Abstract
Study Design
Narrative Literature Review.
Objective
To provide a comprehensive literature review of neurologic complications in Adult Spinal Deformity (ASD) surgery in the pre-operative, peri-operative and post-operative periods.
Methods
A broad review of the literature was conducted using the multiple databases including Pubmed, Embase, Scopus and the Cochrane library. Individual studies of relevance were appraised and included at the discretion of the authors on the basis of pertinence, impact on practice and scientific merit.
Results
The evidence regarding epidemiology, classification of complications, pre-operative evaluation of patients, peri-operative strategies to mitigate risk, intra-operative management of neuromonitoring changes and post-operative management of neurologic injury was critically appraised. Patients with the highest risk of neurologic complication include those with pre-surgery neuroaxis abnormality, high Deformity Angular Ratio, 3 column osteotomies and increased blood loss. Accurate and timely identification of intraoperative neuromonitoring changes is critical to ensuring appropriate response depending on whether changes are perfusion based (maintain adequate MAP and Hb, reverse corrective maneuvers) or traumatic (decompression of neural elements, assessment of instrumentation, reversal of corrective maneuvers, steroids). Surgical checklists can help surgeons navigate these stressful events to ensure appropriate steps and interventions are taken.
Conclusion
Neurological injuries occurring during the course of ASD surgery are potentially devastating complication, with regards to both patient morbidity and economic impact. Pre-operative identification of high risk patients, perioperative strategies to improve safety, timely recognition and management of intra-operative neuromonitoring changes and post-operative supportive measures can potentially reduce the incidence and significance of neurological injuries.
Keywords
Introduction
Surgical treatment for adult spinal deformity (ASD) is associated with high rates of complications, including new neurological deficits. There is considerable variability in the reported rates of new neurological deficits due to several factors, including the lack of a commonly accepted method for assessment of neurological deficits, heterogeneous patient populations and procedures, and data collection methods. Therefore, the incidence and associated risk factors of these complications remain unclear. While retrospective studies have documented low incidences of intra-operative neurological injuries, prospective databases and studies have reported rates of new deficits of over 20%.1-6 Smith and colleagues reported an overall new neurological deficit rate of 27.8% based on the International Spine Study Group (ISSG) database with minimum 2-year follow-up. 7 82 patients with a three-column osteotomy reported an overall new neurological deficit rate of 29.3%, including radiculopathy (9.8%), motor deficit (9.8%), nerve root injury (3.7%), and sensory deficit (2.4%). 8 Lenke and colleagues reported the results of the Scoli-RISK-1 prospectively documenting lower extremity motor score (LEMS) in complex ASD surgeries. 9 In 271 patients from 15 centers, a rate of 23% LEMS decline at the time of hospital discharge was recorded, which decreased to 17.1% at 6 weeks, 9.9% at 6 months, and 10.0% at 2 years.10-12 Buchowski and colleagues reported on new neurological deficits based on a single-institution experience of 108 lumbar pedicle subtraction osteotomies. 1 New deficits, defined as motor loss of 2 grades or more or loss of bowel/bladder control, occurred in 12 patients (11.1%) but were permanent in only 3 patients (2.8%). Nine of these patients underwent additional surgical intervention that included central canal enlargement and further decompression.
Clinical trials for ASD surgery are typically focused on quality of life, functional, and radiographic outcome measures, whereas neurologic outcomes are less intensely studied.13,14 Furthermore, the body of literature in ASD surgery, and spine surgery in general, lacks consistent methods in the assessment and classification of neurologic complications. Many studies utilize vague terminology when reporting neurologic complications, such as “motor deficit” or “sensory deficit” that does not specify the involved structure (e.g. spinal cord vs nerve root) or severity of neurologic injury. Standardized assessment and reporting of complications are essential to accurately estimate the risk of spinal procedures. 15
Neurologic complications after ASD surgery can have significant impact on a patient’s quality of life as well life expectancy and direct and indirect costs. 16 One study found increased cost of a neurologic deficit of $29,561, while another the total hospital charges for patients with a neurologic complication were more than 2-fold higher, while length of stay tripled.17,18 Indirect costs of neurologic complications are more difficult to estimate, but the Christopher & Dana Reeve Foundation estimates the cost of living with a motor incomplete spinal cord injury of $347,484 in the first year, and $42,206 for each subsequent year. 19
There are many controversies in the peri-operative and intra-operative management of high-risk neurological cases. Pre-operatively identifying high risk patients and high-risk procedures is important for the surgical planning and ensure appropriate measures are in place to reduce their occurrence and severity. The utility of intra-operative neuromonitoring (IONM), interpretation and direct protocols needed in response to different IONM changes are still not clear, leaving tremendous variability in the management of patients with potential real-time neurological injuries.
The purpose of this review is to highlight key issues in the neurological management of spinal deformity patients. We will provide an overview of the literature related to the current knowledge as well as identify gaps in knowledge that need to be addressed to minimize neurological risks in the treatment of these complex cases.
