Abstract
Objective
To evaluate the effectiveness and safety of the MI-R1 lower-limb walking assist device for gait training in subacute stroke patients.
Methods
The experimental group received lower-limb function training using the lower-limb training device. In contrast, the control group received lower-limb function training provided by a therapist. After 4 weeks, the patients were evaluated for certain indicators. The primary outcome was the Fugl–Meyer Assessment Lower Extremity score on day 28 (±7 days). The secondary outcomes included the Functional Ambulation Category score, Modified Barthel Index score, safety indicators, and incidence of adverse events.
Results
After treatment, the noninferiority statistic between the experimental and control groups was −0.1976 (−1.5051, 1.1099). The lower limit of the 95% confidence interval was greater than the noninferiority margin. There was no significant difference in the Functional Ambulation Category or Modified Barthel Index scores between the experimental and control groups at 0, 14, and 28 days (p > 0.05). There was no statistically significant difference in vital signs or adverse event rates between the experimental and control groups (p > 0.05).
Conclusions
The MI-R1 lower-limb walking assist device is not inferior to traditional rehabilitation in achieving lower-limb function recovery in subacute stroke patients and is associated with good patient safety.
Introduction
Stroke is the second leading cause of disability and death worldwide. Approximately four of five patients retain varying degrees of neurological impairment after stroke, which not only causes limb motor dysfunction but also seriously affects their quality of life. 1 Despite optimal treatment efforts, >70% of stroke patients experience hemiplegia, and 60% develop lower-limb dysfunction. Most stroke patients continue to experience walking dysfunction at 6 months after stroke. Fewer than 20% of stroke survivors have access to community-based rehabilitation, 2 resulting in stroke being the main cause of adult disability and a considerable burden on families and society.3,4 Meanwhile, falls are a common complication in individuals with stroke. 5 Up to 70% of community-dwelling stroke survivors experience at least one fall annually. 6 The reported prevalence rates of falls and fall-related injuries in stroke survivors are 0.88 and 0.25 per person-year, respectively. 7 Falls in stroke survivors may result in physical injury or death, with fracture rates in this population being up to 4-fold higher than that in the general population. 8 Falls also impose a significant psychological, social (social deprivation), and economical (high medical costs) burden on stroke survivors and their caregivers.
Walking dysfunction and falls after stroke are commonly related to a lack of lower-limb muscle strength, lower-limb support, and balance dysfunction. Currently, traditional rehabilitation treatment can improve these functions through long-term, continuous training; however, such methods are associated with high labor costs. With increasing number of patients, physical therapists experience greater physical strain and fatigue, which may also affect the effectiveness of the rehabilitation treatment. In recent years, with continuous advancements in artificial intelligence technology, lower-limb walking assist rehabilitation training robots have been able to compensate for the limitations of traditional rehabilitation treatment. 9 This technology has achieved remarkable results in lower-limb and walking function training among hemiplegic stroke patients. 10 Based on the principles of neuroplasticity and motor learning, exoskeleton robots and functional aids to assist lower-limb function or walking training have been used in rehabilitation because they maximize sensory and motor inputs around the joint to provide specific stimulation to the central nervous system. Moreover, functional training aids can help provide functional compensation or training for patients with upper or lower extremity dysfunction. 11 During training, these devices can reduce joint loading; provide support to patients with strength deficits; and increase, maintain, or improve function, which can help achieve normal gait. In addition, they can be used to develop an individualized training program for patients, further improving functional recovery.12,13 However, the continuously evolving rehabilitation devices need further investigation to determine their clinical adaptability. Therefore, we hypothesized that the effectiveness and safety of lower-limb walking assist devices is not inferior to traditional rehabilitation therapy in stroke survivors receiving gait training to improve lower-limb function. This study aimed to evaluate the safety and efficacy of a new rehabilitation method and provide a basis for further restoration of the locomotion abilities of subacute stroke patients while ensuring training safety.
Methods
This trial was retrospectively registered on clincialtrials.gov with the unique identifier NCT06564480.
