Abstract
Study Design
Systematic Review Study.
Objectives
The objective of this study is to assess the cost-effectiveness and cost-utility of rhBMP-2 in spine surgeries.
Methods
PubMed, MEDLINE, OVID and CENTRAL (Cochrane Central Register of Controlled Trials) databases were systematically searched for studies reporting the use of rhBMP-2 for spinal procedures in comparison to autograft and/or allograft and alternative graft materials under consideration of cost-effectiveness, cost-benefit, cost minimization, and/or cost-utility analysis. Relevant data points were extracted and compiled. The standardization of the selected studies was graded using the CHEERS criteria.
Results
Of the 88 eligible publications, 17 met the inclusion criteria. Overall, 8 studies showed a favorable cost-analysis profile. Three studies performed cost-consequence analyses, none of which reported positive findings. Among 7 cost-utility analyses and 7 cost-effectiveness analyses, 3 and 5, respectively, reported a likely benefit to the usage of rhBMP-2. The CHEERS scores for the included studies ranged from 15 to 23 out of 24 points, with a median score of 19.
Conclusions
There is no consensus regarding the cost-effectiveness or cost-utility of rhBMP-2 in spinal fusion. rhBMP-2 may function as a cost-effective adjunct in spinal fusion surgeries in select cases.
Introduction
The rise in spinal fusion procedures for degenerative conditions has been attributed to the increase in the aging demographic within the United States and other developed countrie. 1 There has been accompanied by an increasing rate of complex spine procedures necessitating large fusion constructs for revision and/or adult spinal deformity (ASD). 2 Traditionally, iliac crest bone graft (ICBG) has long been considered the gold standard for achieving spinal fusion. 3 Although ICBG functions as an excellent bone graft due to its combined osteoinductive, osteoconductive, and osteogenic properties, its use has been increasingly limited by reports of significant donor-site morbidity, such as structural impairment of the pelvic ring, site infection and chronic pain.4,5 Recombinant human bone morphogenetic protein-2 (rhBMP-2) is a biologic agent which has been gaining popularity as an ICBG substitute, where acts as potent inductor of osteogenesis without requiring additional procedures at the donor site.6,7 RhBMP-2 is a soluble, local-acting signaling protein that acts in the SMAD group signaling pathway to induce and promote the differentiation of mesenchymal cells into cartilage and bone-forming cells.8,9 RhBMP-2 can change the cell phenotype (e.g. fat and muscle cells) into that of an osteoblastic lineage. 10
A number of industry-sponsored studies reported no “adverse events” with rhBMP-2 which has led to increased utilization in spinal fusion procedure.11-13 This risk assessment has since been contradicted. Larger studies, such Glassman et al, showed a complication rate of 0.1-0.6 % in 1037 posterolateral fusion cases. 14 According to the “minimal risk” profile, rhBMP-2 use increased from 0.7% of all fusions in 2002 to more than 50% of anterior lumbar interbody fusions and 43% of posterior lumbar and transforaminal lumbar interbody fusions in 2007.15,16
The baseline cost of rhBMP-2 for hospitals can range from $900 to $5500 depending on multiple factors including the specific indicated surgical procedure and patient bone quality. 16 Few studies have conducted cost analyses, comparing rhBMP-2 to other graft options, most single institution studies. Past literature reveals conflicting results regarding cost. While some studies suggest that rhBMP-2 is the more cost-effective choice over iliac crest bone graft, local bone alone, or local bone with demineralized bone matrix or corticocancellous allograft chips, other studies indicate that autograph alone may actually be more economically viable under certain conditions.7,17
Despite its touted safety profile, the cost-effectiveness profile of rhBMP-2 in spinal fusion is yet to be established. The aim of this study is to perform a systematic review of the literature examining the cost-utility, cost consequence and cost-effectiveness of rhBMP-2 in spinal fusion to evaluate whether use of rhBMP-2 is economical in spinal fusion surgery.
Methods
This systematic review was conducted according to formal PRISMA (Preferred Reporting Items for Systematic Reviews) guidelines.