Methods
A broad review of the literature related to neurological issues related to spinal deformity surgery was conducted using the multiple databases including Pubmed, Embase, Scopus and the Cochrane library. Individual studies of relevance were appraised and included at the discretion of the authors on the basis of pertinence, impact on practice and scientific merit. While there is some variability in the literature, for the purpose of this review a neurological deficit was defined as a decrease in the lower extremity motor score (LEMS) of at least 1 point and/or a new sensory deficit.
Results and Discussion
The review yielded assessments of the overall incidence of neurological complications, classifications of neurological adverse events, the cost of neurological injury, identification of high-risk cases, peri-operative management of neurological deficits as well as the management and interpretation of intraoperative neuromonitoring.
Assessment and Classification of Neurologic Events
Neurologic function is complex and notably difficult to measure, and therefore neurologic complications present perhaps the greatest challenge in AE classification for spinal surgery. Arguably, the most important assessment is a standardized complete neurologic exam, which should test all relevant spinal cord and nerve root functions, including motor function (power, reflexes, bulk, tone, involuntary movements, and coordination), sensory function (light touch, vibration, proprioception, pin prick, and temperature), gait function, balance, and sphincter function (continence, bladder emptying). However, the neurologic exam is largely subjective, and complete assessment is often impractical in an inpatient hospital setting; as a result, most neurologic assessments in clinical practice may not fully represent the true neurological function of the patient.
Recently, several efforts have been made to address the knowledge gaps in the assessment and reporting of AEs in spine surgery, including neurologic complications.20,21 In 1 study, Mirza et al created definitions for 176 unique AEs and established methods for reporting their etiology, preventability, and severity, however did not include specific categories for neurologic injuries. 22 The Spine Adverse Events System (SAVES) provides a framework to classify AEs, with severity defined based upon on the need for intervention, increase in length of stay, or death. 23 Furthermore, SAVES provided a concrete definitions of neurologic injury as “new or increased neurologic deficit” and classified these as complete cord injury, incomplete cord injury, root motor injury, root sensory injury (including neuropathic pain), bladder dysfunction, and bowel dysfunction. An updated system, SAVES-V2, divides adverse events (AEs) into intraoperative and postoperative categories. 24 Among intraoperative AEs, the following neurologic complications are listed: cord injury and nerve root injury, while the following post-operative complications are listed: neurologic deterioration >= 1 motor grade in ASIA motor scale and postop neuropathic pain. Most recently, Klineberg et al reported on the ISSG-AO Multi-Domain Spinal Deformity Complication Classification System for Adult Spinal Deformity, which divides AEs into medical and surgical categories, and then evaluates them in terms of neurologic impact, timing, intervention, and resolution. 25 This system was by far the most comprehensive, including detailed collection of neurologic data (ISNCSCI motor scores, reflexes, sensory scores) to compare pre-operative and post-operative neurologic deficits, while also classifying injuries to many specific structures (e.g., recurrent laryngeal nerve, C5 palsy, femoral cutaneous neuralgia). However, it is unclear if this complex data collection is feasible to implement in clinical practice or even research studies, given the high resource requirements to collect these data.
