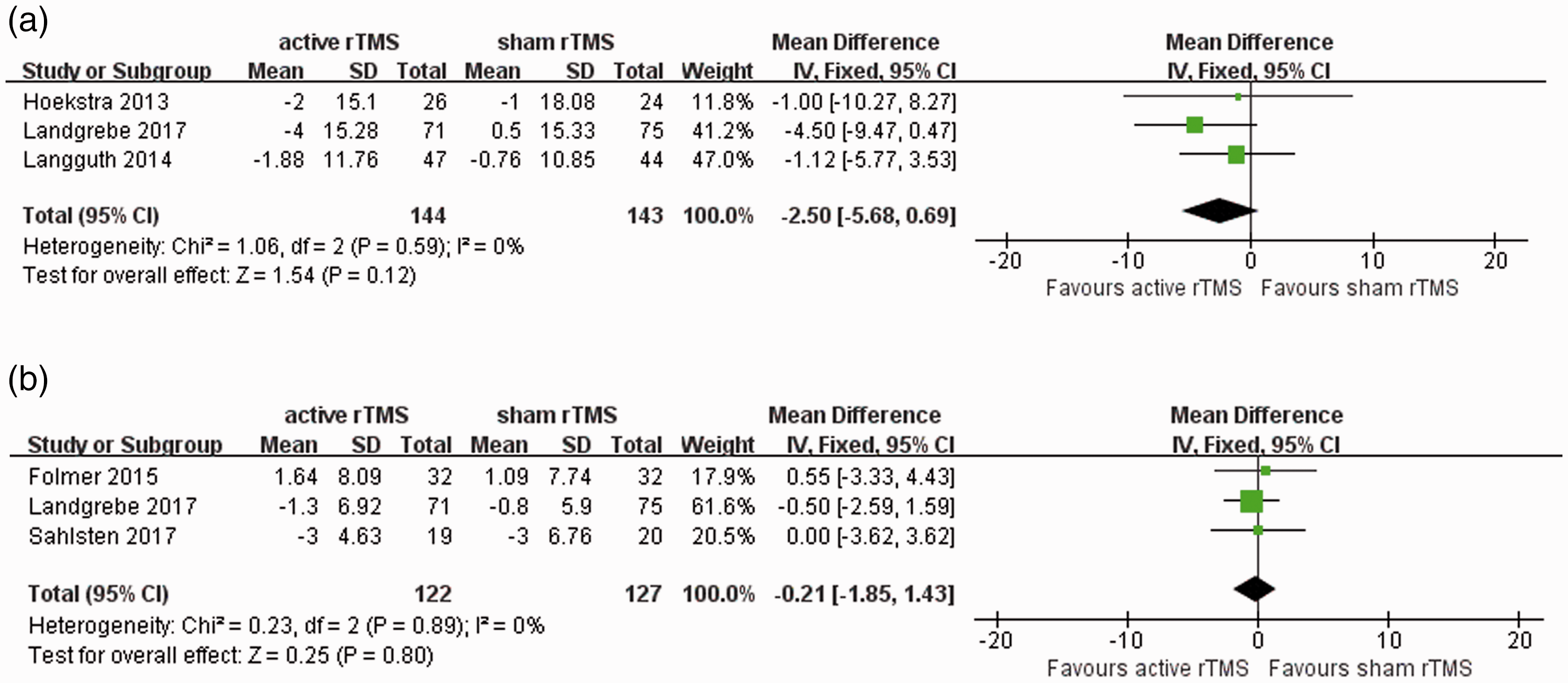
A high degree of heterogeneity was observed among studies analysed for the short-term (

Funnel plot of studies included in this meta-analysis in order to assess publication bias. The included studies were mainly focused on the top of the funnel and symmetrically distributed on both sides, indicating that there was a low publication bias. The analysis included the 11 studies that used the tinnitus handicap inventory score.21,26–28,35–41
The short-term post-rTMS effect on the THI score showed that active rTMS was superior to the sham rTMS, but the heterogeneity was relatively high. A subgroup analysis was conducted based on the geographical regions of the study participants. The results indicated that both subgroups had significant findings: studies from the Chinese mainland (MD –17.14; 95% CI –21.01, –13.27;
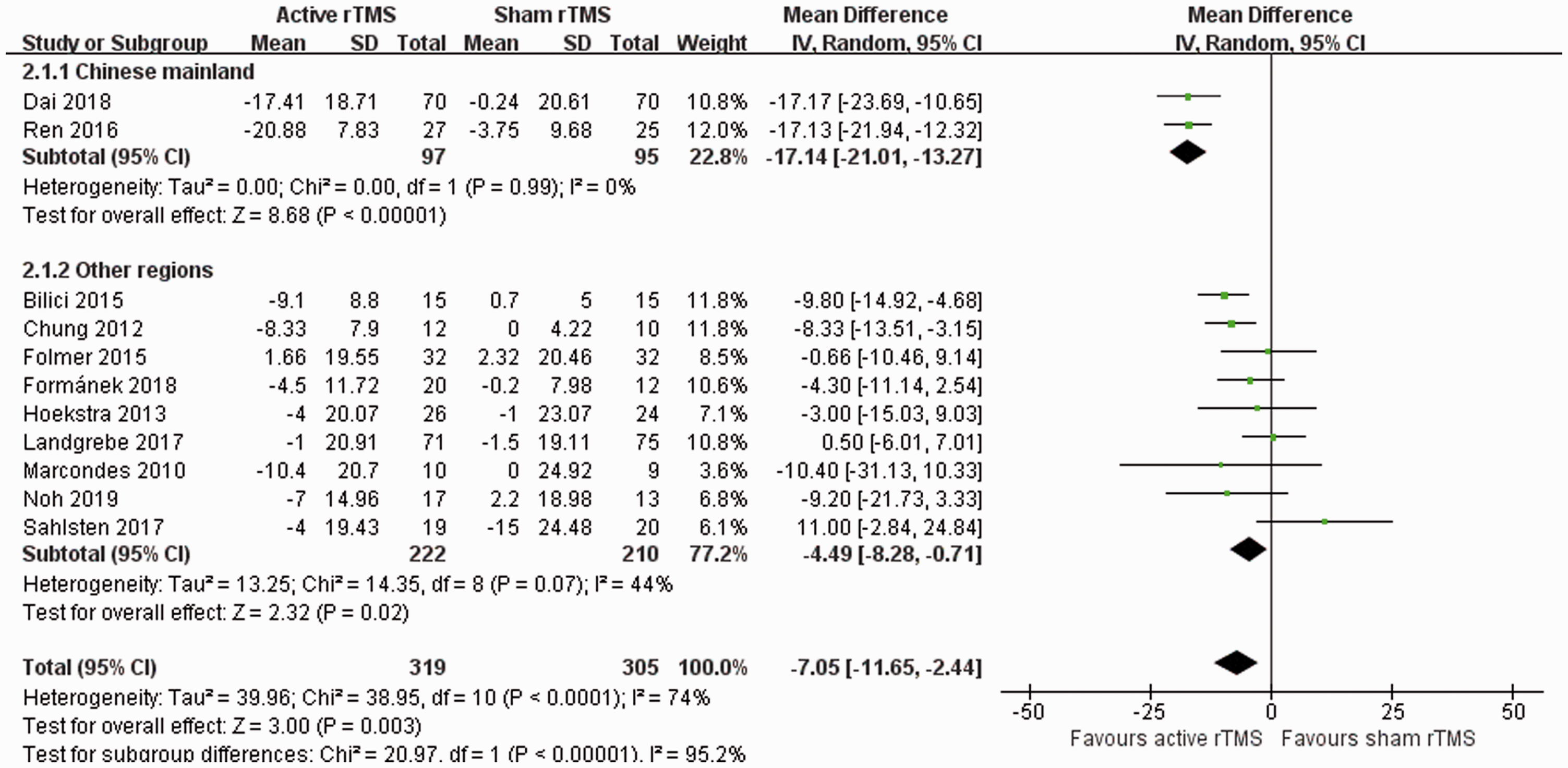
Forest plots of subgroup meta-analyses to evaluate the effect of repetitive transcranial magnetic stimulation (rTMS) on the post-rTMS tinnitus handicap inventory score based on the geographical regions of the study participants. The analysis included the 11 studies that used the tinnitus handicap inventory score.21,26–28,35–41
Discussion
Given that the results obtained by several randomized controlled trials were not completely consistent,26–28 using rTMS as a treatment for tinnitus remains a controversial and long-standing issue. Compared with a previous meta-analysis, 25 the current study added seven trials and conducted a meta-analysis of 12 randomized sham-controlled clinical trials that aimed to determine the efficacy of rTMS for tinnitus. The current meta-analysis found that the use of active rTMS on patients with tinnitus showed a meaningful short-term effect on the THI score; and it had a positive effect on the THI assessment at 1 and 6 months. These findings suggest that rTMS had a possible long-term effect up to 6 months. Furthermore, the analyses demonstrated an increased value of MD between the active rTMS and sham groups over time. In contrast, the immediate effect of rTMS on the TQ and BDI scored was insignificant.
