Abstract
Study Design
Retrospective cohort study.
Objective
In general, Multiple Myeloma (MM) patients are treated with systemic therapy including chemotherapy. Radiation therapy can have an important supportive role in the palliative management of MM-related osteolytic lesions. Our study aims to investigate the degree of radiation-induced remineralization in MM patients to gain a better understanding of its potential impact on bone mineral density and, consequently, fracture prevention. Our primary outcome measure was percent change in bone mineral density measured in Hounsfield Units (Δ% HU) between pre- and post-radiation measurements, compared to non-targeted vertebrae.
Methods
We included 119 patients with MM who underwent radiotherapy of the spine between January 2010 and June 2021 and who had a CT scan of the spine at baseline and between 3-24 months after radiation. A linear mixed effect model tested any differences in remineralization rate per month (βdifference) between targeted and non-targeted vertebrae.
Results
Analyses of CT scans yielded 565 unique vertebrae (366 targeted and 199 non-targeted vertebrae). In both targeted and non-targeted vertebrae, there was an increase in bone density per month (βoverall = .04; P = .002) with the largest effect being between 9-18 months post-radiation. Radiation did not cause a greater increase in bone density per month compared to non-targeted vertebrae (βdifference = .67; P = .118).
Conclusion
Our results demonstrate that following radiation, bone density increased over time for both targeted and non-targeted vertebrae. However, no conclusive evidence was found that targeted vertebrae have a higher remineralization rate than non-targeted vertebrae in patients with MM.
Introduction
Multiple myeloma (MM) is the second most diagnosed hematologic malignancy and is rising in incidence in developed countries.1,2 It is a malignant disease characterized by monoclonal plasma cell proliferation in the bone marrow. 1 In 60%–90% of the MM cases, these malignant plasma cell proliferations cause lytic bone lesions and/or diffuse osteopenia due to increased activity of osteoclast and decreased osteoblastic activity.3,4 This leads to lytic lesions and therefore to a loss of structural integrity of the spine, increasing the risk of pathologic fractures and severe pain with a negative impact on quality of life.3,5
In general, MM patients are treated with systemic therapy including high-dose chemotherapy, in some cases followed by stem cell transplantation. 6 Although systemic MM treatment has improved greatly over the years with the emergence of novel therapies such as monoclonal antibodies and cellular therapies, the disease remains incurable. 7 Radiation therapy can also have an important supportive role in the palliative management of MM related osteolytic lesions. 4 It has been demonstrated that even low doses of radiation offer significant symptom relief for tumor deposits in the bone or soft tissue.4,8 However, radiation can result in myelosuppression and irreversible bone marrow fibrosis. This is detrimental, because a sufficient bone marrow reserve is necessary for eligibility for systemic therapy and future stem cell transplantation.9,10 Hence, the selection of patients for radiation therapy should be based on careful consideration of the potential risks and benefits.
Previous clinical trials have shown the potential of radiotherapy to induce radiation-associated remineralization in patients with solid tumor metastases (renal cell, breast, prostate, lung, urothelial cell, stomach, etc.), i.e. increasing bone mineral density (BMD) and thus (partially) restoring the structural integrity of the vertebrae.11-15 In patients with MM, however, the success rate was lower; one study measured the size of lytic lesions before and after radiation and found that 48% of lesions showed some remineralization. 16 Although, the studies including MM patients did not include quantification of the effect on BMD using DEXA or Hounsfield Unit measurements. Thus, in patients with MM, the impact of radiation on the trabecular and cortical bone surrounding the lytic lesions, as well as the potential for bone reforming within these lytic areas, remains largely unknown. MM often leads to widespread bone disease rather than isolated lesions, adding further complexity to the understanding of the effect of radiation. Therefore, our study aims to investigate the degree of radiation-associated remineralization in MM patients to gain a better understanding of its potential impact on BMD and, consequently, fracture prevention.
Methods
Study Design
This retrospective cohort study was conducted at two affiliated tertiary care hospitals in the United States. Institutional review board approval was obtained prior to initiation of this study (registration number 2018P000688).
Patient Selection
Baseline and Treatment Characteristics.

Values are presented as number (%) and mean [standard deviation] for normally distributed data or median [interquartile range] for non-normally distributed data.
Abbreviations: BMI, Body Mass Index; ISS, International Staging System; NRS, Numeric Rating Scale; ECOG, Eastern Cooperative Oncology Group; ASIA, American Spinal Cord Injury Association, VCF, Vertebral Compression Fracture; R, Revlimid; V, Velcade; d, Dexamethasone; D, Daratumumab, P, Pomalidomide; K, Carfilzomib; C, Cyclophosphamide; AP/PA, Anteroposterior-posteroanterior; 3D, three-dimensional; VMAT, Volumetric modulated arc therapy; IMRT, Intensity modified radiation therapy, SRS, Stereotactic radiosurgery; Gy, Gray.