Search Strategy and Eligibility Criteria
PubMed, Medline, OVID and CENTRAL (Cochrane Central Register of Controlled Trials) were queried in January 2024 for studies comparing intraoperative use of spinal fusion using rhBMP-2 with autograft and/or allograft alongside cost-effectiveness, cost-benefit, cost minimization, and/or cost-utility analysis. These databases were searched using the terms [key MeSH terms], “lumbosacral region” [MeSH Terms] OR lumbar OR thoracic OR “Spine” [Mesh] OR SPINE [TIAB] OR SPINAL [TIAB] OR “Thoracic Vertebrae/surgery” [Mesh] OR “Cervical Vertebrae/surgery” [Mesh], Spinal Fusion” [Mesh] OR FUSION [TIAB] OR ARTHRODESIS [TIAB], bone morphogenetic proteins [mesh] OR “bone morphogenetic protein” OR “rhBMP” OR “rhBMP-2, and economics” [MeSH Subheading] OR “economics” [All Fields] OR “cost” [All Fields] OR “costs and cost analysis” [MeSH Terms] OR (“costs” [All Fields] AND “cost” [All Fields] AND “analysis” [All Fields]) OR “costs and cost analysis” [All Fields]. Studies published in English in any year that evaluated the clinical outcomes of spinal fusion surgeries using rhBMP-2 with autograft and/or allograft, as well as their cost effectiveness, were included. Studies published in languages other than English or with no primary outcome or no full text availability were excluded. After initial evaluation of titles and abstracts, the reviewers independently analyzed the full text, and disagreement between the 3 reviewers was resolved by an external moderator blinded to other aspects of the paper other than inclusion and exclusion criteria. PRISMA flow diagram was used to record of number of studies identified, included and excluded, and the reasons for exclusions (Figure 1). The references examined were screened for standardization on economic reporting using the CHEERS criteria.
18
PRISMA flow diagram of number of studies identified, included, and excluded, and the reasons for exclusions.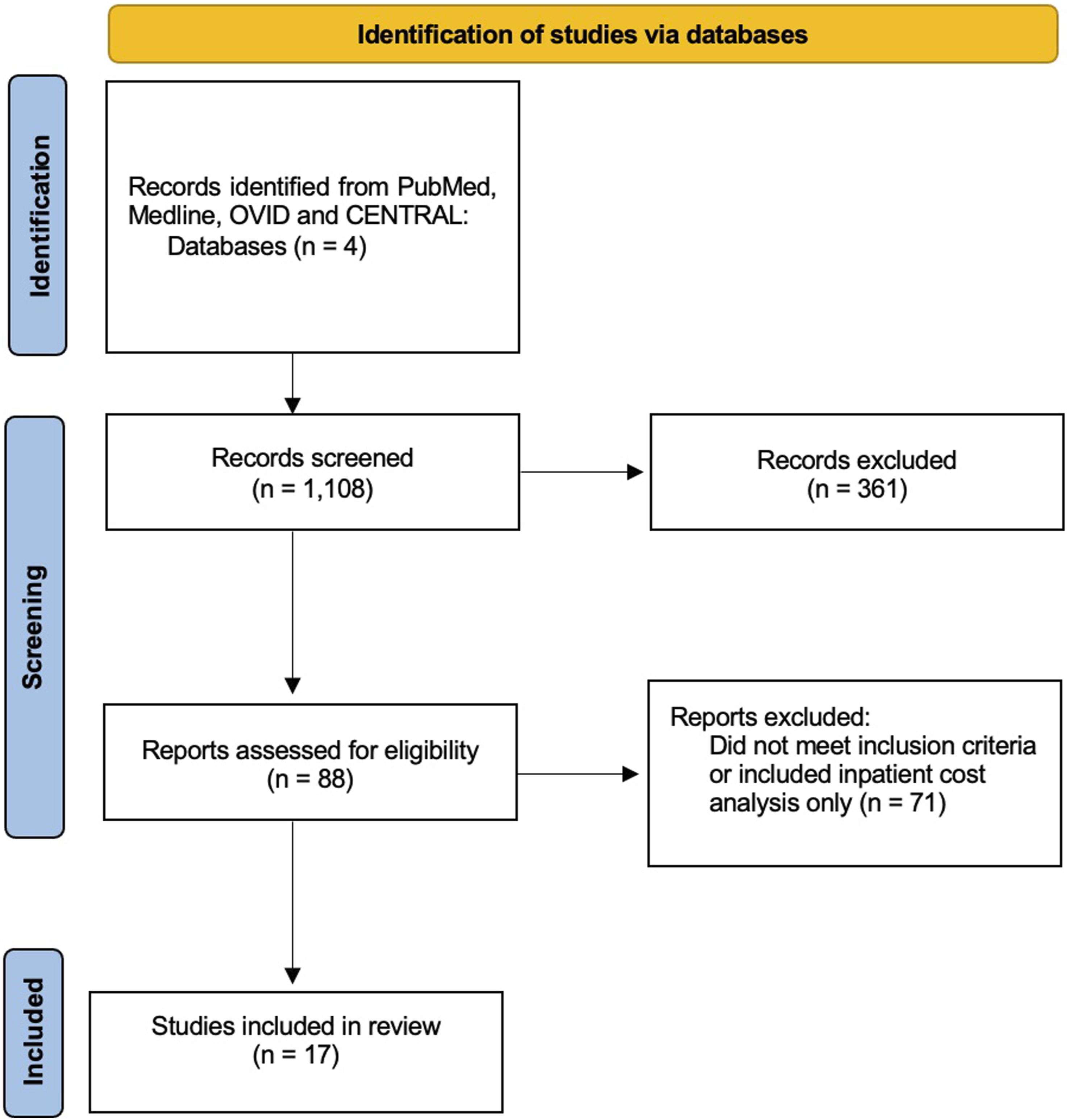
Data Extraction
Three authors independently conducted the initial search and review of literature for inclusion of papers in the systematic review. Data was extracted in the primary format: first author, year of publication, funding, study design, statistical technique, perspective, time horizon, diagnosis, procedure, implants, rhBMP-2, other grafts utilized, clinical data source, population (N), gender discrepancy, age, clinical data points, currency, cost source, cost included, indirect costs, primary findings, and sensitivity analysis. Studies were then categorized by cost-utility, cost-effectiveness and cost-consequence. Cost utility compares 2 or more options based on their costs and outcomes, with the outcomes accounting for both the impact on lifespan and quality of life. Cost effectiveness measures 2 options cost and outcomes measured in a single unit (e.g., rate of revision, complications) whereas in cost-consequence, the outcome is measured with multiple units (e.g., rates of post-operative complications, reoperation, or readmissions). 19
