Abstract
Study Design:
Retrospective case series.
Objectives:
Postoperative urinary retention (POUR) represents a common postoperative complication of all elective surgeries. The aim of this study was to identify demographic, comorbid, and surgical factors risk factors for POUR in patients who underwent elective thoracolumbar spine fusion.
Methods:
Following institutional review board approval, patients who underwent elective primary or revision thoracic and lumbar instrumented spinal fusion in a 2-year period in tertiary and academic institution were reviewed. Sex, age, BMI, preoperative diagnosis, comorbid conditions, benign prostatic hyperplasia, diabetes, primary or revision surgery status, narcotic use, and operative factors were collected and analyzed between patients with and without POUR.
Results:
Of the 217 patients reviewed, 54 (24.9%) developed POUR. The average age for a patient with POUR was 67 ± 9, as opposed to 59 ± 10 for those without (
Conclusions:
POUR was a common complication in our patient cohort, with an incidence of 24.9%. Our findings demonstrate that patients who developed POUR are significantly older and have larger constructs. Patients who developed POUR also had longer in-hospital stays. Although our study supports other findings in the spine literature, more prospective data is needed to define diagnostic criteria of POUR as well as its management.
Keywords
Introduction
With rates of up to 70%, postoperative urinary retention (POUR) represents a common postoperative complication of all elective surgeries. 1 The incidence of POUR has been reported to be between 5.6% and 30% for patients undergoing spine surgery. 2 -5 POUR has been associated with increased risk for further complications such as urinary tract infections (UTIs) and urinary bladder overdistension. 1 Consequently, increased hospital length of stay (LOS) and inpatient postoperative procedures have been associated to POUR. 6 In health care, this translates to an increase in overall costs for a single operative intervention. In addition to the economic burden placed on patient and health care systems, increased LOS has also been linked to increased risk of hospital acquired infections and deep-vein thrombosis. 7 Therefore, developing POUR could potentially increase the risk of patient complications, longer LOS, and poor outcomes.
Typically, the adult bladder has a capacity of 400 to 600 mL; when volume reaches approximately 300 mL, the sensation of bladder fullness and detrusor muscle contraction begin. 1 Once the sensation of fullness is achieved, the patient is typically able to void. For this reason, if the patient is unable to void with a bladder scan volume of 300 mL, they are considered to have failed a trial of void (TOV) and, thereby, have urinary retention. 4,5 Urinary retention for an extended period of time can result in discomfort, bladder distention, detrusor atony, UTI, and renal injury. 1
The goal of this study was to identify risk factors for POUR in patients who underwent elective thoracolumbar spine fusion. Demographic, comorbid, and surgical factors that may increase a patient’s risk for this condition were investigated. Based on prior literature, we hypothesized that factors including sex, age, narcotic use, duration of surgery, and benign prostatic hyperplasia (BPH) would correlate with an increased incidence of POUR.
Materials and Methods
Study Population
This a retrospective patient series study that did not receive any funding. After obtaining institutional review board approval, 586 patients were obtained by querying our surgical billing database from the tertiary referral center to determine a consecutive series of patients who underwent posterior thoracic and/or lumbar spinal instrumentation and fusion surgery between January 2012 and December 2017. The following CPT codes were used to identify the study population: 22 800, 22 802, 22 804, 22 812, 22 842, 22 843, 22 844, 22 845, 22 846. Of the 586 patients identified from the database, 253 were from the 2016-2017 timepoint. Electronic medical records were completely incorporated at our institution at the end of 2015. Therefore, patients from the 2016-2017 period were best suited for further chart review. Exclusion criteria were end-stage renal disease, cauda equina, spinal infection (osteomyelitis, epidural abscess), spinal hematoma, nonambulators, neurogenic bladder, history of prostate cancer, pelvic floor dysfunction or genitourinary disorder potentially causing retention, spinal injury caused by trauma, and prior surgery for metastatic cancer of the vertebral column. A total of 217 patients were included in the study (Figure 1).