Outcome Measures
Due to the difference in absolute HU values across vertebral levels and across different patients, our primary outcome measure was percent change in bone density (Δ% HU) between pre- and post-radiation measurements, compared to non-targeted vertebral levels that functioned as reference. The Δ% HU was calculated for each vertebral level using the following equation: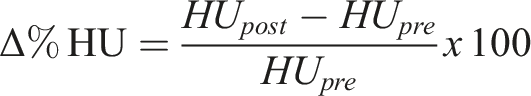
All outcome measurements were pooled into five consecutive follow-up intervals of 3-6 months, 6-9 months, 9-12 months, 12-18 months, and 18-24 months.
HU Measurements
The hospital eUnity PACS software system was used to access imaging and to measure the bone density values in HU. Mean HU score for each vertebra was calculated by placing a circular Region of Interest (ROI) on the mid-axial plane of CT-scans. The center of the ROI was placed on the estimated 3D centroid of the vertebral body, maximizing the diameter while still excluding the vertebral cortices to prevent volume averaging due to the normally high-attenuating cortex (See Appendix, Figure A2). Careful attention was taken to ensure that the ROIs drawn on the axial slices were in the mid-axial portion of the vertebral body by cross-checking on the sagittal slices. The baseline measurement was performed on CT scans obtained within 12 weeks prior to radiotherapy. The post-radiation measurements were done in the same way and as close to the original measured location and diameter as possible, on CT scans performed between 3-24 months after radiation. These measurements were only done if a comparable follow-up CT scan was available (e.g. CT Thorax without contrast agent compared with a CT Thorax without contrast agent). If multiple comparable CT scans were acquired within this timeframe, a maximum of three follow-up measurements were done, ensuring the measurements were at least three months apart. Vertebral levels C1, C2 and S2-S5 were excluded because of their different anatomy. For each patient, two reference measurements were performed on one cranial and one caudal vertebra outside the radiated area (if visible on CT-images) to detect changes in bone density of non-targeted vertebrae. These control measurements were taken from the nearest non-adjacent vertebra to allow the adjacent vertebra to serve as a buffer for some potential imprecision of radiation delivery and the scattered effect of radiation. In the presence of any implants (e.g. cement or metallic implants), the first non-adjacent implant-free vertebra was chosen.
Statistical Analysis
All HU measurements were performed by a single rater (JPV). In order to ensure validity of these measurements, a random sample of 20% of the entire patient cohort was also measured by a second rater (HZ). Subsequently, inter-rater reliability was assessed with an intraclass correlation coefficient (ICC) which was calculated using a single-measurement, absolute agreement, two-way random effects model. Measurement of Δ% HU was considered reliable, with an ICC of .96 (95% CI .94-.97; P < .001), indicating excellent reliability. 17
To compare HU values of radiated vertebrae to non-targeted vertebrae per timepoint, unpaired t-tests were used. In order to assess changes across time within the same vertebrae, paired t-tests were used to compare mean HU values from baseline to five consecutive follow-up intervals. To test if there is a difference in remineralization rate between radiated and control vertebrae, a linear mixed effect model using the restricted maximum likelihood estimator (REML) approach was fitted, since multiple vertebrae per patient were included, and these ‘clusters’ of vertebrae cannot be treated as uncorrelated measurements. In mixed models, a random subject and cluster effect can be added to account for the correlation of the data. 18 Fixed effects are variables that are constant across individuals whereas random effects vary. To estimate the effect of radiation on remineralization rate over time, variables radiation (yes/no) and time from radiation to HU measurement (in months) were fitted as fixed effects and together in an interaction term to compute the remineralization rate. Δ% HU was fitted as dependent variable. To account for multiple measurements (vertebrae) per patient, a random intercept was fitted per patient with vertebral level as a nested effect within patient. The effect of three covariates (chemotherapy, bisphosphonates, and radiation fractionation scheme) were evaluated because of their presumed effect on bone mineral density, by adding them as a fixed variable, and in an interaction term with time and a three-way interaction term with time and radiation.
A two-tailed P-value of less than .05 was considered statistically significant. All statistical analyses were performed using Python programming language, version 3.9.7 (Python Software Foundation, https://www.python.org/) and the MixedLM procedure in the Statsmodels package. 19
Results
Patient Cohort
The mean age of patients at the time of radiation was 64 years (standard deviation (SD) 11.7) and 66% were male (Table 1). ISS 1 was the most common disease stage (n = 47, 39.5%), most patients were capable of self-care (ECOG scores 0-2; n = 106, 89.8%) and fully neurologically intact (ASIA E, n = 96, 80.7%). Forty-two patients (36.2%) presented with at least one vertebral compression fracture (VCF) in the radiated area, of which 20 were classified as severe according to the Genant criteria. 20 A combination of Revlimid, Velcade and Dexamethasone (RVD) was the most common chemotherapy regimen, used in 56 patients (47.1%, Table 1). Sixty-nine patients (65.7%) had already started chemotherapy before initiation of radiotherapy. Bisphosphonates were administered in 78 patients (65.5%), most of which received them in regular intervals (n = 64, 62.1%). The median survival time was 33 months (95%CI 23-46).
Remineralization
Measurements at Baseline and Five Consecutive Timepoints for the targeted (n = 366) and Non-targeted (n = 199) Vertebrae.