The previous meta-analysis from 2016 reported moderate efficacy of low-frequency rTMS as a treatment for chronic tinnitus, in which the treatment outcomes were evaluated by TQ and THI scores. 25 This current meta-analysis showed that active rTMS had a positive effect at 1 and 6 months after the intervention compared with sham rTMS, which was in accordance to the previous meta-analysis. 25 The MD in THI at 1 month decreased from –6.71 in the previous meta-analysis to –6.81, 25 but among the three newly added trials,21,26,28 only one showed clinically relevant changes in THI. 21 The MD of THI at 6 months increased from –12.89 in the previous meta-analysis to –7.01, 25 but none of the three newly added trials showed a clinically relevant effect on THI.26–28 In addition, this current meta-analysis also analysed the short-term and immediate effects of rTMS on the THI scores. There was no significant immediate effect of rTMS on the THI scores, which might have been due to the high levels of heterogeneity among the available studies. The subjectivity of the THI scale might have been an important source of bias. For the short-term effects of rTMS, the MD of the included 11 studies was –7.05, which demonstrated that rTMS had a positive clinical effect on the THI scores.
Physiological and psychological problems are often associated with moderate-to-severe tinnitus, with 10–60% of patients with chronic tinnitus experiencing major depression, 43 so the current meta-analysis took account of the TQ and BDI scores to evaluate any improvement in the patients’ psychological problems. 44 However, the observation that there was no difference in the in TQ and BDI scores between active rTMS and sham treatment immediately after the intervention was confusing. These current findings suggest that rTMS had little benefit in improving the psychological problems of patients with tinnitus. These current results might, in part, be explained by the small number of studies that were included in the analysis. Another possible explanation for these findings was that the THI, TQ and BDI scores may have different sensitivities and specificities in assessing tinnitus severity. 45 Additionally, the long-term effect of rTMS on TQ and BDI remains to be verified, which suggests that the duration of follow-up should be increased in future studies.
The funnel plot showed that the studies were mainly distributed at the top of the funnel and symmetrical on both sides of the plot, indicating that there was low publication bias, which had little effect on the results of meta-analysis.46–48 However, the results of this study should be interpreted with caution. Firstly, approximately one-third of studies were unclear in their reporting of random sequence generation36–39 and one-third were considered at high risk of bias about blinding.28,35,37,40 In addition, three-quarters of the studies were classified as having an unclear risk of bias in allocation concealment,26,28,35–40 which could result in an overestimated treatment effect of active rTMS. Secondly, relatively high heterogeneity was observed for the studies included in the analysis of the short-term effect of rTMS on THI. Sensitivity analysis found that two studies were the main source of the heterogeneity.35,40 Therefore, subgroup analysis was performed and showed only small-to-moderate heterogeneity, which may be due to differences in the patients' constitution, living environment and lifestyle as a result of them living in different geographical regions. Alternatively, unreported blinding and allocation concealment could have been the source of heterogeneity. Other factors that might have influenced these results were differences in the baseline characteristics between the included trials, such as age, disease severity and duration of tinnitus; and differences in the various rTMS parameters such as the brain region that was stimulated, frequency of stimulation, number of pulses and treatment duration. Similarly, the high level of heterogeneity observed for the studies included in the analysis of the immediate effects of rTMS on THI was due to one article. 35
Characteristics of the randomized clinical trials (

MT, motor threshold; THI, tinnitus handicap inventory; LTPC, left temporoparietal cortex; TQ, tinnitus questionnaire; AC, auditory cortex; LT, left temporal; LTP, left temporoparietal; TFI, tinnitus functional index; RT, right temporal; VAS, visual analogue scores; LAC, left auditory cortex; LTC, left temporal cortex; BDI, Beck depression inventory; L-DLPFC, left dorsolateral prefrontal cortex; PAC, primary auditory cortex; TPC, temporoparietal cortex; NA, not available.
Footnotes
Acknowledgement
We would like to thank Dr Shujie Ma, Second Rehabilitation Hospital of Shanghai, for her help with the language used in this manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by the National Science Foundation for Young Scientists of China (no. 81603713).
