Abstract
Study Design:
Retrospective multicenter case series study.
Objective:
Because cervical dural tears are rare, most surgeons have limited experience with this complication. A multicenter study was performed to better understand the presentation, treatment, and outcomes following cervical dural tears.
Methods:
Multiple surgeons from 23 institutions retrospectively identified 21 rare complications that occurred between 2005 and 2011, including unintentional cervical dural tears. Demographic data and surgical history were obtained. Clinical outcomes following surgery were assessed, and any reoperations were recorded. Neck Disability Index (NDI), modified Japanese Orthopaedic Association (mJOA), Nurick classification (NuC), and Short-Form 36 (SF36) scores were recorded at baseline and final follow-up at certain centers. All data were collected, collated, and analyzed by a private research organization.
Results:
There were 109 cases of cervical dural tears among 18 463 surgeries performed. In 101 cases (93%) there was no clinical sequelae following successful dural tear repair. There were statistical improvements (P < .05) in mJOA and NuC scores, but not NDI or SF36 scores. No specific baseline or operative factors were found to be associated with the occurrence of dural tears. In most cases, no further postoperative treatments of the dural tear were required, while there were 13 patients (12%) that required subsequent treatment of cerebrospinal fluid drainage. Analysis of those requiring further treatments did not identify an optimum treatment strategy for cervical dural tears.
Conclusions:
In this multicenter study, we report our findings on the largest reported series (n = 109) of cervical dural tears. In a vast majority of cases, no subsequent interventions were required and no clinical sequelae were observed.
Introduction
Unintentional dural tears complicate cervical spine surgeries in 0.5% to 3.7% of cases. 1 -9 There have been 2 single-center studies that reported on the outcomes following the unintentional cervical dural tears, both finding no long-term sequelae when they are appropriately managed. 6,9 Despite reviewing thousands of patients in these studies, the overall number of patients with a dural tear was low. Because of the rarity of this complication, even experienced surgeons with large cervical spine practices may have limited experience with cervical dural tear management. Furthermore, severe complications from cervical dural tears are possible, including fistula formation, 10 meningitis, 1 or life-threatening airway compromise. 11 Therefore, in order to understand the presentation, treatment, and outcome of this complication, a multicenter study was performed to pool collective experiences with cervical dural tears.
Methods
Patient Cohort
We conducted a retrospective multicenter case series study involving 21 high-volume surgical centers from the AOSpine North America Clinical Research Network, selected for their excellence in spine care and clinical research infrastructure and experience. Medical records for 17 625 patients who received cervical spine surgery (levels from C2 to C7) between January 1, 2005, and December 31, 2011, were reviewed to identify occurrence of 21 predefined treatment complications including dural tear. Trained research staff at each site abstracted the data from medical records, surgical charts, radiology imaging, narratives, and other source documents for the patients who experienced one or more of the complications from the list. Data was transcribed into study-specific paper case report forms. Copies of case report forms were transferred to the AOSpine North America Clinical Research Network Methodological Core for processing, cleaning, and data entry.
Descriptive statistics were provided for baseline patient characteristics. Past surgical history was determined, and revision surgery was defined as a surgery with an approach (anterior vs posterior) and surgical level previously utilized. Operative details were determined, including the specific intraoperative treatment of the dural tears. Clinical outcomes following surgery were assessed, and any reoperations to control cerebrospinal fluid (CSF) drainage were recorded. Other postoperative treatments of the dural tears, such as bedrest and lumbar subarachnoid drain placement, were additionally recorded. Failure of the primary treatment of a dural tear was defined as continued leakage of CSF requiring either reoperation or delayed subarachnoid lumbar drain placement (unplanned beyond postoperative day 1). 9 For those failing the initial treatment, any subsequent interventions were determined. Neck Disability Index (NDI), modified Japanese Orthopaedic Association (mJOA), Nurick classification (NuC), and Short-Form 36 Physical Component Score (SF36-PCS) and Short-Form 36 Mental Component Score (SF36-MCS) were recorded at baseline and final follow-up at certain centers. Scores were reported from 7 centers for NDI, 15 for NuC, 5 for mJOA, and 4 for SF36. All data were collected and collated by a private research organization.
