Abstract
Study Design
Retrospective cohort study.
Objectives
To identify imaging predictors on pre- and perioperative imaging that are associated with a future revision surgery for adjacent segment disease (ASD) following lumbar fusion.
Methods
Patients undergoing open posterior lumbar fusion between 2014-2022 were followed-up for >2-year. The initial MRI was evaluated for disc degeneration by Pfirrmann (grade 1-5); Modic changes (0-3); as well as fatty infiltration and functional cross-sectional area (fCSA) of the multifidus, erector spinae, and psoas. Spinopelvic alignment was measured pre- and postoperatively.
Results
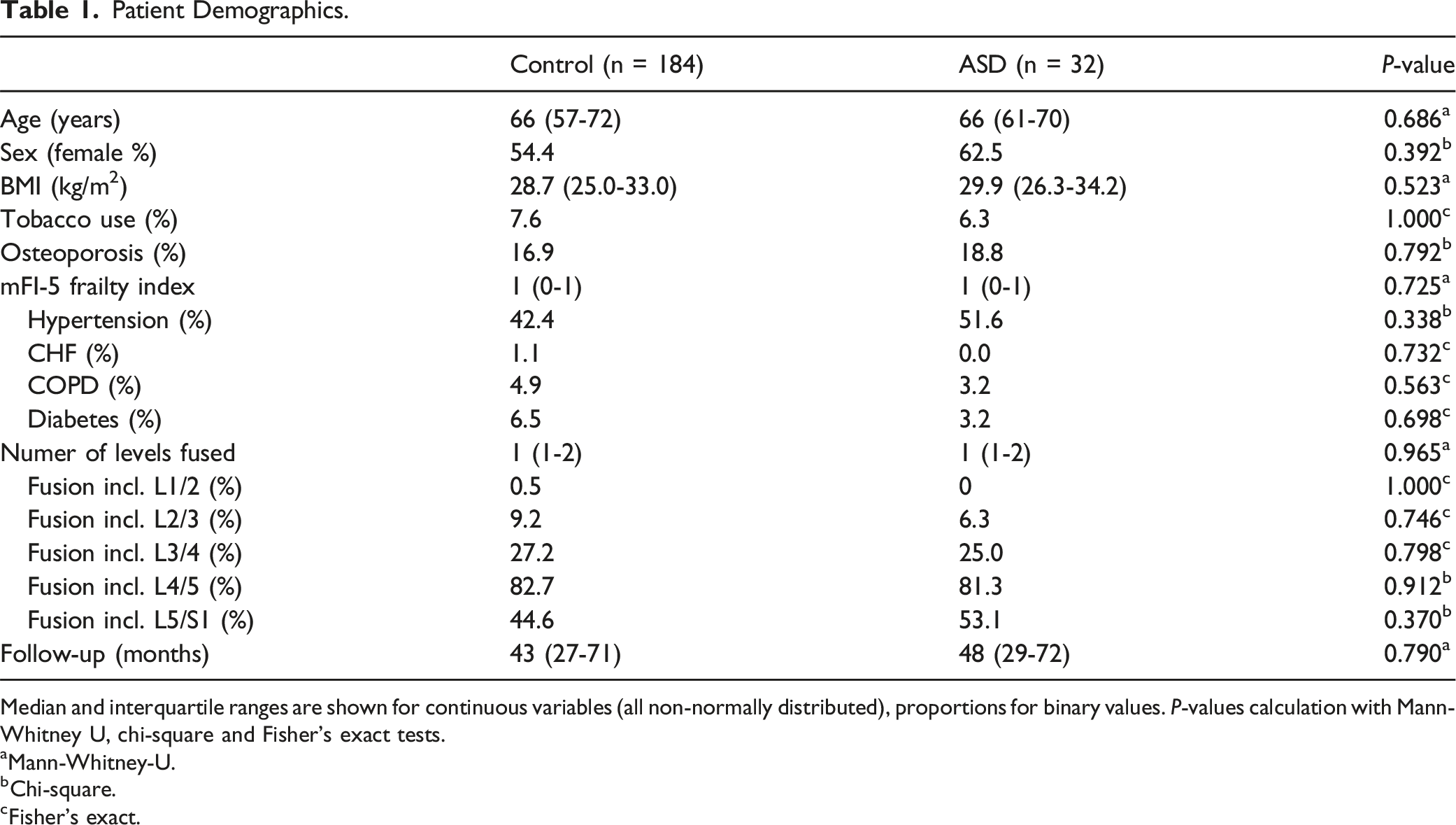
Of 216 patients, 14.8% (n = 32) required revision surgery for ASD in the further course. There were no significant differences in Modic changes or Pfirrmann grades between the ASD group and controls. Fatty infiltration was significantly higher in the ASD group for both the erector spinae (40.5% vs 36.9%, P = 0.043) and multifidus muscles (61.0% vs 53.9%, P = 0.003). Additionally, the multifidus fCSA was smaller in patients who developed ASD (5.7 cm2 vs 6.7 cm2). Pre- and postoperative spinopelvic parameters were not significantly different between groups, and no other MRI measurements showed a significant difference. In a multivariable logistic regression model adjusting for age and sex, multifidus fatty infiltration emerged as the only significant predictor of ASD revision surgery (P = 0.004). Patients with more than 58% multifidus fatty infiltration had over double the risk of developing ASD (OR 2.7, 95% CI: 1.1-6.5, P = 0.032).
Conclusions
Multifidus fatty infiltration is the key imaging predictor for the development of ASD requiring surgical revision. Disc degeneration and spinopelvic alignment appear to have less impact.
Keywords
Introduction
Adjacent segment disease (ASD), the degeneration of the segment adjacent to the instrumentation, is a common debilitating long-term sequela of spinal fusion and represents the primary indication for revision surgery.1,2 Incidence of radiographic ASD ranges between 0% and 100%, with and annual and 10-year revision surgery rates of 2.5% and 22.2%, respectively.3,4 The incidence of ASD is influenced by multiple factors, including ASD definition, pre-existing degeneration of adjacent discs, preoperative pathologies, surgical technique utilized, number of segments fused, and the follow-up period. 5 Although not every ASD is of clinical relevance, if defined on a radiological basis, symptomatic ASD represents the major driver for worse patient-reported outcomes and revision surgery. 6 These revision surgeries are not only undesirable for patients but also represent more challenging procedures with a higher complexity and risk profile than the index surgery.
Various factors have been associated with poor postoperative outcomes, including age, body mass index (BMI), frailty, osteoporosis, and sagittal imbalance.7-9 Although radiographic spinopelvic alignment parameters have been well studied in association with ASD development, magnetic resonance imaging (MRI) biomarkers for patient-reported outcomes and complications after spinal fusion surgeries are less well understood. Some studies have indicated that preoperative disc and facet joint degeneration at levels adjacent to fusion sites are risk factors poor outcomes. 10 In addition, paraspinal muscle status is increasingly being recognized as an important outcome predictor. 11 However, previous research has focused on the outcome of radiographic ASD and less on clinically relevant ASD, particularly those requiring revision surgery. Espacially, in-depth analysis of disc and paraspinal muscle status has not been conducted in relation to ASD revision surgery risk to date.
This study aimed to identify imaging predictors on pre- and perioperative imaging for surgically relevant ASD development following a primary lumbar fusion.
Methods
We hypothesized that adjacent segment-level disc degeneration, paraspinal muscle degeneration, and spinopelvic malalignment are associated with future ASD revision surgical procedures.
