Abstract
Study Design
Retrospective cohort study.
Objective
De novo postoperative urinary retention (POUR) after lumbar posterior decompression surgery for lumbar spinal canal stenosis (LSCS) is a statistically known but uncommon complication for both patients and spine surgeons. The aim of this study is to review clinical data and imaging findings and identify preoperative predictors of de novo POUR.
Methods
The subjects were 738 surgically treated patients with LSCS, without preoperative bladder dysfunction or perioperative complications. Univariate and multivariate analyses using propensity score matching were performed to identify prognostic factors for POUR lasting for at least 1 week after postoperative urinary catheter removal.
Results
POUR occurred in 23 patients (3.1%). The median recovery time was 41 days and only 12 patients (52.2%) showed improvement within 3 months. Patients with POUR were significantly older, and the lumbar Cobb angle, location of compressed dura mater (ventral or dorsal), and type of cauda equina redundancy (curve-type) were identified as independent prognostic factors. POUR had no association with sex, comorbidities, surgical procedures, number of decompressed segments, or degree of dura mater compression.
Conclusions
This study suggests that older age and curve-type stenosis with ventral or dorsal compression of the dura mater are risk factors for development of de novo POUR. Such preoperative imaging findings may indicate a higher risk of intraoperative thermal and nerve injuries, and possible uneven cauda equina flow improvement after decompression.
Keywords
Introduction
Lumbar spinal canal stenosis (LSCS) is a common degenerative disease that has increased in parallel with aging of society, with one cross-sectional study in Japan showing a symptomatic LSS rate of 9.3%. 1 A nationwide survey of patients aged ≥65 years who underwent spinal surgery for LSCS indicated an incidence of surgical complications of 10.8%, with dural tear having the highest incidence (3.6%), followed by deep wound infection (1.4%), neurological complications (1.3%), and epidural hematoma (1.3%). 2 Among neurological complications, de novo postoperative urinary retention (POUR) is an unexpected complication for both patients and spine surgeons, and is associated with augmented medical costs, including the need for intermittent voiding, increased risk of urinary tract infection, and a prolonged hospital stay. The reported incidence of POUR associated with LSCS varies widely from 5% to 70% due to differences in definitions.3-6 In this study, we focused on patients who developed POUR that lasted for more than 1 week after lumbar posterior decompression surgery, and who had no particular perioperative problems. The aim of the study was to review clinical and imaging findings and identify preoperative predictors of de novo POUR in this patient population.
Methods
Study Population
In this retrospective cohort study, medical records of 975 patients with LSCS underwent lumbar posterior decompression, either with (n = 274) or without (n = 701) fusion at our hospital, at our hospital between 2005 and 2020 were reviewed. Patients with severe preoperative bladder dysfunction (i.e., urinary incontinence or retention), prior urological surgery, or perioperative complications other than POUR (e.g., intraoperative dural tears, epidural hematoma, and long-term urinary catheterization) were excluded. Finally, a total of 738 patients underwent posterior decompression with (n = 214) or