Control Group
In order to assess for possible risk factors for the occurrence of cervical dural tears, a control group was identified from the CSM-NA and CSM-I studies 12,13 by meeting the following criteria: from North America, having the same surgery type as the comparison group, and not having a cervical dural tear. All cases in this control group were primary surgeries that were done in the treatment of cervical myelopathy.
Statistics
Statistical analyses were performed by the same independent group. The results were presented as mean (standard deviation) and number (%) for continuous and categorical variables, respectively. Significance (P value) was calculated using 2-sample t test for continuous variables and χ2 tests or Fisher exact tests for categorical variables, with statistical significance taken as P < .05. Statistical analyses were performed using SAS, version 9.4 (SAS, Cary, NC).
Results
Patients With Dural Tears
There were 109 cases of cervical dural tears identified among the 18 463 patients reviewed (0.6%). The median incidence rate among the 21 centers was 0.4%, with an average ± standard deviation of 0.9 ± 1.14%. There were 4 centers that reported no cases of dural tears. Patients had an average age of 57 years and 57% were male (Table 1). The most common diagnoses were myelopathy (64%) and radiculopathy (22%), and anterior approaches were more common (61%). An average of 3.8 levels were addressed during surgery, and the average length of stay was 6.5 days.
Patient Characteristics of Those With Dural Tears.

Abbreviations: BMI, body mass index; OPLL, ossification of the posterior longitudinal ligament.
aData presented as average ± standard deviation and n (% of total).
Intraoperative treatments of the dural tear included direct suture repair (n = 17, 16%), use of sealant or patch materials (n = 62, 57%), or a combination of suture repair with a sealant/patch (n = 16, 15%). No specific treatment was utilized or able to be determined in 14 cases (13%). Sealant/patch materials varied and included DuraGen (Integra LifeSciences Co, Plainsboro, NJ), DuraSeal (Covidien, Minneapolis, MN), Tisseal (Baxter Healthcare Co, Deerfield, IL), Surgicel (Ethicon, Somerville, NJ), Evicel (Ethicon, Somerville, NJ), Surgifoam (Ethicon, Somerville, NJ), Gelfoam (Pfizer Inc, New York, NY), and/or local muscle. Additionally, a lumbar drain was placed in 15 patients (14%), which was continued for an average of 5.0 ± 3.5 days (median = 4 days) at rates of 10 to 20 mL/h. Bedrest was prescribed in 35 cases (32%) and continued for an average of 2.7 ± 2.1 days (median = 2 days). The specific protocol for the bedrest period varied widely from the bed remaining flat to having the head of the bed at 60°. The most common protocol used was keeping the head of the bed at 30°.
A majority of patients required no further treatment following the initial surgery, while 13 patients (12%) required subsequent interventions to control CSF drainage. There were 6 cases that required a revision operative dural repair (6%), 5 cases that required delayed (beyond postoperative day 1) lumbar drain placement (5%), and 2 that had both a revision surgery and lumbar drain placement (2%). There were 10 patients (9%) that had revision operations for reasons other than recurrent CSF drainage, including postoperative hematoma (n = 2), infection (n = 1), revision decompression, and/or fusion (n = 7).
Outcomes
Following successful control of the CSF drainage, a majority of patients had no symptoms attributable to the occurrence of the dural tear (n = 102, 94%). There were 4 patients (4%) that experienced residual effects from the dural tear, and 3 patients (3%) whose outcome could not be ascertained. The possible residual effects attributable to dural tear occurrences were unable to be characterized.