Patient Population
This is a retrospective cohort study of prospectively enrolled patients who underwent lumbar decompression or fusion surgery for degenerative conditions at a single spine unit between 2014 and 2022. All patients were followed up through a study-specific phone-call more than 2 years postoperatively. The phone call aimed to identify potential revision surgeries that may have been performed outside our institution. In addition, patient charts were retrospectively re-reviewed for potential revision procedures after the study-specific follow-up. Follow-up time was defined as the period from the index procedure to either the last clinical follow-up or the study-specific phone call. For patients who underwent revision surgery in the further course (extending the follow-up period), the follow-up time was measured from the index procedure to either the revision procedure or the study phone call. Patients were included in this study if they had an open posterior instrumentation, whereas decompression only, standalone anterior or lateral fusion procedures were not included. Further exclusion criteria included previous lumbar fusion, previous laminectomy, and missing or low-quality MRI. Patient characteristics, surgical details, and comorbidities including the five-factor frailty index (mFI-5) were retrieved from the hospital archiving system. 12 ASD revision surgery was defined as any procedure adjacent to the primary instrumentation construct, including decompression alone, as well as decompression and fusion instrumentation for disc collapse, listhesis, herniations, and central and foraminal stenosis. The hospital’s institutional review board (IRB) approved this study (#2019-2137). Written and informed consent to participate in this study and to publish their data was obtained from all individual participants included in the study. The reporting was in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
MRI Assessment
Preoperative MRI was performed prior to the primary lumbar fusion procedure. Disc degeneration according to Pfirrmann (grades 1-5)
13
and Modic changes (0-3)
14
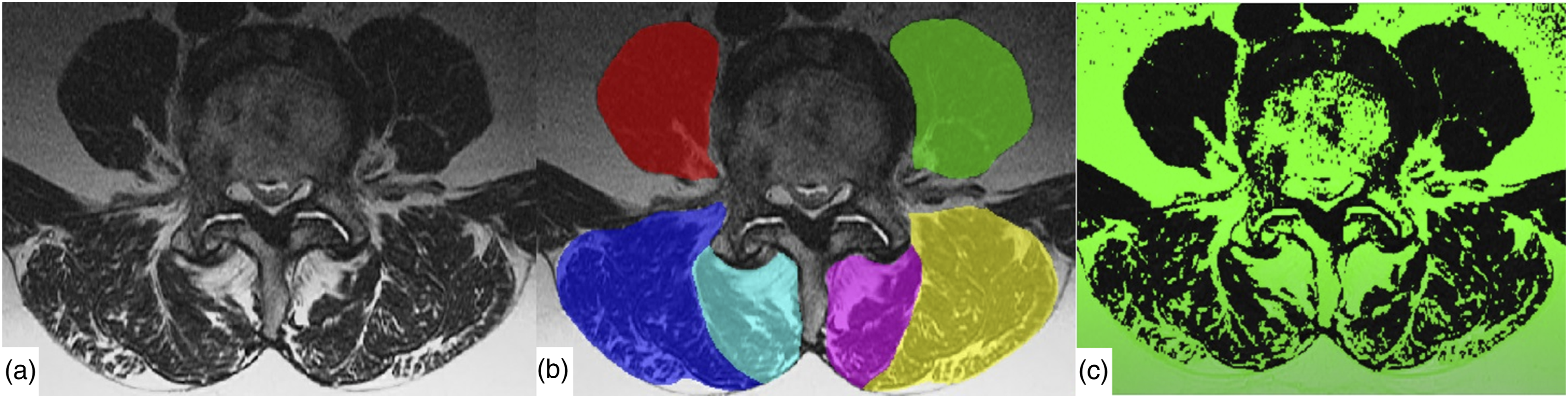
was analyzed on all lumbar segments, as well as the proximal adjacent (herein referred to as the adjacent segment), upper and lower instrumented segments (Figure 1). For instance, in a two-level fusion procedure from L4-S1, the adjacent segment was considered L3/4, the upper instrumented segment L4/5, and the lower instrumented segment L5/S1. Consequently, in single-level procedures, the upper and lower instrumented segments were the same segments. Paraspinal muscle measurements were conducted on axial slices parallel to and at the level of the upper endplate of L4 using ITK SNAP version 3.8.0 software (https://www.itksnap.org/)
15
(Figure 2). Multifidus, erector spinae, and psoas muscles were segmented using dedicated software (MuscleEval). The fat area within the segmented muscle was determined using a previously described method employing custom software (MATLAB version R2019a, The MathWorks, Inc., Natick, MA, USA). This software utilizes automated pixel intensity thresholding to classify pixels as either fat or muscle, based on whether they fall above or below the threshold.
16
The cross-sectional (CSA) of each muscle was divided into muscle area = functional CSA (fCSA) and fat area (CSAfat). The percentages of fatty infiltration of each paraspinal muscle were measured and defined as fatty infiltration = CSA
fat
/(fCSA + CSA
fat
). MRI measurements. Pfirrmann grades (I-V), and Modic changes (M0-M3) were measured on the preoperative MRI (a&b). The postoperative sagittal and coronal postoperative radiograph (c&d) shows the definition of the adjacent segment (AS), upper instrumented segment (UIS) and lower instrumented segment (LIS). Paraspinal muscles measurement. (a) T2-axial slices at the upper endplate of L4 were evaluated. (b) The psoas (red and green), erector spinae (blue and yellow) and multifidus (turquoise and pink) were assessed. c) With dedicated software, the functional component (black) and fat component (green) were segmented, to measure the functional cross-sectional area and percentage fatty infiltration of each muscle.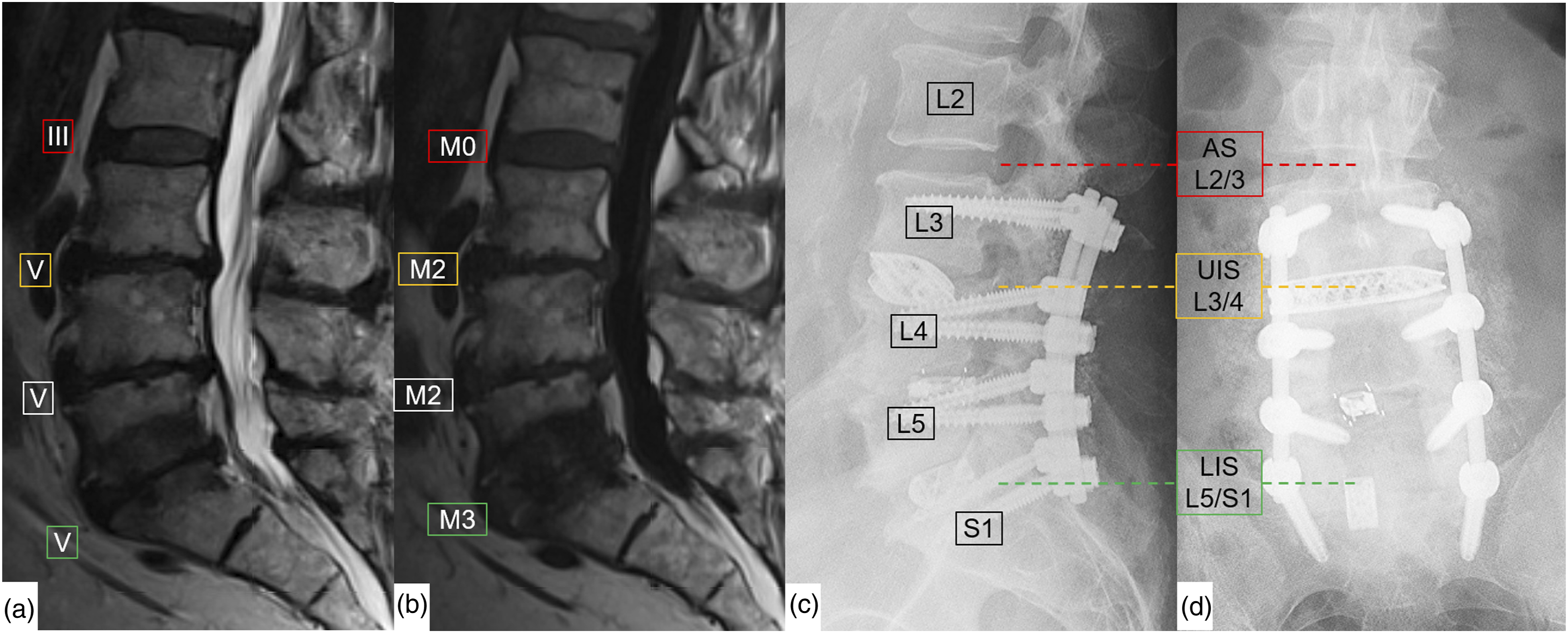
Radiographic Measurements of Sagittal Balance
The sagittal balance was assessed on the last preoperative and on the first postoperative radiographs, on which the femoral heads were displayed. The following parameters were evaluated and compared between groups: pelvic incidence, lumbar lordosis, pelvic-incidence lumbar lordosis mismatch (=PI-LL-mismatch), 17 pelvic tilt, sacral slope, distal lumbar lordosis between upper endplate L4 and sacral endplate (=L4-S1-lordosis), 18 lordosis distribution index (=L4-S1 lordosis / lumbar lordosis) and lumbar pelvic angle (LPA). 19
Statistical Analysis
Statistical analyses were conducted using STATA (STATA/BE 18.0, Stata Corp LLC, TX, USA). The Shapiro–Wilk test was performed to assess normal distribution. Means ± standard deviations represent normally distributed parameters. The medians with interquartile ranges shown in square brackets represent non-normally distributed data. Differences in central tendencies and variances between ASD and control patients were analyzed using Student’st test, Mann-Whitney U test, chi-square test, and Fisher’s exact test, as appropriate. Univariate logistic regression was performed with ASD revision surgery as the dependent variable. Imaging variables with a P-value <0.05 in univariate analysis or group comparisons were included in a linear regression model to assess potential covariates, followed by a multivariable logistic regression model. Receiver operating characteristic (ROC) analysis was used to identify optimal cutoff values for independent predictors, which were then re-evaluated in a multivariate regression model. Statistical significance was defined as P < 0.05.