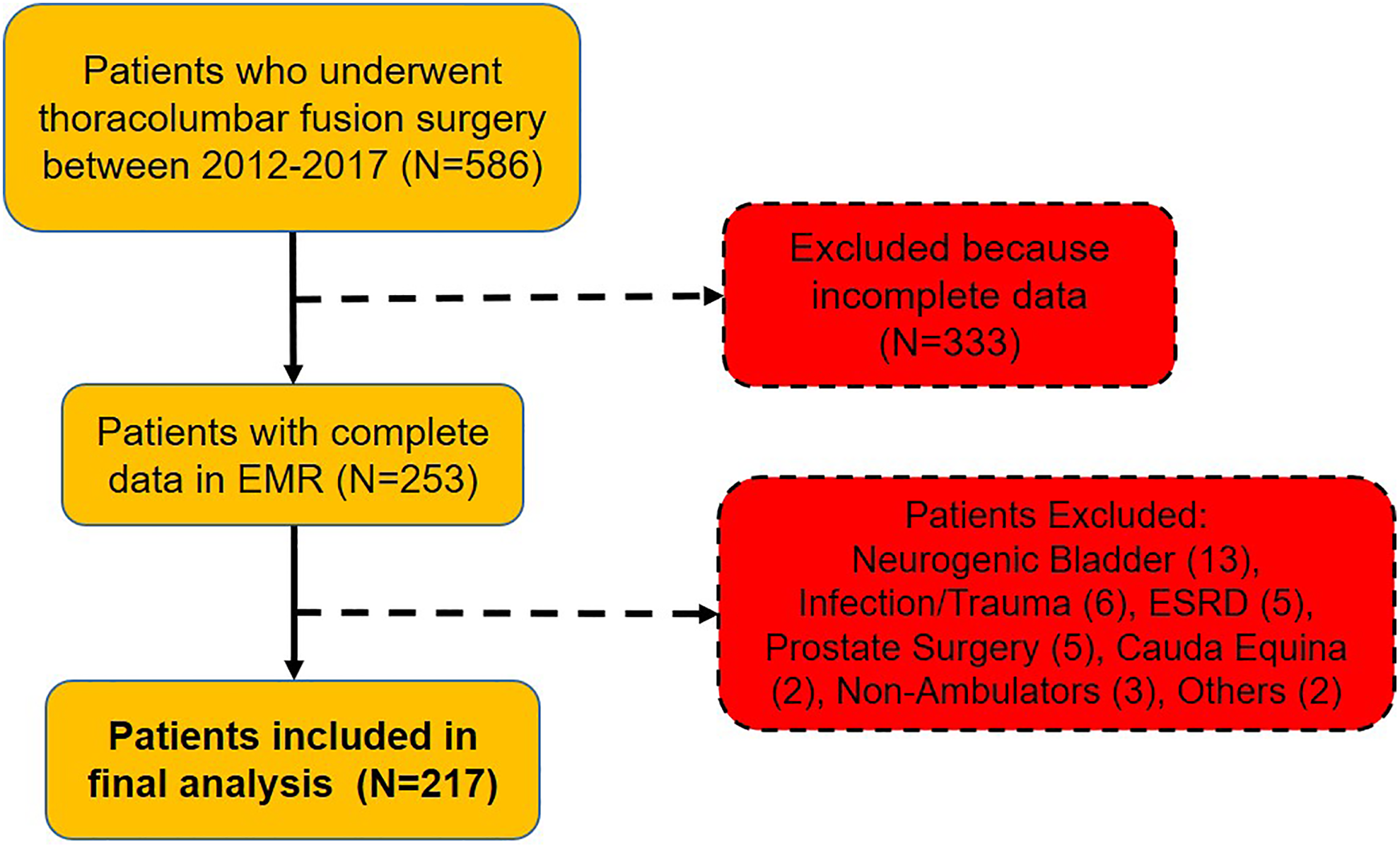
Patient inclusion and exclusion into the study. A total of 586 patients were identified from the database and 217 were included in the study.
Data Collection
We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines to enhance the quality and minimize the bias of this observational study. 8 Only the in-hospital postoperative time period was evaluated, unless the patient had a documented secondary procedure. From chart review, we obtained prior medical history, demographic data, operative details, details of anesthesia, fluid management intraoperatively, length of hospital stay, postoperative Foley and pain management, and urinary retention. Nursing notes, rectal tone notes, and progress notes were used to identify patients with urinary retention. Operative data included procedure type, length of surgery, estimated blood loss (EBL), transfusions, construct length, dural tears, and use of bone morphogenetic protein (BMP) for fusion.
Urinary Retention Protocol
All patients undergoing elective spine surgery received a Foley catheter intraoperatively. The Foley was maintained after the operation, and patients were transported to the floor for postoperative management. The Foley was usually removed 1 to 2 days after surgery or when the patients were ready to ambulate. After removing the Foley, patients had a TOV for 4 hours. If TOV failed, a bladder scan was performed, and rectal tone was evaluated with a digital rectal exam. If the bladder scan showed more than 300 mL, the patient was straight catheterized. After the straight catheterization, the patient was given another TOV for 4 hours. If TOV failed again, the above process was repeated. If they failed to void after 3 attempts, a Foley catheter was placed, and they were started on tamsulosin, if applicable. Alternatively, if the bladder scan showed less than 300 mL on the first scan, they were given IV fluids and given an additional 2 hours to void. If they failed to void in the next 2 hours, a bladder scan was performed, and the above-described procedures were followed again. Finally, for patients who voided successfully but voided less than 300 mL, a bladder scan was performed. If the bladder scan showed an amount larger than the urinated amount, the patient was straight catheterized (Figure 2). If the patient had persistent symptoms, they were discharged with the Foley catheter in place and scheduled for urology consultation.