A statistically significant improvement in myelopathy severity was noted postoperatively, as determined by mJOA and NuC scores (Table 2). No significant difference was noted between baseline and follow-up NDI, SF36-PCS, or SF36-MCS scores.
Patient-Reported Outcome Scores in Patients With Dural Tearsa.
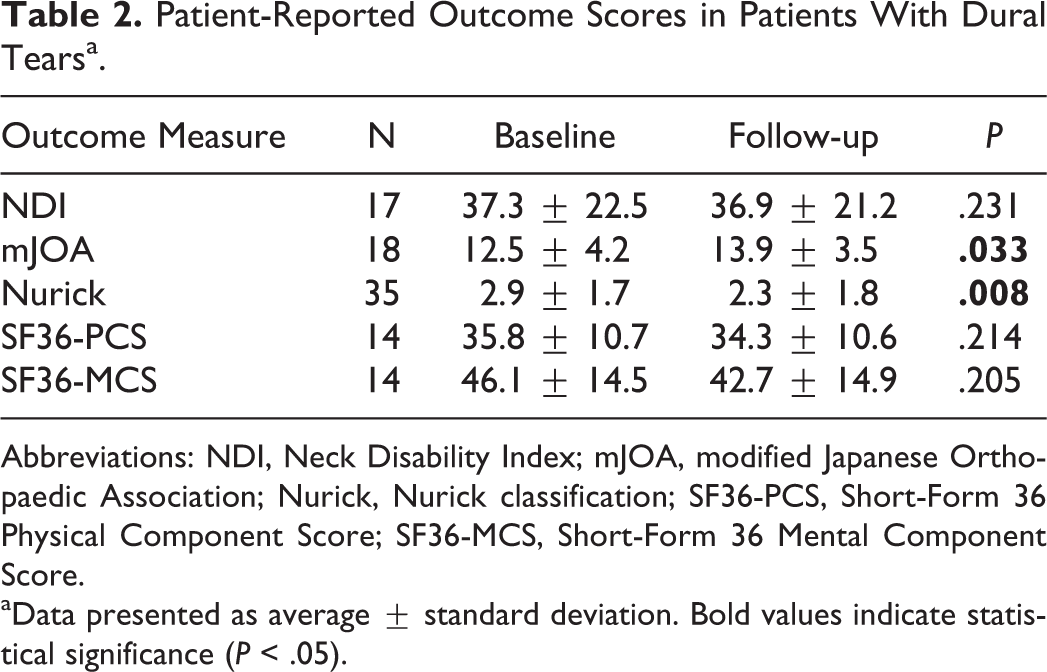
Abbreviations: NDI, Neck Disability Index; mJOA, modified Japanese Orthopaedic Association; Nurick, Nurick classification; SF36-PCS, Short-Form 36 Physical Component Score; SF36-MCS, Short-Form 36 Mental Component Score.
aData presented as average ± standard deviation. Bold values indicate statistical significance (P < .05).
Risk Factor Analyses
No significant differences were identified between patients found having sustained a dural tear and the historical control group (Table 3). As a result, no potential risk factors for the occurrence of dural tears were identified.
Comparison Between Patients With Dural Tears and Control Groupa.