Results
In this study, 269 patients were prospectively enrolled between 2014 and 2022, of whom 237 (88.1%) were followed-up over a phone call after a minimum of 2 years and a median follow-up of 4 years (Figure 3). Due to missing or poor-quality preoperative MRIs, 21 patients were excluded, leaving 216 patients (80.3%) for analysis. The median age at time of primary surgery was 66 [58-74] years, 55.6% were female, and BMI was 28.9 kg/m2 [25.1-33.0]). The median number of fused segments was 1 [1-2], ranging between 1 and 4 levels, and the follow-up time was 45 [28-72] months. 93.6% of the procedures included the L4/5 (81.9%) and/or L5/S1 (45.8%) levels. Study flow chart.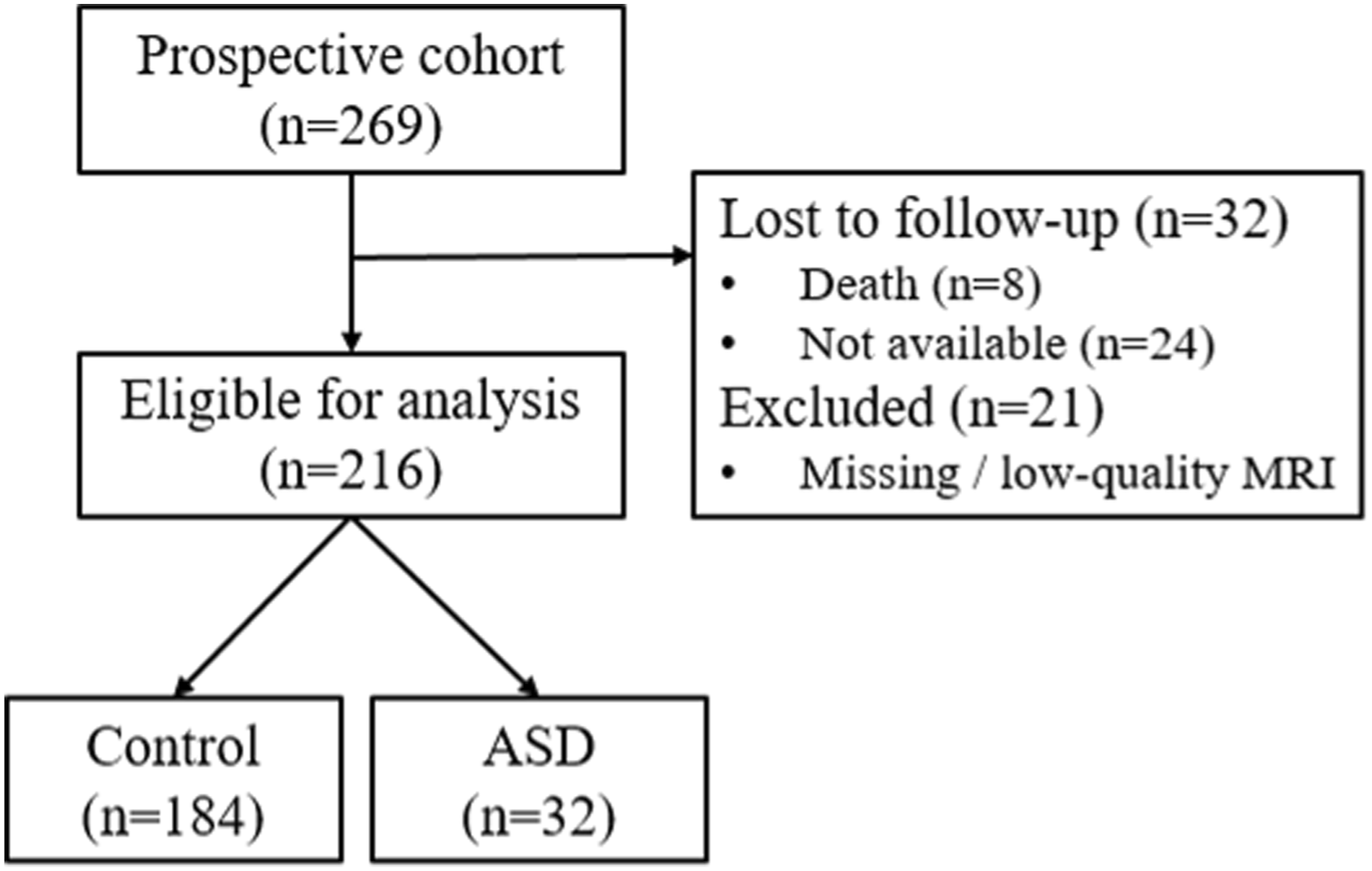
In total, 38 lumbar revision procedures (17.6%) were performed during the follow-up period. Of these, 32 patients (14.8%) required revision surgery for ASD, the majority being proximal ASD (n = 28), with 2 cases of distal ASD and 2 cases involving both proximal and distal ASD. The median time between the primary fusion and ASD revision surgery was 32 months [IQR 17-57]. Revision surgeries involved decompression alone in 12.5% (n = 4) of cases and decompression with fusion extension in 87.5% (n = 26) (Figure 4). Additional revision procedures included screw loosening and pseudarthrosis in 2 patients, irrigation and debridement for hematoma or seroma in 2 patients, a mechanical complication with screw-rod disintegration in 1 patient, and restenosis requiring revision decompression