without (n = 524) fusion for LSCS and had a minimum follow-up period of one year were enrolled in the study (Figure 1). All the patients had clinical signs and symptoms of LSCS. The indications for surgery for LSCS were symptoms of persistent neurological signs, such as neurogenic intermittent claudication at the level of stenosis on high-resolution magnetic resonance imaging (MRI), that were refractory to conservative treatment. The study protocol was approved by the Human Ethics Review Committee of our University Medical Faculty (Approval Number 20220210) and adhered to the Clinical Research Guidelines of the Ministry of Health, Labor, and Welfare of the Japanese Government. Cohort selection flowchart for patients with and without de novo postoperative urinary retention.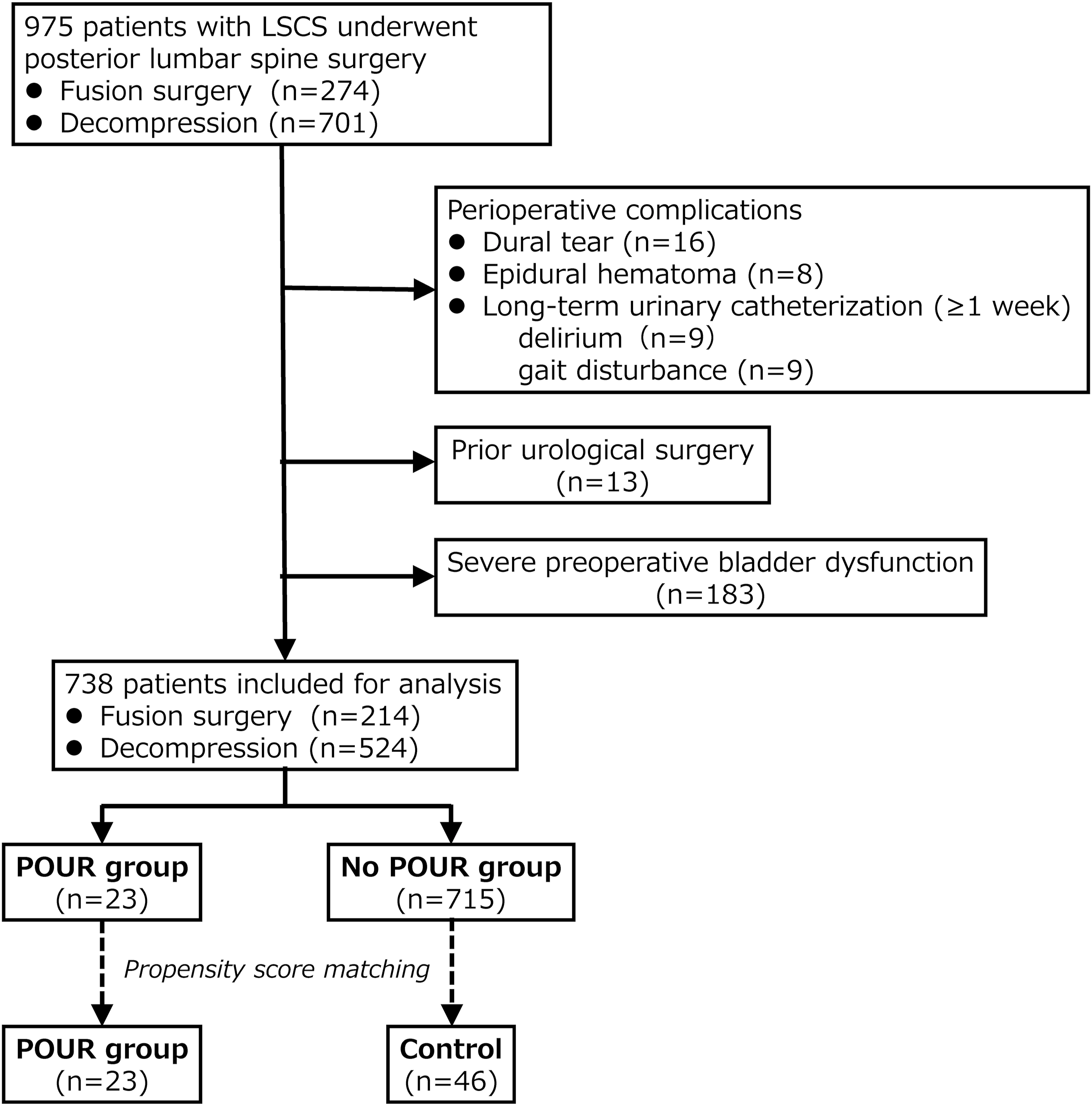
Surgical Procedure
In 2015, we modified the surgical approach for lumbar decompression at our institution, transitioning from conventional laminotomy to lumbar spinous process-splitting (split) laminotomy. 7 The conventional laminotomy approach involved detaching the paraspinal musculature from the spinous process, and then excising a portion of the lamina between its superior and inferior segments. The split laminotomy approach entailed the longitudinal division of the cephalad spinous process, along with its attached bilateral paraspinal muscles, into two equal parts starting from the lamina base. Once decompression was achieved, the separated halves were repaired with sutures, which also reattached the supra- and interspinous ligaments. The lumbar fusion procedure (i.e., posterior lumbar interbody fusion) involved a conventional open midline approach for complete bilateral facetectomy. 8 Following disc material removal and vertebral endplate preparation, two cages were inserted into each intervertebral space filled with locally resected autologous bone.
POUR Definition and Management
De novo POUR was defined as urinary retention for at least 1 week after postoperative urinary catheter removal or the need for intermittent urinary drainage without preoperative bladder dysfunction. A diagnosis of POUR was established by urologists if urinary drainage could not be confirmed after urinary catheter removal, and if clinical signs and symptoms such as lower abdominal distention and pain or residual urine greater than 200 mL were observed. Urinary tract management involved intermittent urinary drainage and/or oral medications such as bethanechol chloride. POUR was considered improved when the residual urine volume decreased to less than 100 mL, intermittent catheterization was gradually discontinued, and the patient demonstrated successful spontaneous urination.
Outcomes and Radiological Measurements
For overall comparisons, clinical data for the affected level, age, sex, symptom duration, preoperative Japanese Orthopaedic Association (JOA) score, and number of decompressed segments were acquired from medical charts. The JOA score has a maximum of 29 points from 3 subscales: subjective symptoms (maximum score of 9 points), clinical signs (6 points), and activities of daily life (14 points). Patient background (body mass index (BMI), smoking history, comorbidities (hypertension, diabetes), laboratory data (creatinine, total cholesterol, and triglycerides), surgical factors (operation time, blood loss, intraoperative infusion volume, history of lumbar surgery), and imaging findings were compared between cases with postoperative bladder dysfunction and matched controls.
