Abstract
Study Design
Retrospective cohort study.
Objective
This study aimed to compare postoperative pain and surgical outcomes of open-door laminoplasty (LP) and three types of muscle-sparing laminoplasties, namely unilateral muscle-preservation laminoplasty (UL), spinous process splitting double-door laminoplasty (DL) and intermuscular “raising roof” laminoplasty (RL) for multilevel degenerative cervical myelopathy (MDCM).
Methods
Consecutive MDCM patients underwent LP or modified laminoplasties (UL, DL, RL) in 2022 were enrolled. Patients’ preoperative baseline data and surgical characteristics were collected. Postoperative transient pain (TP), the axial pain and Japanese Orthopedic Association (JOA) score and neck disability index (NDI) at 6-month and 12-month follow-up were documented.
Results
A total of 154 MDCM patients were included and a 12-month follow-up was completed for 148 patients (LP: 36, UL:39, DL: 37, RL:36). No significant difference was observed in the baseline data. Four groups presented favorable and comparable surgical outcome. The RL group reported significantly the least severe TP on the first three days following surgery. However, no significant difference was found in the axial pain and axial symptoms at both follow-ups. After regression analysis, RL group exhibited significantly better efficacy in alleviating Day-1 TP (P = 0.047) and 6-month axial pain (P = 0.040). However, this superiority was not observed at 12-month follow-up.
Conclusion
All the three muscle-sparing laminoplasty procedures showed similar short-term surgical outcomes compared to LP. The RL procedure demonstrated superiority in alleviating TP and 6-month axial pain compared to LP. The RL and DL groups showed less C5 palsy compared to LP.
Keywords
Introduction
Expansive open-door laminoplasty (LP) is one of the most widely used procedures for treating multilevel degenerative cervical myelopathy (MDCM). Abundant investigations have proven that LP has favorable efficacy, high safety and a low incidence of complications.1-3 However, the postoperative pain after LP remains challenging, impacting patient satisfaction and overall quality of life. Postoperative pain following LP can be categorized into two types: transient pain (TP), which is experienced immediately after surgery, and postoperative axial pain (AP), which is often accompanied by stiffness and collectively referred to as axial symptoms (ASs). The incidence of AS after cervical LP ranges from 7% to 58%.4,5 Recent studies have suggested that intraoperative damage to the posterior extensor muscles and ligaments, which constitute the muscle-ligament complex (MLC), may play a role in the development of post-LP ASs.6,7
To address this concern, several modified laminoplasty techniques, such as unilateral muscle-preservation laminoplasty (UL) and muscle-sparing spinous process splitting double-door laminoplasty (DL), have been developed to protect the MLC.8–10 (Figure 1). These procedures tried to utilize the natural intermuscular gap and minimize paraspinal MLC detachment from bony structures, thus subsequently preserving the function of deep extensor muscles and reducing the risk of axial symptoms and post-LP kyphosis. Additionally, following the same principle, we have devised the intermuscular “raising roof” laminoplasty (RL) using the natural gap between semispinalis capitis and semispinalis to protect the bilateral muscles. Although investigations have reported the efficacy of these muscle-sparing techniques in preserving posterior extensor muscles and cervical curvature and preventing post-LP ASs,
11
limited information is available regarding post-LP pain, especially the transient pain following these muscle-sparing procedures. Furthermore, the surgical outcomes of the RL procedure have not been adequately described. The cross-sectional MR image of posterior muscles on C3-4 Level. MF: multifidus muscle; SSCap: semispinalis capitis; SSCer: semispinalis; SPCap: splenius capitis.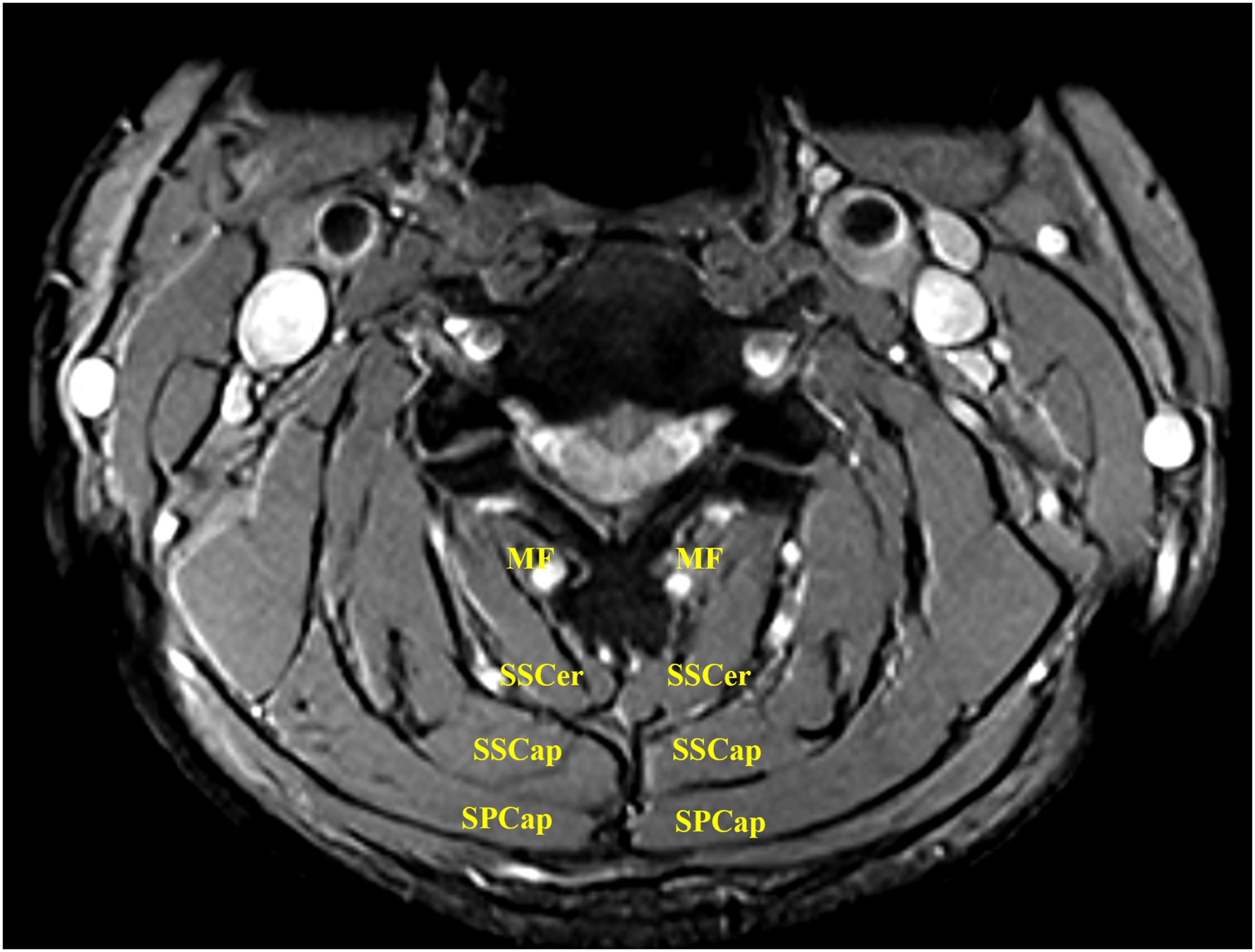
As one of the largest cervical spine centers in China, there are several different types of muscle-sparing laminoplasties were performed in the same surgical ward. From our clinical experience, we have observed that though all these modified surgeries can achieve favorable neurological outcomes, the degree of TP may vary. Additionally, the occurrence of AP and ASs following different muscle-sparing laminoplasties remains unclear. Therefore, we designed this study to investigate the TP, AP, ASs and short-term surgical outcomes of three different muscle-sparing LP techniques compared to conventional laminoplasty. We hypothesized that the muscle-sparing procedures can reduce the postoperative pain and the incidence of axial symptoms.
