Abstract
Study design
Technical note and case series.
Objectives
3D-printed implants (3DPI) for spinal surgery are a relatively recent development. We report on our experience with the rapid creation and regulatory approval of patient-specific 3DPI for use in complex spinal reconstruction, including a novel expedited turnaround time for implant creation.
Methods
Four patients underwent placement of 3DPI to replace osseous anatomy during complex spinal reconstructions. These implants were created and used to replace patient-specific anatomy created by either en bloc tumor resection or by severe neurogenic spinal arthropathy. The surgical planning, implant creation, and postoperative outcomes are outlined.
Results
All patients underwent successful implantation of 3DPI, which was confirmed on postoperative imaging at most recent follow-up. The time to plan, create, obtain regulatory approval, and use the first 3DPI was 28 days. Subsequent 3DPI could be planned, approved, and used in surgery in as little as 4-5 days, which is faster than previously-published reports. Thus, a 3DPI could be generated based on osseous defects created during stage 1 of a multistage surgical plan and implanted during a subsequent stage in an especially expedited manner.
Conclusions
3DPI may be used to effectively replace patient-specific anatomy during complex spinal reconstructions, including for osseous defects that are generated after the initial surgical procedure. These 3DPI may be created, approved, and used in surgery over much faster timelines than have been previously reported. Additional cases utilizing these custom 3DPI will further elucidate their utility during complex reconstructions.
Keywords
Introduction
Complex spinal surgeries such as en bloc tumor resection may involve removal of significant segments of the spine, sacrum, and/or pelvis. In many cases, complex reconstruction is subsequently required to maximize postoperative function. These reconstructions can be challenging if they require prefabricated implants that do not fit the patient’s anatomy. Ideally, implants would be tailored to a patient’s specific anatomy and needs 1 – for instance, designed to fit the defects created during stage 1 of a multistage surgical plan and then manufactured in an expedited manner for implantation during the next stage. The options for designing implants in this way were previously limited by available technology. However, fully custom implants can now be created on rapid timescales via 3-dimensional (3D) printing.
3D printing enables the creation of physical objects from computer models or imaging studies.2,3 Its use in spine surgery was initially limited mainly to the preoperative process where it was used to create anatomic models that could be used to guide tumor resections, instrumentation placement, or surgical education.4,5 More recently, it has been applied to the creation of patient specific 3D-printed implants (3DPI).6-9 The idea to produce metallic 3DPI for orthopedic procedures has existed since the early 2000s, 10 but the technology and processes involved have been significantly improved and refined. These implants may be based on imaging obtained during any part of the perioperative process and can be created from metals such as titanium so that they are inert, stable, and long-lasting. Implant density and inherent stress moduli can also be customized. Furthermore, innovations in printing have allowed for porous metallic implants that facilitate osseus integration.
The use of 3DPI has seen significant growth in degenerative spine disease with the development of custom patient interbody devices,11,12 but their applications remain relatively uncommon for complex spinal and spinopelvic reconstructions. Importantly, further studies are required to illustrate the full range of conditions and implant types for which 3DPI may be applicable. 6 To that end, this study presents the cases of four patients from the authors’ institution who received patient-specific titanium 3DPI for various pathologies involving the spine, sacrum, and/or pelvis. The implants received expedited regulatory approval for compassionate use for these cases. Uniquely, these 3DPI were generated through an innovative methodology that enabled them to be designed based on osseous defects made during stage 1 of a multistage surgery, manufactured, shipped, and implanted during subsequent stages over the course of 4-5 days rather than weeks, which is the fastest timescale yet reported.
Methods
All patient information was deidentified before inclusion in this study. Permission was granted by these patients to study their cases and outcomes. The implantation of and use of follow-up data regarding the 3DPI, instead of conventional institutional review board approval, required the more rigorous Food and Drug Administration (FDA) compassionate use review and approval (compassionate use numbers U220386, U230605, U230020, and U220706).
Patient Selection
Patient Demographic and Clinical Details.
3D Implant Manufacturing
All 3DPI were created following similar steps. High-quality CT scans were obtained for the regions of interest. These included CT of the cervical, thoracic, or lumbosacral spine; CT of the chest/abdomen/pelvis; or angiographic imaging when relevant. The Digital Imaging and Communications in Medicine (DICOM) data were assessed using a radiology workstation by the 3D Printing Center at Washington University. Images were analyzed using Materialise Mimics and SolidWorks for computer-aided design (CAD) modeling derived from CT imaging. Based on the modeling and relevant anatomic defects, as well input from surgeons, a bespoke 3D-reconstructed model was created. The predicted load, fatigue, or other biomechanical forces that the implant would need to sustain were simulated using finite element analysis. These analyses included scenarios where there was no support given by the patient’s remaining bone (i.e. the implant and associated screws would need to withstand the entire force across the construct).
The 3DPI were created using a proprietary industry protocol from a material called PURI-TI (DeGen Medical, Charleston, SC). PURI-TI implants are manufactured, shipped, and sterilized for surgery on a more rapid timeline than is used for conventionally manufactured 3DPI. The PURI-TI 3DPI manufacturing process is largely proprietary and the nuances unfortunately cannot be discussed in this paper. The implants are additively printed and do not require cutting oils or material transfer. They are made of titanium and thus are appropriate substitutes for ordinary titanium implants. Each implant was subjected to further load and deformation testing before it was used in surgery. The implants were designed to be incorporated into spinal fusion constructs using conventional anterior or posterior spinal instrumentation (rods, screws, or connector pieces) or fusion adjuncts (bone graft, osteo-inductive/osteo-conductive materials).