Imaging findings were obtained from plain lumbar radiography and high-resolution MRI. The lumbar Cobb angle (L1-L5), intervertebral disc wedge angle at the level of the largest compression segment, lumbar lordotic angle (L1-S1), and pelvic incidence (PI) were measured on preoperative plain lumbar radiographs. The transverse area of the dura mater at each segment between L1-2 and L5-S1, the stenosis severity classification (Grade A-D),9,10 location of compressed dura mater based on the line of the intervertebral joint (ventral, middle, and dorsal), type of cauda equina redundancy (serpentine-shaped and loop-shaped),11,12 and stenosed cauda equina morphology (soft, sharp/straight, and curve type)
13
(Figure 2) were determined on preoperative sagittal and axial T2-weighted MRI. The transverse area of the dura mater was measured preoperatively using a Picture Archiving and Communication System. All measurements were performed in triplicate by two observers and average values were used. (A) Dural cross-sectional area was measured using T2-wighted MRI. (B) Severity grade classification. Grade A: cerebrospinal fluid is noticeably present within the dural sac, and the arrangement of the roots is irregular. Grade B, nerve roots are spread throughout the entire thecal sac, but can still be distinguished from one another. Grade C, individual single roots are no longer discernible. An epidural triangle of adipose tissue is observed between the lamina and dura mater. Grade D, unlike grade C, the fat has been completely squeezed out. (C) The location of the compressed dura mater was classified based on the intervertebral joint line (dotted lines). (D) Redundant nerve roots are divided into two groups on the basis of their morphologic appearance: serpentine-shaped or loop-shaped. (E) The morphology of the compressed dural sac was classified into 3 types based on the line connecting the upper and lower edges of the posterior margin of the vertebral body (dotted lines).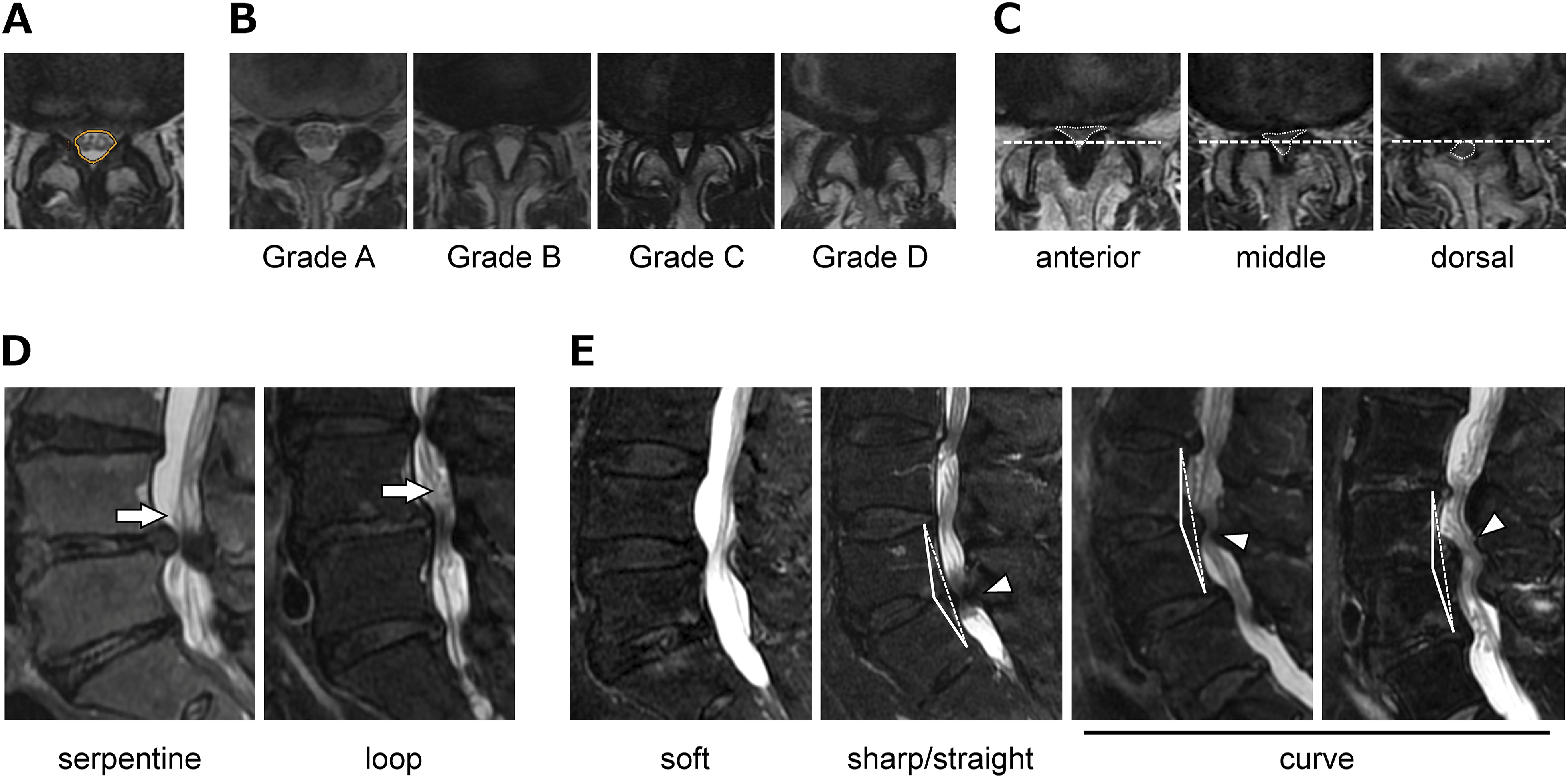
Statistical Analysis
Data are presented as the median [interquartile range] or patient number (%). Categorical variables were compared by Mann-Whitney U-test or chi-square test, with
Results
Demographics of Patients
Background Data for 738 Patients With and Without Postoperative Urinary Retention.