Materials and Methods
Study Design and Population
This study received approval from the ethics committee of the University’s hospital (IRB00006761-M2023296) and was conducted in accordance with the principles of the Declaration of Helsinki. To ensure data consistency, the study included consecutive MDCM patients who underwent either a conventional laminoplasty (LP) or one of three modified muscle-sparing laminoplasty procedures within the same surgery ward at our hospital in 2022. All patients met the inclusion criteria for posterior laminoplasty surgery.
The inclusion criteria for patient enrollment were as follows: (1) confirmed diagnosis of MDCM based on radiological findings and symptoms, (2) received conventional laminoplasty or three kinds of modified laminoplasties. Exclusion criteria were as follows: (1) obvious cervical instability, (2) severe kyphosis, (3) history of spinal trauma or cervical surgery, (4) laminoplasty or laminectomy with posterior fixation, (5) diagnosis of depression or anxiety and (6) obvious facet arthrosis.
The patients were divided into four groups based on the procedure performed: LP group, UL group, DL group, and RL group. The flow diagram illustrates patient recruitment and postoperative follow-ups (Figure 2). The flow chart of the study design, patient cruiment and follow-up.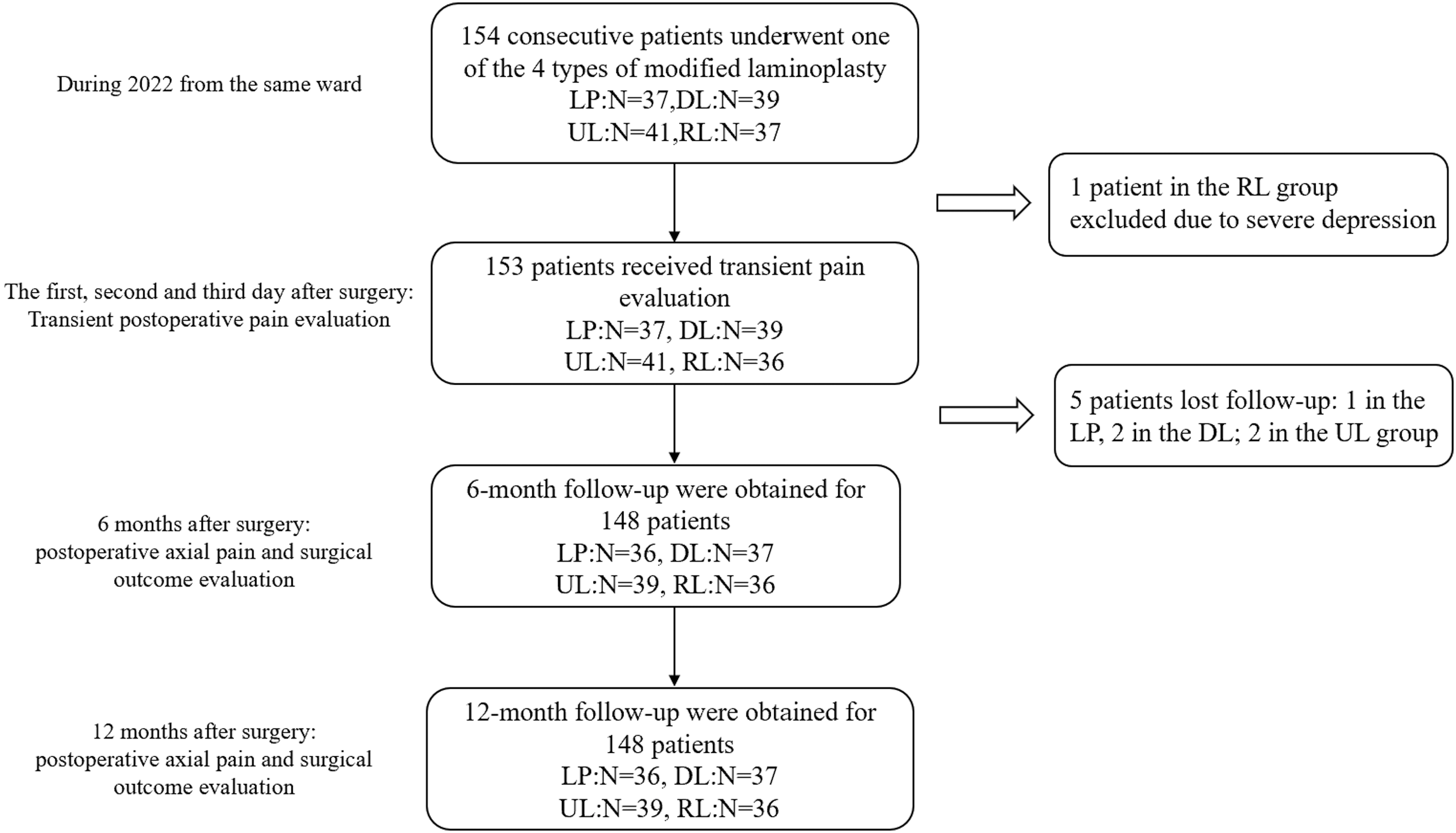
Surgical Techniques
All surgeries were performed by senior spine surgeons from the same team in the Department of Orthopedics. The surgeons chose specific surgical procedure based on their individual preferences. General anesthesia was administered for all surgeries. The patient was positioned prone with the cervical spine in mild flexion, and the head was immobilized using a Mayfield headrest. A midline incision was made in the skin and a subcutaneous fascia layer along the extended neck. The specific procedures for the four different laminoplasty techniques were as follows (Figure 3): Surgical schematic diagrams of four types of modified laminoplasties. (A) The open-door laminoplatsy. (B) The unilateral muscle-preserved laminoplasty. (C) double-door laminoplasty. (D) intermuscular “raising roof” laminoplasty.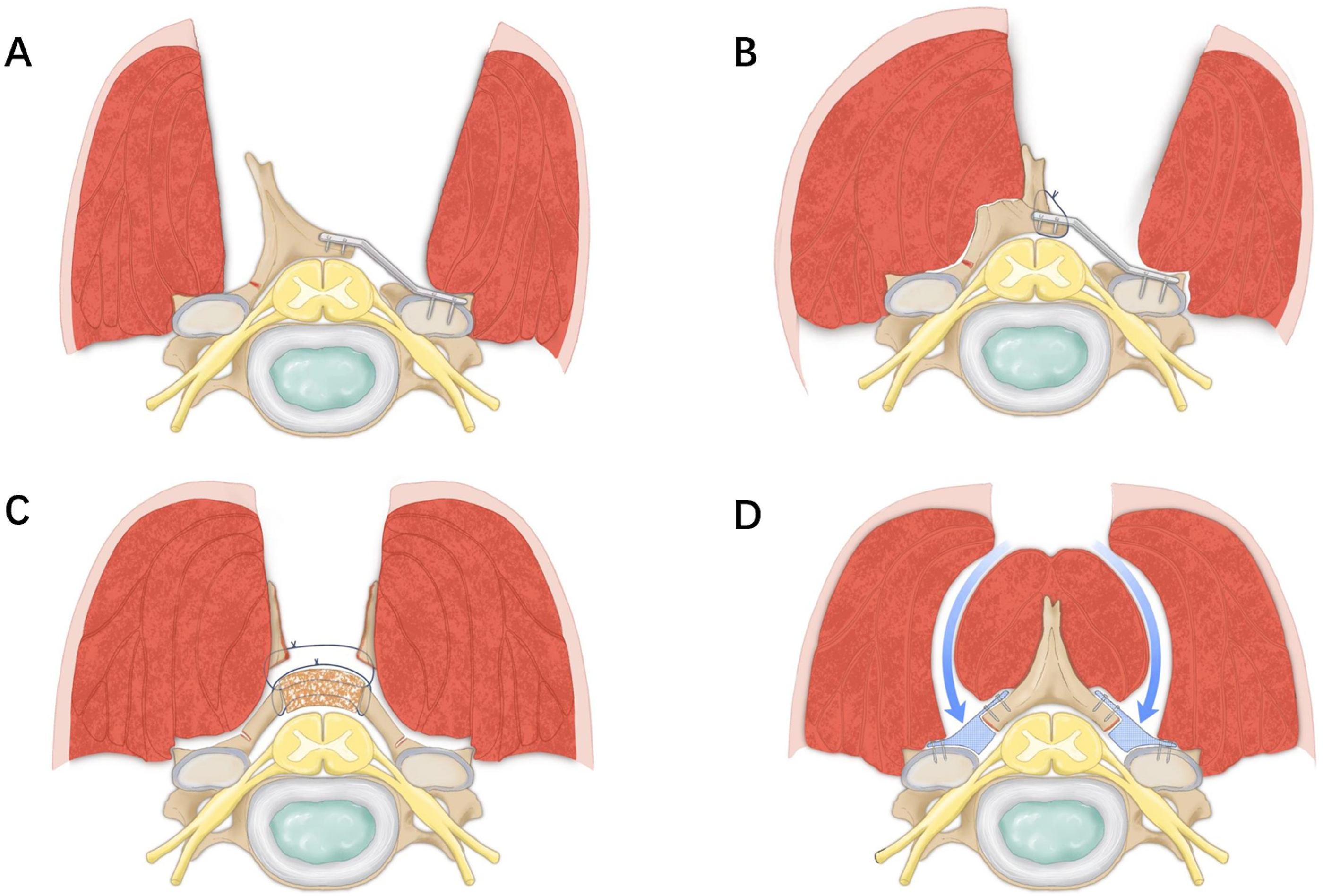