Details of Patient-specific 3D-printed Implants.
Illustrative Cases
Case #1: Advanced Neuropathic Spinal Arthropathy
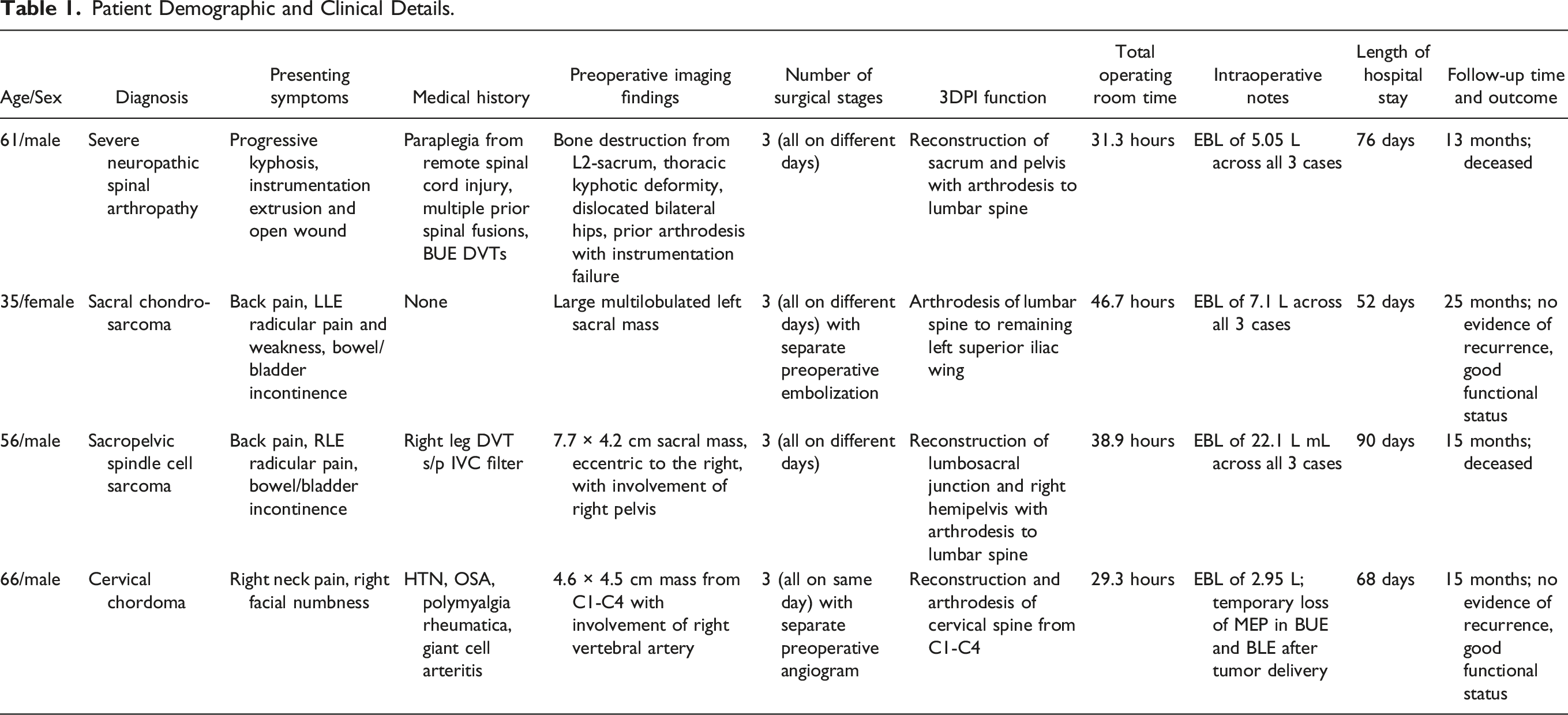
A 61-year-old male with past medical history of a complete spinal cord injury in the 1980s presented with severe neuropathic spinal arthropathy (Charcot spine). He had received multiple prior spinal surgeries, most recently a T4-sacrum posterior fusion 8 years ago. In the interim he had experienced fracture of one of his rods and progressive disintegration of his lumbar spine and sacrum with severe kyphotic deformity (Figure 1A). Ultimately, his lumbar spine became entirely separated from his pelvis. As his kyphosis worsened, his instrumentation began extruding out of his back. On exam, he was ASIA A with a T6 sensory level and an open deformity of the lumbosacral area where there was visualized instrumentation but no bone. He was unable to support his torso and was forced to lean forward with his chest abutting his pelvis. Due to his debilitation, he opted for surgery in a final attempt to restore what he deemed acceptable quality of life. Preoperative sitting radiograph demonstrating kyphotic deformity due to instrumentation failure and dissociation of pelvis from spinal column (A). Schematic for custom 3D-printed sacropelvic and lumbar implant (B). Postoperative radiograph demonstrating 3DPI and revision instrumentation (C). Follow-up radiograph demonstrating improved posture and proper positioning of 3DPI (D).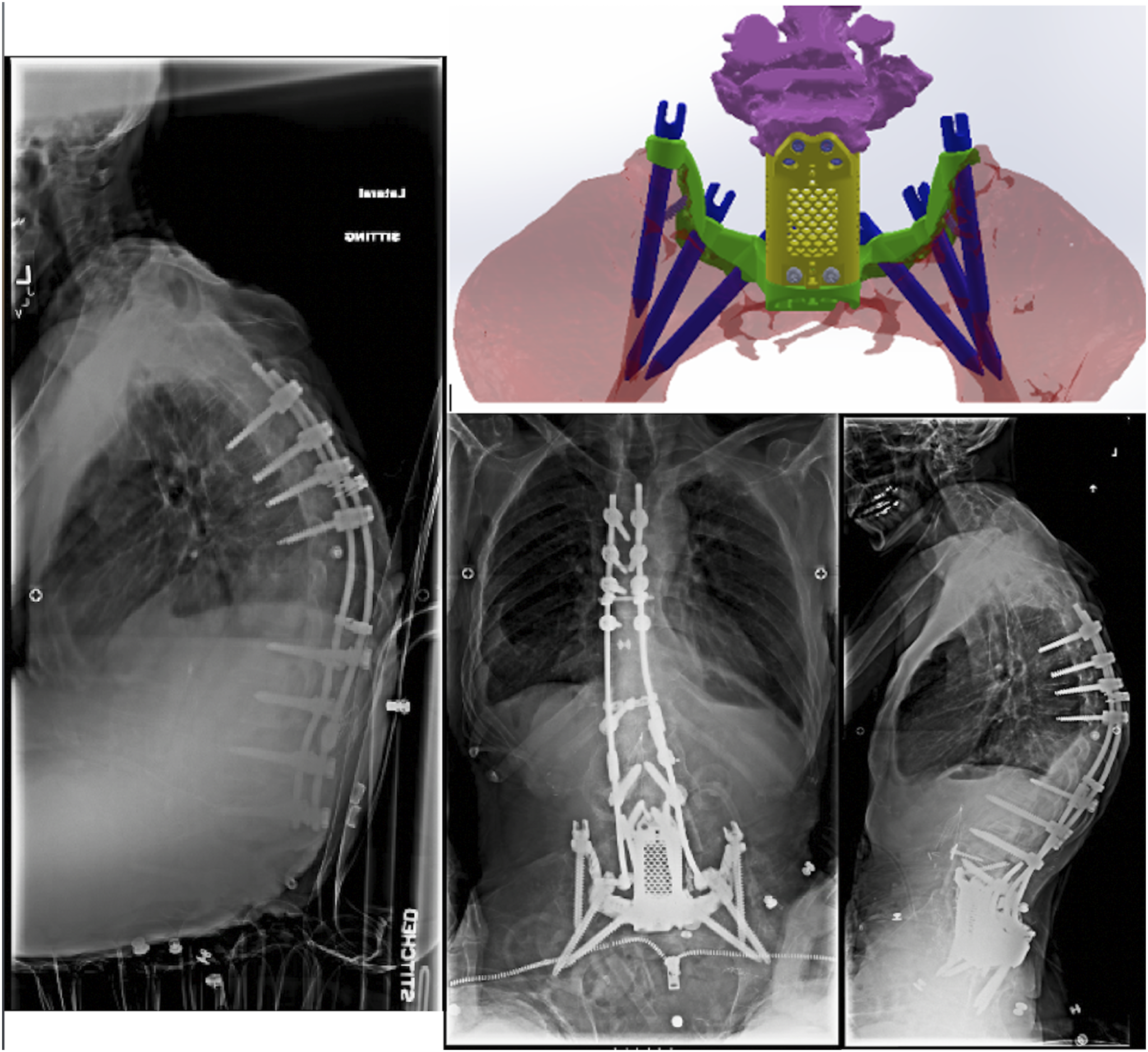