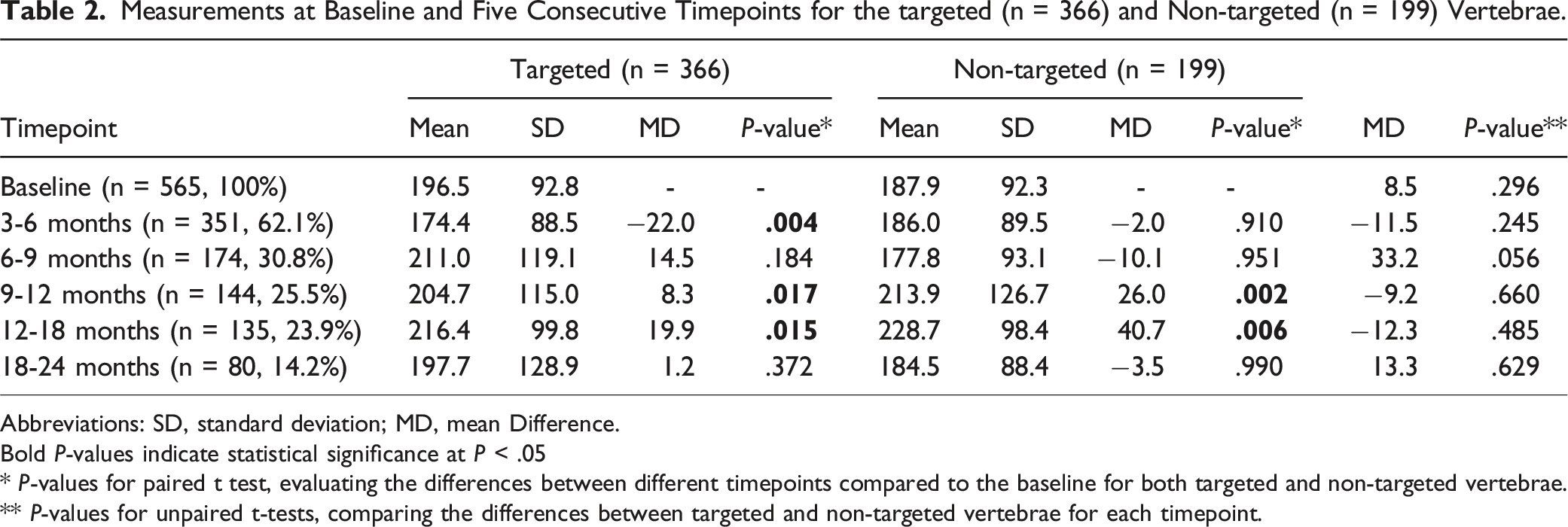
Abbreviations: SD, standard deviation; MD, mean Difference.
Bold P-values indicate statistical significance at P < .05
* P-values for paired t test, evaluating the differences between different timepoints compared to the baseline for both targeted and non-targeted vertebrae.
** P-values for unpaired t-tests, comparing the differences