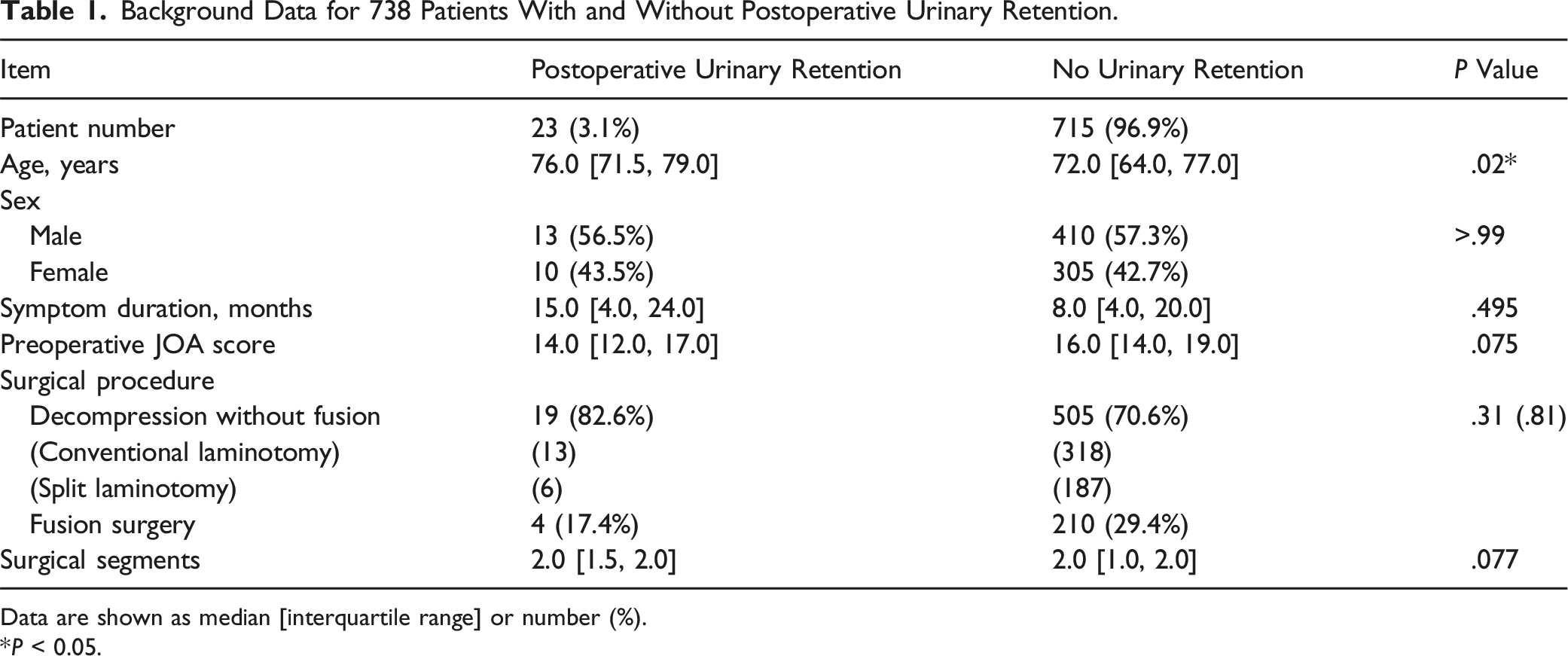
Data are shown as median [interquartile range] or number (%). *
Prognosis of de Novo Postoperative Urinary Retention
Prognosis of 23 Patients With de Novo Postoperative Urinary Retention.
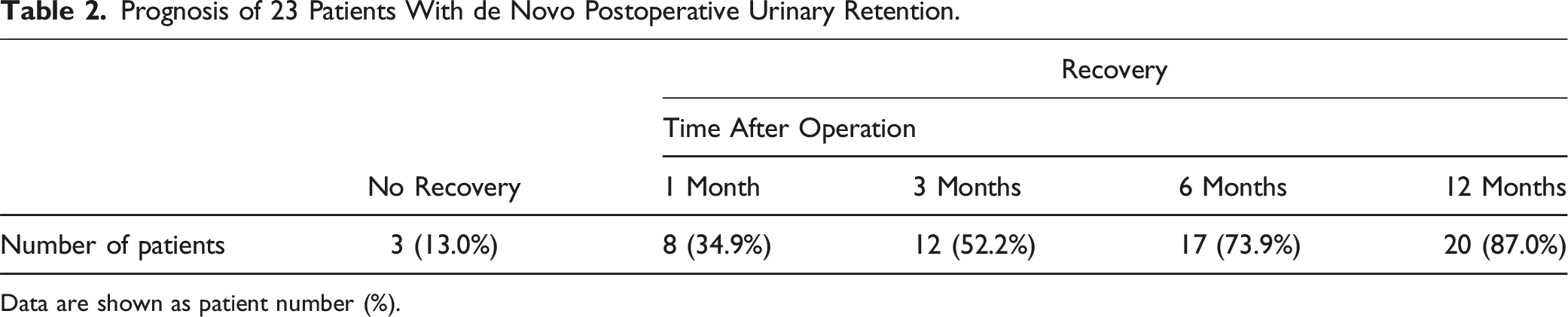
Data are shown as patient number (%).
Preoperative Characteristics and Imaging Findings in Patients With and Without POUR
Background Data for Patients With Postoperative Urinary Retention and Propensity-Matched Controls.