This patient required a complex lumbar, sacral, and pelvic 3DPI (Figure 1B). The complete construct was comprised of a sacropelvic platform with iliac screws, a modular multilevel corpectomy cage, and an anterior plate system with screws. This 3DPI required FDA custom device exception and approval for compassionate use and was the first device of this nature to undergo this process at the authors’ institution. This process, from design of the 3DPI to its use in surgery, took a total of 28 days. 7 The 3DPI was designed from CT scans obtained during the preoperative planning process. A custom 3DPI was necessary given the unique anatomic defects that the patient had sustained over time. Given his extreme degree of bone disintegration and his spinopelvic dissociation, conventional instrumentation was thought to be inadequate for reconstruction.
The surgical plan for reconstruction of the patient’s severely disintegrated spine was carried out in three stages, each on a different day. Stage 1 involved reconstruction of the iliac wings and sacrum using a portion of the 3DPI construct via a posterior approach. The sacropelvic aspect of the implant was fixed in place to the iliac wings bilaterally. Stage 2 involved anterior access to the lumbar spine to insert the anterior component of the 3DPI. This enabled fixation of the lumbar spine to the reconstructed pelvis (Figure 1C). Plastic Surgery then placed a free fibular flap to bridge the lumbar spine to the sacrum. Stage 3 involved revision of his existing posterior instrumentation with two additional rods to connect the posterior lumbar spine to the sacral aspect of the 3DPI, which were bridged via the fibular free flap. Across all three procedures, the total operative time was approximately 31 hours.