between targeted and non-targeted vertebrae for each timepoint.
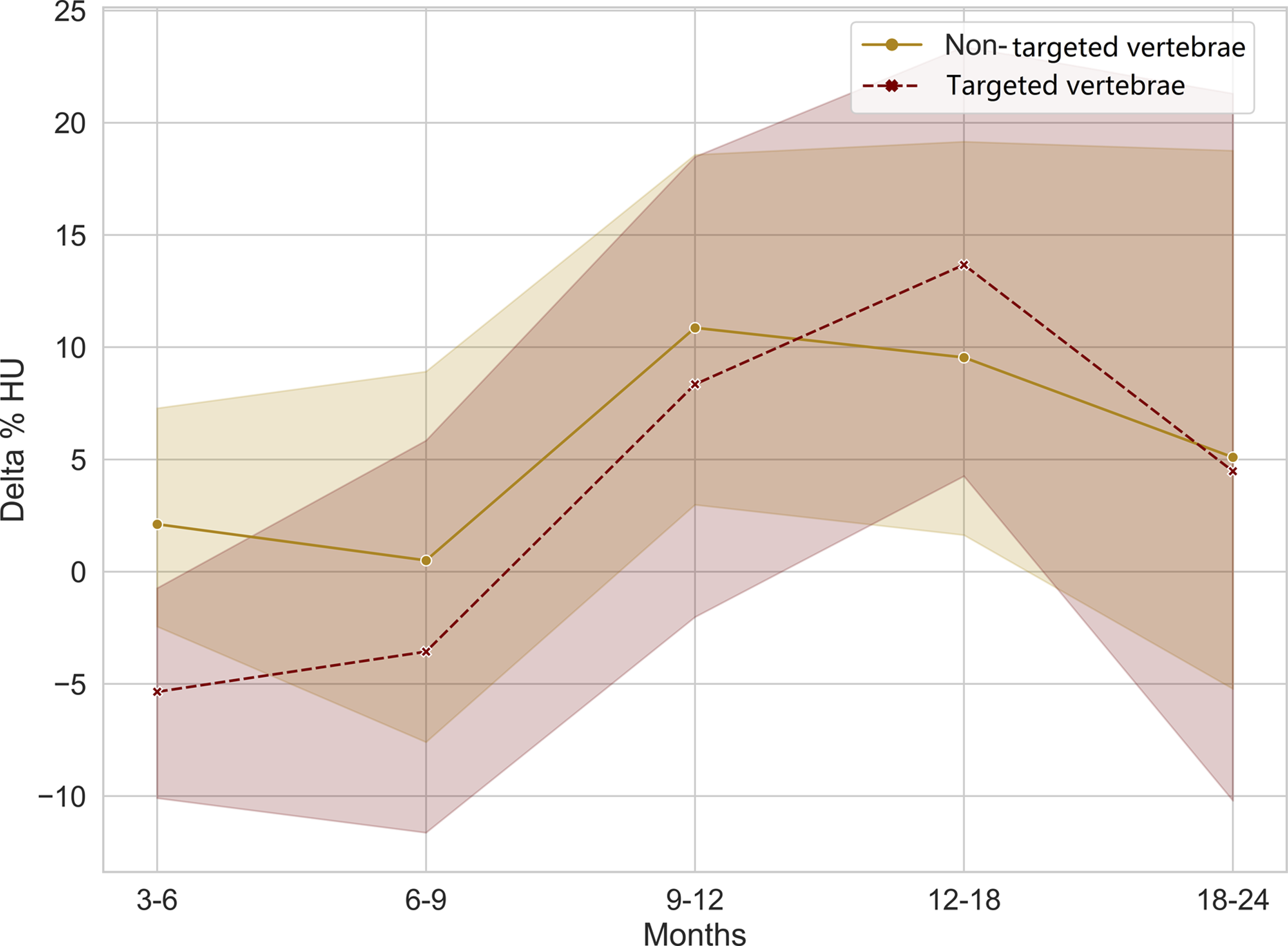
Change in bone density with 95% confidence intervals after radiation until 24 months.
Results of Multivariable (Adjusted) linear Mixed Models.
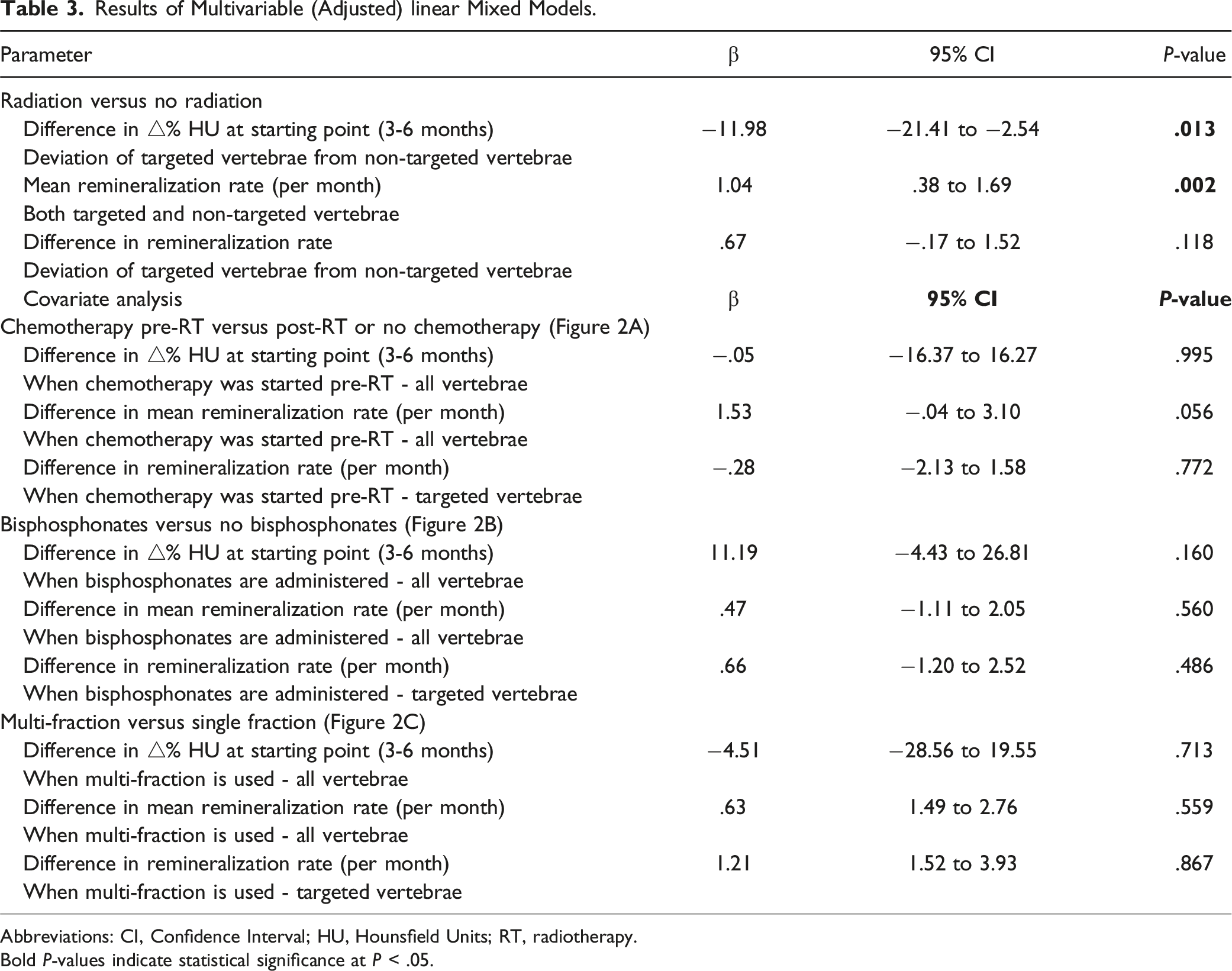
Abbreviations: CI, Confidence Interval; HU, Hounsfield Units; RT, radiotherapy.