Quality Assessment
Three reviewers independently assessed the standardized reporting of each health economic studies utilizing the CHEERS criteria (Consolidated Health Economic Evaluation Reporting Standards). The CHEERS checklist evaluates 24 independent points of economic evaluation, including study design, perspective, costs, outcomes, and analysis. Twenty-four items split into 6 broad categories–1) title and abstract, (2) introduction, (3) methods, (4) results, (5) discussion, and (6) other (source of funding and conflict of interest)–were evaluated and for each article, a point was given for the presence of each item.
To assess bias, various reporting tools were used based on study design. The revised Cochrane risk-of-bias tool for randomized trials (RoB 2), 20 A MeaSurement Tool to Assess systematic Reviews (AMSTAR 2) 21 and Newcastle-Ottawa Quality Assessment Form 22 for cohort studies were all used. Each checklist has various domains that evaluate different aspects of study quality, such as selection bias, performance bias, detection bias, and reporting bias. By applying these validated instruments, potential sources of bias were identified, allowing for a more accurate interpretation of the evidence and strengthening the reliability of the study findings.
Outcome Measures
The primary outcome measures obtained from each study included the overall costs (direct and indirect) of spinal fusion surgeries utilizing rhBMP-2 relative to those procedures using an alternative osteoinductive agent or no osteoinductive agent. Secondary outcome measures included length of hospitalization, rates of revision and complications, health status questionnaire scores, and productivity loss.
Statistical Analysis
Statistical analyses were carried out by a statistician. Mean and standard deviations were used to describe continuous data. Chi Square Test or Fisher’s Exact Test were used to compare frequencies. Continuous variables were compared using the ANOVA F-Test and Kruskal Wallis Test, depending on variable distribution. Following univariate analysis, a multivariate analysis was also performed on variables that showed statistical significance. Two-tailed P-values of <.05 were considered significant.
Results
Study Selection
Inclusion/Exclusion Criteria for PICOT.

Summary of Included Studies.