Postoperatively, the patient remained sedated and prone for two weeks postoperatively to offload the posterior reconstruction and flap. He took several more weeks to be weaned off the ventilator and required 10 total weeks in the intensive care unit due to his frail preoperative status. He was discharged from the hospital after a total length-of-stay of 75 days. Both his 10-week and 6-month postoperative imaging demonstrated proper positioning of instrumentation and 3DPI. (Figure 1D). He was ultimately able to sustain an upright sitting position and had better quality of life. Unfortunately, despite these improvements, he passed away 13 months after surgery due to respiratory issues from a severe COVID infection.
Case #2: Sacral Chondrosarcoma
A 35-year-old female with no past medical history presented with several months of low back pain with radicular pain. She subsequently developed bladder incontinence and occasional bowel incontinence. On exam, she demonstrated full strength and sensation throughout the bilateral lower extremities with exception of 4/5 strength with plantarflexion on the left side. An MRI demonstrated a large multilobulated T2-hyperintense, heterogeneously-enhancing sacral mass with internal calcifications and extension into the left S1-S2 nerve roots and abutment of the left S3 nerve root (Figure 2A). The mass invaded the left piriformis muscle, extended into the sciatic notch, abutted the sigmoid colon and the rectum, and invaded the left common iliac vein with encasement of the internal iliac neurovascular bundle. The patient underwent needle biopsy of this lesion that confirmed a diagnosis of chondrosarcoma. Preoperative T1 postcontrast MRI of sacral chondrosarcoma (A). Schematic for custom 3D-printed left iliac and sacral implant (B). Postoperative radiograph demonstrating 3DPI and spinal instrumentation (C). Follow-up radiograph demonstrating proper positioning of 3DPI (D).
The patient’s 3DPI consisted of a single module designed to bridge the ultimate arthrodesis between the lower lumbar spine and the left superior iliac wing (Figure 2B). This was secured to the L5 inferior endplate via two 5 × 32 mm screws and to the left iliac wing via one 7.5 × 70 mm screw and one 8 × 80 mm screw. As the exact cuts and structural mobilizations to deliver this tumor en bloc were impossible to predict precisely during preoperative planning, the 3DPI had to be designed based on imaging of the osseous defects created during stage 1. Thus, implant manufacturing was not possible until after the initial two surgical stages. Traditional implants could not fit the needed specific configurations required for reconstruction. The total time from acquisition of the osseous defect defining CT scan to implantation of the 3DPI during subsequent surgery was 120 hours.
The patient’s surgical plan was conducted over three procedures, each on a separate day. Stage 1 involved dissection of the tumor capsule from surrounding tissues as well as other preparations for later en bloc tumor resection and reconstruction. Neurolysis of the left lumbosacral plexus was required. Stage 2 involved en bloc resection of the tumor from a posterior approach. Temporary L4-L5 pedicle screw and rod fixation was required to stabilize the spine after delivery of the tumor and in anticipation of stage 3. Stage 3 involved L4-pelvis posterior instrumented fusion with anterior left-sided L5-pelvis fusion using the 3DPI to bridge the anterior column extending from the inferior L5 endplate to the superior aspect of the remaining left iliac wing (Figure 2C). In sum, the total operative time across the three procedures was approximately 47 hours.
Postoperatively, she was transferred out of the ICU on postoperative day 7 and was discharged to a rehabilitation facility 32 days after stage 3 (52 days total length-of-stay). At 2-month follow-up, she was able to walk with assistance with intermittent lower back pain. Standing scoliosis and pelvic radiographs demonstrated stable positioning of her 3DPI and other instrumentation with excellent overall alignment (Figure 2D). At 1-year follow-up her instrumentation remained in proper positioning. She walked without assistance and could participate in daily activities without difficulty. She continues to demonstrate no evidence of recurrence and has good functional status over 2 years after surgery.