Bold P-values indicate statistical significance at P < .05.
Covariates
When patients were started on chemotherapy before radiation compared to when patients received no chemotherapy or this was started after radiation, there was an increase in bone density around 9-12 months for both radiated and non-targeted vertebrae (Figure 2A). Receiving bisphosphonates also seemed to increase bone density in all vertebrae, regardless of radiation (Figure 2B). A multi-fraction radiation scheme resulted in a higher BMD around 9-18 months for targeted vertebrae, but it did not seem to affect BMD for non-targeted vertebrae (Figure 2C). When calculating remineralization slopes for these three covariates, there was no difference in the remineralization rate over time between radiated or non-targeted vertebrae or within radiated vertebrae (Table 3). Bone density over time, stratified for different covariates.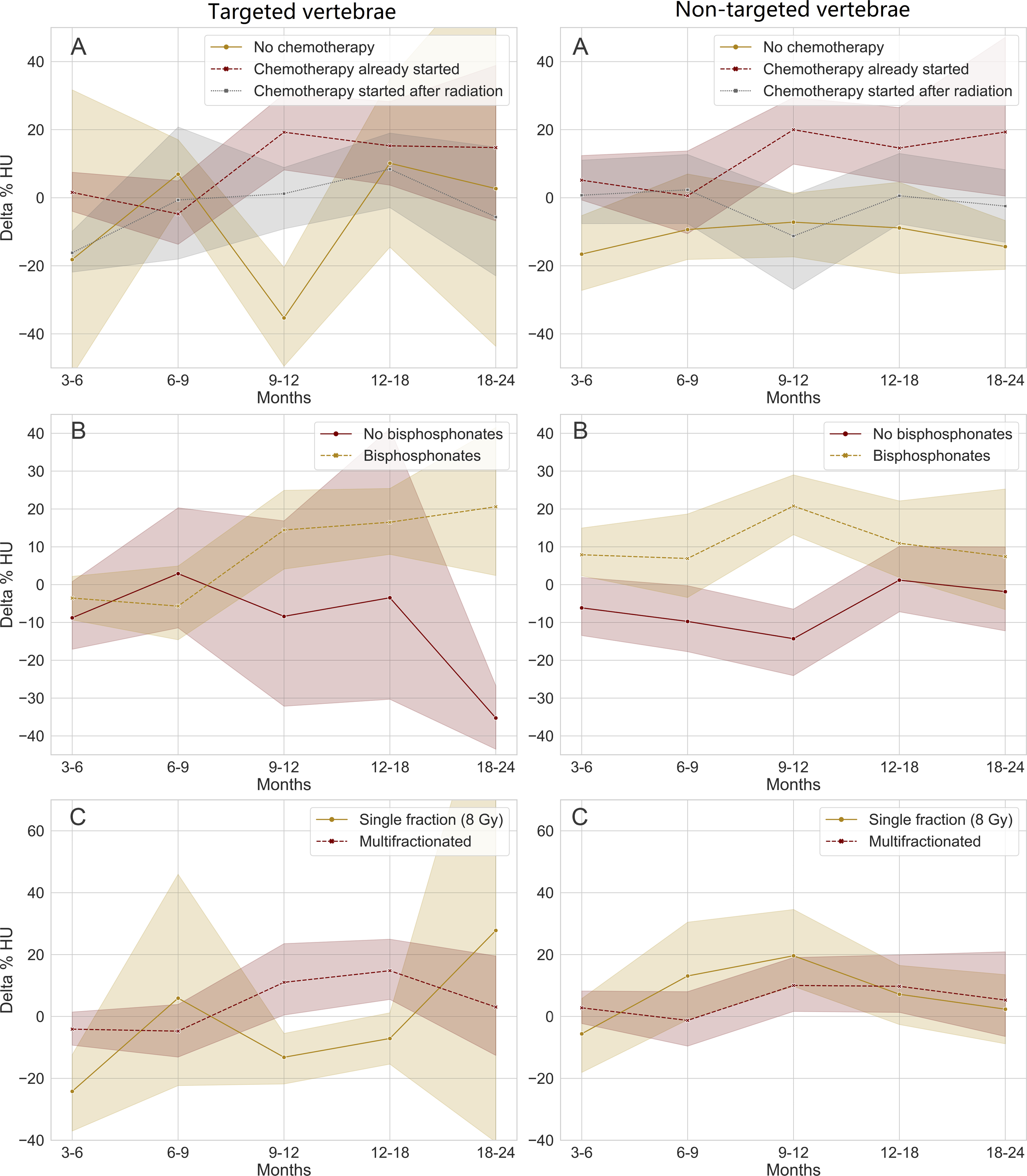
Retreatments
Within 2 years following radiation therapy, a subset of our study cohort underwent subsequent treatments, including additional radiation therapy or surgical interventions. Of the analyzed patients, 15 (12.6%) received further treatment after the follow-up CT scan. Specifically, eight patients underwent kyphoplasty due to fracture or persistent pain, four patients underwent laminectomy (two with fusion and two without fusion), and three patients received a second course of radiotherapy.