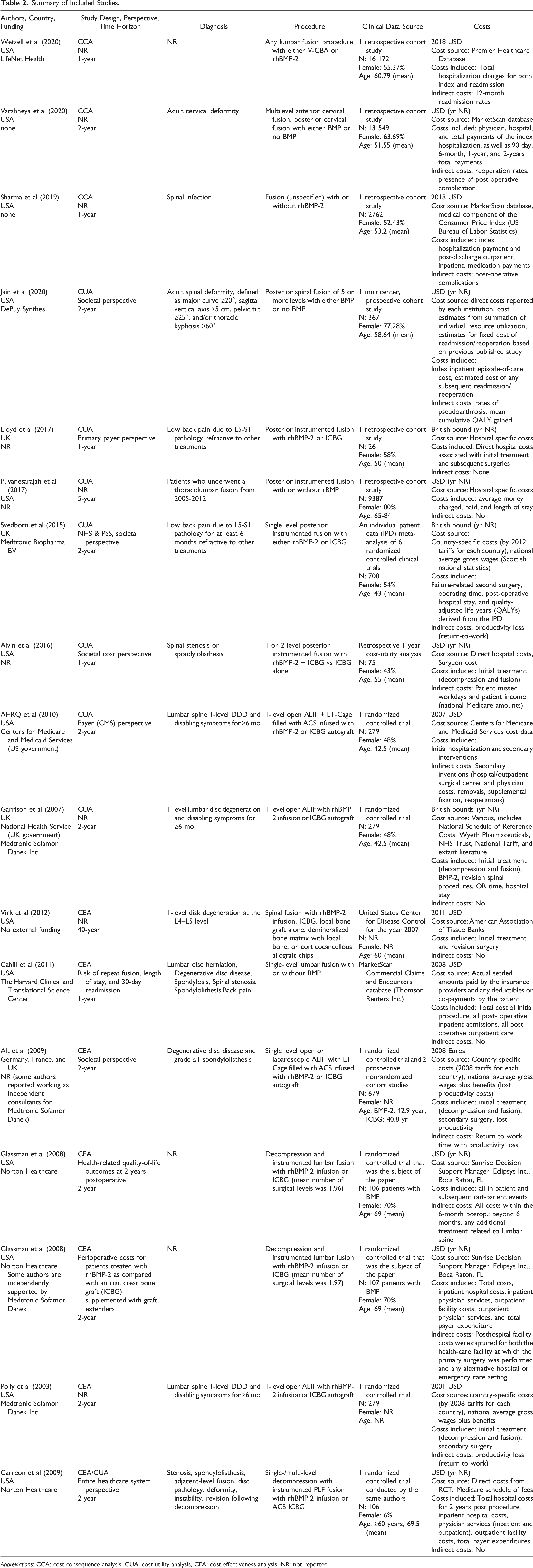
Abbreviations: CCA: cost-consequence analysis, CUA: cost-utility analysis, CEA: cost-effectiveness analysis, NR: not reported.
Bias Assessment
Overall, the quality of included studies varied based on the assessment tools used (Table 4). Cohort studies evaluated with the Newcastle-Ottawa Scale demonstrated generally good quality, with most scoring between 7/9 and 8/9, indicating a low risk of bias. Two of the systematic reviews included (Lloyd 2015, Svedbom 2015) were considered of lower quality based on AMSTAR 2 assessment due to their own inability to report bias in their chosen studies. All randomized trials evaluated using the Cochrane Risk of Bias Tool showed a low risk of bias. Four studies were unable to undergo bias assessment because there was no formal bias tool created for the particular study type (i.e., Statistical decision model study).
Studies of Cost-Consequence
Three studies (Wetzell et al 2020, Varshneya et al 2020, and Sharma et al 2019) were identified as cost-consequence analyses.
Varshneya et al used the MarketScan© Claims Dataset to extract cost data relating to the use of rhBMP-2 in multilevel anterior or posterior cervical fusions in the United States. Results showed while rhBMP-2 usage was associated with lower reoperation rates, it was associated with higher total payments (including after discharge) at the 90-day ($97 917 vs $85 838, P < .0001), 1-year ($103 279 vs $92 357, P = .0004), and 2-year ($107 975 vs $97 620, P = .0021) timepoints.
Sharma et al similarly extracted cost data from the MarketScan© Claims Dataset, assessing the use of rhBMP-2 in patients with spine infections, and found no difference in the rates of post-operative complications, reoperation, or readmissions at 30 days and 6, 12, and 24 months. While rhBMP-2 was associated with increased cost of the index hospitalization ($60 951 vs $47 251, P < .0001), there were no significant difference in total costs at 30 days and 6, 12, and 24 months among patients undergoing fusion for spinal infections.
Wetzell et al compared Premier Healthcare Database cost data for lumbar fusion procedures using either viable lineage-committed bone cells (V-CBA) or rhBMP-2. Results showed the adjusted mean initial procedure and 12-month follow-up hospital charges were significantly lower in the V-CBA group than the rhBMP-2 group ($109 061 and $108 315 vs $160 191 and $130 406, respectively; P < .0001 and P < .0001).
Among 3 cost-consequence analyses, 2 studies found increased costs associated with the use of rhBMP-2 while 1 study of patients with spine infections found no difference.
Studies of Cost-Utility
Seven studies (Jain et al 2020, Lloyd et al 2017, Puvanesrajah et al 2017, Svedbom et al 2015, Alvin et al 2016, AHRQ 2010, and Garrison et al 2007) employed cost-utility analysis techniques.