Case #3: Sacropelvic Leiomyosarcoma
A 56-year-old male with no significant past medical history presented with several months of low back and right buttock pain with radiation down his right lower extremity. He subsequently developed urinary retention. On exam, he demonstrated full strength throughout the bilateral lower extremities with exception of 4/5 strength in the right S1 myotome. Sensation was similarly intact in all lower extremity dermatomes with exception of decreased sensation in the right S1 dermatome. Lumbopelvic MRI demonstrated a right pelvic mass with extension into the right sciatic notch and sacrum from the S1-S3 levels (Figure 3A). The patient underwent needle biopsy of this lesion, which provided a diagnosis of leiomyosarcoma. Preoperative T1 postcontrast MRI of sacropelvic spindle cell sarcoma (A). Schematic for custom 3D-printed sacropelvic implant (B). Postoperative radiograph demonstrating 3DPI and spinal instrumentation (C). Follow-up radiograph demonstrating proper positioning of 3DPI (D).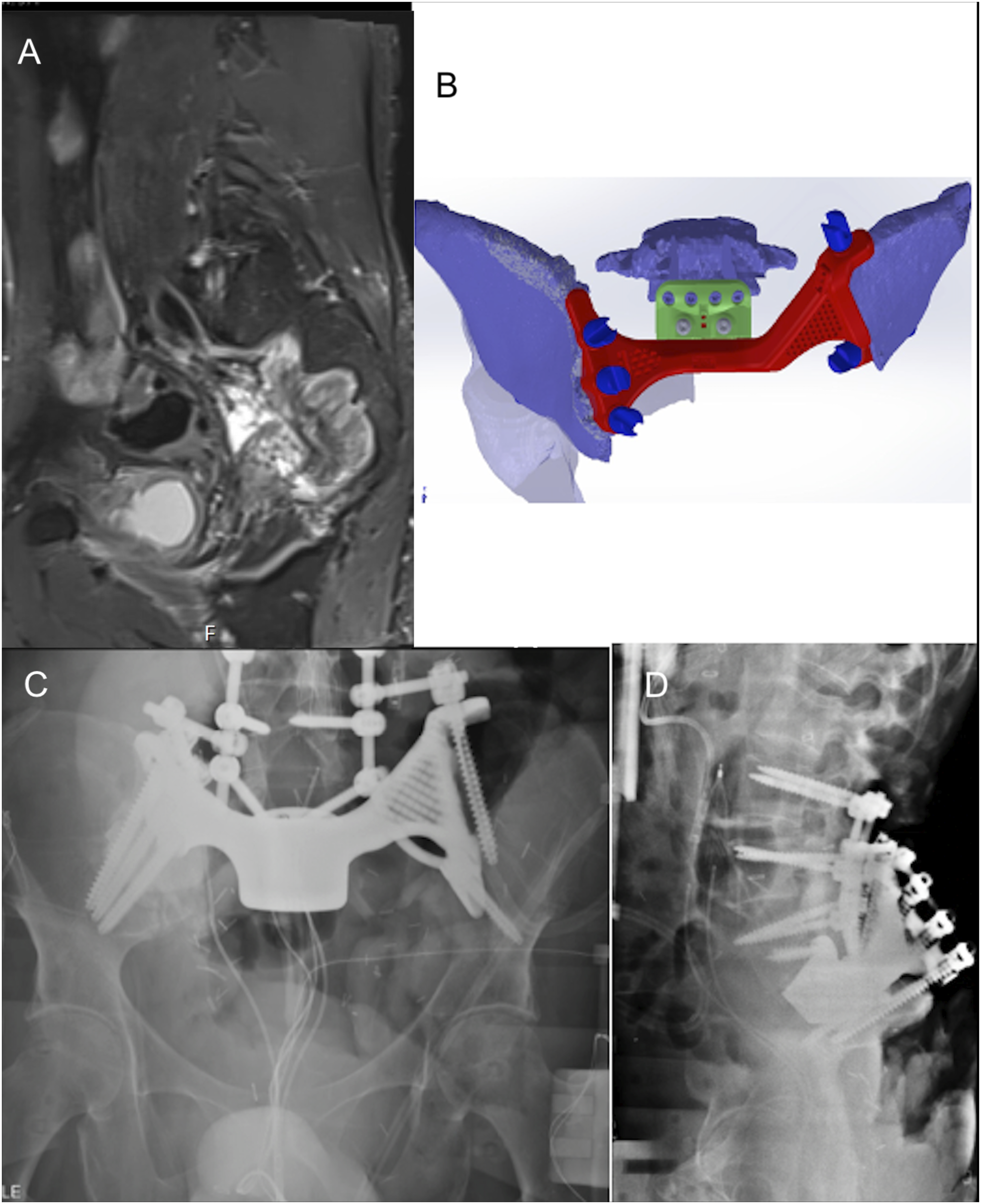
The patient’s 3DPI consisted of two components (Figure 3B). The first was a pelvic cradle that attached to the bilateral iliac wings to provide a secure foundation for reconnection of the pelvis to the lower aspect of the spine. This was secured to the pelvis via three screws (one 8 × 120 mm and two 8 × 130 mm) on the left and two screws (one 7.5 × 70 mm and one 8.5 × 75 mm). The second was a sacral module that bridged the pelvic cradle to the L5 inferior endplate. This was secured to the L5 inferior endplate via four 5 × 32 mm screws and to the pelvic cradle by two 5 × 30 mm screws. This 3DPI was also created based on imaging of the osseous defects created during stage 1 surgery given the difficult nature of predicting the required osseous cuts during the preoperative phase. Accordingly, this implant also had to be designed and manufactured after the initial surgical stages and traditional instrumentation was considered inadequate. The total time from acquisition of the osseus defect defining CT scan to subsequent implantation of the 3DPI was 96 hours.
The patient’s surgical plan was conducted over the course of three procedures, each on a different day. Stage 1 involved separation of the mass from surrounding tissues with preparations for en bloc removal during a separate stage. Stage 2 involved en bloc resection of the tumor via sacrectomy and right-sided internal hemipelvectomy. Through a posterior incision, the spine was exposed from L4 through the coccyx. L4-S1 laminectomies and right-sided facetectomies were performed. The right L5 nerve root was sacrificed and an L5-S1 diskectomy was performed. The sciatic nerve was then traced distally and divided. The patient’s spine was temporarily stabilized and he was placed on bed rest before stage 3. Stage 3 consisted of an L3-pelvis posterior instrumented fusion with left sacroiliac joint fusion and placement of the custom 3DPI. L5-S1 anterior arthrodesis was performed using the sacral component of the 3DPI, which was secured to the L5 vertebral body (Figure 3C). In aggregate, the three surgical stages took approximately 39 hours.
Postoperatively, the patient experienced issues with wound healing, which required multiple procedures from Plastic Surgery and Orthopedic Surgery to salvage the flaps used in his closure. The patient remained in the hospital for a total of 90 days, after which he was discharged to a rehabilitation facility. Approximately six months after surgery, surveillance imaging demonstrated multiple growing bilateral pulmonary nodules as well as a soft tissue mass in the right pelvis, both representing potential metastatic disease. He received postoperative radiation to the pelvic mass, underwent multiple cycles of chemotherapy, and was placed on a clinical trial to address his metastatic burden. Neurologically, he remained with strong proximal movement in his LLE. His instrumentation remained in good position (Figure 3D). However, due to continued systemic disease progression, he ultimately passed away 15 months after surgery.