Role of the Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Discussion
Multiple myeloma often causes osteolytic lesions in the spine. 3 These destructive lesions change the biomechanical nature of the spine and have been known to result in painful compression fractures as well as unstable patterns of injury. 5 Treatment for these spinal lesions is multidisciplinary in nature often involving both systemic therapies as well as radiotherapy. 21 There is little knowledge regarding the effect of available treatments on the structural stability of the spine. Data regarding the structural integrity of the spinal column in the setting of MM lesions could potentially be helpful for both prognostication and treatment decision purposes. Therefore, in this study, we set out to measure the influence of radiation on remineralization of radiated MM vertebrae as measured by bone density in HU on CT scans before and after irradiation. Our results demonstrate that following radiation, BMD increased over time for both targeted and non-targeted vertebrae. However, no conclusive evidence was found that targeted vertebrae have a higher remineralization rate than non-targeted vertebrae in patients with MM.
The process of bone modeling and remodeling is primarily regulated by the RANKL/RANK/OPG pathway (Figure 3A).22-24 The primary roles of receptor activator of nuclear factor-kappa B (RANK) and its ligand, RANKL, (two proteins of the tumor necrosis factor [TNF] superfamily) are regulating bone remodeling by promoting osteoclast differentiation and activation of T cells.
23
Osteoprotegerin (OPG) is released from osteoblasts and regulates the bone remodeling process by inhibiting osteoclast activation and development through blocking the binding of RANK and RANKL.23,24 MM patients have decreased OPG concentrations in the bone marrow micro-environment, caused by intracellular interactions with osteoblasts and downregulating OPG release by bone marrow stem cells.
24
In addition, myeloma cells show upregulated RANKL expression.24,25 Furthermore, B-cell maturation antigen (BCMA) is a transmembrane glycoprotein (also part of the TNF superfamily) which is highly overexpressed on the surface of plasma cells in MM patients.26,27 Along with other proteins, BCMA normally supports B-cell proliferation, survival and maturation and differentiation into long-lived plasma cells. However, in MM it promotes proliferation and survival of malignant myeloma cells. Additionally, BCMA interacts with osteoclasts in the bone marrow microenvironment, leading to their growth and activation.27,28 The BCMA overexpression and the imbalance of the RANKL/OPG axis promotes pathological activation of osteoclasts and therefore underlies the development of the disease-defining lytic lesions in myeloma patients.
23
A. (1) 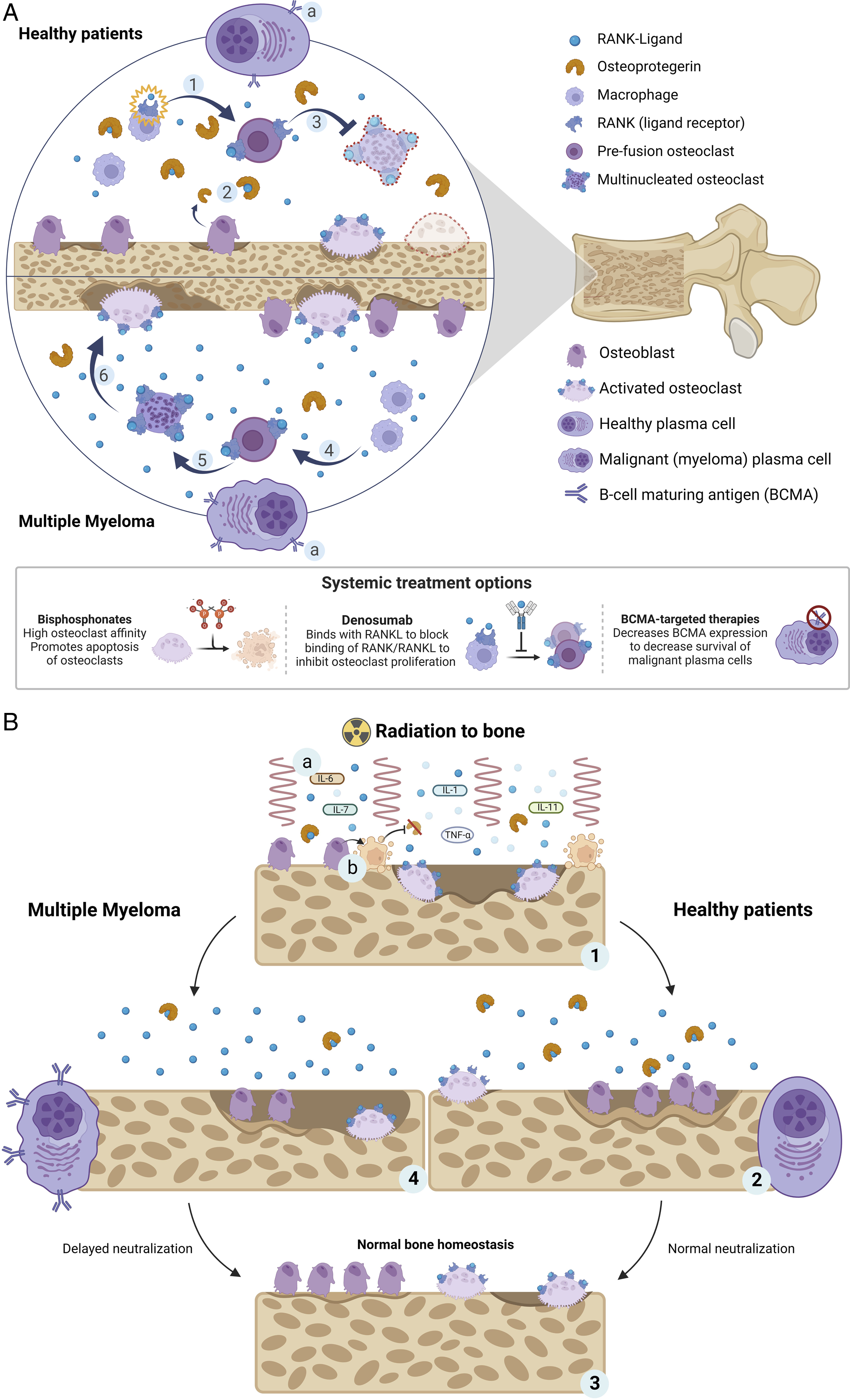