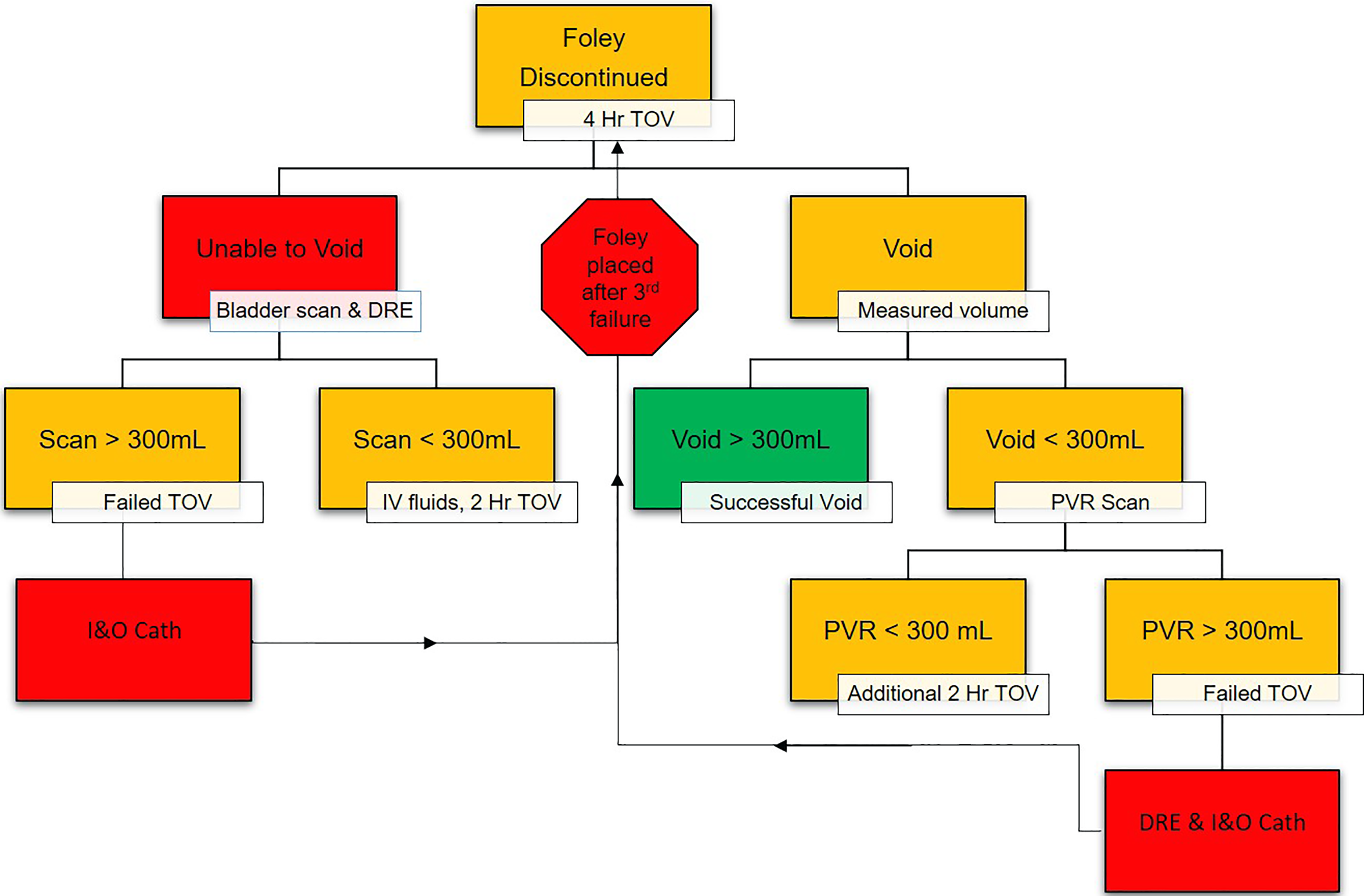
Foley catheter removal protocol: after 2 I&O catheterizations, the Foley catheter is reinserted and the patient is started on tamsulosin.
Statistical Analysis
Data was entered into Microsoft Excel for data cleaning and management (Microsoft Office Professional Plus 2016, Microsoft, Redmond, WA). Stata 11.1 software (StataCorp LP, College Station, TX) was used for all statistical analyses. Means and SDs or medians and interquartile ranges were calculated for continuous variables. Frequencies were calculated for categorical variables. Bivariate comparisons were made between patients with POUR and those without using the Student
Results
A total of 217 patients were included in the final analysis. The average age was 61.3 years, with a BMI of 30.9 kg/m2, and 45.2% of patients were male. Nearly half of the patients (48.9%) had prior lumbar surgery. Hypertension (53%), hyperlipidemia (41.5%), depression (25.8%), former smoking status (25.5%), and diabetes (20.7%) were the most common comorbid conditions (Table 1). The majority of patients (56.2%) had 2-level fusion, and 5.1% had 6 or more levels fused. The posterior approach was most common (89.8%), and only BMP was used in only 6.9% of patients (Table 2).