This multisite, assessor-blinded, randomized controlled trial was conducted at five clinical trial institutions, including the Zhejiang Hospital, Tongde Hospital of Zhejiang Province, Affiliated Hangzhou First People’s Hospital, Sir Run Run Shaw Hospital, and the Second Affiliated Hospital of Zhejiang University School of Medicine. This study was approved by the following institutional review boards: the Ethics Review Committee of Zhejiang Hospital, the Ethics Committee of Tongde Hospital of Zhejiang Province, the Ethics Committee of Hangzhou First People's Hospital, the Ethics Committee of Run Run Shaw Hospital, and the Ethics Committee of Human Research at the Second Affiliated Hospital of Zhejiang University School of Medicine. The study received ethical approval from the following institutes: Zhejiang Hospital (2021 Temporary review No. (60G), approved on 22 October 2021); Tongde Hospital of Zhejiang Province (Zhejiang Tongde Kuai Shen Zi [2021] No. 075, approved on 20 October 2021); Affiliated Hangzhou First People’s Hospital (2023 Medical Ethics Review No. (121)-02, approved on 28 September 2023); Sir Run Run Shaw Hospital (Lunshen 2023 Equipment No.0526, approved on 22 September 2023); and the Second Affiliated Hospital of Zhejiang University School of Medicine: (2023 Lunshenxie No. (519), approved on 27 September 2023).
The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT). 14 This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024.
Sample size and participants
Participants were recruited from five clinical trial institutions. The participants provided written informed consent and met the following inclusion criteria: (a) age between 18 and 85 years; (b) diagnosis of stroke confirmed on computed tomography or magnetic resonance imaging (MRI), hemiparesis on only one side, no residual neurological deficits after the first-ever or any previous strokes, stroke damage in the cerebral hemisphere, and disease duration ≤3 months; (c) no further progression of neurological symptoms after 48 h, without severe cognitive dysfunction or aphasia; and (d) lower-limb muscle tone of ≤level 2, as assessed using the Modified Ashworth Scale (MAS). 15 The MAS is used to grade muscle spasticity. The scale is as follows: 0, no increase in muscle tone; 1, slight increase in muscle tone, with a catch and release or minimal resistance at the end of the range of motion when an affected part(s) is moved in flexion or extension; 1+, slight increase in muscle tone, manifested as a catch, followed by minimal resistance through the remainder (less than half) of the range of motion; 2, marked increase in muscle tone throughout most of the range of motion, but easy movement of the affected part(s); 3, considerable increase in muscle tone, with difficult passive movement; and 4, affected part(s) rigid in flexion or extension, with standing balance function ≥ level 1. 16 The standing balance function is divided into three levels: static balance, dynamic balance, and reactive balance. The exclusion criteria included the following: (a) diseases of the bones, joints, or muscles or lower-limb disabilities, such as severe osteoporosis, bone fracture, spinal instability, or diseases directly affecting the ability to balance and walk; (b) diseases of the cardiovascular, pulmonary, hepatic, renal, or hematopoietic systems or other serious primary diseases as well as critical or psychiatric diseases; (c) other contraindications or complications that may affect walking training; (d) lower-limb local skin injury or local pressure ulcers; I lower-limb peripheral neuropathy; (f) body shape unsuitable for robotic training (height <150 or >190 cm or weight >85 kg); (g) severe cognitive impairment; and (h) location of stroke damage is in the cerebellar hemisphere or brainstem. The withdrawal criteria included the following: (a) fulfillment of the termination criteria in the trial; (b) dysfunction of important organs; (c) poor training compliance; (d) disease aggravation or serious adverse reactions; and I patient willingness to undergo other treatment methods or voluntarily withdraw from the trial.
The primary outcome measure was the Fugl–Meyer Assessment Lower Extremity (FMA-LE) score on day 28 (±7 days). After a review of the literature, we aimed for a relative, clinically relevant, improvement in FMA-LE score on day 28 (±7 days), with a noninferiority margin (δ) of −3, common standard deviation (σ) of 4.14, and an effect size of 0.5, resulting in a sample size of approximately 40 in each group to achieve 80% power with a one-tailed α of 0.025, As follows:
Considering a dropout rate of 20%, we increased our sample to 50 participants in each group.
Randomization and masking
One hundred patients were randomly divided into the experimental and control groups in a ratio of 1:1. After the age and sex of the participants were stratified, each research center was assigned random numbers, and block randomization was used for grouping (block size = 4). Using the random envelope method for allocation concealment, we placed each grouping scheme into an opaque envelope, using sequentially encoded, opaque, and sealed envelopes, with the code written on the outside of the envelope, which was sealed and handed over to the researcher. The personnel who enrolled and those who assigned participants to the interventions could not access the random allocation sequence. The random numbers were obtained from a random number table generated using Statistical Analysis System (SAS; version 9.4). Patients and therapists were not blinded throughout the study because of the nature of the intervention. The pre- and post-treatment assessor was a third person independent of the trial.