Conventional laminoplasty (LP, Figure 3A) was conducted by standard procedure with titanium miniplates and screws. 12
Unilateral muscle-preservation laminoplasty (UL, Figure 3B) was performed as Chen et al 9 had reported. The left paraspinal muscles were dissected to expose the left spinous process. The spinous process was cut at its base using an electric saw and was flipped to the right side along with the attached muscles, exposing the right spinous process. At the junction between the spinous process and the lateral mass on one side, a bone nibbler and burr were used to remove the outer cortical bone and some cancellous bone, creating a hinge. The entire layer of the lamina was cut at the junction between the spinous process and the lateral mass on the other side and some ligamentum flavum was simultaneously severed. The spinous process was flipped to the right side and stabilized with titanium plates and screws. Then holes were drilled at the base of the spinous process and the left side of the flipped spinous process, and titanium cables were used to reconstruct and fix the spinous process to the lamina.
The spinous process splitting Double-door laminoplasty (DL, Figure 3C) was performed as Nakashima has reported with some modifications. 13 After locating the apex of the spinous process, the spinous process was split into a “T-shape”, the muscular attachment on the spinous process tip can be preserved. Then the bilateral paraspinal muscles on the lamina were bluntly peeled off and the junction between the lamina and the lateral mass on both sides were exposed. A burr was used to remove the outer cortical bone and some cancellous bone, creating hinges on both sides. Then the base of spinous process was split at the midline and flipped to both sides. Finally, the split spinous processes on both sides were fixed with a graft by suture.
Intermuscular “raising roof” laminoplasty (RL, Figure 3D) was performed as Goto has reported with some modifications via the intermuscular gap.
14
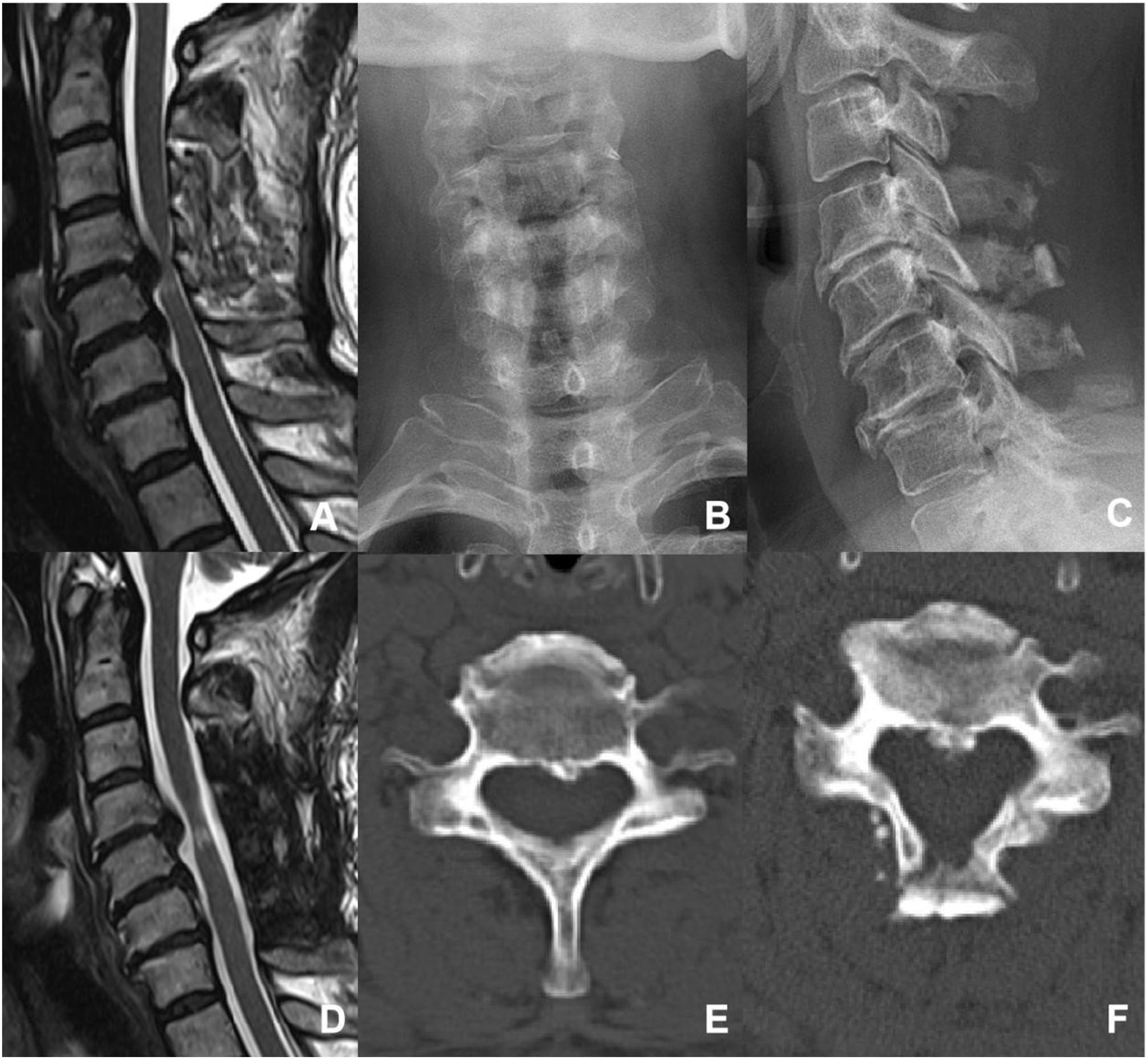
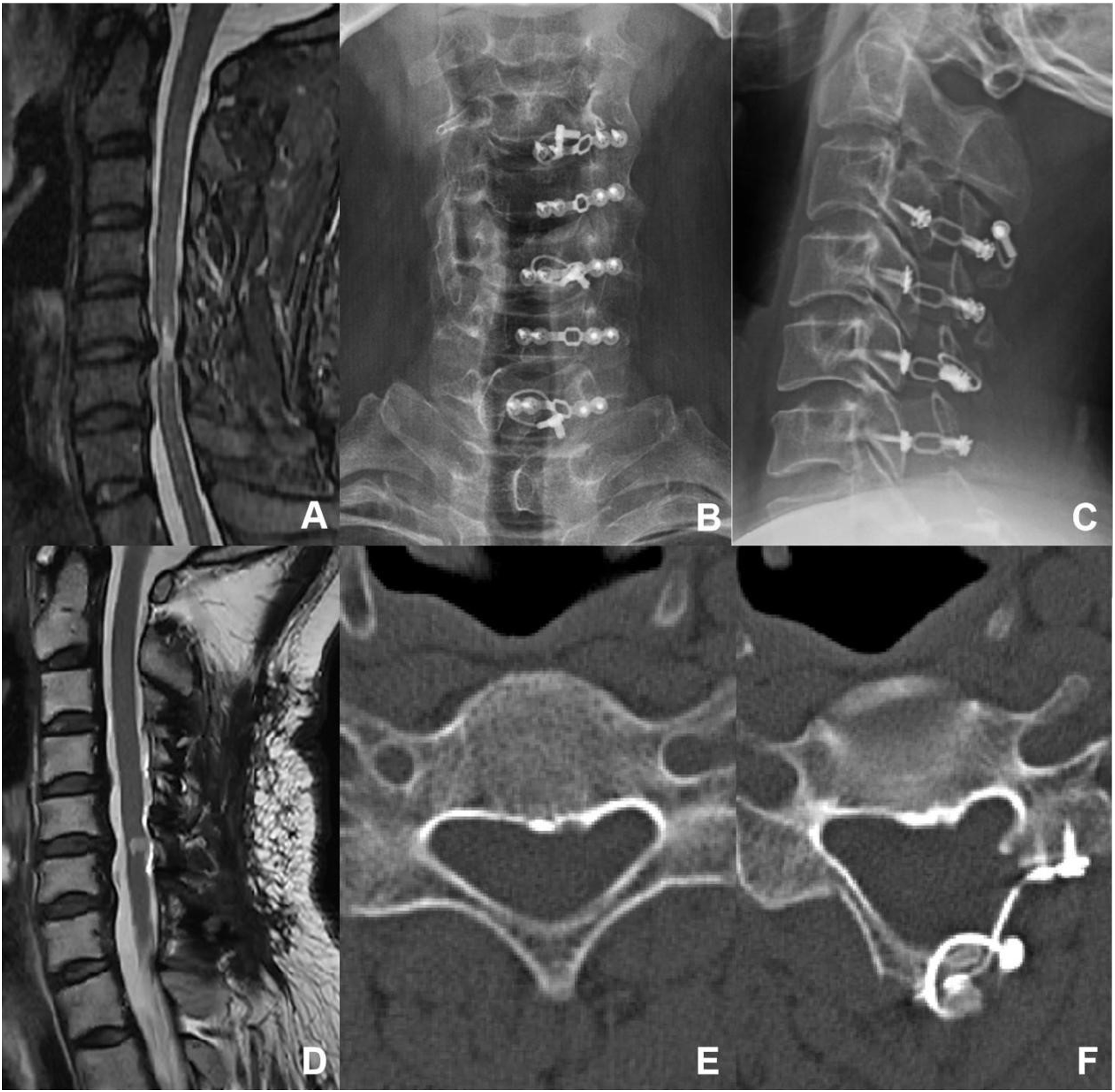
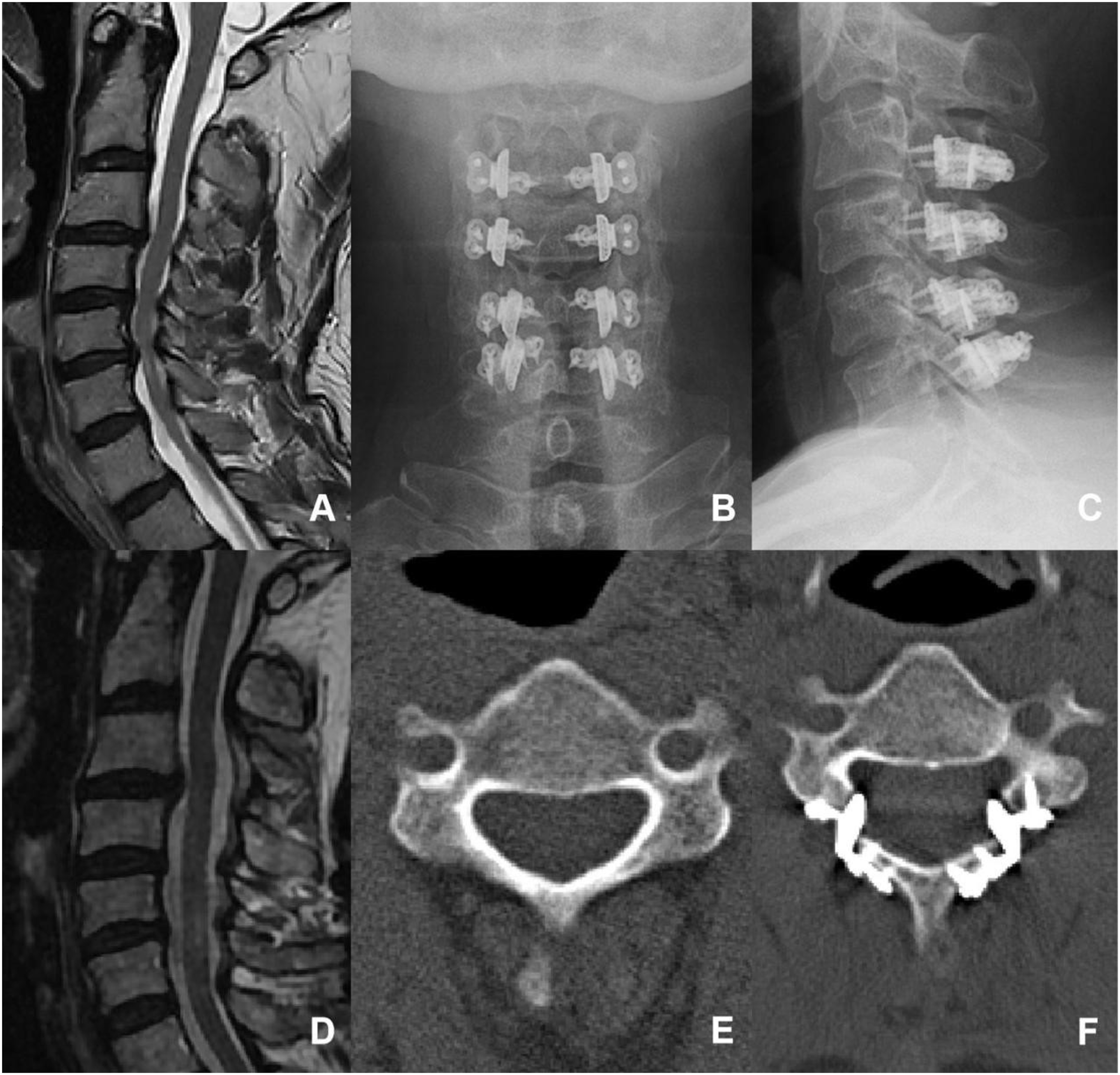