Pre-operative Identification of High-Risk Patients
Pre-operative Identification and Recommendations for High Risk Patients.
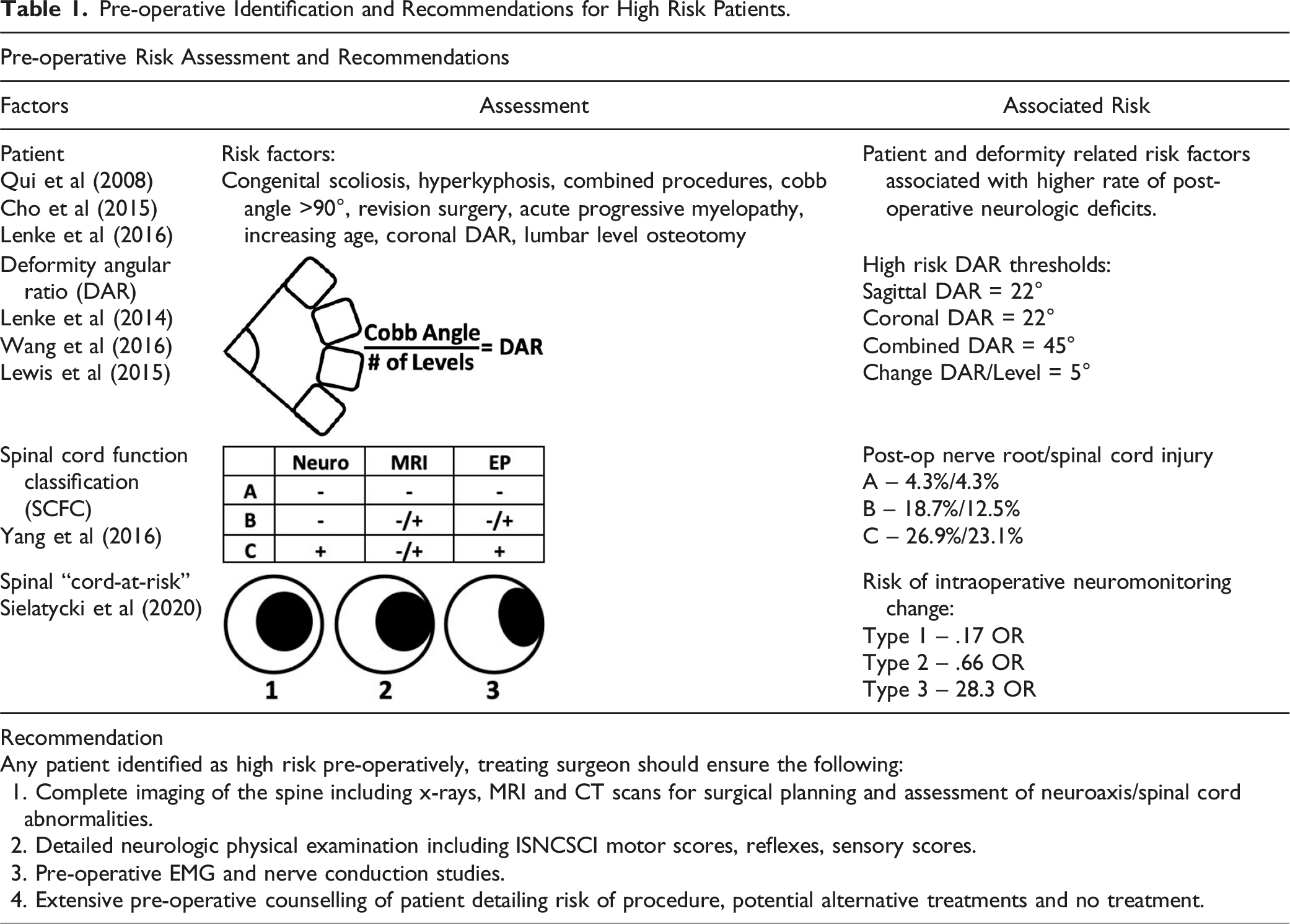
Patient characteristics which have been shown to increase neurologic risk are frequently associated with severe spinal deformity and increase potential for pre-existing neurologic deficits and neuroaxis abnormalities. In a single institution study of 1373 patients, Qui et al identified congenital scoliosis, hyperkyphosis, combined procedures, Cobb’s angle >90°, and a revision surgery as risk factors for neurologic deficits. 4 Cho et al (2015) found that patients with angular kyphosis, acute progressive myelopathy and unobtainable SCM during osteotomy to have higher rates of postoperative transient spinal cord deficits. 26 SCOLI-Risk identified age, coronal DAR and lumbar level osteotomy key factors associated with post-operative neurologic decline, however further retrospective analysis of the same cohort failed to implicate 3CO’s as a factor associated with neurologic decline.27,28
The Deformity Angular Ratio (DAR) as first described by Lenke et al is the maximum cobb angle of a curve divided by the number of levels over which the curve is distributed. 29 It is a unitless measure and can be calculated for both the coronal and sagittal planes as well as summed for a total DAR. Multiple studies have shown that increasing DAR is associated with increased risk of changes to spinal cord monitoring (SCM) at time of surgery and increased risk of neurologic deficit post-surgery. Lenke et al found DAR SCM alert was 13.48°/level (±6.78) and 16.27°/level (±8.81) for coronal and sagittal deformities respectively. For every 5°/level increase, SCM alert rate during surgery increased 39.6% (Odds ratio 1.396) for coronal deformity and 46.8% (Odds ratio 1.468) for sagittal deformity. 30 Lewis et al categorized high risk pediatric patients as those with sagittal DAR scores above 22° per level and a total DAR of 45° per level and demonstrated a positive linear relationship between increasing DAR and the rate of intraoperative SCM alert. 31
In 2016, Yang et al proposed the Spinal Cord Function Classification (SCFC) for stratifying patients undergoing posterior vertebral column resection for severe spinal deformity by assessing pre-operative neurologic status, presence of spinal cord abnormality on MRI and evoked potentials. 32 Their study showed that Types B and C patients had significantly higher rates of post-operative nerve injury [4.3 % (2/47), 18.7 % (3/16), and 26.9 % (7/26); Group A, B, and C, respectively, P = .02] and postoperative cord injury [4.3 % (2/47), 12.5 % (2/16), and 23.1 % (6/26); Group A, B, and C, respectively, P = .05].
A more recent study by Li et al (2018) combined the Spinal Cord Function Classification and Deformity Angular Ratio which supported both their utility in predicting neurologic complications. 33 In Type C patients with DAR >31, new neurologic deficits were present in 100% (4/4) patients. The study also found that Type A patients usually experienced better recovery. The rates of monitoring events and new neurologic deficits increased exponentially with an increase in S-DAR angle in those with type B and C but not type A.