Case #4: Cervical Chordoma
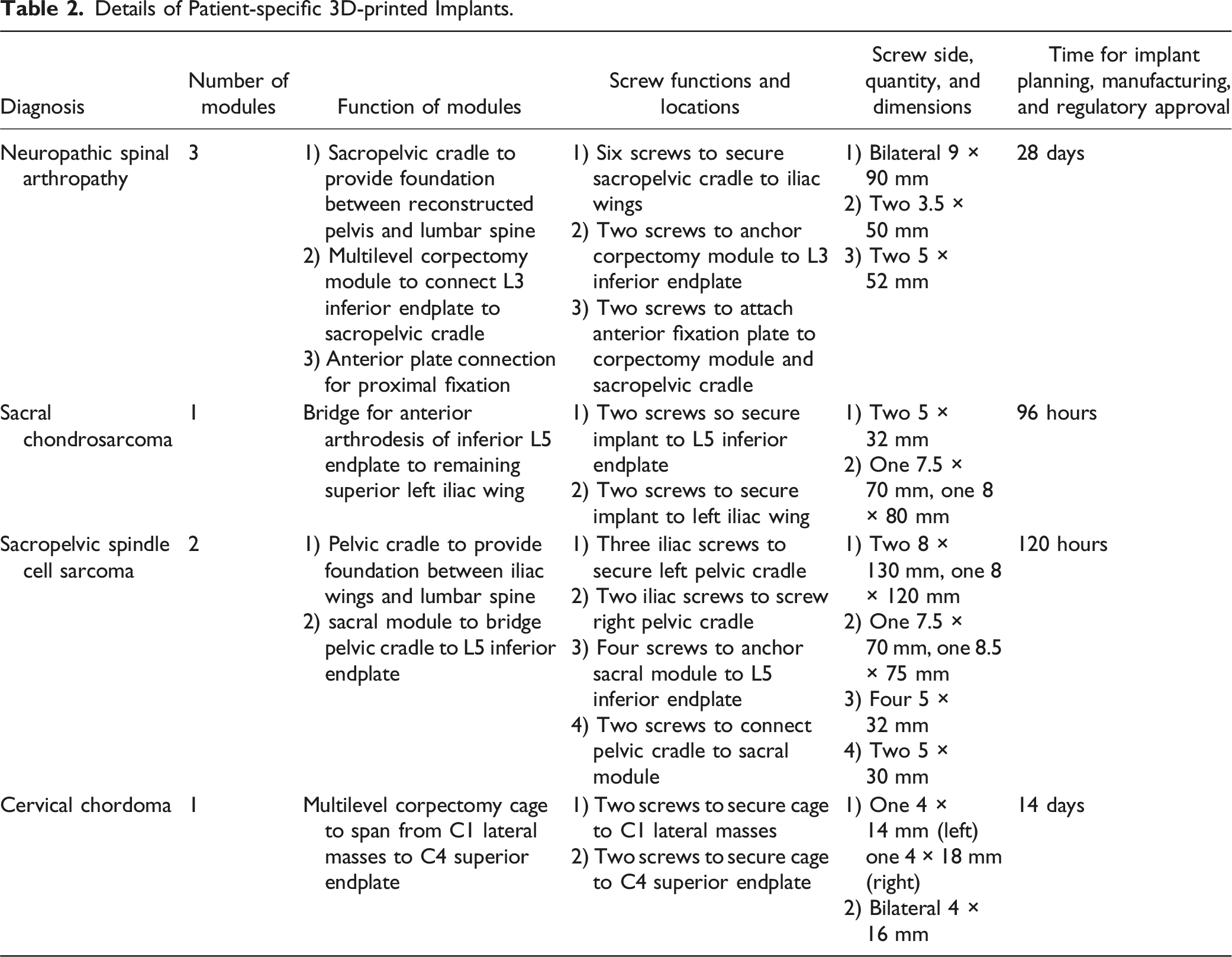
A 66-year-old male with past medical history of hypertension, sleep apnea, giant cell arteritis, and polymyalgia rheumatica presented with neck pain and right facial numbness that had been progressively worsening over years. On exam, he was neurologically intact. Cervical MRI demonstrated an approximately 5 cm mass centered in the right C2 vertebral body that extended into the C2 posterior elements, the C3 vertebral body, and the lateral epidural space (Figure 4A). CT angiography showed that the mass encased the distal V2 segment of the right vertebral artery. The patient received a needle biopsy of the lesion demonstrating a diagnosis of chordoma. Preoperative T1 postcontrast MRI of cervical chordoma (A). Schematic for custom 3D-printed cervical implant (B). Postoperative CT scan demonstrating 3DPI with fibular graft (C). Follow-up radiograph demonstrating proper positioning of 3DPI (D).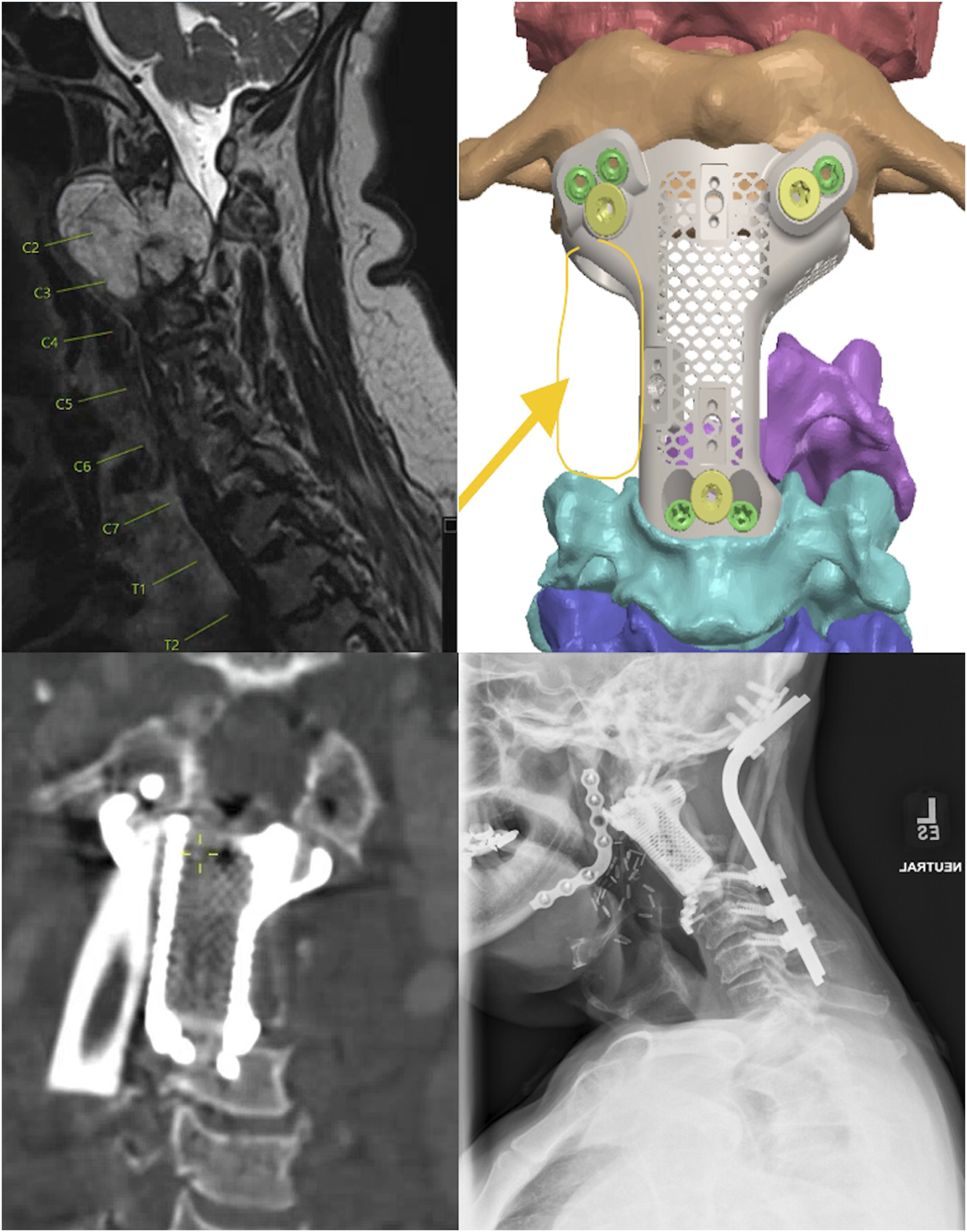
The patient’s 3DPI consisted of a custom multilevel corpectomy cage that was designed to span the bilateral C1 lateral masses to the C4 superior endplate (Figure 4B). It was one module with a lateral space to accommodate a fibular graft. It was secured to the C1 lateral masses using one 4 × 14 mm screw and one 4 × 18 mm screw and was secured to C4 using two 4 × 16 mm screws. In this case, the 3DPI was already printed in anticipation of all surgical stages being performed in one day. This was possible due to the ability to connect the implant to expected native anatomical structures – e.g. the bilateral C1 lateral masses and C4 superior endplate. Therefore, the manufacturing