Outcome assessment
All questionnaires were evaluated under uniform conditions at each center by the third person. After uniform preassessment training, a standardized assessment protocol was used.
The primary outcome was the FMA-LE score on day 28 (±7 days). The scale has 17 items, including lower extremity synergistic movement, separation movement, coordination and speed, and reflex activity. Each item is scored as 0, 1, or 2; the maximum total score is 34 points. The higher the score, the better the lower extremity motor function. The intrarater reliability of the expert rater for the FMA-LE score was high for the motor and sensory scores, ranging from 0.95 to 1.0. The interrater agreement (intraclass correlation coefficient (ICC): (2, 1)) between the expert and therapist raters for the FMA-LE scores was high for the motor scores (total, 0.98; upper extremity, 0.99; and lower extremity, 0.91).17,18 The secondary outcomes included the Functional Ambulation Category (FAC) and Modified Barthel Index (MBI) scores on days 0, 14, and 28. The FAC scale was used to assess the walking ability of patients and was scored from 0 to 5 according to the amount of walking assistance needed by the patients. The higher the rating, the better the patient’s walking ability. Research shows the FAC has excellent reliability, good concurrent and predictive validity, and good responsiveness in patients with post-stroke hemiparesis. There was high test–retest reliability (Cohen kappa = 0.950) and interrater reliability (kappa = 0.905). 19 The MBI scale is used to assess activities of daily living and includes 10 items, including feeding, bathing, dressing, transfers, personal hygiene, bowel and bladder control, toileting, walking on a level ground, and walking up and down the stairs, with a minimum score of 0 and a maximum score of 100. The higher the score, the stronger the ability to take care of oneself and the lower the degree of dependence. The ICC and minimal detectable change (MDC) for the MBI were 0.94 and 15.4, respectively. 20
The safety evaluation outcomes included vital signs (temperature, respiratory rate, blood pressure, and pulse). Half an hour before and after the treatment, we used a thermometer to monitor the changes in the above indicators.
Adverse events and serious adverse events were evaluated. Adverse events are unfavorable medical events that occur during a clinical trial and may be unrelated to the medical device used in the trial. A serious adverse event is an event that occurs during a clinical trial that results in death or serious health complications.
Training interventions
Both groups underwent conventional rehabilitation training at the Department of Rehabilitation, including muscle strength and endurance training, trunk control training, and bridge training. The experimental group received training using the MI-R1 lower-limb walking assist device for lower-limb functional training, and the control group received traditional lower-limb functional rehabilitation training under a therapist’s guidance.
MI-R1 lower-limb walking assist device training
This device, developed and produced by Zhejiang Fubon Technology Co., Ltd., is a lower extremity rehabilitation training device consisting of a main control package, lumbar support fixation component, leg component, handheld device, battery charger, and mobile support frame. Through the limb straps and motor drive on the patient’s lower limb, continuous passive movement triggers hip and knee joint movement, while the handheld device assists in simulating normal sitting, standing, and walking. The height parameters of the lower-limb walker were first adjusted according to the length of the patient’s lower limbs before training initiation. The lower-limb straps were then fixed, and the equipment was adjusted to a reasonable and comfortable position before starting the rehabilitation training. The lower-limb walking assist device was selected according to the patient’s specific condition to perform rehabilitation training, including center-of-gravity transfer training, stepping training, walking training, and sitting and standing training. The training time was 30 min, 5 times a week, once a day, for a total of 4 weeks.
Conventional rehabilitation training
According to the patient’s specific condition, lower-limb functional rehabilitation training, including center-of-gravity transfer training, single-legged weight-bearing training, stepping training on the balance bar, and walking training on the balance bar, was selected under the guidance of the rehabilitation therapist. The training time was 30 min, 5 times a week, once a day, for a total of 4 weeks.
Statistical analyses
Statistical Analysis System version 9.4 was used for statistical analysis. The test results were statistically analyzed for validity indicators, using both full analysis set (FAS) and per protocol set (PPS), and for safety indicators using the safety set (SS). For one-sided test, the α-value was 0.025, and for two-sided test, the α-value was 0.05; if the p value was ≤α, the difference was considered statistically significant. Quantitative indicators were presented using mean, SD, median, minimum, and maximum values, while categorical indicators were presented as the number of cases and percentage. According to the characteristics of the variables, a paired t-test was used to compare quantitative data between the two groups. The chi-square test/Fisher’s exact probability method was applied to compare the qualitative data between the two groups. Missing data were processed based on their respective datasets. For the main outcome data, the last observation carried forward (LOCF) method was used.
