Abstract
Study Design
Retrospective cohort analysis.
Objective
(1) Develop a novel computed tomography (CT)-based assessment of endplate bone density (EP-HU), (2) Determine if EP-HU was a stronger predictor than trabecular HU for subsidence after transforaminal lumbar interbody fusion (TLIF).
Methods
All adult patients who underwent single-level TLIF for lumbar degenerative conditions at an academic center between 2017-2022 were retrospectively identified. EP-HU was calculated from a 2 mm superior and inferior endplate region on the preoperative mid sagittal CT scans, accounting for surface undulations. Lumbar vertebral HUs (trabecular region) were determined in a standard fashion on axial CT. EP-HU + vertebral HU served as an aggregate bone quality metric. Interbody subsidence (≥2 mm threshold) was directly measured on the endplate-facing surface of 1 year CT scans. Univariate and multivariate analysis compared subsidence based on CT bone metrics.
Results
A total of 114 patients met the inclusion/exclusion criteria. There was no significant difference in fusion or reoperation rate based on subsidence occurrence. Both vertebral HU (P = .012) and EP-HU (P < .001) were associated with subsidence. In receiver operating curves, EP-HU was more optimal for subsidence prediction than vertebral HU, but the aggregate metric further optimized the specificity and total area under the curve. In a predictive logistic regression model EP-HU + vertebral HU (aggregate HU < 515 odds ratio: 7.82, P < .001) was a strong independent predictor of subsidence.
Conclusion
Preoperative calculation of EP-HUs in addition to vertebral HUs may enhance prediction of TLIF subsidence where aggregate endplate and vertebral HU < 515 can be used to identify high risk patients.
Introduction
Interbody cages are often utilized in spinal fusion to maximize indirect decompression, improve regional sagittal alignment, enhance immediate mechanical stability, and increase fusion rate.1-3 In lumbar fusion, transforaminal lumbar interbody fusion (TLIF) may be preferred relative to other lumbar interbody approaches as its single-incision posterior approach is often associated with increased operative efficiency compared with two-stage interbody and posterior pedicle screw fixation procedures. However, the incidence of subsidence is increased after TLIF relative to lateral and anterior interbody approaches due to the anatomic constraints of the facetectomy window requiring smaller cage footprints.4,5 As such, subsidence represents a common TLIF complication with an incidence ranging from 15% to 59% in prior studies.6-12 These discrepancies in TLIF subsidence rates not only reflect variations in patient populations and surgical techniques, but also may be attributed to differences in subsidence measurement methods.13,14 The majority of TLIF subsidence assessment in the literature is limited to indirect measurement of subsidence from postoperative lateral X-ray which relative to computed tomography (CT) may underpredict subsidence rates based on variations in patient positioning and rotation, magnification error, and inadequate resolution for millimeter scale measurements. 13
Subsidence after TLIF that has been shown to correlate with pseudoarthrosis, loss of segmental lordosis, recurrence of preoperative symptoms, higher reoperation rates, and decreased overall improvement in pain and disability scores.13,15-18 Prior literature has implicated numerous medical and operative risk factors for TLIF subsidence including smoking, advanced age, obesity, disc space over-distraction, aggressive endplate preparation, and decreased bone density.8-10,12,19 Of these risk factors, low bone density is commonly cited as a strong risk factor that is independently predictive of TLIF subsidence. However, there remains substantial discrepancy regarding the ideal method that should be utilized to assess bone quality. CT vertebral Hounsfield units (HU) relative to DEXA T-score and Z-score have demonstrated stronger association with interbody subsidence.12,20-23 This may reflect that DEXA has less regional specificity to the instrumented vertebrae and can be subject to false elevations in the presence of arthritis.12,24 Vertebral HUs, however, only assess trabecular bone density, which may be an inaccurate proxy for the density and thickness of endplate cortical bone in contact with intervertebral cage. Endplate bone quality could provide a more site-specific predictor of the resistance to fracture propagation and deformation of the vertebral cortex, which dictates early and late subsidence risk. The present investigation developed a CT-based assessment to isolate lumbar endplate bone quality (EP-HU) and aimed to determine if EP-HUs, trabecular HUs, or a combined assessment of lumbar endplate and trabecular HUs was most effective for predicting TLIF subsidence, as classified on postoperative CT.