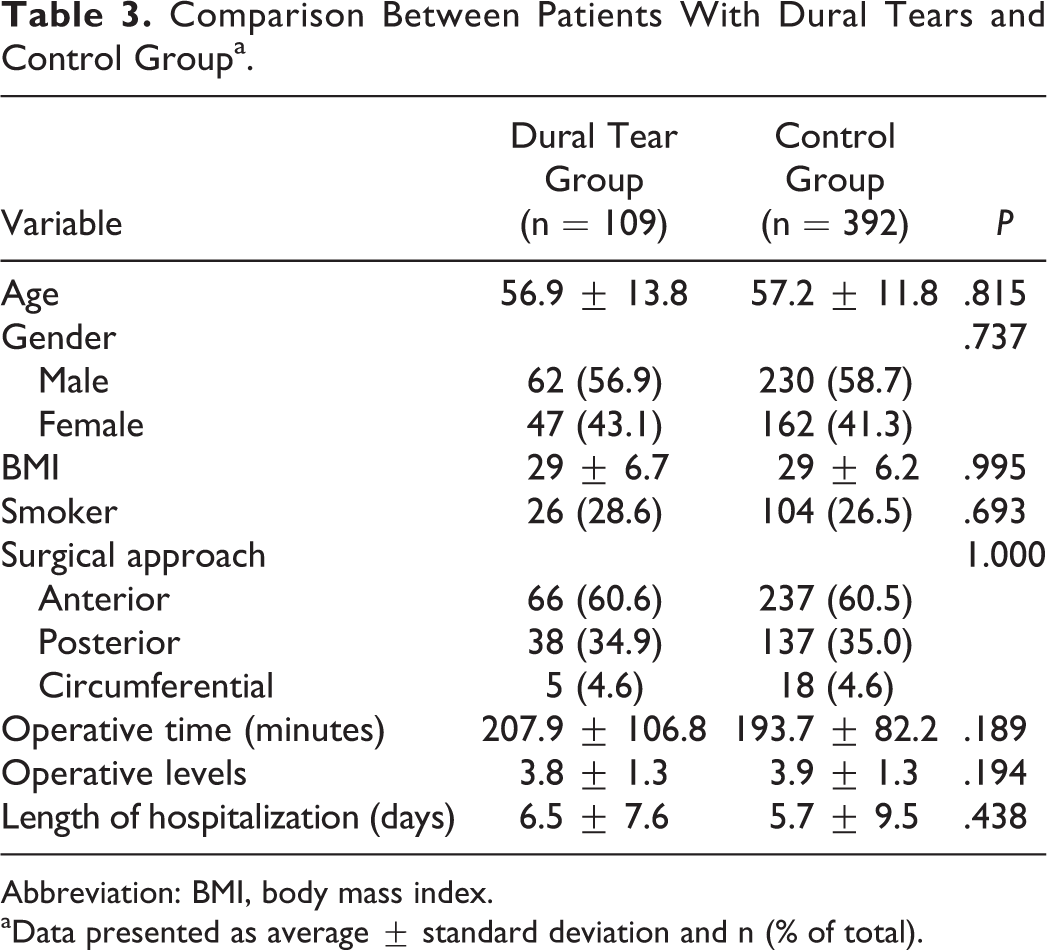
Abbreviation: BMI, body mass index.
aData presented as average ± standard deviation and n (% of total).
Male gender was found to be the only significantly different factor between those who had successful primary treatment of the dural tear with those that required subsequent surgery or later lumbar drain placement (Table 4). The group requiring subsequent treatment of CSF drainage also had significantly longer hospital stays.
Comparison Between Patients With Dural Tears and Control Group.