Results
This trial was conducted at five clinical trial institutions and included a total of 106 patients. Seven patients were excluded. One hundred patients who met the inclusion criteria and did not meet the exclusion and withdrawal criteria from May 2022 to February 2024 were selected, enrolled, and followed up. Six patients did not meet the inclusion criteria. One patient was excluded due to concomitant knee arthritis. Seven patients were discharged. One patient underwent lumbar MRI, which revealed a recent compression fracture at T12, and an orthopedic consultation recommended absolute bed rest. One patient could not tolerate walking training. Two patients requested early discharge and voluntarily withdrew from the trial. Another patient requested to withdraw from the trial. One patient withdrew due to concerns about the inconvenience caused due to trial participation, and one patient’s family member refused to provide their ID card and requested to withdraw from the trial (Figure 1). The first patient entered the trial on 24 May 2022, and the final patient completed the trial on 1 February 2024, spanning a period of 620 days. After the analysis, the FAS, PPS, and SS included 99, 92, and 99 cases, respectively.

CONSORT 2025 flow diagram demonstrating the number of participants at each stage and outlining the procedural steps leading to the final data analysis. CONSORT: Consolidated Standards of Reporting Trials.
The general information of patients in the two groups, including age, sex, ethnicity, height, weight, and educational level, was statistically analyzed. The FAS revealed that the differences between the two groups were not statistically significant (p > 0.05) (Table 1).
Analysis of demographic information (FAS).

p < 0.05 indicated a statistically significant difference.
FAS: full analysis set.
As shown in Table 2, PPS analysis revealed that the FMA-LE scores (mean ±SD) on day 28 (±7 days) were 21.73 ±6.99 in the experimental group and 22.13 ± 6.82 in the control group, with a noninferiority statistic of −0.1976 (−1.5051, 1.1099). The lower limit of the 95% confidence interval (−1.5051) was greater than the noninferiority cutoff value (−3). The test results reached the expected target value.
Statistical analysis of the noninferiority of the FMA-LE score on day 28 (±7 days) of rehabilitation training (PPS).
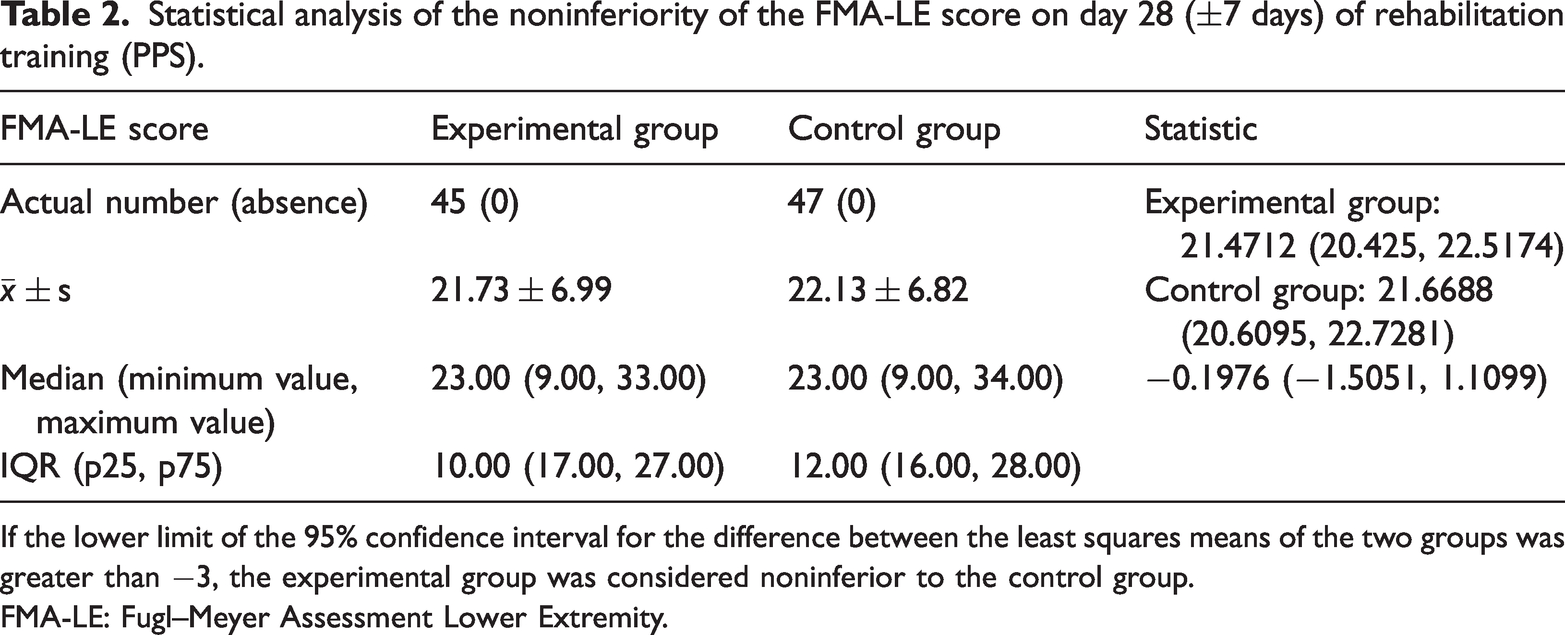
If the lower limit of the 95% confidence interval for the difference between the least squares means of the two groups was greater than −3, the experimental group was considered noninferior to the control group.