The deep layer of the trapezius muscle was dissected laterally, aiming to locate the gap between the splenius capitis muscle and the splenius cervicis muscle, and dissection was continued along the gap until the surface of the lamina. Multifidus and the rotator muscles were then bluntly separated to expose the junction between the lamina and the lateral mass. Initially, on the right side, a bone nibbler and burr were used to remove the outer cortical bone and some cancellous bone, creating a hinge. On the left side, the entire layer of the lamina and some ligamentum flavum was cut at the junction between the lamina and the lateral mass. Then the lamina was flipped to the right side, and a 3D-printed artificial spacer was placed and fixed with titanium screws, but the screws were not tightened. Then, on the right side at the hinge, the entire layer of the lamina and ligamentum flavum was cut, another artificial laminar spacer was placed and fixed with titanium screws, and the screws on both sides were tightened Figures 4-7. An illustration of open-door laminoplasty. A 68-year-old female presented with a 6-month history of numbness, weakness, and pain in upper limbs, with a preoperative JOA score of 14.5/17. (A): Preoperative sagittal view of cervical spine MRI, revealing multiple segmental disc protrusions and cervical spinal canal stnosis. (B) and (C): Postoperative X-ray images in antero-posterior and lateral views. (D): Postoperative sagittal view of cervical spine MRI, revealing an enlarged spinal canal and no obvious compression of the cervical spinal cord. (E) and (F): Preoperative and postoperative axial view of CT images, the surgery was done with open-door laminoplasty (LP). At the final follow-up, the JOA score was 16/17. An illustration of double-door laminoplasty. A 43-year-old female presented with numbness in both hands for 5 years and right upper limb pain for 6 months, with a preoperative JOA score of 13.5/17. (A): Preoperative sagittal view of cervical spine MRI, revealing multiple segmental disc protrusions and cervical spinal canal stenosis. (B) and (C): Postoperative X-ray images in antero-posterior and lateral views. (D): Postoperative sagittal