A new classification to identify “cord-at-risk” patients uses MRI to assess spinal cord morphology at the apical vertebrae was published by Sielatycki et al. 34 Spinal cords were classified as types 1, 2 or 3 depending upon its relationship to the concave pedicle. In type 1, the spinal cord was non-deformed with circumferential CSF, whereas type 2 was a non-deformed spinal cord abutting the pedicle with no visible CSF. Type 3 spinal cords were deformed, or tethered, over the apical concave pedicle, and in their study, had an Odds Ratio of 28.33 of intra-operative loss of neuromonitoring data.
The use of 3 column osteotomies has been shown to increase the risk of neurologic injury in multiple studies. This increased risk can be attributed to not only to the risk of direct trauma to the neural elements (accidental, during osteotomy or correction/shortening), but also to the increased risk of blood loss and severity of deformity. Neurologic complications can be divided into 3 groups; those with positive spinal cord monitoring (SCM) alert, transient post-operative neurologic deficit and permanent neurologic deficit. SCM alert positive cases have been reported with an incidence of up to 60%. 31 Transient post-operative neurologic deficits have been reported to occur up to 13.8%. 2 Permanent post-operative deficits following 3 column osteotomy range from 0%-7.7%.2,3,35-44
Preventative Perioperative Strategies to Mitigate Neurologic Risk
Non-surgical
Perioperative Strategies and Recommendations to Reduce Risk of Neurologic Complications.

Patient positioning is an important aspect of ASD surgery given the length of surgery as well as the potential risks and benefits from positioning. A rare and devasting complication of surgery is a spinal cord injury as a result of over extension of the cord level spine in the setting of regional degenerative change. Preoperative assessment of myelopathy as well as radiographic images of the cervical spine can help identify patients at risk.45,46 Peripheral nerve injuries can occur if care is not taken to ensure nerves are tension free and without compression. Positioning of the shoulders should be no more than 90/90 of abduction/external rotation to reduce tension on the brachial plexus and the elbows and hip bolsters should be well padded to protect the ulnar and lateral femoral cutaneous nerves. Lastly, thoracic compression may increase intrathoracic pressure leading to higher airway pressures and lower cardiac output and the abdomen should be free to ensure low venous pressures promoting blood return to the heart to minimize bleeding. Increased intrathoracic or intraabdominal pressures in the setting of obesity or malpositioning can have a deleterious effect on circulating blood volume, subsequently impacting spinal cord perfusion and increasing the risk of neurologic changes. 47
Choice of aneasthetic agents impacts the ability to use intra-operative neuromonitoring (IONM). Total intravenous aneasthesia (TIVA) with propofol is the aneasthetic agent of choice. Avoidance of halogenated aneasthetics or neuromuscular blockade agents is important for reliable IONM. Hypotensive aneasthesia has been used to minimize blood loss in deformity surgery, however risk of hypoperfusion of the spinal cord and neurologic changes as a result is increased. If hypotensive aneasthesia is a strategy employed by the surgical team, understanding the potential for neurological complications and the need for accurate IONM is necessary. Hemodynamic monitoring is usually performed with an arterial line, which is mandatory in long (>4 hours) surgeries or in cases with anticipated higher blood loss. Blood pressure goals are usually within 10%-20% of baseline. Hypotension may particularly arise during or after the induction phase of anesthesia. This may cause worsening of spinal cord perfusion limiting autoregulation of the spinal cord. 45
Routine use of glucocorticoids in ASD surgery is not well studied. Administering around 20 milligrams (mg) (.3 mg/kilogram (kg)) of dexamethasone preoperatively, followed by 10 mg (.15 mg/kg) after eight and 16 hours, did not have an influence on intraoperative blood loss or delayed extubation in a prospective, randomized, double-blind study of 66 patients after multilevel cervical corpectomy surgeries. 48 Similarly, the use of systemic steroids does not have an influence on dysphagia after cervical spine deformity correction. 49
Surgical
Placement of freehand thoracolumbar pedicle screws carries an implicit risk of neurological injury, even in experienced hands. With respect to freehand screw malposition, rates in the literature vary, from as low as 10% to as high as 40%.50-56 Likewise, Amaral et al performed postoperative CT scans to evaluate 2229 pedicle screws placed by freehand technique in 106 ASD patients, and noted that 25% of their screws put the viscera at risk. 57 By contrast, a number of studies have shown dramatically higher rates of pedicle screw accuracy by CT-based navigation of pedicle screws.58-64
With regards to robotic-assisted pedicle screw placement, Devito et al noted in a retrospective review of 3271 robotic screws in 635 cases that screws assessed by intraoperative fluoroscopy were found to be clinically acceptable in 98% of cases, and on postop CT scans 98.3% of pedicle screws fell within the safe zone. 65 Lieberman et al demonstrated improved accuracy with use of robotics, as well as reduced operative time and radiation exposure. 66 Likewise, Perdomo-Pantoja et al demonstrated improved pedicle screw accuracy with both CT-based navigation and robotics over freehand placed pedicle screws. 67
There is an associated risk of neurological injury related to pedicle screw malposition, 68 although the rate of neurological injury varies across the literature. Faraj & Webb reported a rate of 1.09% of neurological complications due to malpositioned pedicle screws in their study of 648 cases. 69 Lonstein et al evaluated 4790 screws in 875 patients and noted 115 (2.4%) complications related to the pedicle screws. 70 In a meta-analysis, Gautschi et al found 32 of 5654 patients experienced neurological problems related to pedicle screws, and required revision; however, no comparisons were made between freehand and navigated screws. 71 In 1 study that evaluated thoracic pedicle screw placement by freehand technique in 208 deformity patients, there were no significant neurological injuries related to screw placement. 72 Within the body of ASD literature, however, it is unclear if the use CT-based intraoperative navigation or robotics reduces the risk of neurological injury related to malpositioned pedicle screws.