FMA-LE: Fugl–Meyer Assessment Lower Extremity.
As shown in Table 3, PPS analysis revealed no statistically significant difference in the FAC scores between the experimental and control groups on days 0, 14, and 28 of rehabilitation training (p > 0.05).
Comparison of the FAC scores between the experimental and control groups (PPS).
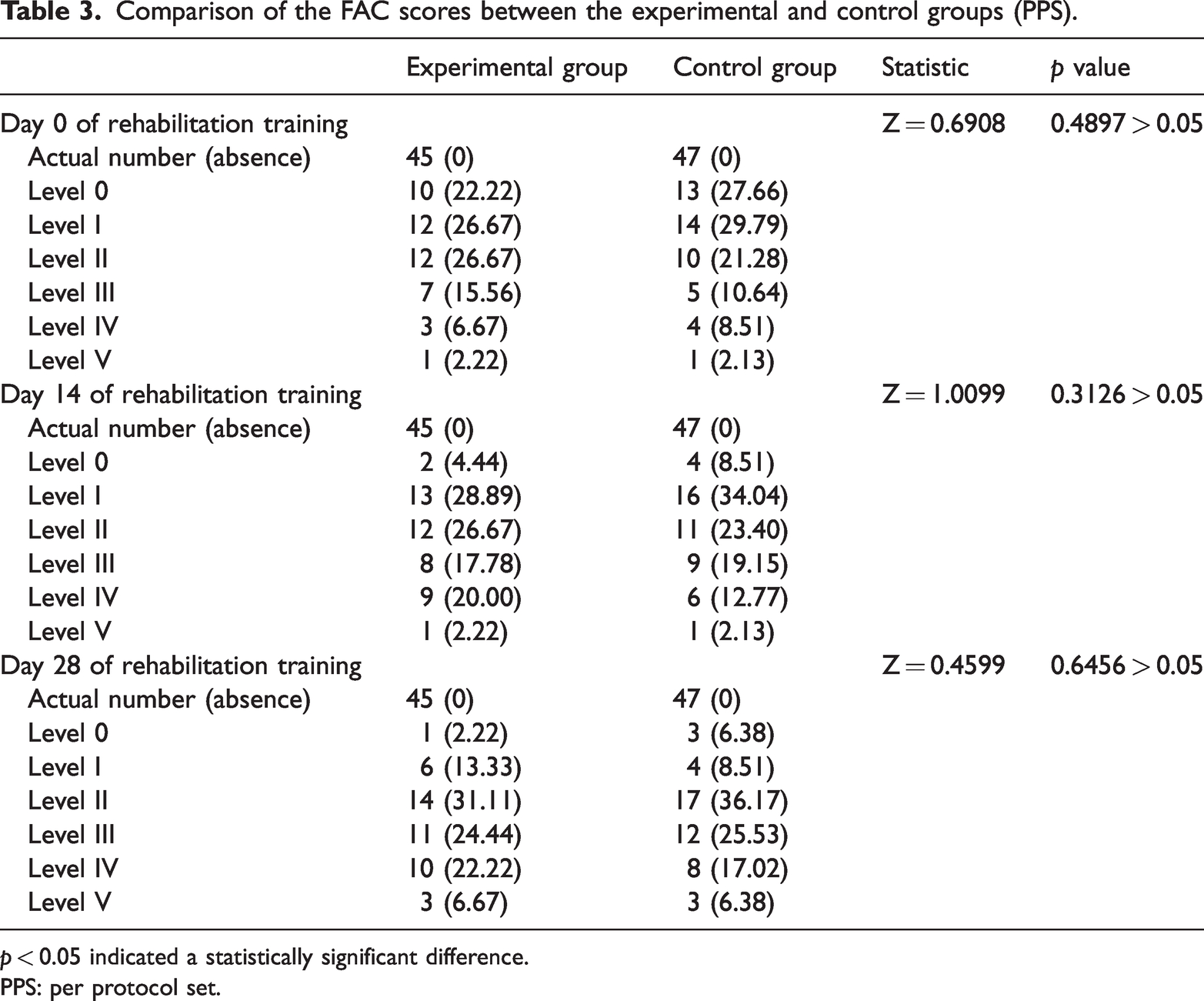
p < 0.05 indicated a statistically significant difference.