Jain et al employed a so called ‘Monte Carlo decision-analysis model’ to assess the cost utility of rhBMP-2 use in adult spinal deformity surgeries performed in the United States in which there was a posterior spinal fusion of 5 or more levels compared to when no rhBMP-2 used. The rates of pseudoarthrosis were significantly lower with rhBMP-2 usage although rhBMP-2 usage was associated with increased mean direct costs. Notably, there was no significant difference in cumulative QALYs gained between rhBMP-2 and non- rhBMP-2 groups. The author’s analysis demonstrated that rhBMP-2 was associated with positive incremental utility in 67% of patients and considered favorable at a willingness-to-pay threshold of $150 000/QALY in >52% of patients.
Alvin et al examined the cost-utility of adding rhBMP-2 in single- or two-level posterior instrumented fusions for spinal stenosis or spondylolisthesis to ICBG autograft relative to ICBG without rhBMP-2. Costs included in the analyses were those associated with the initial treatment as well as indirect costs related to missed employment and loss of income. The 1-year cost-utility ratio (total cost/DQALY) for the control cohort was significantly lower ($143 251/QALY gained) than that of the rhBMP-2 cohort ($272 414/QALY gained) (P < .05). Incremental cost-effective ratio (ICER) analysis at 1-year follow-up demonstrated that the control cohort (autograft) far outpaced the rhBMP-2 cohort.
Svedborn et al examined the cost utility of rBMP-2 in the United Kingdom, comparing rhBMP-2 to ICBG for single-level posterior instrumented fusion surgeries in patients presenting with lower back pain due to L5-S1 degenerative disc disease (DDD) for at least 6 months. Patients treated with rBMP-2 achieved higher mean health utility following surgery relative to ICBG and over 4 years accrued an additional 0.055 QALYs. These findings resulted in an increase in the incremental cost per QALY of £13 523. ICER remained stable while varying a range of inputs in deterministic sensitivity and scenario analyses. The authors concluded that rhBMP-2 is cost-effective for use in lumbar interbody fusions compared to ICBG at a willingness-to-pay threshold of £20 000 per incident case.
Lloyd et al examined the cost-utility of rhBMP-2 utilization in posterior instrumented fusions for lower back pain due to DDD in the United Kingdom relative to ICBG. Notably, indirect costs were not included. Under the assumption that rhBMP-2 is associated with a lower reoperation rate compared to ICBG, the analysis found that using rhBMP-2 in all instrumented lumbar fusions to prevent a single reoperation was calculated at £26 923 while the average direct costs of a failed instrumented lumbar fusion with ICBG followed by re-operation with rhBMP-2 totaled £47 734, a difference that, however, was not statistically significant. Therefore, the authors concluded that using rhBMP-2 in all patients undergoing a posterior instrumented fusion is not recommended on a cost-utility basis.
The AHRQ study conducted a similar analysis using data in the United States and assessed rhBMP-2 vs ICBG in single-level open anterior lumbar instrumented fusions. This study determined that rhBMP-2 is favored from the payer perspective on a cost per QALY basis over ICBG assuming that rhBMP-2 use does not incur additional costs. rhBMP-2 was associated with a $592 savings per QALY relative to ICBG (rhBMP-2: $31 159 per 1.218 QALY; ICBG: $31253 per 1.194 QALY). Sensitivity analysis determined that if rhBMP-2 was an added cost of $3000 or greater, it was no longer the dominant treatment. However, if rhBMP-2 did not incur additional treatment cost, for variations done on utility values, costs other than of rhBMP-2 administration, fusion rates, secondary intervention rates, and donor site pain rates, rhBMP-2 dominated.
Puvanesrajah et al examined the cost-utility of rBMP-2 in patients who underwent a thoracolumbar fusion from 2005 to 2012 in the United States. Increased weighted averages of hospital charges and paid amounts were observed with rhBMP-2 use, even after adjusting for differing risks of revision surgery, in both 3-7 level ($177 850 vs $148 158) and 8 or greater level fusions ($302 624 vs $255 828). The authors established that while rBMP-2 reduced the risk of non-union in certain patient populations, it could not be recommended for routine administration on a cost-utility basis.
Garrison et al examined the cost-utility of single-level open anterior lumbar instrumented fusions using an LT-Cage device with rhBMP-2 relative to ICBG in the United Kingdom. ALIF with rhBMP-2 was determined to be associated with an increased cost of £120 390 per QALY gained compared to ICBG and therefore is unlikely to be recommended on a cost-utility bases. However, the use of rhBMP-2 was associated with shorter OR time, inpatient length of stay, and reduced need for revision surgery.