of this 3DPI differed from the two prior resection cases where the 3DPI had to be manufactured to perfectly fit post-resection defects that would have been impossible to precisely predict preoperatively. The entire 3DPI was created before stage 1 surgery during the preoperative planning phase. Given this less expedited schedule, the time from conception to sterilization of the implant for use was 14 days.
The patient’s surgical procedure was composed of multiple stages that were all performed under the same anesthesia. Stage 1 was via a posterior approach to perform initial tumor mobilization and separation from surrounding structures. The left vertebral artery was skeletonized at the level of C2-C3. The right vertebral artery was skeletonized and ligated at the level of the sulcus arteriosus and the C4 transverse foramen. The patient was then closed and placed supine for stage 2. Stage 2 involved en bloc resection of the tumor including delivery of the anterior arch of C1, the C2 vertebral body and dens, and C3 vertebral body. A submucosal division of the right side of the mandible, performed by Otolaryngology, was required to access the most rostral aspects of the tumor. The 3DPI was then placed to connect the head and cervical spine. A vascularized fibular autograft was placed and anastomosed across the gap to assist with arthrodesis. The mandible was plated, the patient was closed, and he was rotated prone for stage 3. Stage 3 involved a posterior instrumented fusion from the occiput to C6. Remaining fibular autograft was used to span the fusion construct posteriorly. In sum, these three stages required a total operative time of approximately 29 hours.