Patient Demographic Characteristics and Comorbidities.

a Smoker (current): quit less than 1 year ago.
b Smoker (Former): Quit more than 1 year ago.
Abbreviations: BMI, body mass index; OSA, obstructive sleep apnea; UTI, urinary tract infection; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; BPH, benign prostate hyperplasia.
Patient Surgical Characteristics.

In the study, 54 (24.8%) patients experienced POUR. Comparison of the characteristics of the patient groups with and without POUR revealed no significant differences, with the exception of age. Patients with POUR were significantly older than patients without POUR (67 vs 59.4;
Comparison of Characteristics Between Patients With and Without Postoperative Urinary Retention (POUR).
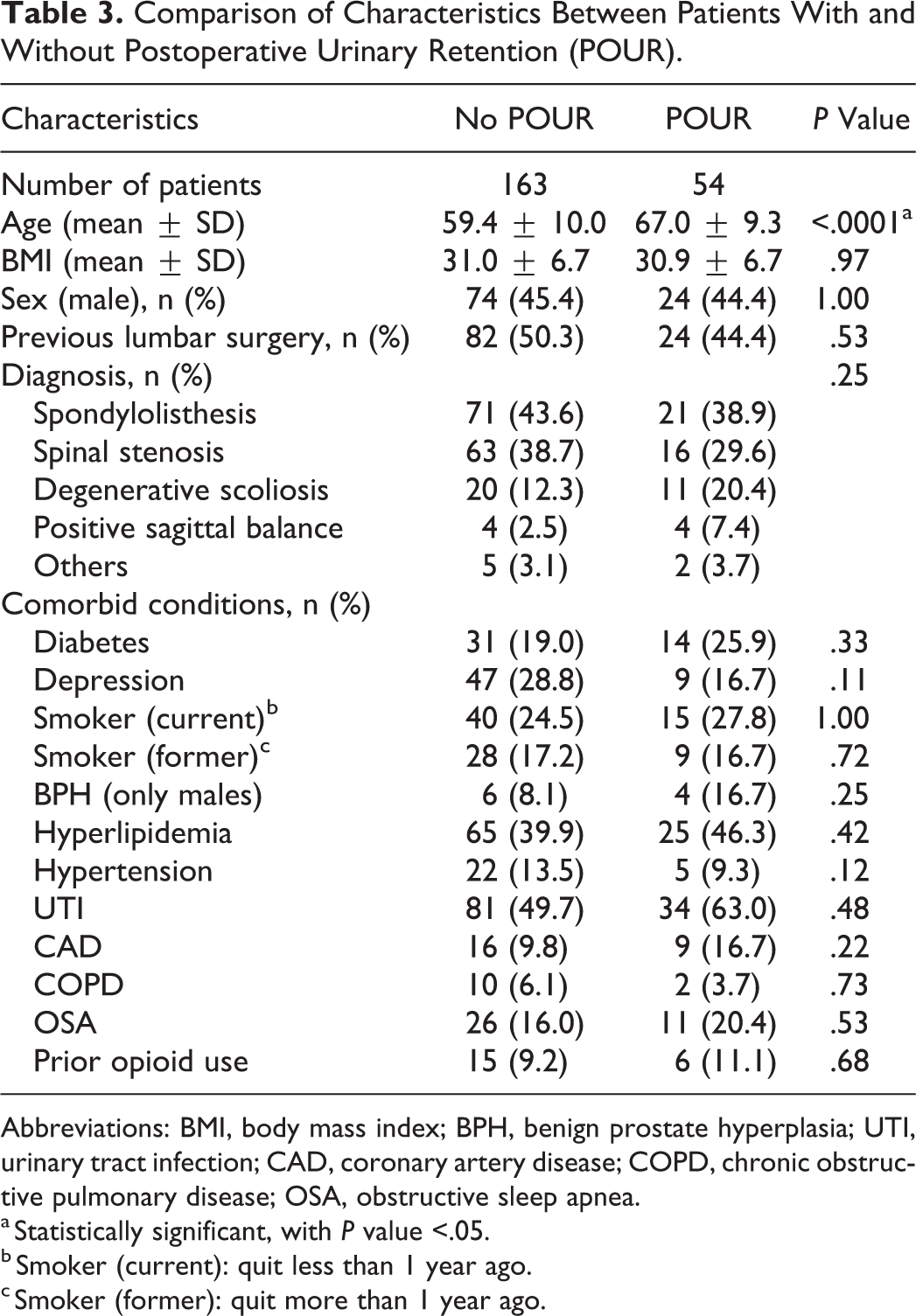
Abbreviations: BMI, body mass index; BPH, benign prostate hyperplasia; UTI, urinary tract infection; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; OSA, obstructive sleep apnea.
a Statistically significant, with
b Smoker (current): quit less than 1 year ago.
c Smoker (former): quit more than 1 year ago.