PPS: per protocol set.
As shown in Table 4, PPS analysis showed that the MBI scores (mean ± SD) on days 0, 14, and 28 of rehabilitation training were 51.19 ± 19.87, 60.47 ± 21.78, and 68.08 ± 21.53 in the experimental group and 49.09 ± 20.83, 57.29 ± 20.90, and 65.08 ± 20.12 in the control group, respectively. The difference between the two groups was not statistically significant (p > 0.05). The MBI scores of the experimental and control groups gradually increased with rehabilitation training.
Comparison of the MBI scores between the experimental and control groups (PPS).
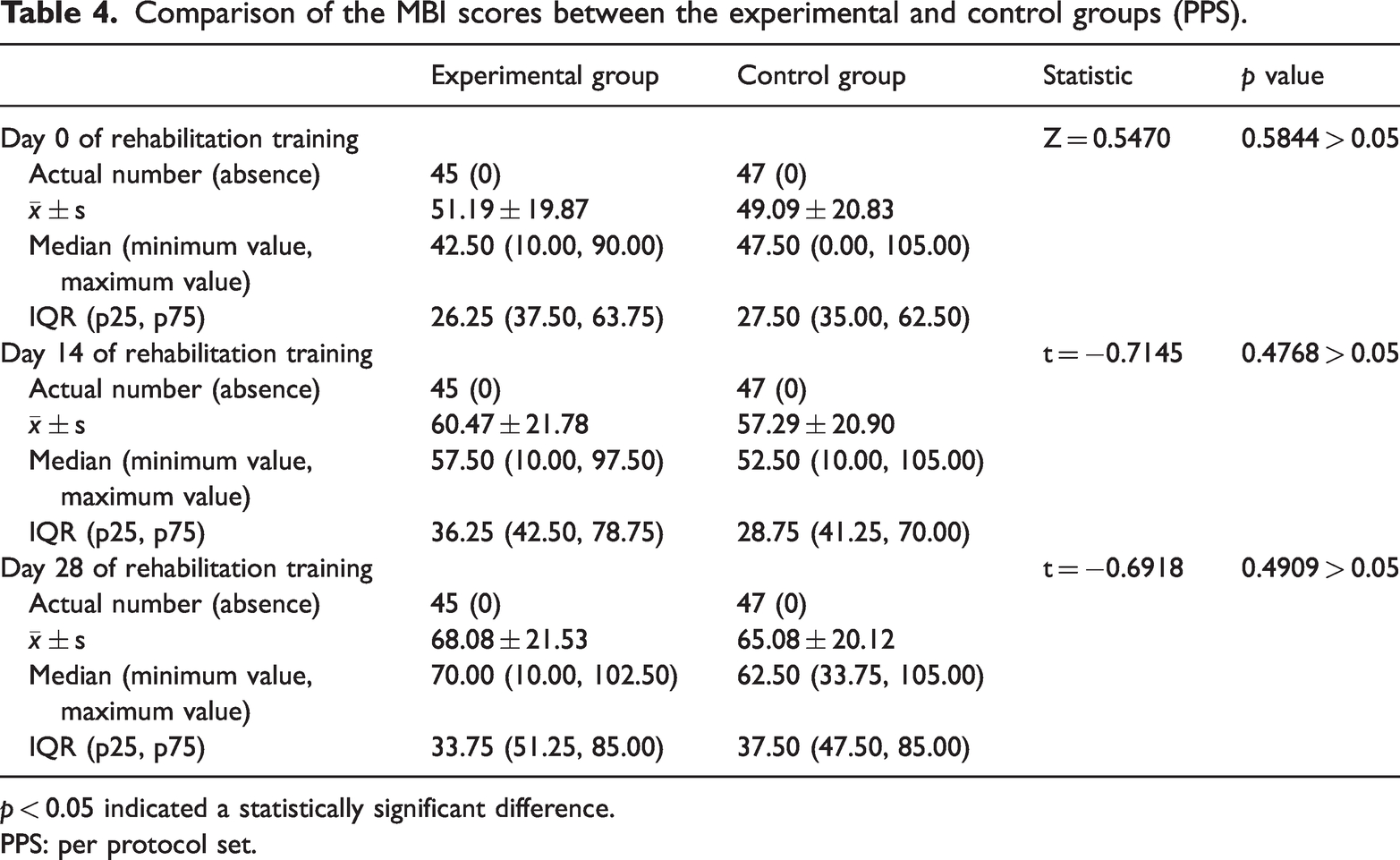
p < 0.05 indicated a statistically significant difference.