Among 7 cost-utility analyses, 3 studies reported a likely benefit to usage of rhBMP-2, while 4 studies failed to find a benefit regarding cost-utility.
Studies of Cost-Effectiveness
Seven studies (Virk et al 2012, Cahill et al 2011, Alt et al 2009, Glassman et al 2008, and Glassman et al 2008) reported cost-effectiveness analyses.
Virk et al compared the cost-effectiveness of rhBMP-2 Infuse®, ICBG, local bone graft alone (LBG), demineralized bone matrix with local bone (DBM), and corticocancellous allograft chips for spinal fusion surgeries performed for patients diagnosed with single-level disc degeneration at the L4-L5 level. rhBMP-2 was determined to be the most cost-effective graft option largely due to the reduced rate of revision spine surgery. ICER analysis demonstrated that rhBMP-2 cost $16 595 per additional QALY which represented significant savings relative to the second lowest cost (LBG: $21 204/QALY). The most common alternative to rhBMP-2–ICBG–cost $21 308 per additional QALY.
Polly et al compared two-year costs associated rhBMP-2 and ICBG in single-level ALIF surgeries for DDD. Over a 2-year period, the total direct costs of rhBMP-2 and ICBG were nearly equivalent. The increased cost of rhBMP-2 was offset by prevention of pain and complications associated with ICBG, less operating room time, shorter total LOS, and autograft extenders/harvesters. The higher success rate (94.5% vs 88.7%) of the index surgery in rhBMP-2 patients offset the remaining difference in costs. Sensitivity analysis confirmed these findings. Notably, high-risk patient populations such as smokers were associated with rhBMP-2 being more cost-effective.
Glassman et al published 2 randomized clinical trials in 2008 examining the cost-effectiveness of rhBMP-2 relative to ICBG for lumbar spine decompression in combination with instrumented fusion. One study explored rhBMP-2 cost-effectiveness relative to ICBG in patients greater than 60 years of age and included inpatient costs and subsequent outpatient events. Mean cost of the initial admission was higher for rhBMP-2 patients ($36 530 vs $34 235 for ICBG). However, the total cost after 2 years was less in the rhBMP-2 group ($40 131 vs $42 574) among elderly patients. A second investigation included total perioperative costs (inpatient hospital costs, inpatient physician services, outpatient facility costs, outpatient physician services, and total payer expenditure). Total payer expenditure for the three-month perioperative period averaged $33 860 in the rhBMP-2 group and $37 227 in the ICBG group. The authors noted that the hospital carries the initial cost burden associated with the utilization of rhBMP-2 while the payer in a Diagnoses-Related Group (DRG) model would experience a net saving. Ultimately, rhBMP-2 demonstrated a significant cost-effectiveness benefit in both analyses.
Cahill et al compared rhBMP-2 utilization to a lack thereof in single-level lumbar fusion surgeries with data extracted from the Marketscan© claims dataset. While rhBMP-2 was associated with a decreased risk of revision surgery, cost analysis demonstrated increased up-front and 1-year costs. Total costs associated with the use of rhBMP-2 were $42 627 vs $38 686 without (P < .01). Total one-year costs were similarly increased in the rhBMP-2 cohort ($53 624 vs $49 315, P < .01) with an adjusted increase of 10.1%.
Alt et al examined the cost-effectiveness of single-level lumbar fusions with rhBMP-2 fusion vs with ICBG autograft in Germany, France, and the UK. While rhBMP-2 was associated with increased initial costs, several factors were identified which ultimately resulted in favoring of rhBMP-2. These factors included cost reduction by shorter operating time, lower rates of secondary intervention, faster return to work, and cost of productivity loss. The authors reported a cost-effectiveness benefit per case of €9191 in France, €8483 in Germany, and €8567 in the United Kingdom.
Carreon et al conducted a cost-effectiveness analysis with an additional cost-utility component. Cost data was obtained from the trial as well as the Medicare schedule of fees; costs included total hospital costs for 2 years post procedure, inpatient hospital costs, physician services (inpatient and outpatient), outpatient facility costs, and total payer expenditures. rhBMP-2 costed less than ICBG with an increased improvement in utility score of 0.01 (BMP-2: $39 967/0.11, ICBG: $42 286/0.10 [cost/utility]). A decision-tree analysis noted that while the incidence of complications was not significantly different between groups, the costs associated with management of complications of the ICBG group were greater.