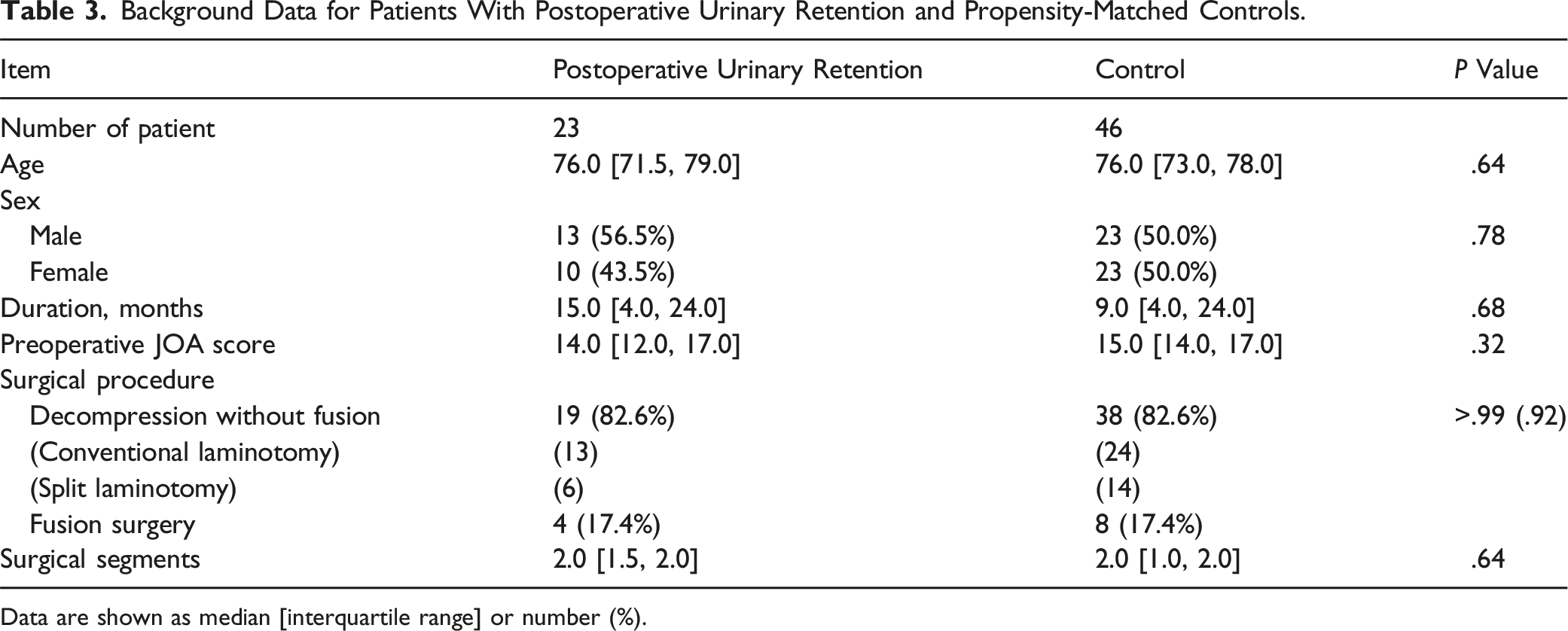
Data are shown as median [interquartile range] or number (%).
Comparison of Clinical and Imaging Findings for Patients With Postoperative Urinary Retention and Propensity-Matched Controls.
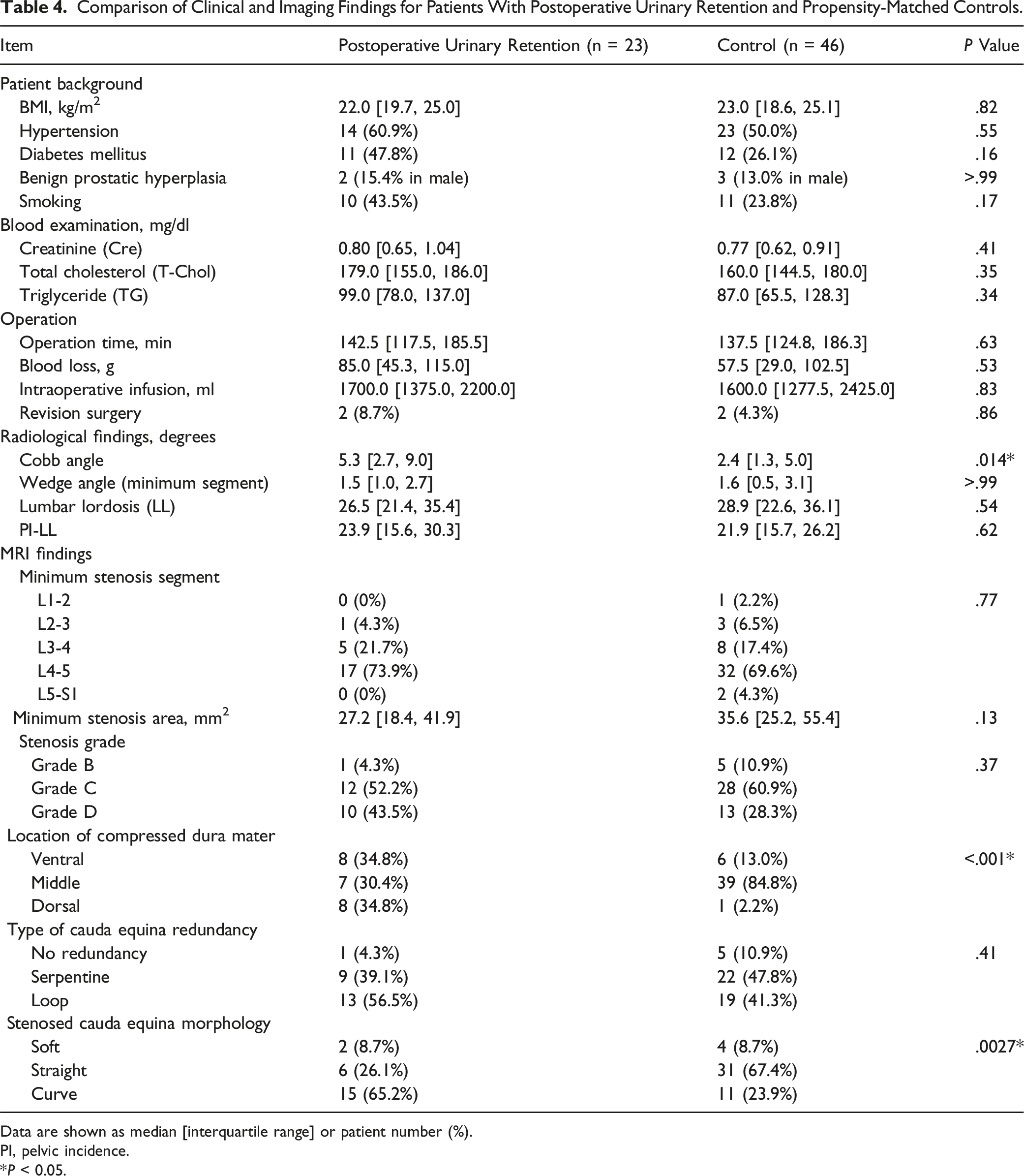
Data are shown as median [interquartile range] or patient number (%).