Methods
Patient Identification
After approval from the Institutional Review Board (23-003878), a retrospective cohort study was conducted on all patients over age 18 who underwent only single-level transforaminal lumbar interbody fusion (TLIF) for lumbar spondylosis associated with neurogenic claudication, radiculopathy, and/or spondylolisthesis at a multicenter academic institution between 2017-2019. Our study was deemed exempt from needing informed consent by the Institutional Review Board. Additional inclusion criteria required complete patient demographic profiles, surgical characteristics, interbody device and fusion construct records, and preoperative radiographs, and preoperative (within 1 year before surgery) and postoperative CT (at a minimum of 6 months preferencing the one-year follow-up) scans. All patients who were initiated on anabolic or anti-resorptive agents within the year prior to surgery for preoperative optimization obtained an updated CT scan immediately before surgery that was utilized for all vertebral bone assessments in this study. Patients with a traumatic injury, infection, malignancy, previous fusion at the index level, combined anterior/posterior procedures, or surgery with greater than one TLIF levels were excluded. Patient demographic and medical data, surgical factors, perioperative radiographic measures, and duration of follow-up were recorded via chart review.
Operative Treatment
All patients underwent an open midline posterior approach to the lumbar spine. Following fluoroscopic confirmation of the TLIF level, a unilateral complete facetectomy was performed and the ventral facet capsule and ligamentum flavum were removed to expose the posterolateral disc space. After distraction was achieved at the disc space, a box annulotomy and disc space preparation were performed. Endplate preparation was achieved with paddle shavers and serrated curettes. The interbody was filled with autograft and/or morselized cancellous allograft and inserted as anteriorly as possible in the disc space under fluoroscopic guidance. If bone morphogenic protein (BMP) was utilized during the TLIF, it was placed in the anterior disc space prior to interbody insertion. All patients had posterior pedicle screw fixation with posterolateral bone grafting. Although care was taken to avoid over aggressive endplate preparation and mitigate the removal of any cortical bone, endplate violation events were determined through a cortical breech visualized on intraoperative or immediate postoperative radiographs.
Endplate and Vertebral Bone Assessment
EP-HU was determined from a 2 millimeter (mm) superior and inferior endplate region of interest (ROI) circumscribed on the preoperative mid-sagittal CT scans (settings: 140 kV, 280 mAs, 0.75 mm resolution) using the free draw function, in ≥500% magnification, to account for endplate surface undulations and scalloping, unique from existing endplate bone assessment metrics (Figure 1). A 2 mm ROI was selected as this corresponded to the upper bound of lumbar endplate thickness from cadaveric studies. Specifically, a micro CT-based assessment of 150 lumbar cadavers found that the average lumbar endplate cortical thickness was 0.91 mm (1.03 mm cranially and 0.78 mm caudally) and ranged from 0.44-2.00 mm.