in 1 patient. Patients undergoing revision for ASD were categorized into the ASD group, while those without revision or with revision for other reasons were assigned to the control group. No significant differences in patient characteristics, comorbidities, surgical levels, or follow-up duration were found between the ASD and control groups (Table 1). Patient example who developed adjacent segment disease (ASD) and had to be reoperated. 30 months postoperative follow-up radiograph (a) and MRI (b)-(d) of the patient illustrated in Fig. 1 after L3-S1 fusion who developed a surgically relevant ASD at L2/3 with disc collapse and protrusion, resulting in central and foraminal stenosis. (e)/(f) The patient was reoperated with a standalone lateral lumbar interbody fusion at that ASD level L2/3. Patient Demographics. Median and interquartile ranges are shown for continuous variables (all non-normally distributed), proportions for binary values. P-values calculation with Mann-Whitney U, chi-square and Fisher's exact tests. aMann-Whitney-U. bChi-square. cFisher’s exact.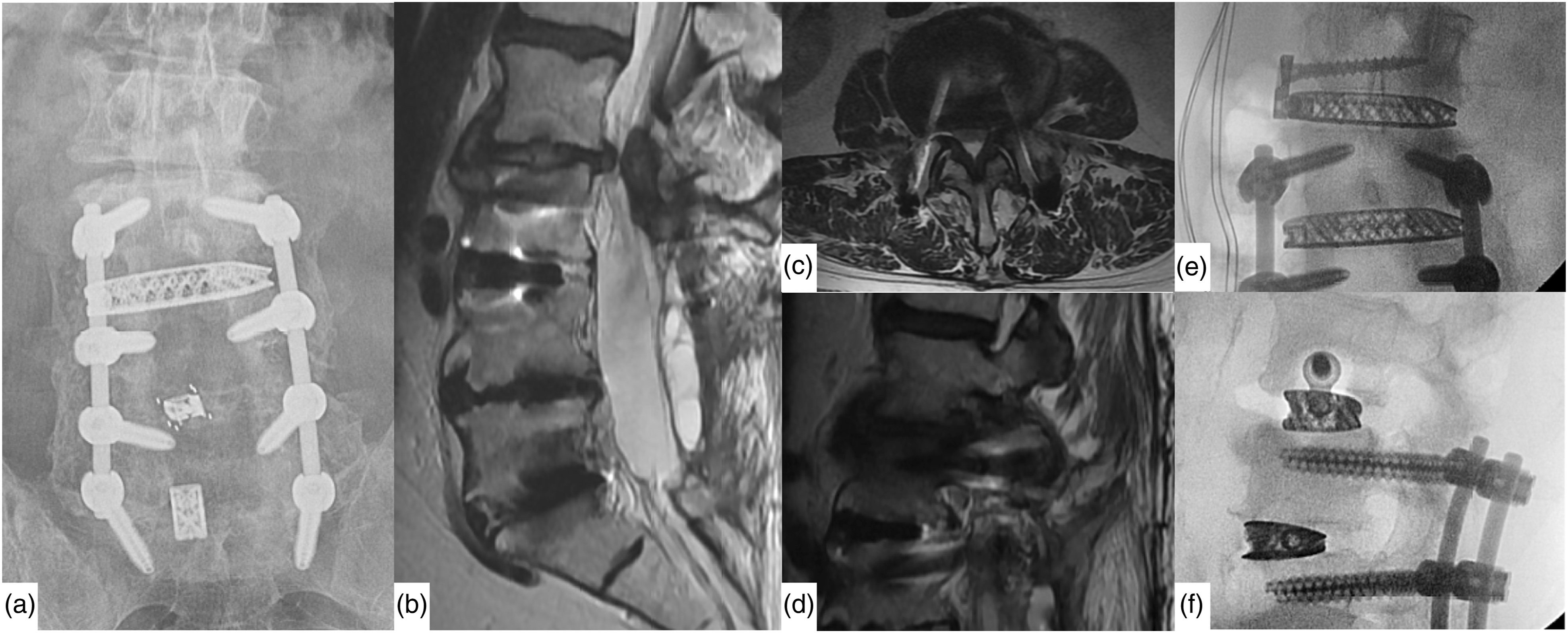
MRI Measurements
Univariate Logistic Regression Analysis for MRI Predictors of ASD Revision Surgery.
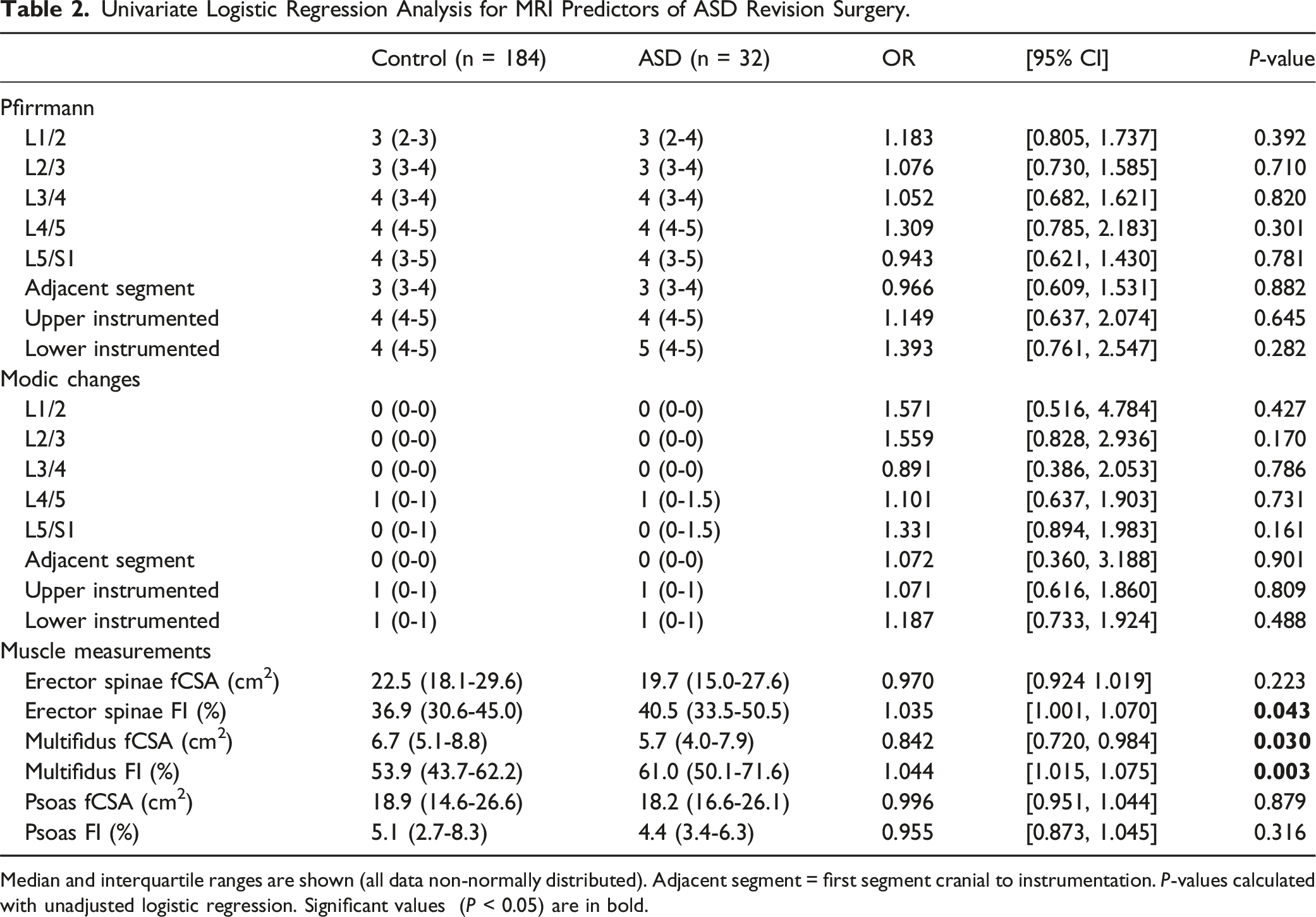
Median and interquartile ranges are shown (all data non-normally distributed). Adjacent segment = first segment cranial to instrumentation. P-values calculated with unadjusted logistic regression. Significant values (P < 0.05) are in bold.