PI, pelvic incidence.
*
Multivariate Logistic Regression Analysis of Predictors of Postoperative Urinary Retention After Lumbar Decompression Surgery.
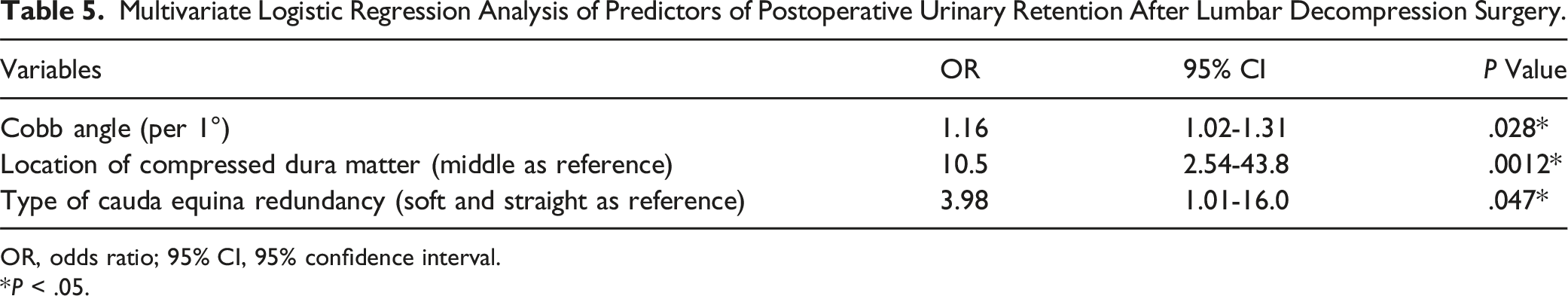
OR, odds ratio; 95% CI, 95% confidence interval.
*
Representative Case
Images of a representative 78-year-old female with POUR after laminotomy at L3-4 and L4-5 are shown in Figure 3. The patient had a chief complaint of intermittent claudication and no preoperative bladder dysfunction. Lumbar decompression surgery was performed without adverse events; however, POUR and perineal paresthesia developed. It took 10 months to gradually reduce the need for intermittent catheterization. Preoperative sagittal MRI showed cauda equina redundancy (loop-type, caudal side of the L3-4 segment) with curved-type dural sac compression. Preoperative transverse MRI revealed that the dura mater was compressed ventrally. Postoperative MRI at 6 months after surgery showed no abnormal findings with adequate decompression and normalization of cauda equina flow. Representative case. A 78-year-old female developed de novo postoperative urinary retention after laminotomy at L3-4 and L4-5. (A) Preoperative MRI showing cauda equina redundancy at L3-4 with curved-type dural sac compression and ventrally located compressed dura mater. (B) Postoperative MRI at 6 months after surgery showing no abnormal findings with adequate decompression and normalization of cauda equina flow.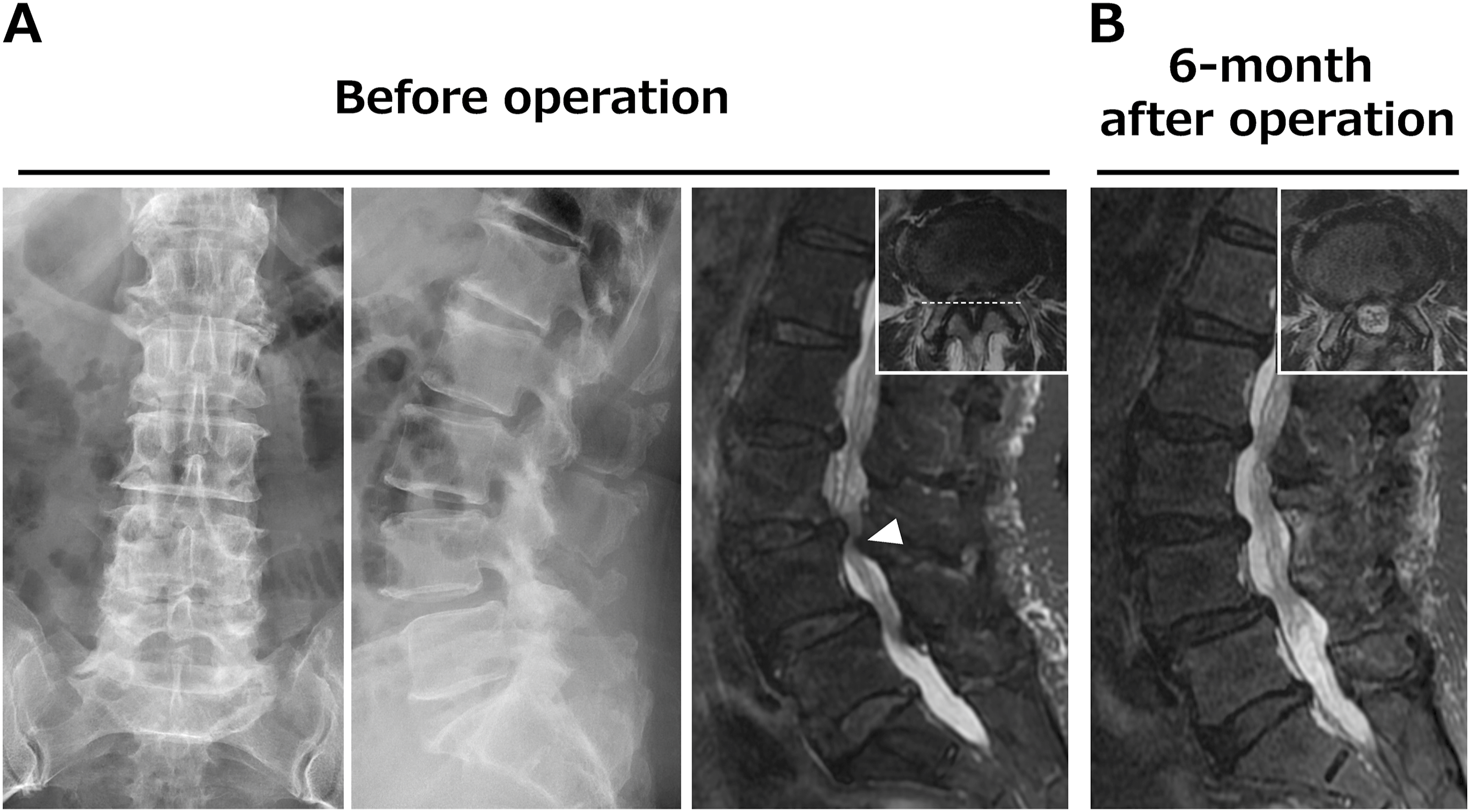
Discussion
POUR occurs frequently after surgical procedures, with an incidence ranging from 3% to 70%. 15 A meta-analysis of POUR following elective spine surgery indicated an overall incidence of 15.1%. 16 There may be a higher risk of POUR following lumbar spinal surgery, with a reported rate of 6%-38% after elective spine surgery.4-6,17 A meta-analysis of POUR following lumbar spine surgery found an incidence of 8.6%. 18 The differences in rates may be partly attributable to definitions of POUR including cases with temporary symptoms. The results of this study showed improvement after 1 and 3 months in only 34.9% and 52.2% of patients postoperatively, indicating that once POUR occurs, it takes time to improve and is burdensome for the patient. To our knowledge, this is the first study to focus on risk factors, including preoperative imaging findings, for postoperative continued (≥1 week) de novo bladder dysfunction without perioperative complications.
Several studies have investigated the relationships of demographic factors with emergence of POUR following LSCS. A meta-analysis of risk factors for POUR after lumbar spine surgery revealed a significant risk for male patients, with an OR of 1.38. Furthermore, the difference in mean age between patients with and without POUR was 5.58 years (POUR + vs POUR−: 64.84 vs 59.51 years). 18 Benign prostatic hyperplasia (BPH) has also been associated with a higher risk of POUR.16,19,20 The conclusion that POUR is more likely to occur in elderly males with BPH is easy to understand. However, the patient population in the current study was older than those in previous studies, and while de novo POUR was more frequent in older patients, there was no effect of sex including the rate of BPH in male patients. A previous study also found no significant sex difference in POUR incidence after lumbar surgery for LSCS. 5