view of cervical spine MRI, revealing an enlarged spinal canal and no obvious compression of the cervical spinal cord. (E) and (F): Preoperative and postoperative axial view of CT images, the surgery was done with double-door laminoplasty (DL). At the final follow-up, the JOA score was 17/17. An illustration of unilateral muscle-preserved laminoplasty. A 53-year-old male presented with Numbness in both hands and weakness in both lower limbs for 5 years, with a preoperative JOA score of 14/17. (A): Preoperative sagittal view of cervical spine MRI, revealing multiple segmental disc protrusions and cervical spinal canal stenosis. (B) and (C): Postoperative X-ray images in antero-posterior and lateral views. (D): Postoperative sagittal view of cervical spine MRI, revealing an enlarged spinal canal and no obvious compression of the cervical spinal cord. (E) and (F): Preoperative and postoperative axial view of CT images, the surgery was done with unilateral muscle-preservation laminoplasty (UL). At the final follow-up, the JOA score was 17/17. An illustration of intermuscular “raising roof” laminoplasty. A 59-year-old male presented with Numbness in both hands and weakness in both lower limbs for 8 years, with a preoperative JOA score of 14/17. (A) Preoperative sagittal view of cervical spine MRI, revealing multiple segmental disc protrusions and cervical spinal canal stenosis. (B) and (C): Postoperative X-ray images in antero-posterior and lateral views. (D): Postoperative sagittal view of cervical spine MRI, revealing an enlarged spinal canal and no obvious compression of the cervical spinal cord. (E) and (F): Preoperative and postoperative axial view of CT images, the surgery was done with intramuscular “raising roof” laminoplasty (RL). At the final follow-up, the JOA score was 16/17.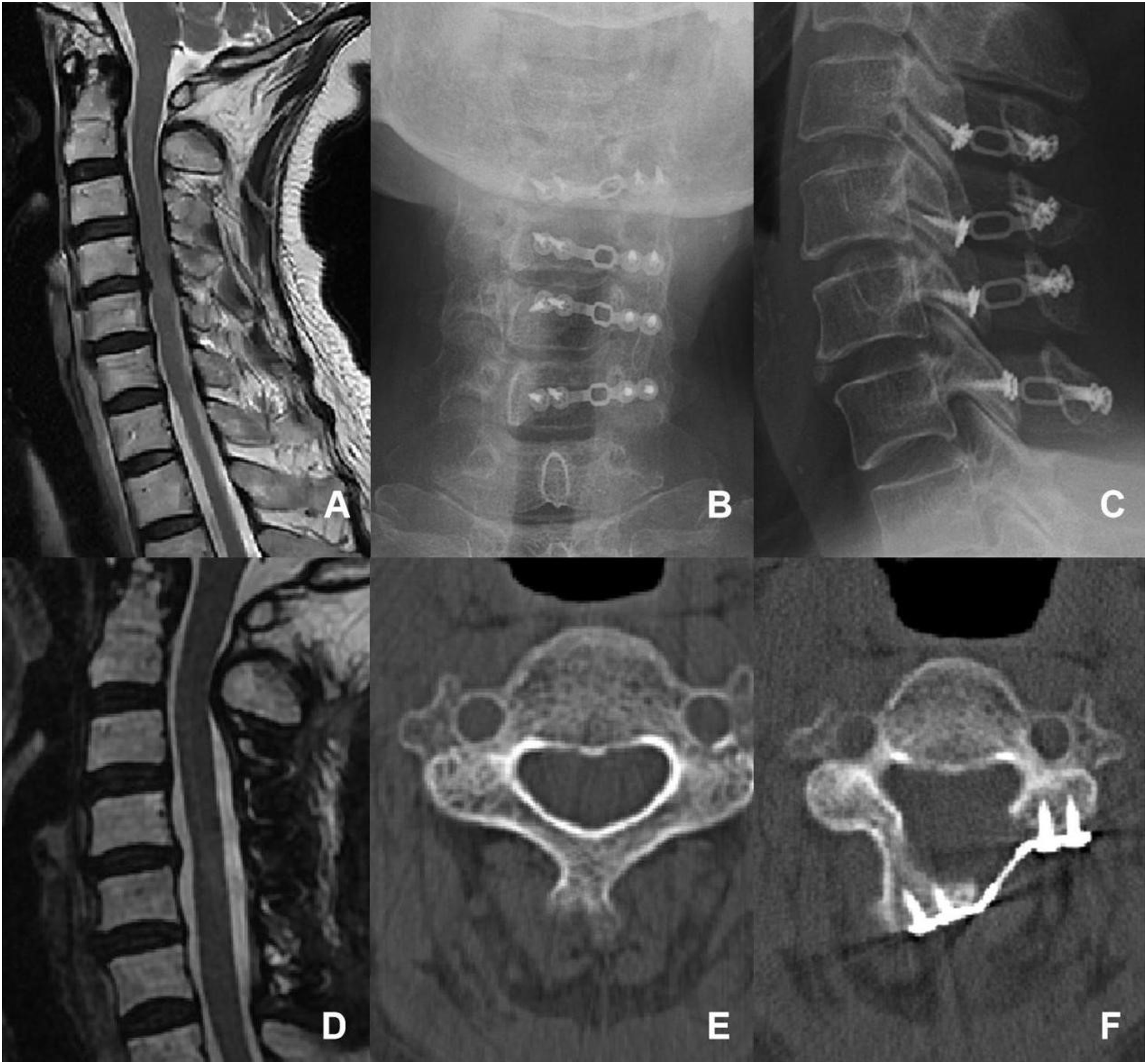
For the closure techniques, bilateral semispinalis cervicis, capitis muscle, and trapezius muscle were interruptedly sutured using #1 absorbable sutures. The fascial layer, subcutaneous tissue, and dermal layer were continuously sutured separately using 2-0 absorbable sutures. Finally, the intradermal suture was applied to the skin.