Surgical details were collected for each of the patients. With regard to the procedural details, urinary retention rates did correlate with the number of levels fused (
Comparison of Surgical Details for Patients With and Without Postoperative Urinary Retention (POUR).
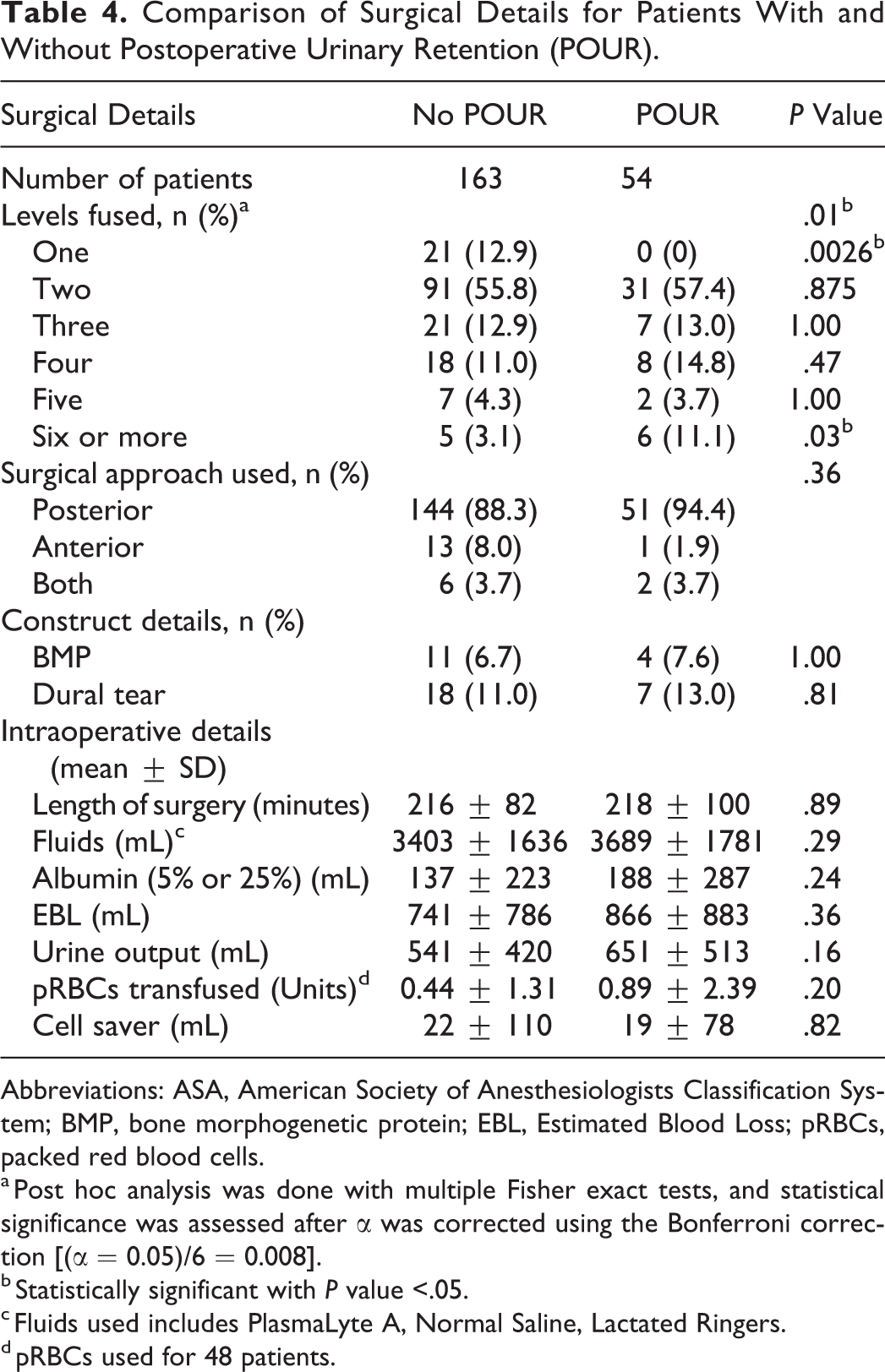
Abbreviations: ASA, American Society of Anesthesiologists Classification System; BMP, bone morphogenetic protein; EBL, Estimated Blood Loss; pRBCs, packed red blood cells.
a Post hoc analysis was done with multiple Fisher exact tests, and statistical significance was assessed after α was corrected using the Bonferroni correction [(α = 0.05)/6 = 0.008].
b Statistically significant with
c Fluids used includes PlasmaLyte A, Normal Saline, Lactated Ringers.
d pRBCs used for 48 patients.