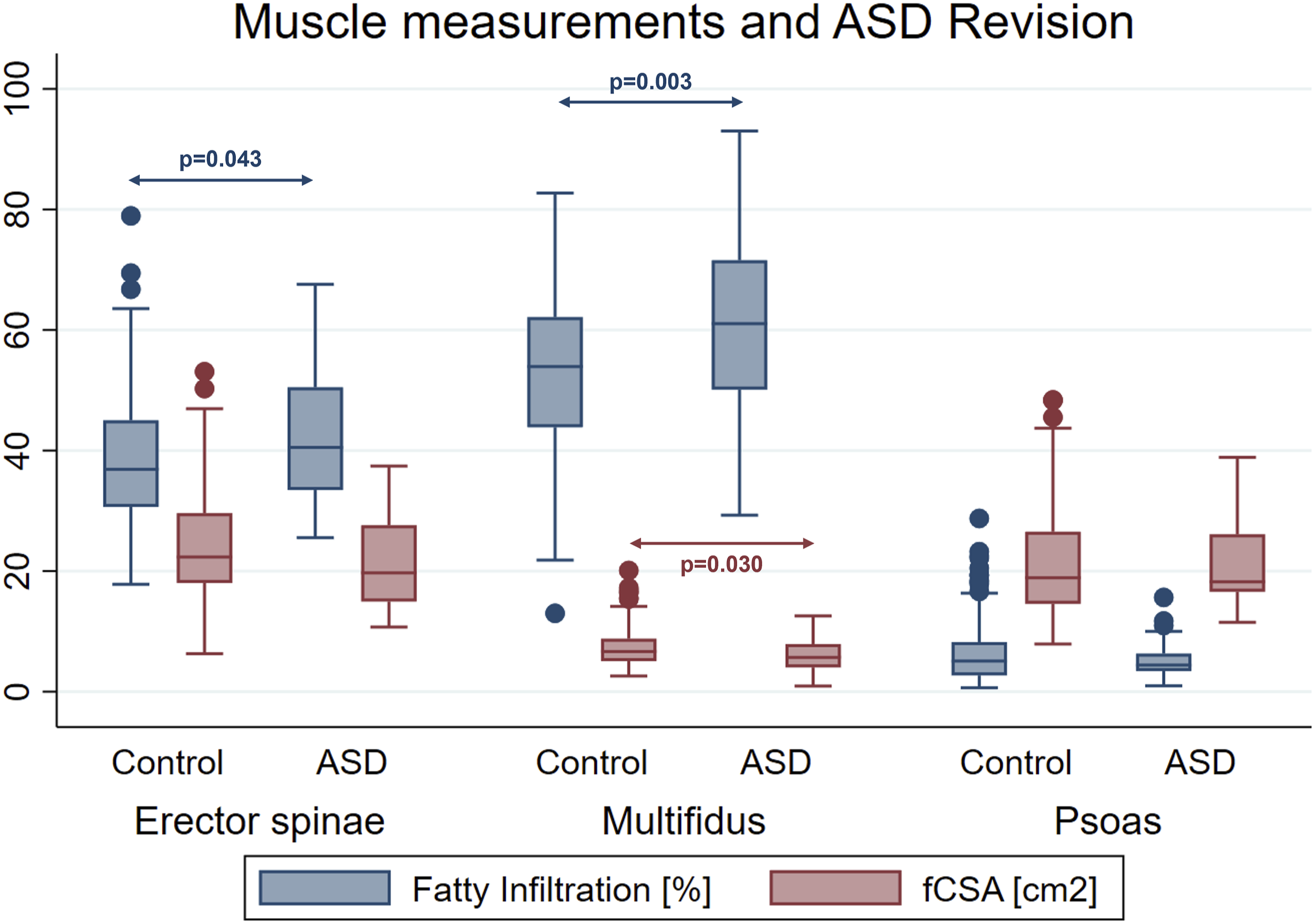
Boxplot of muscle measurements.
Radiographic Measurement of Sagittal Balance
Univariate Logistic Regression for Sagittal Balance Predictors of ASD Revision Surgery.
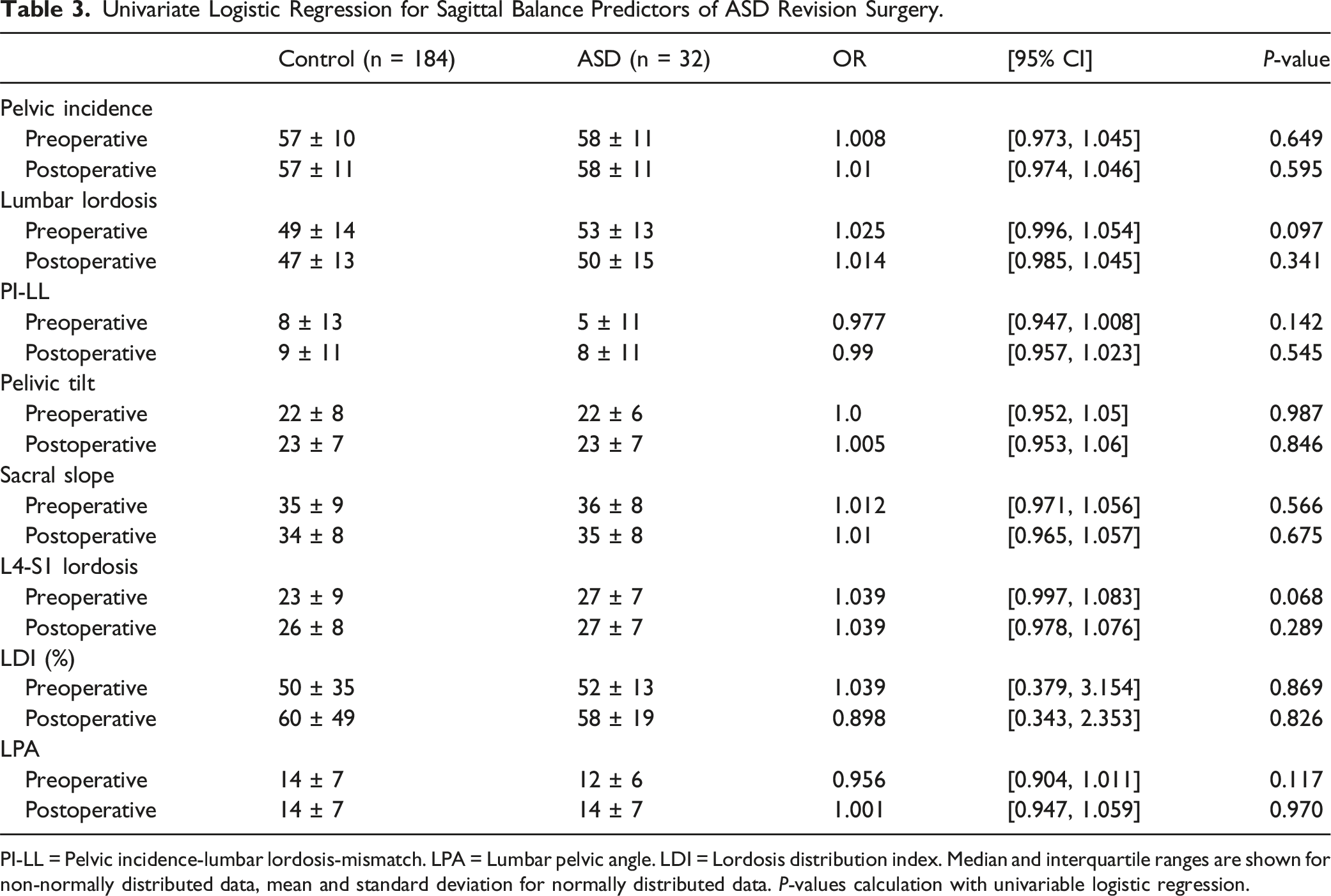
PI-LL = Pelvic incidence-lumbar lordosis-mismatch. LPA = Lumbar pelvic angle. LDI = Lordosis distribution index. Median and interquartile ranges are shown for non-normally distributed data, mean and standard deviation for normally distributed data. P-values calculation with univariable logistic regression.