Some studies have indicated an association of POUR with comorbidities such as diabetes and lifestyle choices such as smoking. A meta-analysis showed that patients with diabetes had a significantly increased risk for POUR (OR 1.53). 18 Patients with diabetes commonly have impaired bladder sensation, increased post-voiding residual urine volume, and decreased detrusor contractility as urodynamic findings. Smoking accelerates progression of endothelial sclerosis, leading to a greater likelihood of bladder ischemia and a higher incidence of lower urinary tract complications. However, these associations were not found in the current study, which included relevant blood tests. Associations of POUR after lumbar spine surgery with surgical factors, including fusion surgery, multi-level lumbar spine surgery, longer operative time, incidental dural tears, higher volume of fluid infusion during surgery, have also been suggested.16-19,21 Excessive intravenous fluid administration can result in bladder overdistension, which may subsequently lead to suppression of detrusor function and POUR. 22 However, no association of POUR with these surgical factors was found in this study. The differences in results may be a consequence of the difference in pathogenesis between temporary POUR and persistent de novo POUR.
An association between redundant nerve roots and the degree of stenosis in patients with LSCS has also been proposed.10,23 In addition, a meta-analysis indicated that a finding of redundant nerve roots on preoperative MRI was associated with negative clinical outcomes.24,25 A previous study suggested that the cutoff size of the minimum cross-sectional area of the dural sac in the overactive bladder was 69 mm2. 26 Contrary to our predictions, however, we found no correlation of POUR with the minimum stenosis area (27.2 and 35.6 mm2 in patients with and without POUR, respectively) or the presence of cauda equina redundancy. In contrast, the morphology and location of the stenosed cauda equina were independent prognostic factors for de novo POUR. The image findings of curve-type stenosis with the dura mater located dorsal or ventral to the spinal canal can be observed with or without spondylolisthesis due to the position of the intervertebral disc and ligamentum flavum hypertrophy, as shown in the representative case. The nerve roots responsible for control of urinary function originate primarily from the lower sacral roots, specifically the S3 and S4 roots, and tend to occupy a more central position. 27 An association between intraoperative dural tear in the central area and POUR has also been reported, 21 but in cases with such imaging findings, decompression procedures without dural damage may induce POUR. The lumbar Cobb angle was also identified as an independent risk factor for POUR, but the median Cobb angle was <10° in both groups, which does not constitute degenerative scoliosis. A greater Cobb angle may indicate increased biomechanical stress or altered spinal alignment, which could affect the neural elements responsible for urinary control. Further investigation is needed to elucidate the mechanisms underlying this association.