Although three-column osteotomies (3COs) are commonly employed for correction of fixed or rigid spinal deformities, these techniques have been shown to carry a risk of neurological injury.1,2,8,36,42,43,73-78 However, much of the literature concerning 3COs is plagued by methodological issues including retrospective design, small sample size and single surgeon/institution series. Smith et al performed a prospective multicenter study of ASD patients undergoing 3COs. Of 106 patients, 9.8% experienced postoperative radiculopathy, and another 9.8% experienced postoperative motor deficits. 8 In the Scoli-RISK-1 study, 21% of the included 273 patients experienced decline in lower extremity motor score after surgery, which declined to 12% by 6 months. 10 However, in a retrospective analysis of 207 patients from the Scoli-RISK-1 study, no statistically significant increase in rates of neurologic deficit were noted when comparing 3COs to posterior spinal fusions without 3COs, although overall higher complication rates were observed in the 3CO group. 28 Likewise, Kim et al studied 564 ASD patients, and noted 13.7% incidence of surgical neurological complications; however, the use of osteotomies, including the use of 3COs, did not increase the risk of neurological complications. 79
Intraoperative Interpretation of Neuromonitoring Changes
In the setting of cord level deformity surgery neuromonitoring includes somatosensory and motor evoked potentials (SSEPs/MEPs), and in the setting of surgery near the brachial or lumbosacral plexus 1 may also include electromyography (EMG). 80
MEPs measure directly from the corticospinal tracts, and indirectly from the spinothalamic tracts, which correlates with perfusion from the anterior spinal artery.81,82 Subdermal needles over the motor cortex stimulate this tract directly, and recordings are taken from various muscle groups distally. Loss of MEPs may lag behind the time of injury depending on when they were triggered last from the neurophysiologist. SSEPs reflect the function of the dorsal columns and are measured continuously with electrodes stimulating peripheral nerves distally, usually the tibial nerve at the ankle, and ulnar nerve at the wrist. 83 Recordings are taken from pre-specified locations on the skull. They too may produce a lag between the time of injury and changes in recording as SSEP recording requires signal averaging which may take several minutes to identify a true change. 84 SSEP/MEP loss of amplitude greater than 50% or latency >10% is generally considered a significant cut-off. 83 EMGs reflect the function of a particular nerve root and are interpreted within the setting of impedance. EMG is also used to interpret pedicle screw placement and the probability of breech. For pedicle screw stimulation, it is assumed that a screw within the bone will have higher impedance than a screw which is near the nerve root for example. Impedance thresholds are controversial but cut-off’s for acceptable pedicle screw placement in relation to the nerve range generally between 6-8.5 milliamperes (mA).85,86 The abdominal wall is innervated by the T6 to T12 nerve roots, and as such, pedicle screw stimulation proximal to T6 will not be accurate as they will not illicit a response in the abdominal muscles where the EMG recordings are made.
Bilateral loss of MEPs with intact SSEPs suggests an anterior cord syndrome, whereby usually a perfusion-based deficit leads to insufficient oxygenation to the anterior spinal cord through the anterior spinal artery. 81 The remainder of the neuromonitoring changes are generally traumatic. Unilateral loss of MEPs with or without unilateral loss of SSEPs generally reflect a Brown-Sequard injury pattern. 81 This commonly occurs during osteotomies of the posterior column. Osteotomies at the concave apex of coronal deformities are at particular risk for these changes. 87 Bilateral loss of MEPs and SSEPs are more likely to reflect a central-cord injury pattern and are frequently associated with the closure of pedicle subtraction osteotomies. 81 Loss of SSEPs alone suggest a dorsal column injury which may occur in the setting of laminar hooks, sublaminar wiring or trauma during decompression. 88 Isolated EMG findings generally reflect injury to a nerve root commonly occurring with inferomedial pedicle screw misplacement, over-shortening or insufficient foraminal decompression in a pedicle subtraction osteotomy. 81
The surgeon should consider timing of the change in neuromonitoring when considering the cause for the loss of MEP/SSEP. In the setting of coronal deformity for example, convex screw breeches may only become a symptomatic Brown-Sequard injury pattern with unilateral loss of MEPs/SSEPs during the deformity correction as the spinal cord shifts and impinges on the hardware within the canal.