The primary goal of radiation in patients with MM is to induce cell apoptosis of malignant plasma cells or deprive them of their multiplication potential to slow progression and achieve local control. 29 Histomorphometric analyses of bone after radiation have revealed higher concentrations of multiple inflammatory cytokines (e.g. IL-1,6,7,11) and TNF-α in targeted and non-targeted contralateral bone shortly after radiation exposure, leading to systemic activation of osteoclastogenesis (Figure 3B).30-33 Additionally, some studies have observed cell cycle arrest and reduced proliferation rate of osteoblasts after radiation, leading to decreased OPG which subsequently prevents inhibition of RANKL.30,33-35 These factors lead to an initial decrease in BMD, which is in line with our results and other studies observing the same effect in both murine models30,34 and in humans.12,36 After this initial dip, bone homeostasis should be restored back to normal after several cycles and osteoblast production is reinitiated. When tumor infiltration in vertebrae is reduced and osteoclasts have excavated an area of bone (Howship lacunae), osteoblasts fill up this bone resorption pit with unmineralized matrix which hardens when minerals (i.e. calcium) are deposited in it. 37 Return to full density may take up to 6 months. 38
A relative increase in bone density compared with the baseline can occur when this remineralization process happens in a tumor cavity. 33 Many authors investigating bone density after radiation in patients with other spinal metastatic diseases have demonstrated this positive effect on BMD and found a greater increase in bone density in targeted vertebrae compared with non-targeted vertebrae.12-14,39 However, due to the overexpression of BCMA and imbalance of the RANKL/OPG axis, neutralization of bone homeostasis is delayed in MM 40 and therefore presumably less effective compared to bone metastases originating from non-B-cell derived malignancies. We presume that this is the reason we did not find a difference in remineralization between targeted and non-targeted vertebrae for patients with MM. Notably, within our initial study cohort, a relatively high proportion of patients (n = 25, 17.6%) required surgery within the first 3-6 months post-radiation (and were therefore excluded from further analyses). While the exact reasons for these surgeries were not uniformly documented, the observed dip in BMD during the initial post-radiation period could potentially contribute to the need for surgical interventions.
Our findings show that both targeted and non-targeted vertebrae show remineralization after radiation and a pronounced increase in absolute bone density compared with the baseline. Targeted vertebrae had a slightly higher correlation coefficient (slope) for remineralization effect per month compared with non-targeted vertebrae, although this difference was not statistically significant. These findings are in line with the aforementioned systemic effect of radiation in MM patients. Previous researchers have investigated this systemic effect by measuring targeted and contralateral-shielded bone in mice.30,34,41,42 In these models, it is evident that radiation causes increased osteoclast numbers in both targeted and contralateral-shielded (non-targeted) bone sites due to the systemic activation of osteoclastogenesis.30-33 Additionally, while we did take into account the possible imprecision of radiation delivery and the scattered effect of radiation by taking the first non-adjacent vertebrae (skipping one vertebral level from the targeted site), it cannot be ruled out that these ‘control-vertebrae’ did receive some radiation.