Perioperative Management
All the patients received the same perioperative pain management plan. Preemptive analgesia was administered in the evening before surgery; local infiltration anesthesia was applied with ropivacaine and lidocaine when the wound was closed; intravenous application of nonsteroidal analgesics was applied within 48 h after surgery; and then oral nonsteroidal analgesics combined with muscle relaxants were administered for 1 week. Patients were instructed to leave the bed first day after surgery and perform early exercises. The drainage tube was removed when the drainage volume was less than 50 mL/24 h. After drainage tube removal, the patients were discharged. The Philadelphia cervical collar was required to be worn for one month after surgery and patients were required to perform cervical extensor muscle exercises 2 weeks after surgery. 8
Clinical Evaluation
Patient demographics, surgical characteristics, preoperative baseline clinical data were collected. The preoperative neck pain was evaluated using the numerical rating scale (NRS) and neck disability index (NDI). Neurological function was evaluated using the Japanese Orthopedic Association (JOA) score. Surgical duration and blood loss were recorded.
Patients were advised to schedule outpatient follow-up appointments at six, and 12 months after surgery. The TP was evaluated in the ward on the first, second and third days after surgery using NRS score. The postoperative AP was defined as postoperative nuchal or periscapular pain and evaluated at each follow-up point using the NRS score. Pain alleviation was calculated by the preoperative NRS score minus the postoperative NRS score. The axial symptoms were evaluated at each follow-up point and were defined as any complaint of nuchal or periscapular stiffness and/or comfort.
Postoperative JOA, JOA recovery rate (JOARR) and neck disability index (NDI) scores were assessed. 2 Perioperative Complications, including transient paralysis, progression of myelopathy, hematoma, wound complications, infection, cerebrospinal fluid (CSF) leakage, and C5 nerve root palsy (C5 palsy), were assessed.
Statistical Analysis
SAS (version 9.2; SAS Institute Inc, Cary, NC) was employed for the statistical analysis. Categorical variables were expressed as percentages and counts, and the continuous variables were expressed as the mean value ±standard deviation. Additionally, T-tests, Chi square tests, ANOVA and nonparametric analysis were employed to compare the differences between different groups. Multivariable linear regression was conducted to control confounding bias. A two-tailed P value less than 0.05 was considered to indicate statistical significance.
Results
Population Characteristics
A total of 154 patients were included in this study based on the criteria. Of the 154 eligible patients, one patient was excluded due to severe depression, five patients were lost to follow-up, and the follow-up obtained for 148 patients at 6 months and 12 months following surgery; of these patients, 36 were in the LP group, 37 were in the DL group, 39 were in the UL group, and 36 were in the RL group.
The Demographic, Clinical and Surgical Characteristics of the Four Groups.
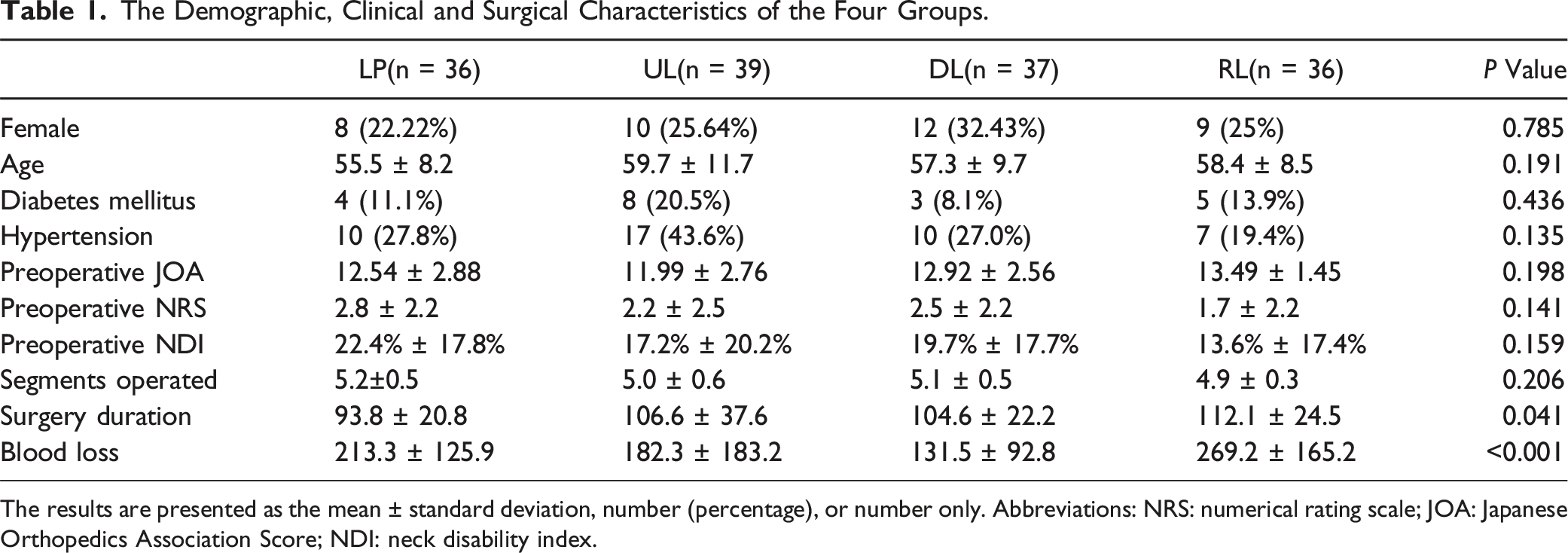
The results are presented as the mean ± standard deviation, number (percentage), or number only. Abbreviations: NRS: numerical rating scale; JOA: Japanese Orthopedics Association Score; NDI: neck disability index.
Surgical Characteristics and Complications
The average operation levels of the LP, DL, UL, and RL groups were 5.2 ± 0.5, 5.1 ± 0.5, 5.0 ± 0.6, and 4.9 ± 0.3, respectively (P = 0.206). The operation duration in the LP group was significantly shorter than that in the other groups (P = 0.041).
There were six patients (16.7%) in the LP group and four patients (10.3%) in the UL group reported C5 palsy at the 6-month follow-up (P = 0.009), whereas only one patient in the LP group reported the remaining C5 palsy at 1 year follow-up. Additionally, there was one CSF leakage in the UL group (P = 0.375). No other complications were found in any of the four groups.
Postoperative TP after Different Surgeries
Transient Postoperative Neck Pain in the Four Groups.