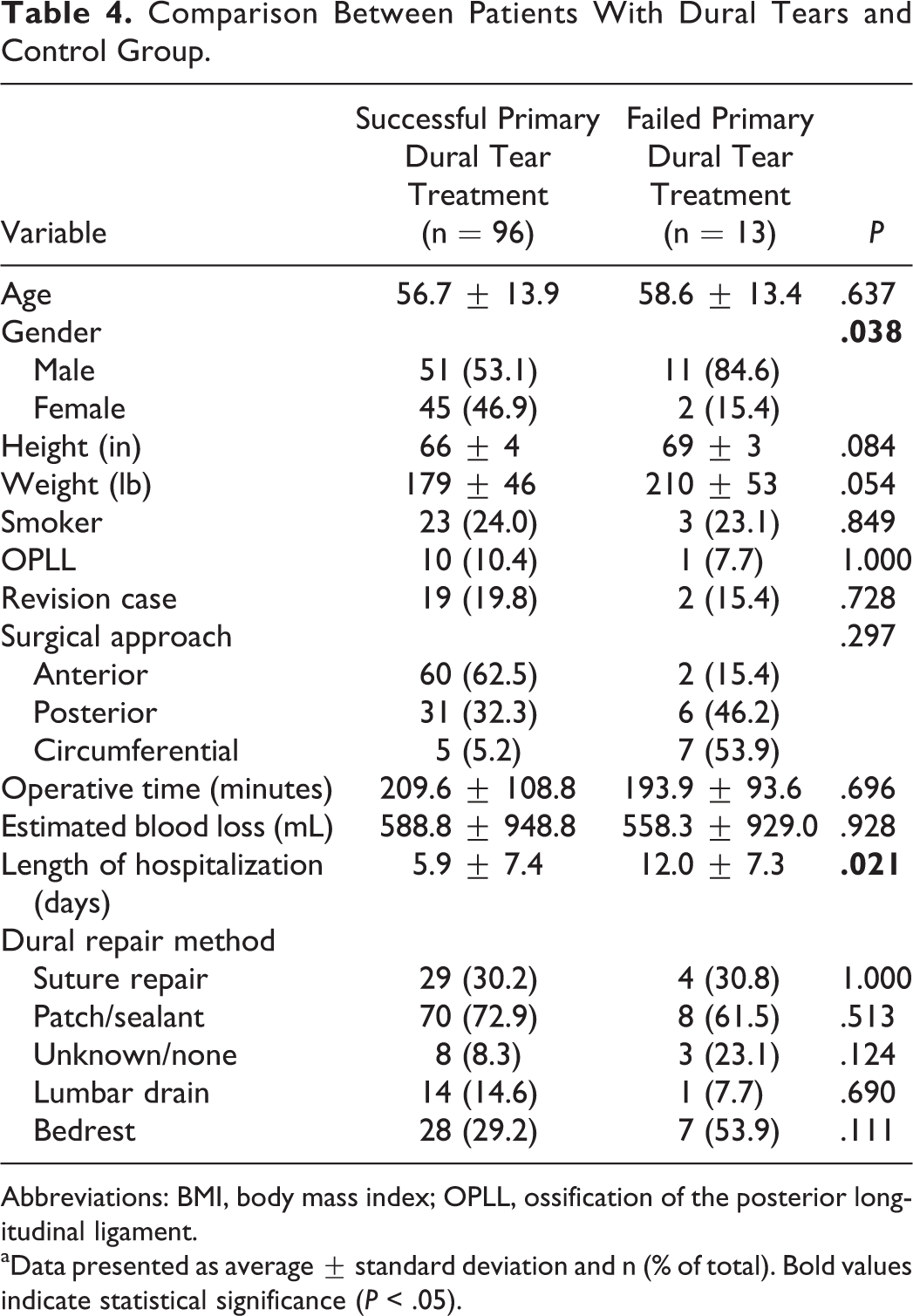
Abbreviations: BMI, body mass index; OPLL, ossification of the posterior longitudinal ligament.
aData presented as average ± standard deviation and n (% of total). Bold values indicate statistical significance (P < .05).
Discussion
The occurrence of an unintentional dural tear during cervical spine surgery is a rare complication. In this multicenter study, we report our findings on the largest reported series (n = 109) of cervical dural tears. In most cases no subsequent interventions to control CSF drainage were required, while 12% required further treatments following the index surgery. In a majority of cases (93%), there were no clinical sequelae directly attributable to the occurrence of a dural tear.
There have been 2 previous studies investigating the outcomes following cervical dural tears. Hannallah et al performed a retrospective review of 1994 cervical spine surgery patients at a single institution. 6 They found dural tears to have occurred in 20 patients (1%), and they identified risk factors for cervical dural tears to be OPLL (ossification of the posterior longitudinal ligament), revision anterior surgery, male gender, and performance of a corpectomy. Age and location of approach (anterior vs posterior) were not found to be significant factors. Though no patient-reported outcomes were reported, the study found no adverse events attributable to the occurrence of a dural tear. Similarly, our study did not find age or location of approach to be associated with dural tears, and a majority of patients had no clinical sequelae from its occurrence. However, we did not find that gender was associated with the occurrence of dural tears.