Imaging Predictors for ASD Revision
Linear Regression Analysis of Patient Demographics Predicting Paraspinal Muscle Degeneration.
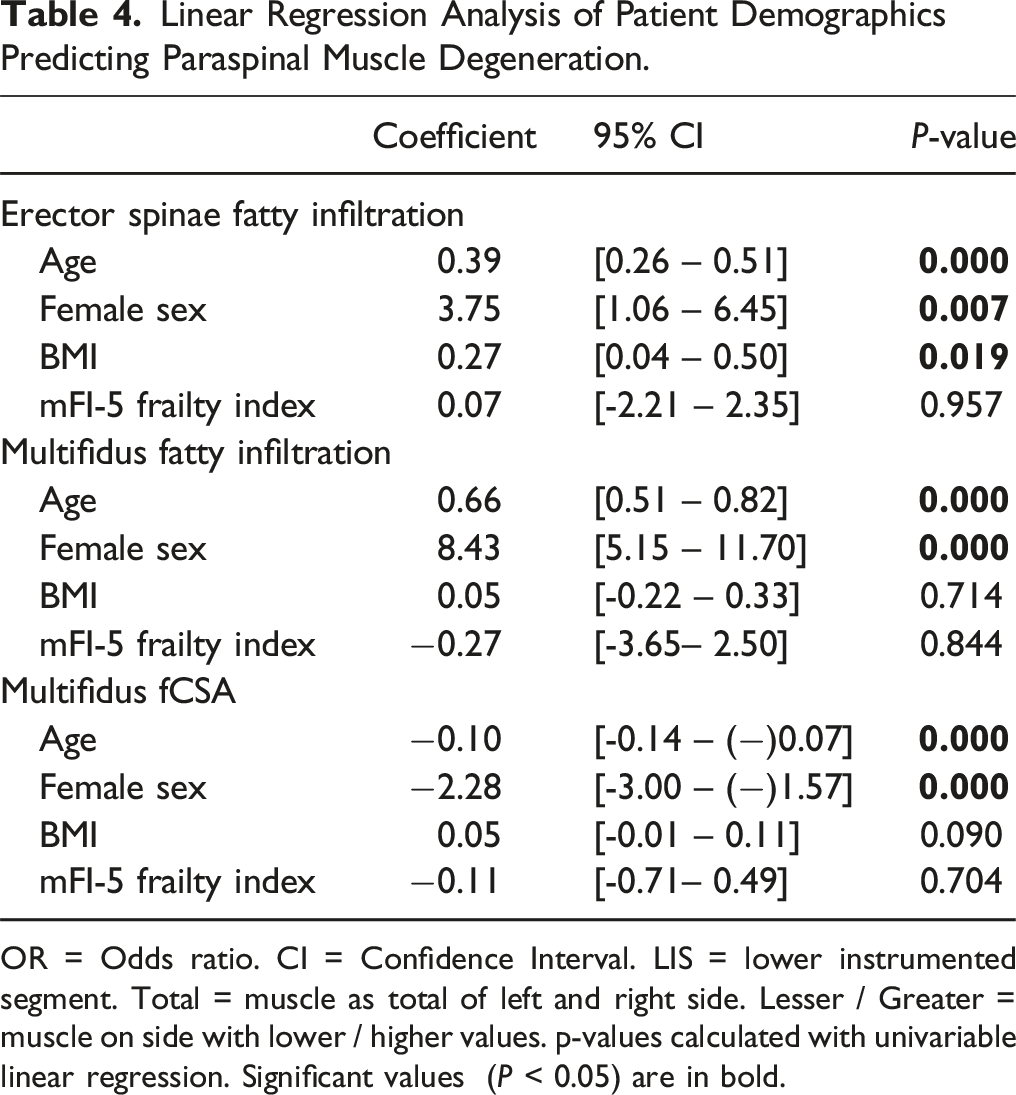
OR = Odds ratio. CI = Confidence Interval. LIS = lower instrumented segment. Total = muscle as total of left and right side. Lesser / Greater = muscle on side with lower / higher values. p-values calculated with univariable linear regression. Significant values (P < 0.05) are in bold.
Multivariable Logistic Regression Analysis of Muscle Parameters Predicting ASD Revision Surgery.
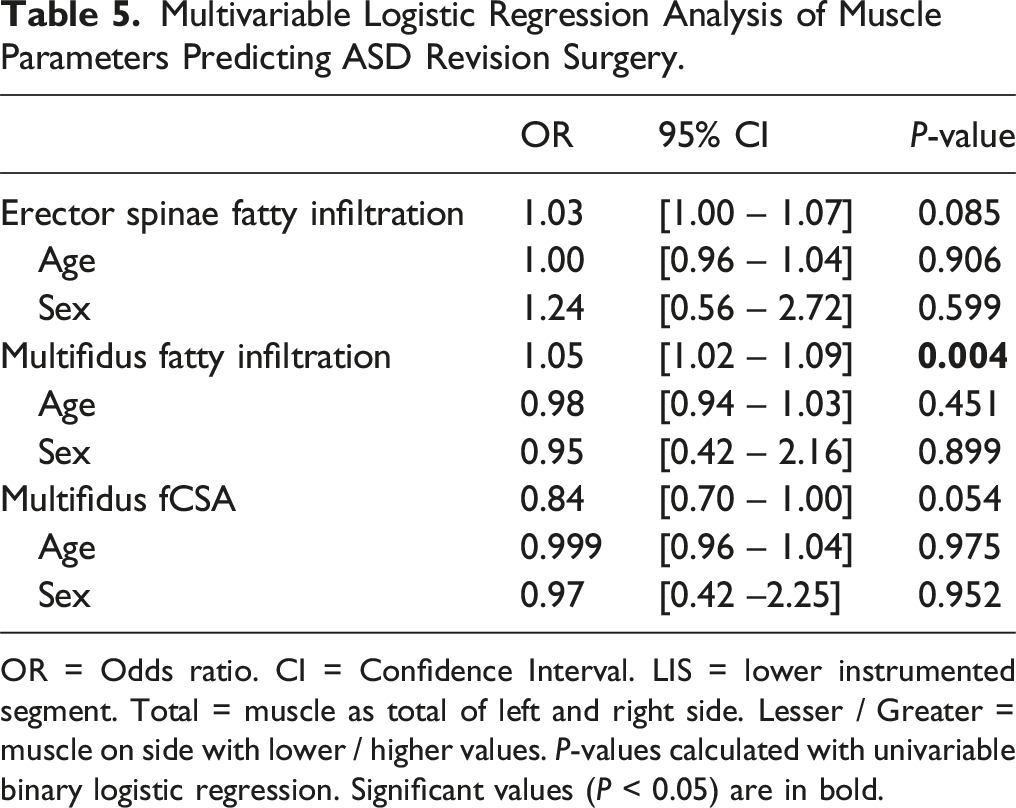
OR = Odds ratio. CI = Confidence Interval. LIS = lower instrumented segment. Total = muscle as total of left and right side. Lesser / Greater = muscle on side with lower / higher values. P-values calculated with univariable binary logistic regression. Significant values (P < 0.05) are in bold.