25
The superior, inferior, and total ([superior + inferior]/2) EP-HUs were calculated as an average of the endplate regions on all views. EP-HUs were performed by two independent reviewers. Average standard lumbar vertebral HUs were determined from circumscribed trabecular bone within cranial, middle, and caudal axial CT cuts at the each TLIF level. The aggregate EP-HU and vertebral HU were also added together as a combined endplate and trabecular bone quality metric. Likewise, the EP-HU and vertebral HU difference (EP-HU- vertebral HU), ratio (EP-HU/ vertebral HU), and normalized ratio ([EP-HU- vertebral HU]/ EP-HU + vertebral HU] were calculated. Depiction of EP-HU Measurement Technique on the Mid-Sagittal CT Cross Section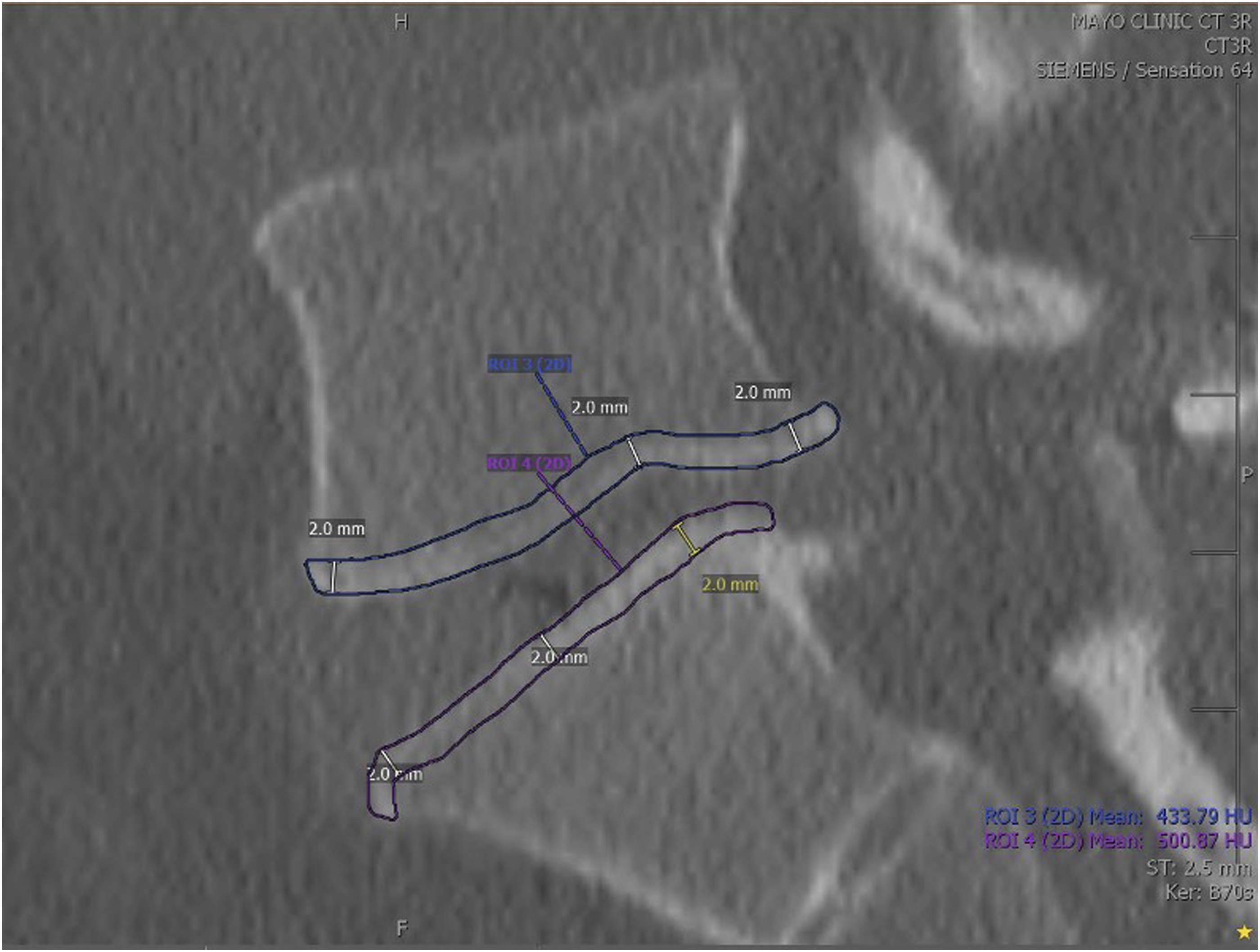
Cage Subsidence Assessment
Interbody subsidence at the superior and inferior endplate of each TLIF level was directly measured on the endplate-facing surface of both mid-coronal and mid-sagittal CT scans (settings: 140 kV, 280 mAs, 0.75 mm resolution), perpendicular to the interbody cage, obtained at greater than 6 months postoperatively, according to previously developed methods (Figure 2).13,14 Subsidence was assessed at this postoperative time interval because it is routine within the institutional practice to obtain CT scans at this interval for fusion monitoring. Only lead surgeons that regularly obtained postoperative CTs on all patients according to their postoperative TLIF protocol were included in the study to mitigate worst case selection bias. Additionally, prior literature has demonstrated that the vast majority of subsidence occurs within the first few months postoperatively.