Intraoperative Management of Neurologic Changes
Intraoperative neuromonitoring (IONM) changes are stressful events which demand a timely and coordinated response by the entire operating team. In these situations, safety checklists have been found to beneficial in helping team members communicate effectively and work through all critical steps. For IONM changes in deformity surgery, several checklists have been proposed which share many common items, the most recent an expert consensus checklist by Lenke et al (Figure 1).89-91 Intraoperative checklist for responding to neuromonitoring alerts.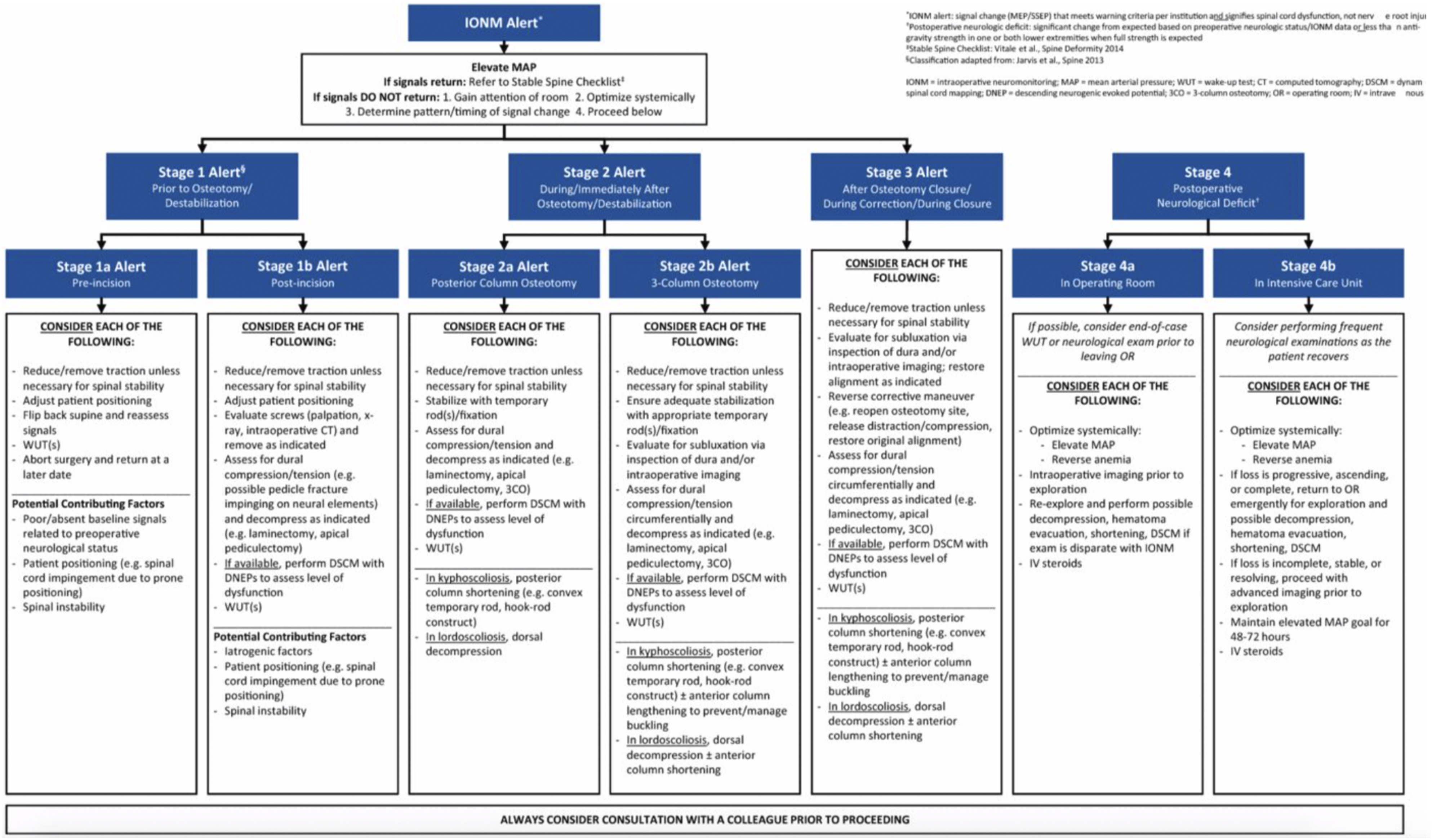
The first step in responding to an IONM change is to determine whether the changes are true or false positives. Common reasons for false positives include technical issues with equipment or changes in anesthetic technique (esp. addition of volatile anesthetic or neuromuscular blockade). Therefore, in the early stages of the response, anesthesia and neurophysiology teams must be involved and work together to verify a true IONM change is occurring.