PPS: per protocol set.
SS analysis (Table 5) revealed that vital signs (temperature, heart rate, respiratory rate, systolic blood pressure, diastolic blood pressure, and pulse) were within the normal reference range before rehabilitation training and on day 28 of rehabilitation training. There was no statistically significant difference between the experimental and control groups (p > 0.05).
Comparison of vital signs between the experimental and control groups (SS).

p < 0.05 indicated a statistically significant difference.
SS: safety set.
In total, 86 adverse events occurred in 41 patients. The prevalence rates of adverse events were 46.94% in the experimental group and 36% in the control group, representing a statistically nonsignificant difference (p > 0.05). The 86 adverse events were not related to the experimental device. No serious adverse events occurred (Table 6).
Comparison of the incidence of adverse events between the experimental and control groups (SS).

p < 0.05 indicated a statistically significant difference.
SS: safety set.
Discussion
This study aimed to verify the clinical effectiveness and safety of lower-limb functional rehabilitation training utilizing an MI-R1 lower-limb walking assist device. According to the analysis of FMA-LE scores on day 28 (±7 days), the lower limit of the 95% confidence interval was greater than the noninferiority cutoff value, indicating that stroke patients who underwent lower-limb functional training with the MI-R1 lower-limb walking assist device were noninferior to those who underwent conventional rehabilitation and that both treatment protocols were effective in recovering lower-limb function. Previous studies have investigated the effectiveness of using lower-limb exoskeleton robots in recovering lower-limb function, gait, and neuroplasticity in patients with subacute stroke.21,22 The results showed that patients trained using lower-limb exoskeleton robots for 4 weeks had better FMA-LE, Berg Balance Score (BBS), and FAC scores than those who underwent traditional rehabilitation training, which is consistent with our research findings. Robot-assisted gait training can achieve more coordinated and effective gait patterns and promote the reorganization of bilateral motion-related networks associated with balance and gait recovery after stroke, thereby achieving more improvement in lower-limb function and walking stability in patients with subacute stroke. In our study, there was no significant difference in the FAC scores between patients who underwent training using the lower-limb walking assist device and those in the control group. Research shows that walking assist training devices are recommended for stroke patients who cannot walk independently because compared with traditional therapy, these devices increase the walking speed, distance, and balance (level 1 evidence). 23 However, a recent Cochrane review 24 revealed that although this type of training increased the walking speed and endurance, it demonstrated a relatively mild effect on improving independent walking ability, which is in line with our results. This may be attributed to our relatively short duration of one-time training and insufficient walking training.
Regarding MBI scores, compared with the control group, rehabilitation training with the MI-R1 lower-limb walking assist device conferred no additional benefit in terms of the ability to perform activities of daily living. Recent studies have shown that patients who underwent robot-assisted gait training did not show significant statistical differences in outcomes compared with those who received regular gait training. 25 This may be because this scale mainly assesses self-care abilities, such as dressing, eating, bowel continence, and transfers. Our training protocol did not include occupational therapy programs and provided limited training for upper limb function. Therefore, this scale was not considered sensitive in this study. The vital signs of both experimental and control groups remained stable before and after training. The incidence of adverse events in patients receiving training with the MI-R1 lower-limb walking assist device did not increase significantly compared with that in the control group. These adverse events were not determined to be serious adverse events.