Postoperatively, he required prolonged time on the ventilator and had a tracheostomy on postoperative day 13. He had a gastrostomy tube placed approximately 1.5 months after surgery due to swallowing difficulties. Two months after surgery, he was walking up to 1000 feet with therapy and was discharged to a rehabilitation facility. At his 6-month follow up, he no longer necessitated a tracheostomy and was full strength throughout. Postoperative radiographs at that time demonstrated proper positioning of his instrumentation with unchanged alignment (Figure 4). He remains without evidence of recurrence and with good functional status 15 months after surgery.
Discussion
Complex spinal or spinopelvic tumor resections are highly complex procedures that often require numerous surgical services and thoughtful surgical reconstruction. In this study, we present the cases of four patients for whom custom 3DPI were used to achieve reconstruction of complex spinal defects. Though prior reports have been published on the use of 3DPI in spine surgery, few studies have outlined the use of these implants for complex multistage, multidisciplinary surgical procedures, and no similar studies have been published in the United States to the best of our knowledge.
Select Examples of prior Studies Examining the use of 3DPI for en Bloc Spinal Tumor Removal or Sacrectomies.

One of the major cited drawbacks of 3DPI is the conventional need to have a 4-6 week lead time for implant design, printing, and post-print processing. Overall, our cases demonstrate advancements in engineering, manufacturing, and execution beyond previously-reported studies. This is particularly highlighted by the speed in which these implants can be designed, printed, processed, and implanted. Through progressions of institutional experience, manufacturing innovations, and regulatory approval processes, we were able to reduce the timeframe from 28 days to 96 hours from the time a CT scan defining the osseus defect is obtained – a significant time reduction in comparison to the conventional lead time that operates on the order of several weeks, expanding our surgical abilities and options when performing complex reconstructions.
Previously, 3DPI had to be processed weeks in advance of the resection and may not have fit as well as planned, depending on bone cuts made during the resection. While cutting jigs and other methods have facilitated accurate resection, near-perfect placement of jigs and subsequent cutting are required and still may not render an exact implant fit. Further, production of these custom implants previously has taken an extended period of time to produce for safe use. At our institution, the time between stages of a large multistage surgical plan is typically a few days (to perhaps a week) when using conventional implants/instrumentation. However, for the cases highlighted in this paper, we required bespoke implants that would fit the anatomical changes that were produced during initial surgical stages. Had we used conventional implants, the patient would have to be on strict bed rest with an unstable spine for potentially weeks. Using the rapid 3DPI, we were able to maintain a timeframe that surely mitigated complications and risks from having patients immobile for so long.
This ability to generate a custom 3DPI in such a short time frame is an innovative solution to the problem of designing hardware in the midst of multistage surgical agendas. Most notably, this expedited process allowed for creating a 3DPI after the bone cuts were made for the tumor resection in stage 1 of multistage procedures. The 3DPI could then be used merely days after the first surgery, at which time the tumor could be delivered en bloc and the implant could seamlessly fit into the reconstruction process. These case illustrations supplement the current literature by demonstrating the advances that are possible regarding the use of 3DPI. These implants can enable more precise surgical care without compromising overall patient management due to unacceptable wait times. One downside, however, is the higher cost of a custom 3DPI vs a non-custom 3DPI or a standard implant. That said, as the technology required to produce 3DPI becomes more commonplace, the cost per custom implant should decrease. Moreover, using a custom 3DPI may help to stave off hardware-related or other complications in the future, thus saving money by preventing future care needs.
While our results are promising for our institution’s use of 3DPI in these complex cases, our study nevertheless has limitations. The first 3DPI was implanted approximately two years before the writing of this manuscript. In that time, we have accrued four patients, but this is still a limited sample size. As more of these cases are performed, we will learn more about the design process and implantation of these devices. The PURI-TI implant manufacturing process is largely proprietary and thus we cannot expand on the exact reasons why such rapid 3DPI manufacturing is feasible using this technology. It is theoretically possible that DeGen Medical can produce implants on such a fast timeline due to limited demand compared to manufacturers of more conventional implants, though this is unproven and DeGen still offers many different conventional and 3D-printed implants that should place high manufacturing burdens on the company. Additionally, more time is needed to assess long term outcomes for these patients, though this may not be feasible for some patients, as exhibited by the patient who passed away after progressive metastatic disease despite en bloc resection. Regarding the use of custom 3DPIs in general, these implants are substantially more expensive than prefabricated devices (up to several-fold more expensive). Because they are so specifically manufactured, they have less intraoperative flexibility regarding how they may be used in the reconstruction process. Finally, the long-term mechanical complications of using complex 3DPI have also not been fully studied given the relative novelty of this technology.
Conclusions
Here we present a case series of four patients who received patient-specific titanium 3DPIs for complex spinal or spinopelvic reconstructions with spinal fusions. The 3DPI maintained proper alignment on postoperative imaging. The first implant was able to be designed, manufactured, approved for use, and implanted in 28 days. This process was ultimately streamlined down to 96 hours, allowing for precision printing of the implant after the first stage of a multistage plan, an innovative approach that created an implant with an exact fit to the resection osteotomies. Further alterations to the 3DPI design and manufacturing process as well as longer term patient outcomes will continue to be analyzed by the authors as the number of patients receiving these 3DPI increases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Regulatory Approval
The use and study of these patients and 3DPI was covered by FDA compassionate use numbers U220386, U230605, U230020, and U220706.