In the event of a true IONM change, the next step is to optimize physiology to ensure sufficient spinal cord blood flow (SCBF). The primary determinant of SCBF is spinal cord perfusion pressure (SCPP) which is defined as the difference between MAP and intraspinal pressure (ISP). 92 Through autoregulation, SCBF in an uninjured spine is maintained at a constant rate when mean arterial blood pressure (MAP) is maintained between 60 to 120 mmHg. However, spinal cord injury (SCI) can lead to loss of autoregulation which means that MAP and ISP need to be directly controlled to drive SCBF. Although an active area of research in the traumatic SCI literature, decreasing ISP as a means to increasing SCPP for neuromonitoring changes in deformity surgery is not common practice. 92 Therefore, elevating MAP is the primary means of driving SCPP.
There is debate over MAP targets in acute and the American Association of Neurological Surgeons’ guideline of a MAP of 85-90 mmHg is still under investigation (MAPS trial, NCT02232165).93,94 In cervical or high thoracic regions SCI hypotension may result from loss of sympathetic tone resulting in both (i) peripheral vasodilation, and (ii) interruption of cardiac sympathetic innervation with unopposed vagal activity. 95 Consequently agents with both alpha- and beta-adrenergic actions such as dopamine or norepinephrine are recommended. However, norepinephrine has been found to be the least arrhythmogenic and is preferred. 96 For low thoracic SCI where vasodilation is the primary cause of hypotension, phenylephrine is used because beta-adrenergic support is not needed. 95 With SCBF addressed, anemia should be corrected, and consideration may be given to transfusion with low hemoglobin in the setting of bilateral MEP loss. 81 The oxygen carrying capacity of hemoglobin is influenced by temperature and acid-base status which should also be normalized.
Following confirmation of a real IONM alert and physiological optimization, the surgeon should diagnose the etiology of alert in order to select the proper corrective interventions. Jarvis et al provide a useful framework for diagnosing the cause of IONM alerts in three-column osteotomies by classifying them as chronologically as type I alert: prior to decompression, type II alert: occurring during decompression and bone resection, type III alert: occurring after osteotomy closure. 88
Type I alerts occur before neural elements are exposed. It is theorized that spinal lengthening can reduce cord perfusion from over stretching of the anterior spinal artery. 87 If used, skull-femoral traction weights should be removed as these forces cause spinal lengthening. If screws were placed at the time of the changes, screw placement should be carefully evaluated with intraoperative imaging, screw stimulation, or screw removal and repositioning.
Type II alerts occur when neural elements are exposed and at risk of injury. The decompression should be completed and widened so that the neural elements can be carefully inspected. In particular, the surgeon should evaluate for compressive pathology such as bone fragments, hematoma, or surgical material. During bone resection, the spine may become unstable and moving of the spinal canal can impinge on the neural elements; temporary rods should be placed to prevent these types of injuries.
Type III alerts occur after the spine has been manipulated. The osteotomy should be opened and neural elements examined as per the response to Type II alerts. The osteotomy site should be inspected to ensure adequate resection of bone elements. The surgeon must then re-consider the impact of the planned deformity correction on spinal cord function. Although spinal shortening is well tolerated and can promote spinal cord blood flow, over-shortening can lead to spinal cord and anterior spinal artery kinking. 97 For Type III alerts, the surgeon should consider reducing the amount of kyphosis correction and/or place a cage (or larger cage) in the osteotomy site to prevent over-shortening. 91
We highlight that if IONM changes occur before osteotomy closure for kyphosis (Type I and Type II alerts), it may be appropriate to proceed with deformity correction as this maneuver can increase spinal cord perfusion through shortening. 88
Pharmacologic interventions to minimize the secondary injury to the spinal cord may also be considered. Given that the timing of the SCI can be inferred from the time of a neuromonitoring change, steroids may be indicated. In subgroup analysis, the NASCIS 2 trial found benefit to methylprednisolone when given within 8 hours of the time of injury. 98 The approximately 5-point motor score benefit must be balanced against the harms which include sepsis and gastrointestinal hemorrhage. Riluzole, a treatment for Amyotrophic Lateral Sclerosis (ALS), which is under Phase 3 trials as a neuroprotective agent for traumatic SCI. 99 This medication has a good safety profile, but there is currently no level 1 evidence in the management of IONM changes.
Post-operative Management of Neurologic Injuries
Post-operative Recommendations for Management of Neurologic Complications.