Comparison of Drugs Used in Anesthesia for Patients With and Without Postoperative Urinary Retention (POUR).
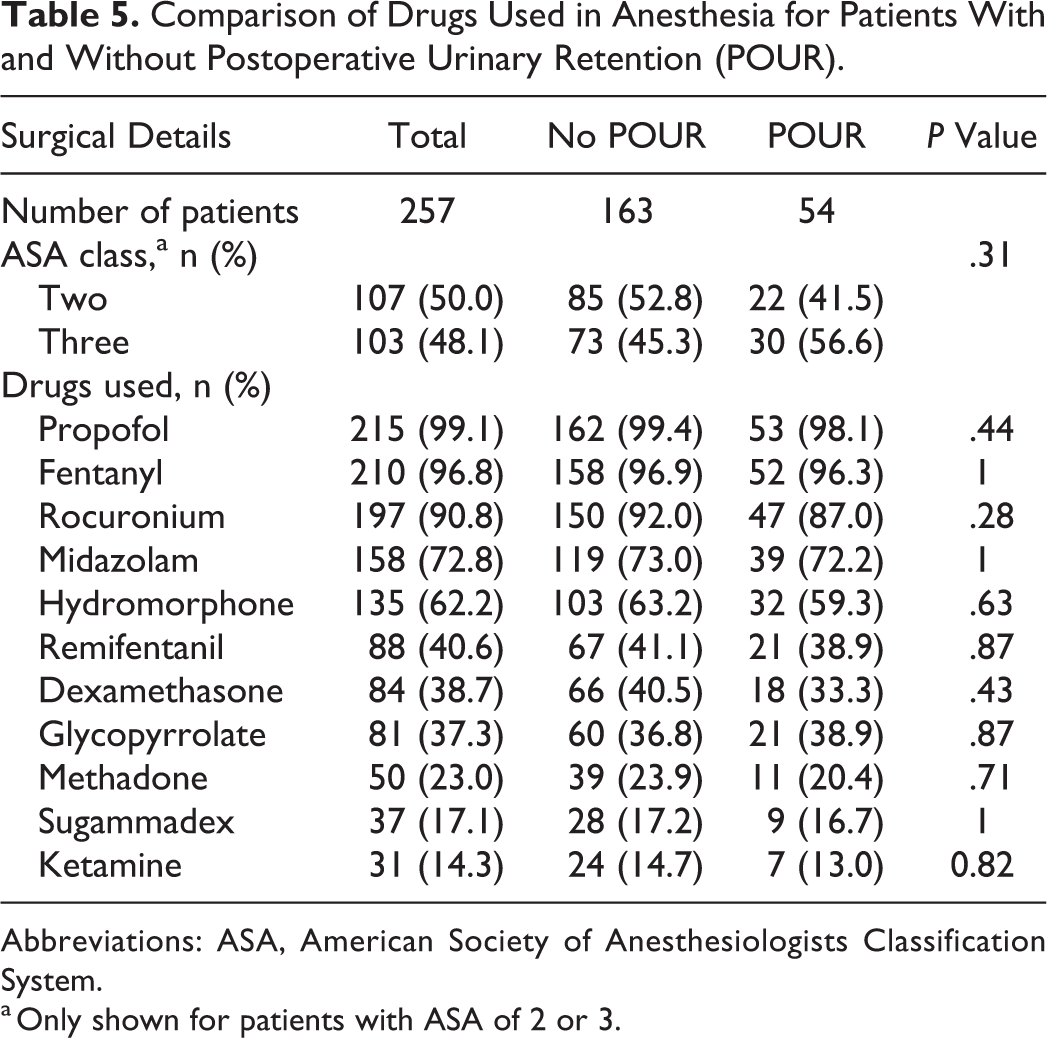
Abbreviations: ASA, American Society of Anesthesiologists Classification System.
a Only shown for patients with ASA of 2 or 3.
The postoperative outcomes for patients with and without POUR were evaluated. Patients with POUR had significantly longer hospital stays than those without (5.8 vs 4.9 days;
Comparison of Postoperative Stay for Patients With and Without Postoperative Urinary Retention (POUR).