Among 7 cost-effectiveness analyses, 5 studies reported a likely benefit to usage of rhBMP-2, while 1 study failed to find a benefit regarding cost-effectiveness. One study reported nearly equivalent cost-effectiveness outcomes.
Study Quality Assessment
Results, Limitations, and CHEERS Scoring of Included Economic Studies.
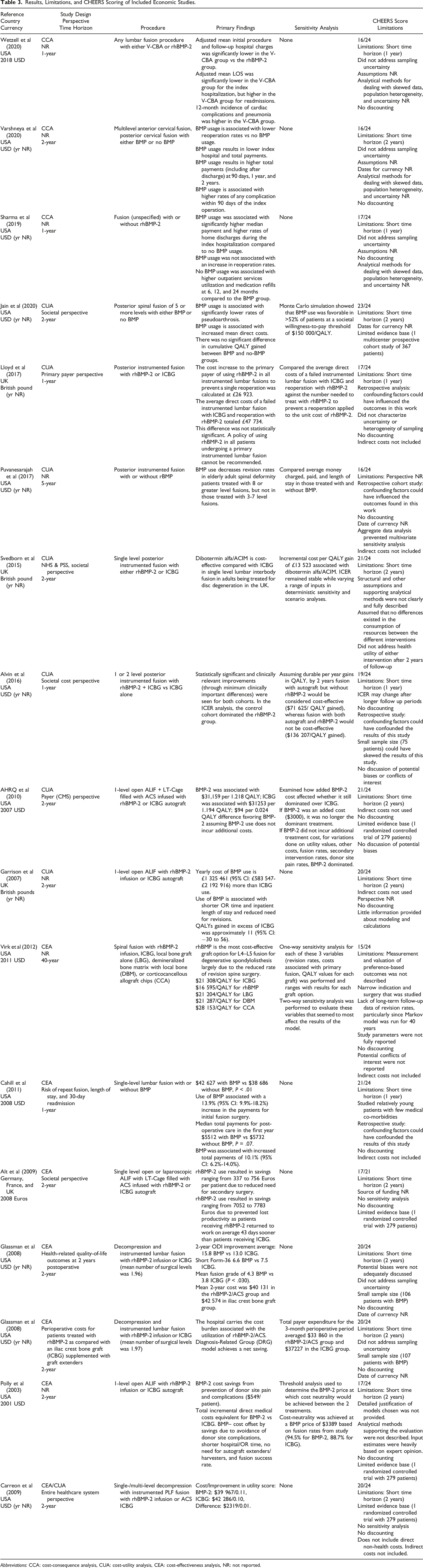
Abbreviations: CCA: cost-consequence analysis, CUA: cost-utility analysis, CEA: cost-effectiveness analysis, NR: not reported.
Discussion
Bias Assessment. All Studies Used in the Systematic Review of Literature, the Bias Tool for Assessment and Their Respective Score. A Good Score Indicates Low Risk of Bias. A Low Score Indicates a Higher Risk of Bias.

Among the 17 studies meeting our inclusion criteria, 8 reported favorable findings in regard to a cost-analysis of the usage of rhBMP-2. However, none of the cost-consequence studies reported positive findings, and among 7 cost-utility studies, 3 reported a likely benefit. Among 7 cost-effectiveness analyses, 5 studies reported a likely benefit to usage of rhBMP-2. The findings of our study demonstrated that there remains no consensus regarding the cost-effectiveness or cost-utility of rhBMP-2 in spinal fusion. In fact, rhBMP-2 may function as a cost-effective adjunct in spinal fusion surgeries if used in a judicious manner in well-selected patients, but not if used in a ubiquitous routine fashion as a fusion adjuvant. Studies that report the cost-effectiveness of rhBMP-2 attribute the difference in cost to decreased pseudoarthrosis and re-operation rates, in addition to the shorter hospital stay during the index hospital visit, fewer post-operative pain medication requirements, and a faster return to work with a decreased loss of income. Studies that examined the use of rhBMP-2 in combination with autologous bone grafts in spinal fusion found varying and sometimes reduced complication rates while frequently higher overall median costs at the 1- and 2-year mark. Garrison et al investigated the clinical efficacy and cost-effectiveness of rhBMP-2 proteins in non-healing fractures and spinal fusion and found rhBMP-2 to be clinically more effective than autologous bone graft for radiographic fusion in patients with single-level spinal fusion. However, Garrison et al report rhBMP-2 to be cost ineffective and advocate for further cost-effectiveness research. It should be noted that the external validity of study by Garrison et al is limited by a small sample size and limited studies of comparison given the recent market release. 34
Further analysis of the studies included in our review suggests that rhBMP-2, if used judiciously, may indeed be cost-effective in specific patient populations and for certain types of spinal fusion procedures. Clinical and patient factors, economic impacts, and long-term cost-utility need to be considered when determining the use of rhBMP-2 in spinal fusion surgeries. Virk et al noted that in a single level (L4-L5) spinal fusion, rhBMP-2 use was the most cost-effective option vs ICBG when considering the avoidance of reoperation, further categorizing $21 308/QALY for ICBG vs $16 595/QALY for rhBMP-2. 7 Carreon et al and Glassman et al also suggests that rhBMP-2 may be more cost effective than ICBG alone in patients over the age of 60, where bone healing potential tends to be declining and the risk of nonunion is increasing, citing a value of $46 852 for revision surgery for incidents of nonunion.35,36 The cost-effectiveness of rhBMP-2 in patients greater than 60 years of age is likely due to higher rates of nonunion in patients receiving ICBG alone compared to those also receiving rhBMP-2. 35