ROC Analysis
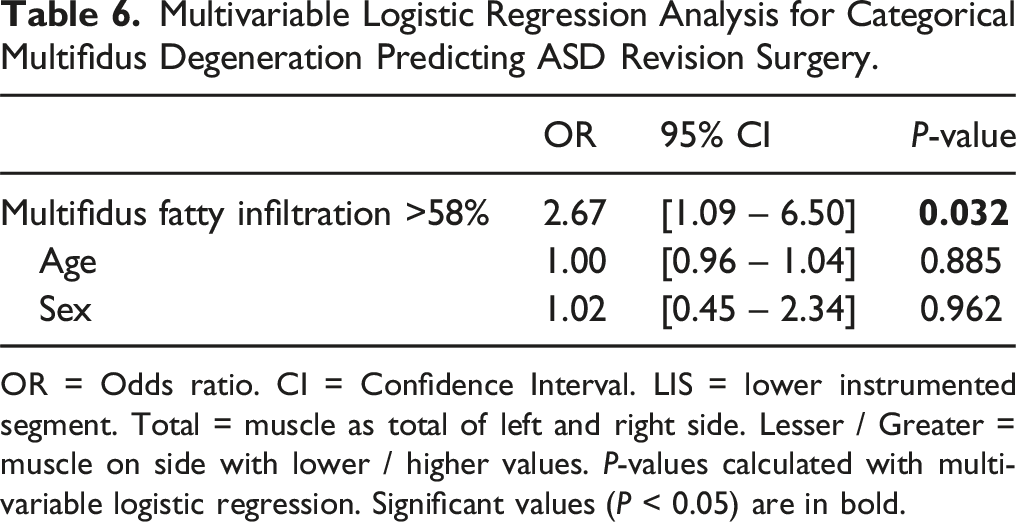
ROC analysis for multifidus fatty infiltration revealed an AUC of 0.648 for total fatty infiltration (Figure 6). A cut-off value of 58.3% was determined, with 59.4% sensitivity and 64.1% specificity for predicting ASD revision in this cohort. When applying this cut-off value in the multivariate logistic regression analysis adjusted for age and sex, multifidus fatty infiltration ≥58.0% was found to be a significant predictor of ASD revision surgery (OR 2.7, 95% CI: 1.1-6.5, P = 0.032) (Table 6). ROC analysis of multifidus fatty infiltration. Multivariable Logistic Regression Analysis for Categorical Multifidus Degeneration Predicting ASD Revision Surgery. OR = Odds ratio. CI = Confidence Interval. LIS = lower instrumented segment. Total = muscle as total of left and right side. Lesser / Greater = muscle on side with lower / higher values. P-values calculated with multivariable logistic regression. Significant values (P < 0.05) are in bold.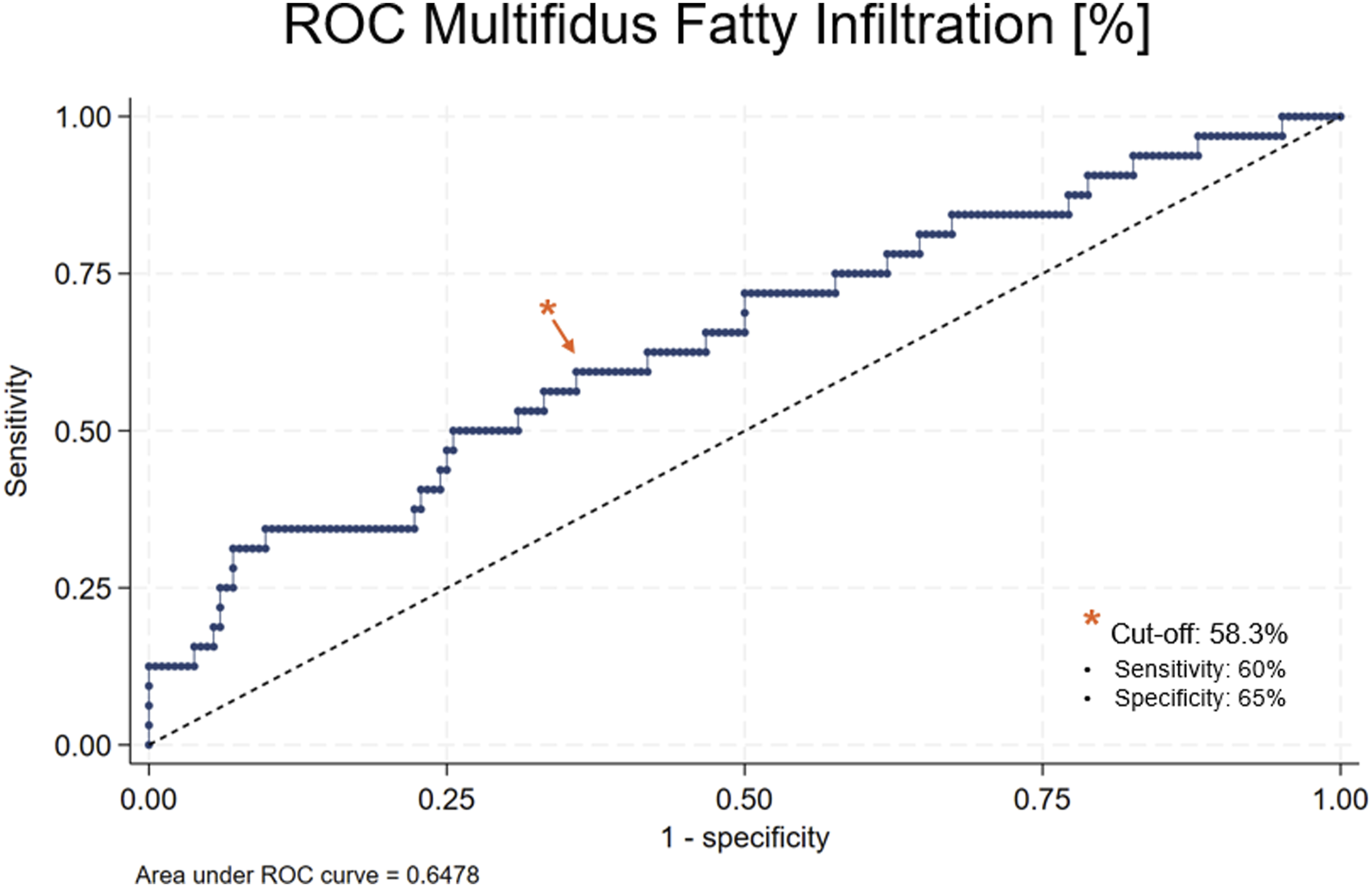
Discussion
This study elucidates preoperative and perioperative imaging factors that predict the development of surgically relevant ASD following elective lumbar fusion surgery. In our study cohort with a median follow-up of 4 years, the incidence of surgical ASD was 14.8%, which aligns with previously published reports.4,20 In contrast to previous research, our study specifically investigated clinically relevant ASD that necessitated a revision surgiery. The key finding is that fatty infiltration of the posterior paraspinal muscles, particularly the multifidus, is the strongest predictor of surgically relevant ASD.
Disc Degeneration and Modic Changes
In this study, disc degeneration and Modic changes were evaluated at both the operated and adjacent levels. Contrary to our hypothesis, we found no significant association between preoperative degeneration of the adjacent disc and future ASD requiring revision surgery. This is in line with Takeda et al., 5 who also found no linke between radiological ASD and Pfirrmann grades at the adjacent level before elective L5-S1 fusion. In contrast, Kiss et al 21 found a correlation between higher Pfirrmann scores in adjacent discs on the initial MRI and ASD. Differences in study design, settings, and especially ASD definitions, such as loss of disc height or segmental lordosis in their study and revision surgery for ASD in our study, may account for this disparity. In our study, Pfirrmann grades of the adjacent disc did not differ, but a trend toward higher degeneration in the operated levels were found in patients undergoing a future ASD Revision surgery (P = 0.282). Although not significant, our results may indicate that the degenerative cascade occurring in the lower lumbar spine translates to adjacent segments once the segments are fixed. Meanwhile, the initial extent of MRI-evident degeneration in the adjacent segments may have less influence on ASD onset.