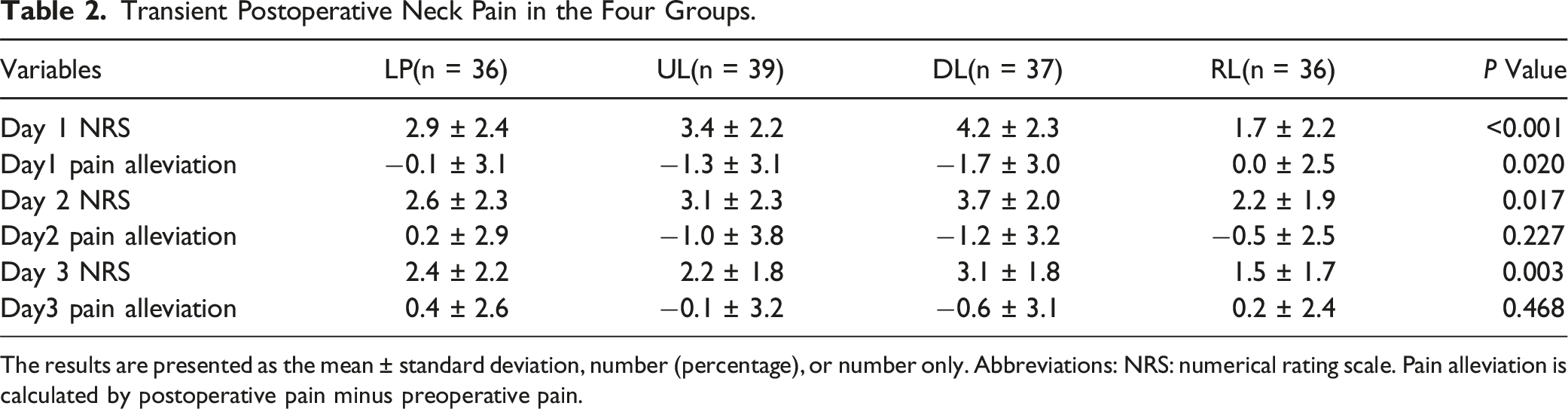
The results are presented as the mean ± standard deviation, number (percentage), or number only. Abbreviations: NRS: numerical rating scale. Pain alleviation is calculated by postoperative pain minus preoperative pain.
Postoperative AP and Neurological Outcomes
Surgical Outcomes and Axial Pain at 6 months and 1 year After Surgery.

The results are presented as the mean ± standard deviation, number (percentage), or number only. Abbreviations: NRS: numerical rating scale; JOA: Japanese Orthopedics Association Score; JOARR, JOA recovery rate; NDI: neck disability index, SF-36-PF: short-form-36 physical function. Pain alleviation is calculated by postoperative pain minus preoperative pain.
At the 12-month follow-up, the four groups presented higher postoperative JOA scores and JOARR than did those at the 6-month follow-up. No significant differences were found in the postoperative JOA scores (P = 0.079), JOARR (P = 0.143). The LP group had higher postoperative NDI scores than the other three groups (P = 0.044), but no statistically significant difference was found in the NDI improvement (n = 0.231). The postoperative axial pain (P = 0.099), pain alleviation (P = 0.211) and the incidence of ASs of four groups did not show significant differences.
Multivariable Regression
Multivariable Regression Results of Transient Postoperative Pain Alleviation During the First Three Days After Surgery.
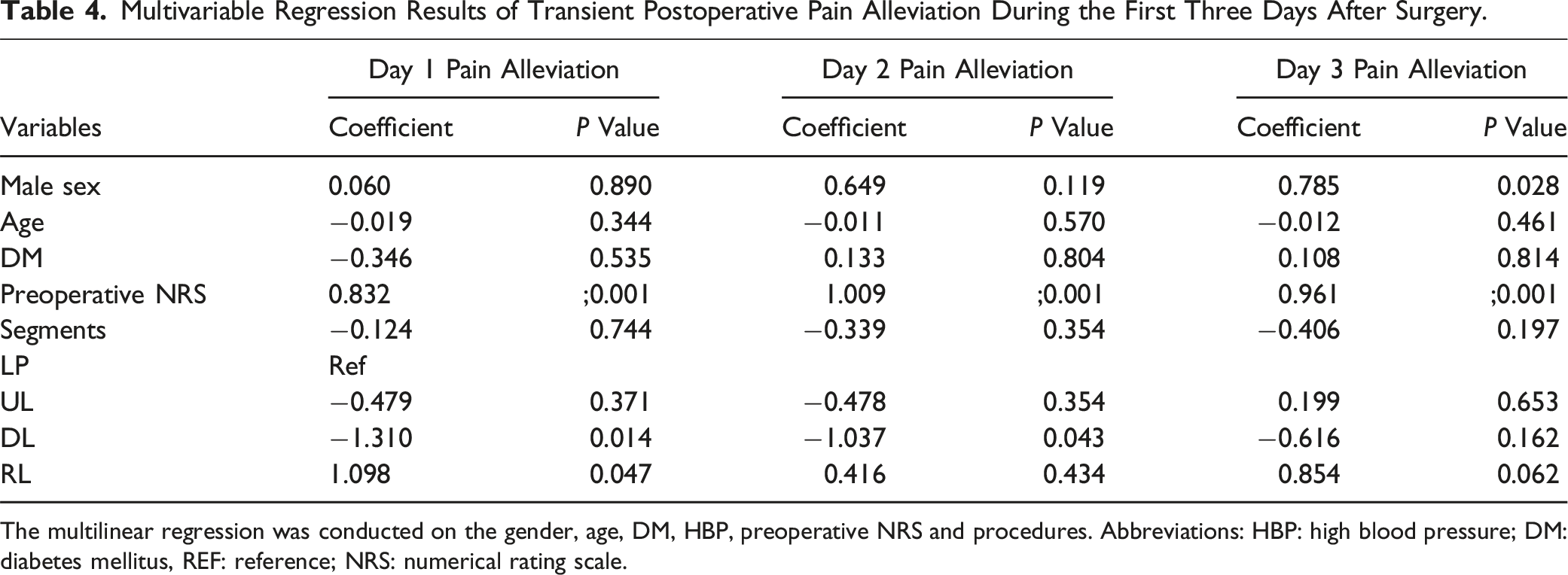
The multilinear regression was conducted on the gender, age, DM, HBP, preoperative NRS and procedures. Abbreviations: HBP: high blood pressure; DM: diabetes mellitus, REF: reference; NRS: numerical rating scale.
Multivariable Regression Results of Postoperative Axial Pain Alleviation and Axial Symptoms at the 6-Month and 12-Month Follow-Ups.