26
Subsidence was measured by two independent reviewers and averaged to determine the final subsidence measurement. Interbody subsidence at each operative level was classified as mild or moderate- severe based on previously documented <2 mm and ≥2 mm thresholds, respectively.13,14,27 The average subsidence measurement with respect to the superior and inferior endplates and coronal and sagittal planes dictated the subsidence classification. Depiction Subsidence Measurement Technique From an Example With Severe Subsidence on (left) Mid-Sagittal and (right) Mid-Coronal CT Slices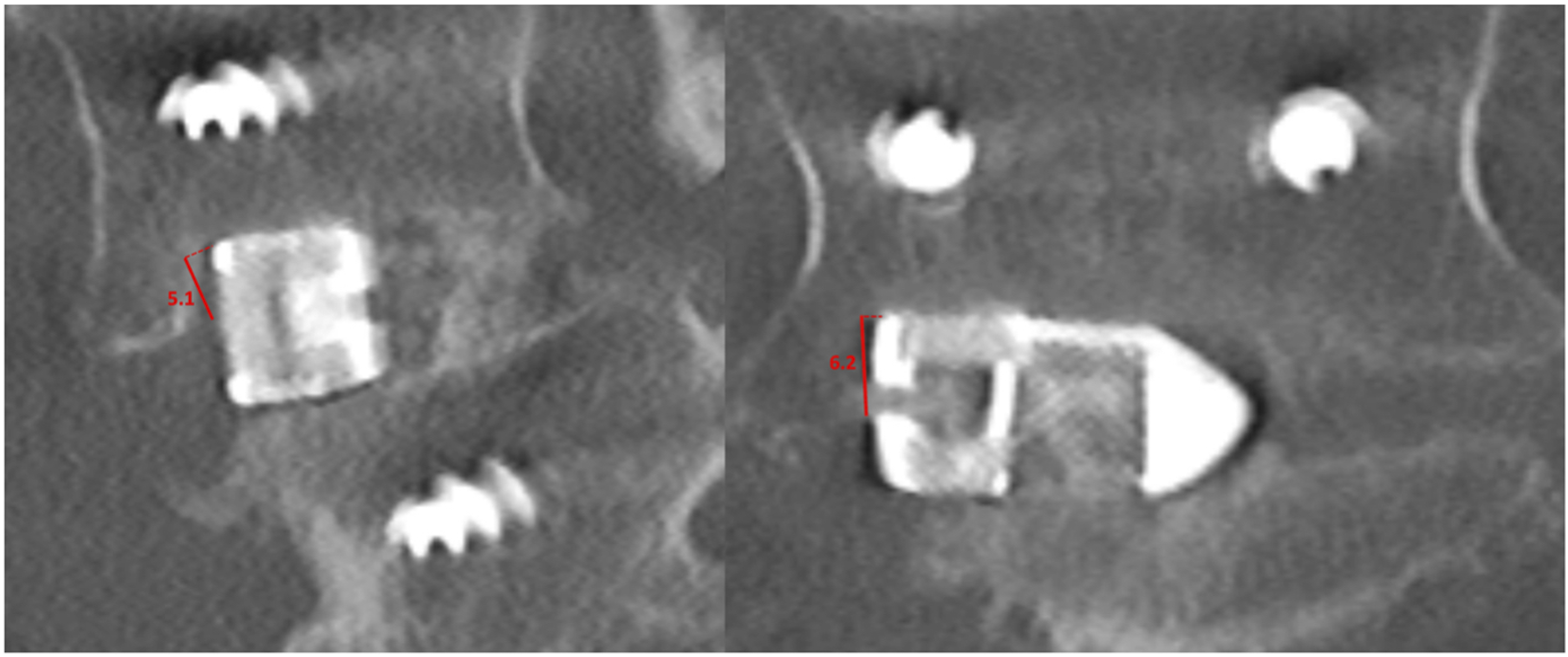
Assessment of Radiographic and Construct-Related Variables
Preoperative disc space height at the TLIF level (average of anterior, middle, and posterior aspect measurements on mid-sagittal view) and preoperative vertebral length (axial view at level of endplate) was determined from preoperative CT scans. Operative notes and device implantation records were reviewed to determine surgery and instrumentation specific parameters including operative indication, TLIF levels, cage length, cage height, and cage degree of lordosis. Fusion assessment was performed on the 1 year postoperative CT scan obtained routinely according to standard institutional practice. Fusion was defined as bony bridging between vertebrae with an absence of continuous radiolucent lines between endplates in at least three cross sections. The all-cause reoperation rate within the 1 year postoperative follow-up was determined.
Statistical Analysis
Agreement in EP-HUs measurement between the two reviewers was determined by interclass correlation coefficients (ICC). ICC was interpreted according to: >0.90 as excellent, 0.75-0.90 as good, 0.50-0.75 as moderate, and ≤0.50 as poor. 28 Patients were dichotomized into groups based on the absence or presence of moderate subsidence defined by a 2 mm threshold. Descriptive statistics were used to compare patient demographics and outcomes in terms of mean and standard deviation, median and interquartile