Paraspinal Muscle
Impaired muscle health has been increasingly recognized as a marker of frailty, decreased quality of life, and adverse perioperative outcomes. 11 Although the muscle volume of the psoas is commonly used as a reference to measure the general muscle status or sarcopenia, spine-specific sarcopenia, as determined on posterior spinal muscles, is increasingly recognized in spine research.22,23 However, literature on paraspinal muscle assessment is scarce in the context of ASD and revision surgery.
Kim et al. 24 investigated fatty infiltration and CSA of the psoas and posterior paraspinal muscles (multifidus and erector spinae combined) and found a significant association between radiological ASD and a decreased CSA and increased fatty infiltration in the paraspinal muscles. However, only a few patients had symptomatic ASD and required surgery in their study. Our study on surgically relevant ASD mirrors their findings that fatty infiltration of the posterior paraspinal muscles is an important factor in ASD development. Furthermore, we demonstrated that fatty infiltration of the multifidus plays a more important role than the erector spinae in ASD development.
Gong et al. 25 also found that multifidus fatty infiltration as well as CSA of the multifidus were risk factors for ASD, which they defined on a radiographic basis. Our study highlights the importance of paraspinal muscle assessment in clinically relevant ASD development, particularly for multifidus fatty infiltration. The significant difference in multifidus fatty infiltration between the ASD and control groups suggests that alterations in the muscular envelope surrounding the lumbar spine may contribute to ASD pathogenesis. The multifidus, acting as a segmental stabilizer, may play a crucial role in preventing excessive stress on adjacent segments. 26
Spinopelvic Alignment
The secondary objective of this study was to assess spinopelvic alignment parameters in association with surgically relevant ASD development. Against our hypothesis and previous literature, the spinopelvic parameters were not associated with ASD in our cohort. 18 This may be attributed to the fact that our patient cohort did not include any major deformity surgery, but were mostly 1-2 level fusion procedures for degenerative disease in the lower lumbar spine. Although our study indicates that other radiologic factors, beyond the traditional radiographic spinal sagittal alignment measurements are even more important for successful long-term outcomes, we do believe that an optimal alignment should always be targeted intraoperatively as much as possible. A functioning paraspinal muscle envelope is crucial for the rehabilitation post-surgery as well as for a physiologic load distribution on the instrumented and adjacent segments. Thus, preservation of multifidus fibers, particularly of fibers attached to the adjacent vertebrae, should be attempted during every fusion procedure. Paraspinal muscle-sparing, minimally-invasive techniques may further show more advantageous ASD risk profiles. 27
Clinicians should recognize that patients with a significant degree of multifidus fatty infiltration (ie, >58%) are at an increased risk for subsequent revision surgery due to ASD. Therefore, it is essential to assess the paraspinal muscles, especially the multifidus, alongside the disc, neural, and bony structures. The findings of our study can aid in patient counseling and surgical decision-making, particularly when dealing with patients who have increased risk profiles based on their characteristics and MRI results. While this study demonstrates an association between preoperative paraspinal muscle degeneration and ASD development, the extent of muscle degeneration that occurs post-surgery—whether due to the surgical approach, muscle fiber detachment, disinnervation, or segment immobilization—remains unknown and is currently being investigated. It is possible that less-invasive, muscle-sparing fusion techniques may present a better risk profile for ASD development compared to open posterior fusion surgery. However, the choice of surgical strategy or approach was not the focus of this manuscript and is part of a separate ongoing study by our group. Additionally, the role of paraspinal muscle degeneration in determining the surgical technique, approach, and selection of upper and lower instrumented vertebrae is still unclear. Primary care physicians and spine specialists should emphasize the importance of preventing sarcopenia progression through regular exercise and a balanced diet.
Limitations
This study has several limitations that must be considered. In the prospectively enrolled cohort, 11.9% of patients did not have a 2-year minimum follow-up and another 7.8% were excluded due to missing MRI or low-quality MRI. Thus, only 80.3% of the patients of interest could be included for analysis, which may have introduced some selection bias. Moreover, the test accuracy of the cutoff values to detect the risk for future ASD revision applies to the cohort investigated in this study and would need to be validated with independent patient cohorts. Although multifidus fatty infiltration was the only independent imaging predictor of ASD, its sensitivity and specificity was moderate to low, indicating that other factors, beyond imaging markers may play a role in ASD development as well. Future multicenter studies with larger cohorts and longer follow-up periods are warranted to validate our findings and to further elucidate the underlying mechanisms that drive ASD development following lumbar fusion surgery.
Conclusion
Preoperative atrophy of the posterior paraspinal muscle is associated with a future ASD revision surgery, with fatty infiltration of the multifidus being the key imaging predictor. In contrast, the severity of disc degeneration at the operated and adjacent segments, as well as spinopelvic alignment, appear to be less influential in short-segment fusions. Assessment of not only bone, disc, and neural structures, but also of the surrounding muscular envelope on MRI, may be key for improved patient counseling and surgical decision-making.
Footnotes
Author contributions
\All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Marco D. Burkhard, Erika Chiapparelli, Ali E. Guven, Krizia Amoroso, and Jiaqi Zhu. The first draft of the manuscript was written by Marco D. Burkhard and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AAS reports royalties from Ortho Development, Corp. DePuy Spine Products/Medical Device Business Services, Clariance, Inc.; private investments for Vestia Ventures MiRUS Investment, LLC, ISPH II, LLC, ISPH 3, LLC, and Centinel Spine (Vbros Venture Partners V); consulting fees from DePuy Spine Products/Medical Device Business Services, Clariance, Inc., Kuros Biosciences AG; speaking and/or teaching arrangements for DePuy Spine Products/Medical Device Business Services; membership of scientific advisory board of DePuy Spine Products/Medical Device Business Services, Kuros Biosciences AG, Clariance, Inc., and research support from Spinal Kinetics, Inc./Orthofix, Inc., outside the submitted work. FPC reports royalties from Accelus; ownership interest for 4WEB Medical/4WEB, Inc.; Healthpoint Capital Partners, LP; ISPH II, LLC; ISPH 3 Holdings, LLC; Ivy Health care Capital Partners, LLC; Medical Device Partners II, LLC; Medical Device Partners III, LLC; Orthobond Corporation; Spine Biopharma, LLC; Tissue Differentiation Intelligence, LLC; Tissue Connect Systems, Inc.; VBVP VI, LLC; VBVP X, LLC; Woven Orthopedics Technologies; consulting fees from Spine Biopharma, LLC, and Accelus; membership of scientific advisory board/other office of Healthpoint Capital Partners, Medical Device Partners II, LLC, Orthobond Corporation, Spine Biopharma, LLC, and Woven Orthopedic Technologies; and research support from 4WEB Medical/4WEB, Inc., Camber Spine, Choice Spine, DePuy Synthes, Centinel Spine, and Royal Biologics outside the submitted work. FPG reports royalties from Lanx, Inc. And Ortho Development Corp.; ownership interest in Centinel Spine, BICMD; consulting fees from Lanx, Inc, Ortho Development Corp, and Sea Spine;and stock ownership in Centinel Spine, Healthpoint Capital Partners, LP; membership of scientific advisory board/other office of Healthpoint Capital Partners, outside the submitted work. APH reports research support from Kuros Biosciences AG and Expanding Innovations, Inc.; private investments in Tissue Connect Systems, Inc.; and fellowship support from NuVasive, Inc., Globus Medical North America, Inc., and Alphatec Spine, Inc., outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
ORCID iDs
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