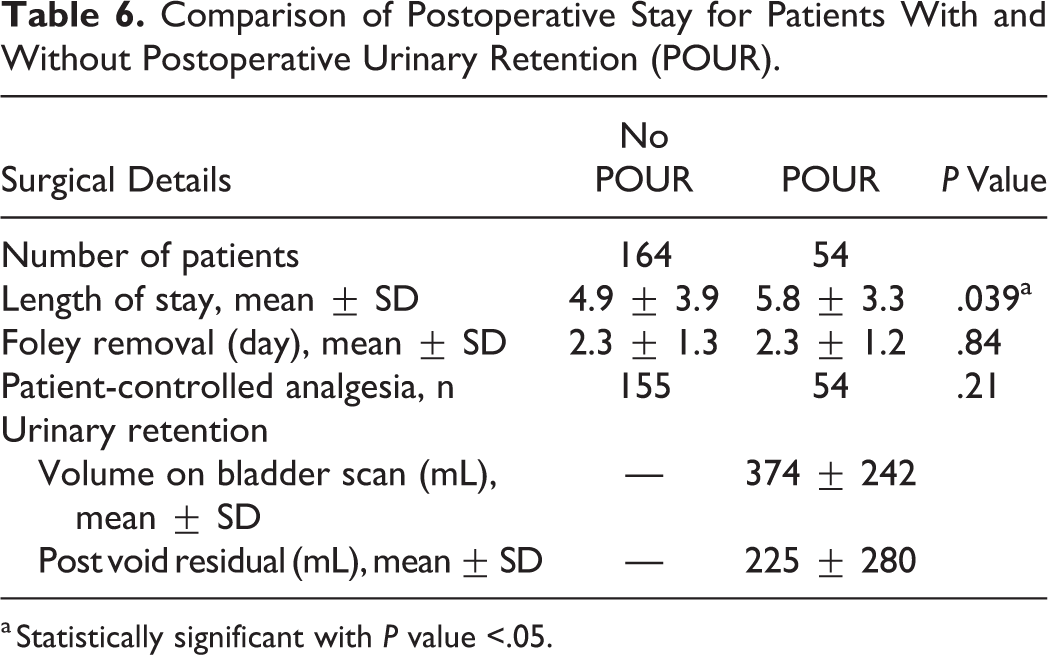
a Statistically significant with
Discussion
Incidence of POUR
In our study, the overall incidence of POUR was 24.8%. This agrees with the results of prior studies, which have looked at the incidence of POUR after elective spine surgery. The range reported in the spine literature is 5.6% to 30%.
3
-5,7,9
In our study, POUR rates were on the higher spectrum of this range; however, it included patients who had revision surgery (48.9%) and excluded those operated at another spine region (cervical spine). In his retrospective analysis of POUR in patients undergoing elective spine surgery, Altschul et al
2
reported an incidence of POUR of 8.8%. Although the reported POUR rates were lower compared with our study, it included patients from all spine levels (cervical, thoracic, and lumbar), via both anterior and posterior approaches, which might have contributed to the difference in rates,
2
particularly because authors reported a significant association between POUR and lumbar surgery when compared with cervical and thoracic (
POUR Risk Factors
In our patient series, age was the only demographic factor identified to be associated with POUR. Patients who developed POUR were older than those who did not develop urinary retention (67 vs 59.4 years;
In the present study, despite controlling for female sex, male patients with a previous diagnosis of BPH were not associated with an increased incidence of POUR. This is contrary to prior published studies, where an increased risk of POUR was reported with the diagnosis of BPH prior to surgery. 4 We suspect that our sample size was not sufficiently powered to demonstrate an effect between POUR and prior history of BPH. There is considerable literature supporting BPH as a risk factor for POUR; therefore, our negative findings could be the result of type 2 error (false negative). 3,4 Thus, in spite of our results, we would suggest that BPH should be considered as a risk factor for POUR in patients with elective lumbar spine surgery. Additionally, it is possible that BPH was underdocumented in our medical record and/or patients did not present with any lower urinary tract symptoms prior to their surgery. Urology literature suggests, from histological studies, that the prevalence of POUR rises from 8% to 50% between the fourth and sixth decades of life. 10 In our study, patients with BPH were older than those without the condition. The possibility of an underreported diagnosis of BPH and not being sufficiently powered could account for why a male-sex association with POUR was not apparent in our study.
The preoperative diagnosis, as well as the status of primary versus revision surgery did not demonstrate any significant association with POUR. Other studies have found that diabetes and depression have been associated with urinary retention, but our analysis did not support this. Gandhi et al 3 identified a protective association between smoking and POUR, but this was not supported by our analysis either.
Surgical Factors
Several surgical factors (Table 4) were investigated in this study. None of the operative factors analyzed, with the exception of levels fused, was associated with an increased incidence of POUR. Constructs with 6 or more levels fused were associated with the development of POUR, although this was a small sample size (5% of total population). In the present study, 66% of the study population had 2 or fewer levels fused. However, none of the patients who underwent single-level fusion developed POUR, whereas more than half of the patients with 6 or more levels fused developed urinary retention.
There are possible explanations of why patients undergoing more extensive surgery had increased retention rates. Prior studies have shown a direct association with longer operative times and the development of POUR. 2,5 In their study evaluating POUR in degenerative lumbar spinal stenosis patients, Lee et al 5 propose that for every minute of duration of surgery there is a 0.3% increased chance of developing POUR. 11 The number of spinal levels is proportional to duration of surgery. 12 Although our results did not reflect an association between POUR and operative times, the number of levels fused may be a surrogate for longer operative times.