Systemic treatment of MM has improved greatly within the recent years and the emergence of novel therapies have changed the therapeutic landscape for MM. While the disease remains incurable, BCMA-targeted (immune) therapies like Chimeric Antigen Receptor (CAR) T-cell treatments have achieved remarkable clinical success in MM, especially for patients who are refractory to standard treatment. 7 Research has shown a rapid decrease of malignant plasma cells in the bone marrow and a remarkable decrease in serum BCMA expression following CAR T-cell treatment. 43 Bisphosphonate treatment (e.g., zoledronate) has been demonstrated to decrease the occurrence of skeletal-related events (such as spinal cord compression, pathological fractures, need for radiation and need for surgery) in patients with myeloma bone disease. 6 Bisphosphonates protect the bone from bone resorption by promoting apoptosis of osteoclasts. 23 In addition, denosumab is becoming increasingly common in the treatment of myeloma bone disease. Denosumab is a monoclonal antibody that had already proven to reduce the frequency in skeletal-related events in patients with advanced solid tumors, and more recent clinical trials have suggested its value in myeloma treatment as well by blocking the RANK/RANKL interaction.23,44 Additionally, it is not known to be nephrotoxic. 44 In short, BCMA-targeted therapies, denosumab and bisphosphonates inhibit bone resorption by osteoclasts and therefore support bone remineralization after radiation. In the current study, our data demonstrate that both radiated and non-targeted vertebrae show remineralization after radiation, which could be the result of systemic treatment. Although the study results suggest a positive effect of radiation on BMD locally and systemically, it is possible that systemic treatment contributes more to this effect. Additionally, the emergence of novel systemic therapies has opened new perspectives for the concomitant administration of radiation and systemic drugs, as possible synergy has been observed in pre-clinical studies in MM. 45 In terms of future research, it would be useful to extend the current findings by evaluating bone density of patients who received novel BCMA-targeted therapies without radiation to any location.
Another explanation for the increased bone density in both targeted and non-targeted vertebrae, is that patients who received radiotherapy experience less pain after radiation, 4 and therefore could become more physically active. Physical activity induces more mechanical stress on the bone, which is important for regulation of the bone metabolism and promotion of bone growth, according to Wolff’s law.37,38 In this study, level of activity was not measured, so no comparative analyses can be done to determine the effect of physical activity on BMD. It would be interesting to include activity level in any further studies investigating BMD after radiation.
Limitations and Recommendations
There are several limitations to this study. First, due to the retrospective nature of this study, CT-scans were not acquired at specific, predetermined timepoints or according to set protocols. In some patients, CT simulation scans acquired just prior to radiation were not available for baseline measurements. In those cases, we had to use a CT scan from an earlier moment. This increases the risk of missing any bone breakdown between that CT scan and the start of radiation. We did ensure that the pre-radiation CT scan was only compared with a follow-up CT using the same protocol. Second, it is still unclear what the effect of systemic therapy is on remineralization. Prospective studies with larger sample sizes and including a group of patients receiving only systemic therapy and a group receiving only radiation and no systemic therapy would be needed to confirm the exact relationship between radiation and bone strength. Unfortunately, data regarding the use of Denosumab was not available so an analysis of the effect of Denosumab was impossible. Nevertheless, despite these limitations, this study is the first to investigate bone density after radiation in patients with MM, using a relatively large patient population.
Lastly, our data revealed that a notable proportion of patients required subsequent surgeries or reirradiation within 2 years post-radiation, underscoring the complex and evolving nature of clinical care in MM-related spinal lesions. These findings highlight the ongoing need for a multidisciplinary approach, including spine surgeons, in the management of MM patients following radiation therapy. Radiation therapy should only be used after careful patient selection, considering its potential benefits (pain reduction) and limitations in bone remineralization. Close follow-up post-radiation is essential to monitor for any emerging complications, including fractures, allowing for timely intervention if needed.
Conclusion
The current study investigated vertebral remineralization rate in patients with MM undergoing radiation therapy. Our data showed that in the early period after radiation, targeted vertebrae demonstrated a decline in bone density compared with non-targeted vertebrae. However, after nine months, both targeted and non-targeted vertebrae showed increases in bone density, with no additional effect of radiation. This could be explained by a systemic effect of local radiation, a higher activity level of patients after radiation or the effect of (newer) systemic therapeutic approaches. The results can be useful for guiding patient selection for the use of radiation in the treatment of MM and highlights the need for future research for evaluating bone remineralization in patients who received novel targeted therapies without radiation to any location in a prospective study with controlled treatment protocols.
Supplemental Material
Supplemental Material - Remineralization Rate of Lytic Lesions of the Spine in Multiple Myeloma Patients Undergoing Radiation Therapy
Supplemental Material for Remineralization Rate of Lytic Lesions of the Spine in Multiple Myeloma Patients Undergoing Radiation Therapy by Hester Zijlstra, Jens P. te Velde, Brendan M. Striano, Olivier Q. Groot, Tom M. de Groot, Noopur Raje, Chirayu Patel, Jad Husseini, Diyar Delawi, Diederik H. R. Kempen, Jorrit-Jan Verlaan, and Joseph H. Schwab in Global Spine Journal
Footnotes
Acknowledgments
We thank Mr. D.L. Hayden, PhD and Mr. H. Lee, PhD, from the MGH Biostatistics Center for their support and advice on design and statistical analysis.
CRediT Roles
Conceptualization: HZ, JH, DD, DHRK, JJV and JHS; Data curation: HZ and JPV; Formal analysis: HZ; Investigation: HZ; Methodology: HZ, JH and JHS; Supervision: BMS, OQG, NR, CP, JH, DD, DHK, JJV and JHS; Visualization: HZ and TG; Writing – original draft: HZ; Writing - review & editing: BMS, OQd, TdG, NR, CP, JH, DD, DHK, JJV and JHS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