Furthermore, several studies have explored the cost effect and utility of rhBMP-2 when combined with other osteogenic pathway augmenters. For instance, Differ et al studied modulators of the nitric oxide pathways in vitro and found that nitric oxide synthase (NOS), nitric oxide, and soluble guanylyl cyclase (sGC) supplementation all enhanced rhBMP-2 signaling. 37 A similar study performed in an in an vivo in rats shows that inhibition of NOS leads to decreased fusion mass and less stable fusion. 38 The use of cartilage oligomeric matric protein (COMP) is also shown to augment rhBMP-2 signaling by binding to rhBMP-2 and increasing its osteogenic activity, both in vitro and in vivo. 39 They also report that COMP plus low-dose rhBMP-2 provided significantly better fusion than low-dose rhBMP-2 alone. 40 These studies suggest that lower doses of rhBMP-2 plus COMP or NOS could decrease the potential dose-dependent side effects and costs of rhBMP-2 in spinal fusion while having comparable fusion rates with higher doses of rhBMP-2. However, additional studies are needed to assess the effects of these rhBMP-2 adjuncts on rhBMP-2’s overall cost-effectiveness in humans.
This review has several limitations. First, while 4 different databases were used, PubMed, Medline, OVID and CENTRAL, they are still limited, and some literature may not be represented in the present study. Second, a quantitative meta-analysis evaluating the cost effectiveness of rhBMP-2 and other biologics for promoting arthrodesis was not possible secondary to heterogenous outcome parameters. Encouragingly, the CHEERS criteria ensures that economic studies are transparent, consistent, and comprehensive, and allows a level of standardization so studies can be properly compared. Additionally, we were able to assess bias in 4 of the studies because they did not fit into a study type category for a given bias assessment tool. Several limitations common to a large proportion of the reviewed studies were identified. All but 2 studies had a time horizon less than or equal to 2 years. Additionally, ten studies did not adequately describe all methods dealing with skewed, missing, or censored data, extrapolation methods, methods for pooling data, approaches to validate model, and methods for handling population heterogeneity and uncertainty or explain why such methods were not necessary. Eleven studies did not report and justify the choice of discount rates used for costs and outcomes, and 9 studies did not report the year of currency used in analysis. Furthermore, 9 studies did not explicitly characterize uncertainty for all input parameters and the consequence of methodological assumptions. Four studies that assessed the cost-effectiveness of rhBMP-2 compared with ICBG for anterior lumbar interbody fusion relied on the same single noninferiority randomized trial that served as the pivotal trial for FDA approval of Medtronic’s Infuse (rhBMP-2).
Of particular note, at least five of the studies included in our systematic review reported that at least 1 author had a financial conflict of interest with the manufacturer of rhBMP-2. Although the mere existence of a conflict of interest does not merit the discounting of results and conclusions, such relationships should be considered when interpreting the data. Despite the aforementioned limitations, all studies fulfilled at least fifteen of the CHEERS criteria, and 8 studies scored twenty or higher, indicating a proper standard of economic reporting.
Conclusion
The findings of this systematic review demonstrate that the true cost-effectiveness or cost-utility of rhBMP-2 in spinal fusion remains unclear. The use of rhBMP-2 may function as a cost-effective adjunct in spinal fusion surgeries if used in a judicious manner in well-selected patients. The extant literature is of overall moderate quality, with several limitations in methodology, although several high-quality studies were identified. Further research, preferably in the form of randomized controlled trials, will be helpful to further elucidate the economic benefit of the use of rhBMP-2 in spinal fusion surgeries in more discretely targeted at-risk populations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