In the other study investigating cervical dural tears, O’Neill et al 9 performed a single institutional review of 3848 operations. In agreement with Hannallah et al, the incidence of dural tear was found to be 1%. Significant risk factors for the occurrence of a dural tear were found to be (1) older age, (2) rheumatoid arthritis, (3) OPLL, (4) cervical deformity, (5) longer operative time, (6) greater number of surgical levels, (7) worse neurologic status, (8) performance of a corpectomy, and (9) revision laminectomy. Interestingly, our results did not find an association between dural tears and age, operative time, or the number of surgical levels.
We found that 12% of patients underwent further treatment of persistent CSF drainage, and no patients required hospital readmission. Similarly, Hannallah et al found that 10% had issues related to CSF drainage beyond the index surgery, with 5% requiring a reoperation. 6 Suture repair and/or patches/sealants were utilized in only 25% of these cases. In contrast, O’Neill et al found 32% of patients required additional treatment of CSF leakage following the index surgery, with 16% undergoing a reoperation and 13% requiring hospital readmission—despite 97% using suture repair and/or patches/sealants. It was postulated that explanations for the apparent improved outcomes by Hannallah et al were more common use of a lumbar drain (20% vs 8%) and use of bedrest on all patients at least for one night. However, in our study, we found no association between the successful primary treatment of dural tears and the use of bedrest, lumbar drains, or the specific type of repair utilized. As a result, we are unable to recommend an optimal treatment strategy following the occurrence of a cervical dural tear.
There were several limitations to our study. First, the retrospective nature of this study may affect overall accuracy. Second, patient-reported outcome scores were available only in a limited number of patients, which limits the power of our study to detect true differences in outcome. However, though not ideal, this is the first available patient-reported outcomes data on patients with cervical dural tears. Finally, the use of a historical control from a different study setting likely diminished the accuracy of our risk factor analysis for the occurrence of dural tears, and precluded us from analyzing certain risk factors that previous studies have identified as significant—such as presence of OPLL and/or performing revision posterior laminectomy.
This study is the largest reported series investigating cervical dural tears. In most cases no subsequent interventions to control CSF drainage were required, while 12% required subsequent treatments. In a majority of cases (93%), there was no clinical sequelae directly attributable to the occurrence of a dural tear. Our analysis of treatment methods utilized following dural tears did not find an optimum treatment strategy.
Footnotes
Authors’ Note
This study was ethically approved by the institutional ethics committees at all participating sites.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kevin R. O'Neill reports grants from AOSpine North America during the conduct of the study; Michael G. Fehlings reports grants from AOSpine North America during the conduct of the study; Thomas E. Mroz reports other from AOSpine, grants from AOSpine North America, during the conduct of the study, personal fees from Stryker, personal fees from Ceramtec, other from Pearl Diver, outside the submitted work; Zachary A. Smith reports grants from AOSpine North America during the conduct of the study; Wellington K. Hsu reports grants from AOSpine North America during the conduct of the study, personal fees from Medtronic, personal fees from Stryker, personal fees from Bacterin, personal fees from Graftys, personal fees from Ceramtec, personal fees from Relievant, personal fees from Bioventus, personal fees from Globus, personal fees from SpineSmith, outside the submitted work; Michael P. Steinmetz reports grants from AOSpine North America during the conduct of the study; Praveen V. Mummaneni reports grants from AOSpine North America during the conduct of the study, other from Depuy Spine, grants and other from AOSpine, other from Globus, other from Springer Publishers, other from Thieme Publishers, other