A hypothesis of the pathomechanism of de novo POUR based on the results of this study is shown in Figure 4. In a curve-type stenosis, the compressed dura mater is located dorsal or ventral to the spinal canal, and there is more concern about thermal injury to the nerve root caused by the heat of friction with a high-speed burr or nerve injury induced by pressure on the compressed dura mater during bone drilling. Furthermore, improvement in cauda equina flow after decompression may not occur as smoothly and evenly in the curve type compared to the straight type. In addition, elderly patients often present with decreased neural plasticity, diminished detrusor muscle contractility, and altered bladder sensations. These factors, coupled with degenerative changes in the spine, likely contribute to prolonged postoperative recovery of urinary function. Thus, a more cautious approach with a vertically wide laminotomy might be preferable in cases with risk factors in preoperative imaging findings to reduce the rate of POUR; however, this remains a hypothesis requiring further investigation to confirm its validity. Pathomechanisms of de novo postoperative urinary retention after lumbar decompression surgery in patients with lumbar spinal canal stenosis. Curve-type stenosis has a higher risk of heat injury and nerve damage during decompression procedures and of irregular improvement of cauda equina flow after decompression, compared to straight-type stenosis.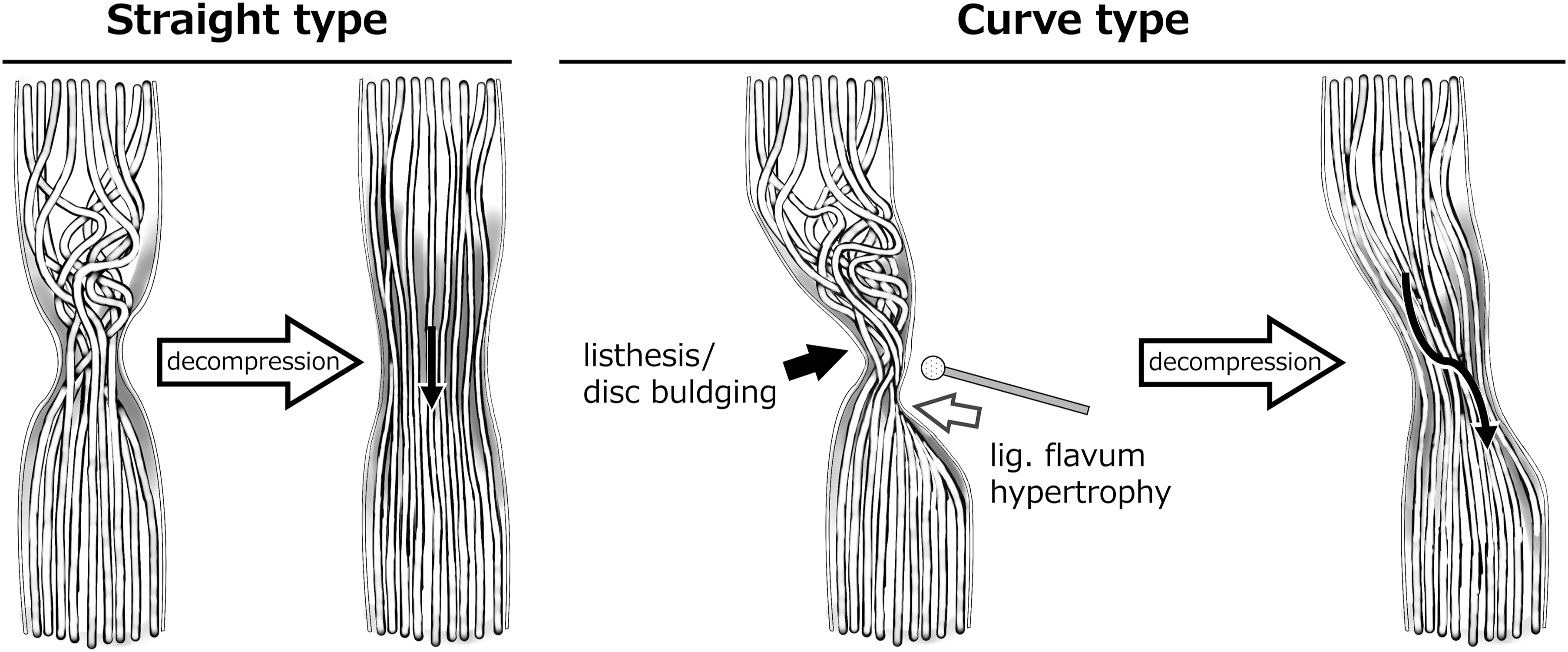
The limitations of the study include its retrospective, single-center design with only 23 patients with de novo POUR. Despite these limitations, we believe that our findings provide important insights and preventive measures for de novo POUR after surgery for LSCS.
Conclusions
The incidence of de novo POUR for at least 1 week after postoperative urinary catheter removal was 3.1% in patients who underwent lumbar decompression surgery for LSCS. The median recovery time was 41 days, and only 52.2% of the patients showed improvement within 3 months after surgery. An evaluation of clinical and imaging features showed that older age and curve-type stenosis with ventral or dorsal compression of the dura mater were risk factors for development of de novo POUR, regardless of sex, comorbidities, surgical procedures, number of decompressed segments, or degree of dura mater compression.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