Patients with longer constructs may have increased narcotic requirement and are at higher risk of developing POUR because of the anticholinergic effects on the detrusor muscle by systemic opiates.
1
Additionally, the use of PCA was demonstrated to have twice the likelihood of developing POUR when compared with intermittent intramuscular injections.
2
In our study, 96.3% of patients received PCA, but there was no significant association between groups (
Insertion of Foley catheter intraoperatively may cause irritation of the urethra and lead to difficulty voiding and development of POUR. 11,13 Although Boulis et al 13 were not able to find any difference in incidence of POUR for those who were intraoperatively catheterized, they found that those who were catheterized and developed POUR had longer duration of their symptoms. We evaluated medications used during anesthesia as well as fluids and blood products administered, but none resulted in a significant difference between groups. Other factors like the use of BMP, intraoperative dural tears, estimated blood loss, or surgical approach did not reflect any difference either.
Postoperative Care and Voiding Protocol
Foley catheters were removed on average after 2.3 days, without any difference between groups. Those who developed POUR did end up having longer hospital LOS. Longer LOS translates to increased costs. Although our voiding protocol could be contributing to the relatively high incidence of urinary retention, we adopted this to avoid potential complications of POUR. We only allow 4 hours to pass after discontinuing Foley catheter for a TOV before performing a bladder scan. If scan is above the threshold of sensation of fullness (300 mL), a straight catheterization is performed. Bladder overdistension can occur with as little as 400 to 600 mL of retained urine. 5 Our goal is to avoid the complications associated with bladder stretch injury, including bladder atony, renal injury, and UTI. It is worth noting that 27 (50%) of the patients who experienced POUR ultimately were discharged with a Foley in place. Patients were scheduled for a urology consultation and had their catheters removed during postoperative care. Patients were monitored for recurrence of genitourinary symptoms, but further evaluation for possible prostate pathology was managed by primary care or by a urologist.
The use of PCA as postoperative pain management did not influence the development of POUR in our study. However, it was difficult to understand its true relationship because full medication dosages could not be obtained. The use of high-dose opioids to achieve postoperative pain management in some patients may have influenced the development of POUR.
Limitations
The retrospective nature of our study and the relatively low sample size limits our findings. This particularly could be the reason why we did not find an association between POUR and male sex and/or BPH. Also, there was no ability to estimate the duration or severity of BPH prior to surgery. The use of preoperative and postoperative medications with autonomic activity could potentially increase the risk of POUR. This is true in the case of opioids and postoperative PCA regimens. There can also be selection bias because of the presence of missing data in medical records and patient population selection. Ultimately, this study is at risk for measurement bias because of the lack of a universal definition for the diagnosis of POUR. Unfortunately, the lack of consistency in both the diagnosis and management of POUR makes it difficult to draw comparisons between the few studies investigating this condition. However, despite these limitations, our study provides adequate data on this common postoperative complication.
Conclusions
POUR is a common postoperative complication in our elective spine surgery patient series, with 24.9% of patient developing urinary retention after surgery. Patients who developed POUR were significantly older, had more levels fused, and, on average, had longer inpatient hospital lengths of stay. Given the potential complications and implications of developing POUR, it is important to identify risk factors that can potentially lead to urinary retention after surgery. Higher-quality prospective data is needed to reach consensus on the definition of POUR, its associated risk factors, and proposed management.
Footnotes
Authors’ Note
The views expressed in the submitted article are our own and not an official position of the institution. Approval was obtained from our institutional review board.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Gelb is a board member and fellowship committee chair for AOSpine NA. He receives payment for lectures and for development of educational presentations from AOSpine NA. He receives royalties from DePuy Synthes Spine and Globus Medical. He has stock in the American Society for Investigative Pathology. Dr Koh receives payment for consultancy from Biomet. His institution receives RO1 grant money from the National Institutes of Health. Dr Ludwig is a board member for Globus Medical, the American Board of Orthopaedic Surgery, the American Orthopaedic Association, the Cervical Spine Research Society, and the Society for Minimally Invasive Spine Surgery. He is a paid consultant for DePuy Synthes, K2M, and Globus Medical. He receives payment for lectures and travel accommodations from DePuy Synthes and K2M. He receives payment for patents and royalties from DePuy Synthes and Globus Medical. He has stock in Innovative Surgical Designs and the American Society for Investigative Pathology. He receives research support from AO Spine North America Spine Fellowship support, Pacira Pharmaceutical, and AOA Omega Grant. He is a board member of Maryland Development Corporation. He receives royalties from Thieme, Quality Medical Publishers. He is on the governing board of Journal of Spinal Disorders and Techniques, The Spine Journal, and Contemporary Spine Surgery. The authors have no further potential conflicts of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