from Taylor and Francis Publishers, other from Spincity/ISD, outside the submitted work; Dean Chou reports grants from AOSpine North America during the conduct of the study, other from Globus, other from Orthofix, other from Medtronic, outside the submitted work; Ahmad Nassr reports grants from AOSpine North America during the conduct of the study; Sheeraz A. Qureshi reports grants from AOSpine North America during the conduct of the study, and is a consultant and receive royalties from Stryker Spine, Biomet Spine, and RTI; Evan O. Baird reports grants from AOSpine North America during the conduct of the study; Justin S. Smith reports grants from AOSpine North America during the conduct of the study, personal fees from Biomet, personal fees from Nuvasive, personal fees from Cerapedics, personal fees from K2M, personal fees from Globus, personal fees from DePuy, outside the submitted work; Christopher Shaffrey reports grants from AOSpine North America during the conduct of the study, personal fees from Biomet, personal fees from Medtronic, from Nuvasive, personal fees from K2M, personal fees from Stryker, outside the submitted work, and Editorial Board Spine, Spinal Deformity and Neurosurgery; Chadi A. Tannoury reports grants from AOSpine North America during the conduct of the study; Tony Tannoury reports grants from AOSpine North America during the conduct of the study; Ziya L. Gokaslan reports grants from AOSpine North America during the conduct of the study, grants from AOSpine North America, personal fees from AO Foundation, grants from AOSpine, outside the submitted work; Jeffrey L. Gum reports grants from AOSpine North America during the conduct of the study, personal fees from Medtronic, Alphatec, Stryker, LifeSpine, Acuity, Pacira, PAKmed, Gerson Lehrman Group, personal fees from OREF, AOSpine, personal fees from Acuity, other from Medtronic, personal fees from MiMedx, Pacira Pharmaceuticals, Alphatec, grants from Fischer Owen Fund, nonfinancial support from American Journal of Orthopaedics, nonfinancial support from American Journal of Orthopaedics, The Spine Journal, outside the submitted work; Robert A. Hart reports grants from AOSpine North America during the conduct of the study, other from CSRS Board, other from ISSLS, other from ISSG Executive Commitee, personal fees from DepuySynthes, personal fees from Globus, personal fees from Medtronic, other from Evans, Craven & Lackie, other from Benson, Bertoldo, Baker, & Carter, grants from Medtronic, grants from ISSGF, personal fees from Seaspine, personal fees from DepuySynthes, other from Spine Connect, personal fees from DepuySynthes, outside the submitted work; Robert E. Isaacs reports grants from AOSpine North America during the conduct of the study, grants and personal fees from NuVasive, Inc., personal fees from Association for Collaborative Spine research, outside the submitted work; David B. Bumpass reports grants from AOSpine North America during the conduct of the study, grants from North American Spine Society, personal fees from Doximity, personal fees from Gerson Lehrman Group, outside the submitted work; Adeeb Derakhshan reports grants from AOSpine North America during the conduct of the study; Eric M. Massicotte reports grants from AOSpine North America during the conduct of the study, grants from Medtronic, Depuy-Synthes Spine Canada, personal fees from Watermark Consulting, grants from AOSpine North America, nonfinancial support from AOSpine North America, outside the submitted work; Jonathan R. Pace reports grants from AOSpine North America during the conduct of the study; Gabriel A. Smith reports grants from AOSpine North America during the conduct of the study; Khoi D. Than reports grants from AOSpine North America during the conduct of the study; K. Daniel Riew reports personal fees from AOSpine International, other from Global Spine Journal, other from Spine Journal, other from Neurosurgery, personal fees from Multiple Entities for defense, plantiff, grants from AOSpine, grants from Cerapedics, grants from Medtronic, personal fees from AOSpine, personal fees from NASS, personal fees from Biomet, personal fees from Medtronic, non-financial support from Broadwater, outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by AOSpine North America Inc, a 501(c)3 nonprofit corporation.