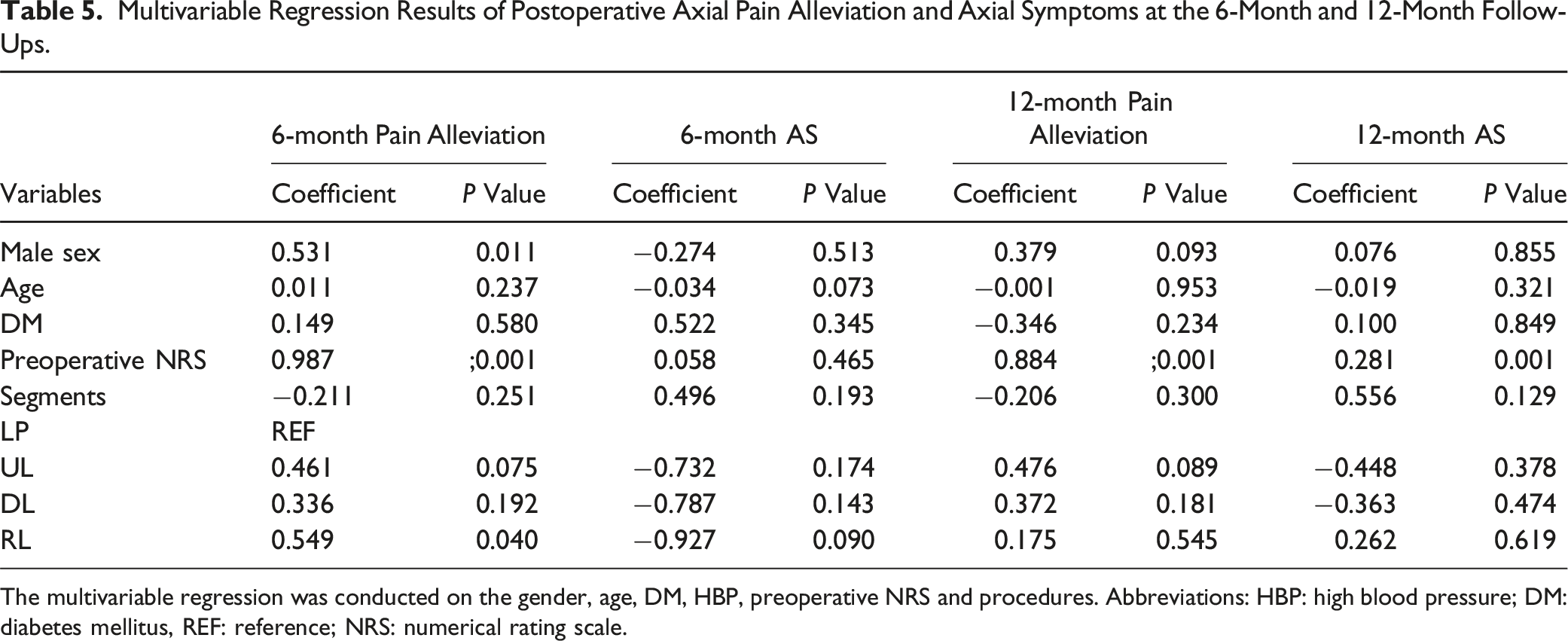
The multivariable regression was conducted on the gender, age, DM, HBP, preoperative NRS and procedures. Abbreviations: HBP: high blood pressure; DM: diabetes mellitus, REF: reference; NRS: numerical rating scale.
Discussion
Concurrent conventional open-door laminoplasty has been successfully performed in MDCM patients for decades, with proven efficacy.1–3 However, the patients commonly faced with post-LP axial symptoms (ASs) and postoperative transient neck pain. Theoretically, the LP procedure unavoidably causes intraoperative damage to the posterior ligament and extensor muscular attachment to the spinous process and lamina, namely MLC, which has been associated to ASs and cervical malalignment.6,7,15 Recently, increasing studies have highlighted the importance of the deep extensor muscles in cervical degenerative diseases, such as the multifidus and semispinalis.16-19 Consequently, several modified laminoplasty techniques aimed at MLC preservation have been developed and been reported with fair efficacy in muscle preservation and post-LP ASs prevention.20-23 However, few studies have specifically focused on the efficacy of TP management of these muscle-sparing procedures, which may also be theoretically influenced by intraoperative damage to the MLC. Furthermore, the outcomes of newly-developed intermuscular “raising roof” laminoplasty (RL) have not been extensively described.
In our clinical experience, all three modified laminoplasty procedures performed at our center, namely DL, UL, and RL, have demonstrated efficacy in terms of neurological function recovery and pain alleviation. In detail, the UL surgery commonly preserves the ligamental-muscular attachments on the spinal process and unilateral lamina-muscle attachments on the hinge side; the DL preserve the muscular attachments on spinous process tip; and the RL surgery preserves paraspinal ligamental-muscular attachments on both sides.9,24 Utilizing these surgical techniques, the surgeons expected decreased trauma of MLC, and thus preserved function of these muscles. Therefore, we designed this study to investigate the postoperative pain and outcomes of the three types of modified muscle-sparing laminoplasty procedures compared to conventional LP. Our hypothesis is that the patients undergoing these muscle-sparing procedures would experience milder postoperative TP and AP and lower incidence of axial symptoms.
In this study, no significant differences were observed in the baseline data among four groups. Regarding surgical characteristics, most patients in all four groups underwent surgery on five segments. However, the RL group had the longest surgical duration and the highest amount of blood loss, which can be attributed to the technical complexity and study curve. After surgery, the RL group reported significantly milder transient pain than the other three groups and greatest pain alleviation on Day 1. Regression analysis revealed that the RL group did demonstrate greater pain alleviation on the first day after surgery, while the DL group presented significantly lower pain alleviation than the LP group. A possible explanation for this difference was that the RL procedure preserved the bilateral muscles, thus leading to milder postoperative pain; while the DL group has more bone resection on spinous process which might lead to higher TP. These findings highlight the potential of the RL procedure in managing postoperative transient pain.
During both follow-up periods, all four groups presented remarkable and comparable neurological recovery, with no reports of reoperations. The RL group exhibited milder AP at the 6-month and 12-month follow-ups. However, no significant intergroup differences were found in pain alleviation or axial symptoms. Regression analysis revealed that the RL group showed higher efficacy at the 6-month pain alleviation, but this superiority was not observed at the 12-month pain alleviation. Interestingly, the LP and UL groups had a higher incidence of C5 palsy than the other two groups. A possible explanation might be that the RL and DL procedure provides limited posterior drift space and thus lower rate of C5 palsy. Furthermore, recent studies have reported the post-LP multifidus swelling might have traction on the C5 nerve through the medial branches of the cervical dorsal rami and lead to C5 palsy, which is also a possible explanation.25,26 Further radiological and biomechanical analysis is needed to explore the underlying mechanism of this phenomenon.
This study is the first study to specifically investigate the TP and AP following muscle-sparing laminoplasty procedures. Our findings demonstrate that the RL procedure has superior efficacy in controlling TP and short-term AP than does LP. As enhanced recovery after surgery has become a consensus in the field of spinal surgery, we believe the effective short-term postoperative pain control is crucial. 27 Mild transient pain allows patients to mobilize early, facilitating early rehabilitation and plays a critical role in preventing postoperative complications associated with prolonged bedrest and improving long-term function. Therefore, it is recommended that surgeons pay greater attention to muscle-sparing procedures for their potential efficacy in perioperative pain control.
This study has several limitations that should be acknowledged. Firstly, this was a single-center study with a relatively short follow-up duration and limited sample, which may introduce biases and limitations in data analysis. Therefore, cautious interpretation of the findings is necessary. Secondly, though we have tried to minimize other confounding bias, such as conducting the study in the same ward and within the same timeframe; ensuring a standardized perioperative pain medication management protocol; excluding the patients with anxiety disorders, depression, and other psychiatric conditions that could potentially influence the results, the bias caused by individual subjectivity to pain and variations in responses to analgesics were hard to be eliminated in the current retrospective study. Multicenter prospective studies involving diverse patient populations with extended follow-up duration would help to validate and enhance the results. Thirdly, this study lacks radiological assessments, such as the measurement of cross-sectional area and functional cross-sectional area of the muscles, as well as the evaluation of cervical curvature. We are currently developing a three-dimensional automatic segmentation and assessment method using convolutional neural networks, with which a precise measurement of posterior muscles could be achieved, and the patients’ risk of postoperative malalignment and adverse patient reported outcome could be better predicted.17,28-30 Furthermore, the intermuscular natural gap between paraspinal muscles could be assessed, and the real effect of muscle-sparing procedures in muscle volume preservation could be validated.
Conclusion
All the three muscle-sparing laminoplasty procedures showed similar short-term surgical outcomes compared to conventional laminoplasty. The RL procedure demonstrated superiority in alleviating transient pain and 6-month axial pain compared to conventional laminoplasty. The DL and RL groups showed lower C5 palsy rate at 6-month follow-up compared to LP.
Footnotes
Acknowledgments
We thank Miss Qianqian Li for her help with the data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data availability statement
Research data supporting this publication are available from the Peking University Third Hospital, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly.