The effectiveness of training using the MI-R1 lower-limb walking assist device may be attributed to the following mechanisms. First, exoskeleton robots or assisted-walking training devices can reduce the load on the lower extremity in stroke patients by partially supporting their body weight and improving walking stability and training efficiency.26,27 Second, lower-extremity–assisted training can provide patients with repetitive, high-intensity, and standardized training by setting a training program according to their individual health status. It is a dynamic, task-oriented training approach with greater continuity and consistency of treatment, which can help improve rehabilitation efficacy.28,29 Chang et al. also demonstrated that 2 weeks of Lokomat-assisted training resulted in a 12.8% increase in the maximal oxygen uptake (VO2 max), improved cardiorespiratory endurance, and significantly improved lower extremity muscle strength in stroke patients. 30
The use of a lower-limb walking assist training device is conducive to improving the treatment efficacy in stroke patients with cognitive impairment. Owing to central nervous system damage, different stroke patients have varying degrees of cognitive dysfunction. In traditional rehabilitation training, this can lead to decreased patient cooperation or difficulty in correctly executing the training content, resulting in reduced training efficacy. A study 31 revealed that lower-limb task-oriented training combined with cognitive training can significantly improve limb and cognitive function, correct abnormal gait, and improve balance in stroke patients. Moreover, a study 32 conducted device-assisted balance training for cognitively impaired patients; the results demonstrated that this training can be widely used for patients with mild cognitive impairment and helps improve postural control in this cohort.
The use of lower-limb walking assist training devices can reduce the burden on therapists. A study on therapists revealed 33 that the majority (67.8%) of physical therapists work for 6–8 h per day, 28.1% work for 8–10 h per day, and 2.1% work for >10 h per day. In 2018, more than 100 million people in China needed rehabilitation management; however, <20,000 professionals provide rehabilitation therapy, 34 which include physical therapists, occupational therapists, and speech and swallowing therapists. The ratio of rehabilitation therapists to the total population in China is approximately 1:28,000, with the average staffing level of physical therapists being even lower. The ratio of physical therapists to the total population is 1:1693 in the United States; 1:1011 in Australia; 1:1344 in the United Kingdom; 1:768 in Switzerland; and 1:485 in Norway, 35 demonstrating a larger gap between the number of physical therapists and the number of patients requiring physical therapy in China. The application of lower-limb walking assist training devices can lower the physical strength and energy demands of therapists, ensure training consistency, and improve treatment safety.
Current traditional rehabilitation methods have demonstrated efficacy in treating lower-limb dysfunction after stroke. However, such methods have certain limitations. For example, traditional rehabilitation techniques tend to be labor- and resource-intensive, involve a monotonous training process, fail to boost patients’ enthusiasm, and involve overreliance of patients on the therapist, resulting in inefficient training.36,37 In most cases, the therapeutic effect on stroke patients is limited. In addition, the frequency and intensity of routine therapy performed in clinical practice are insufficient to maximize recovery. 38 The use of a lower-limb walking assist training device can partially overcome these limitations and increase patient training cooperation, thereby enhancing the effectiveness of rehabilitation training.
Taken together, the results of this study indicate that the MI-R1 lower-limb walking assist training device can be used for rehabilitation training in stroke survivors. Its effect is comparable to that of traditional rehabilitation therapies, and this method is associated with patient safety. The advantages of the MI-R1 lower-limb walking assist device mainly lie in the standardized training environment, adaptive support, sufficient training intensity, and reduced physical burden on therapists. Lower-limb walking-assist training devices may provide a more convenient approach for clinical rehabilitation. Further research and follow-up studies are needed to validate these findings.
Limitations
In this study, double-blind treatment was not possible because the intervention in the experimental group required the use of instruments. Moreover, this study was registered after trial completion. Owing to time and manpower constraints, follow-up studies to explore the duration of treatment efficacy could not be performed.
Conclusions
The MI-R1 lower-limb walking-assist device is not inferior to traditional rehabilitation for achieving recovery of lower-limb function in stroke patients and is associated with good patient safety.
Footnotes
Acknowledgments
The authors thank all those who facilitated this research, especially the patients.
Author contributions
Bing Xiong conceptualized and performed the study as well as conducted the formal analysis. Xinyuan Wang performed the validation, formal analysis, and manuscript writing. Xin Xu supervised the study. Zhilai Lou, Ting Tong, Danlei Gu, Kedan Zhang, and Huanhuan Liu recruited the patients and performed the treatments.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Ethical approval
The submitted manuscript does not contain any previously published materials and is not under consideration for publication elsewhere. Ethics committee approval was obtained from the authors’ institution, and informed consent was obtained from the study participants.
Funding
This study did not receive any funding.