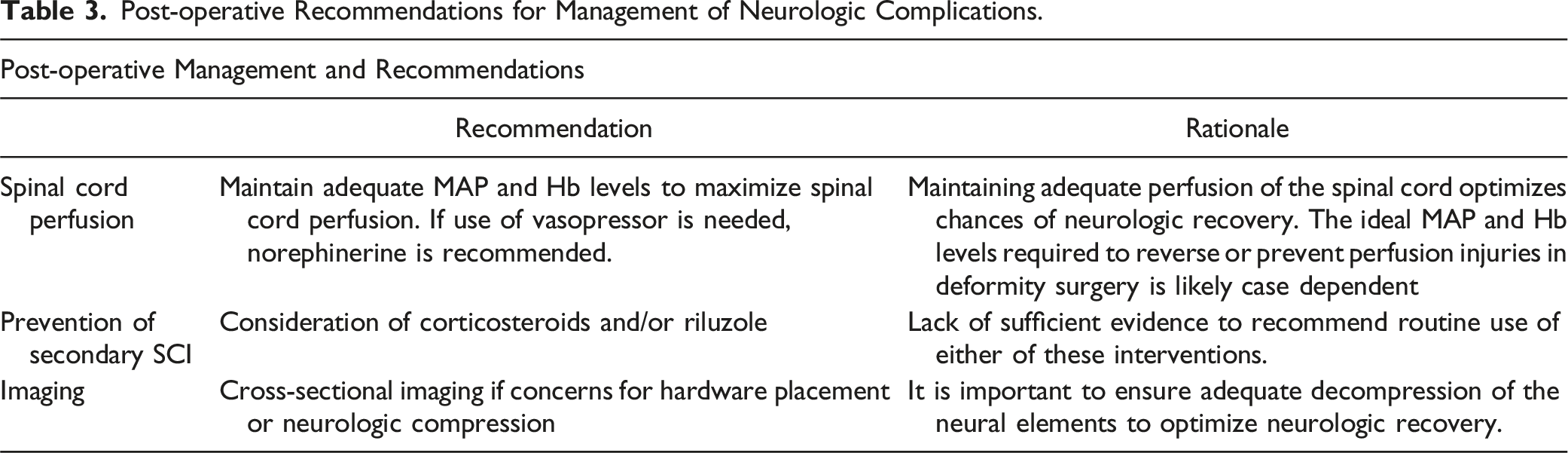
If the etiology of the IONM alert is unclear, the surgeon should consider appropriate cross-sectional imaging. A CT scan should be obtained to evaluate screw position, deformity correction, and bone compression. An MRI scan should be obtained if there is continued concern for compressive pathology after intraoperative exploration.
Maintenance of spinal cord perfusion is imperative in 2 aspects. First, spinal cord contouring may cause the stretch of spinal vasculature including anterior spinal artery resulting in spinal cord hypoperfusion. 100 Secondly, even in traumatic SCI models, studies have shown that local insult to microcirculation results in vasospasm and loss of autoregulation which lead to blood flow alteration in spinal cord. 101 This could be further exacerbated by systemic hypotension related to loss of sympathetic control as well as hypovolemia due to blood loss by trauma or surgical invasion. Thus, spinal cord perfusion is one of the key treatment targets to mitigate post-spinal cord injury neurological damage through MAP and hemoglobin targets.
To achieve the ideal spinal cord perfusion, routine practice in intensive care unit settings consists of volume expansion and vasopressor usage. Particularly in the post-operative status, extracellular fluid administration should be incorporated mainly by crystalloids, with the target systemic blood pressure being above 90 mmHg and mean arterial pressure at 85 to 90 mmHg. 102 These targets can be altered on case-by-case basis depending on the patients’ baseline blood pressure, but the idea is to maintain high-normal arterial blood pressure to secure prefusion of spinal vasculature. Common vasopressors used in traumatic SCI cases are those with both alpha and beta-adrenergic effects such as dopamine and norepinephrine. 103 Previous studies have shown that norepinephrine has less potential for cardiac side effects than dopamine. As in SCI models, use of phenylephrine, which has purely alpha-adrenergic effect, are limited to mid-to low-thoracic injury and should be avoided when high thoracic injury is suspected due to the potential risk of reflex bradycardia.
In cases with acute SCI, alleviation of other secondary injury mechanisms such as local inflammatory process and insult by chemical mediators are also crucial. Among a few key drugs are steroid and riluzole.104,105 The clinical benefits of these drugs have been long debated in traumatic SCI, but unfortunately its effectiveness in post-ASD surgery scenarios have never been proven. Further investigations with solid and robust study design are warranted in this population.
Conclusion
Reducing the rates and severity of neurologic complications during ASD surgery is an important consideration in improving patient safety in these complex procedures. Pre-operative identification of high-risk patients, perioperative strategies to improve safety, timely recognition and management of intra-operative neuromonitoring changes and post-operative supportive measures can potentially reduce the incidence and significance of neurological injuries. Future research should focus on a standardized instrument for the collection and classification of neurologic complications, a tool for the pre-operative stratification of neurologic risk, educating surgeons on appropriate interpretation of IONM events and strategies for both intra-operative and post-operative management of neurologic injury.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CJN receives support for attending meeting from AOSpine. JSS is a Consultant for Cerapedics, Carlsmed, SeaSpine, ZimVie, NuVasive, Medtronic, DePuy Synthes, has royalties with ZimVie, NuVasive, has stock with Alphatec, NuVasive and receives support for attending meeting from AOSpine. SJL is a Consultant for Stryker, Medtronic and AOSpine, and receives payment for lectures and travel for meetings from Medtronic, Stryker, AOSpine, L&K and Depuy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Deformity, a focused group of international Adult Spinal Deformity experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization. Support was provided directly through AO Network Clinical Research.