range, or number of occurrences and percent of total. Continuous and categorical variables were analyzed with t-tests and chi-square tests, respectively, or the corresponding non-parametric tests based on the Shapiro-Wilks test to assess for data normality for each variable of interest. Univariate analysis compared patient demographics, medical comorbidities, preoperative EP-HUs, vertebral HUs, and intraoperative factors, and postoperative outcomes among subsidence groups. Pearson’s correlation was utilized to assess the relationship between HUs metrics. Pearson’s rho was interpreted as follows: 0-0.1 as no correlation, 0.1-0.4 as weak correlation, 0.4-0.7 as moderate correlation, and 0.7-1.0 as strong to perfect correlation. 29 Receiver operating curve analyses determined the probability of subsidence based on EP-HUs and vertebral HUs. Youden’s method was utilized to determine a cutoff HU valuable that maximized sensitivity and specificity for the prediction of subsidence. Predictive logistic regression models, with a P-value threshold of <.01 for inclusion, were developed to isolate independent predictors of subsidence. All significant univariate factors were included in the initial logistic regression model and eliminated in a stepwise fashion to optimize the model’s Akaike information criterion. All statistical analysis was performed with R Studio Version 4.0.2 (Boston, MA). A P-value <.05 was statistically significant.
Results
Cohort Definition and Subsidence Characterization
A total of 114 patients that underwent single-level TLIF met the inclusion/ exclusion criteria, operated on by eight lead surgeons with the majority of surgeries performed by a single surgeon (surgeon 1: N = 93, surgeon 2: N = 11, surgeon 3: N = 4, other surgeons: N = 6). The median radiographic follow-up duration was 1.02 [0.95; 1.29] years postoperatively, with no significant differences based on subsidence group.
Subsidence Patient and Surgical Risk Factors
Demographic and Surgical Parameters Based on Patient Subsidence Group
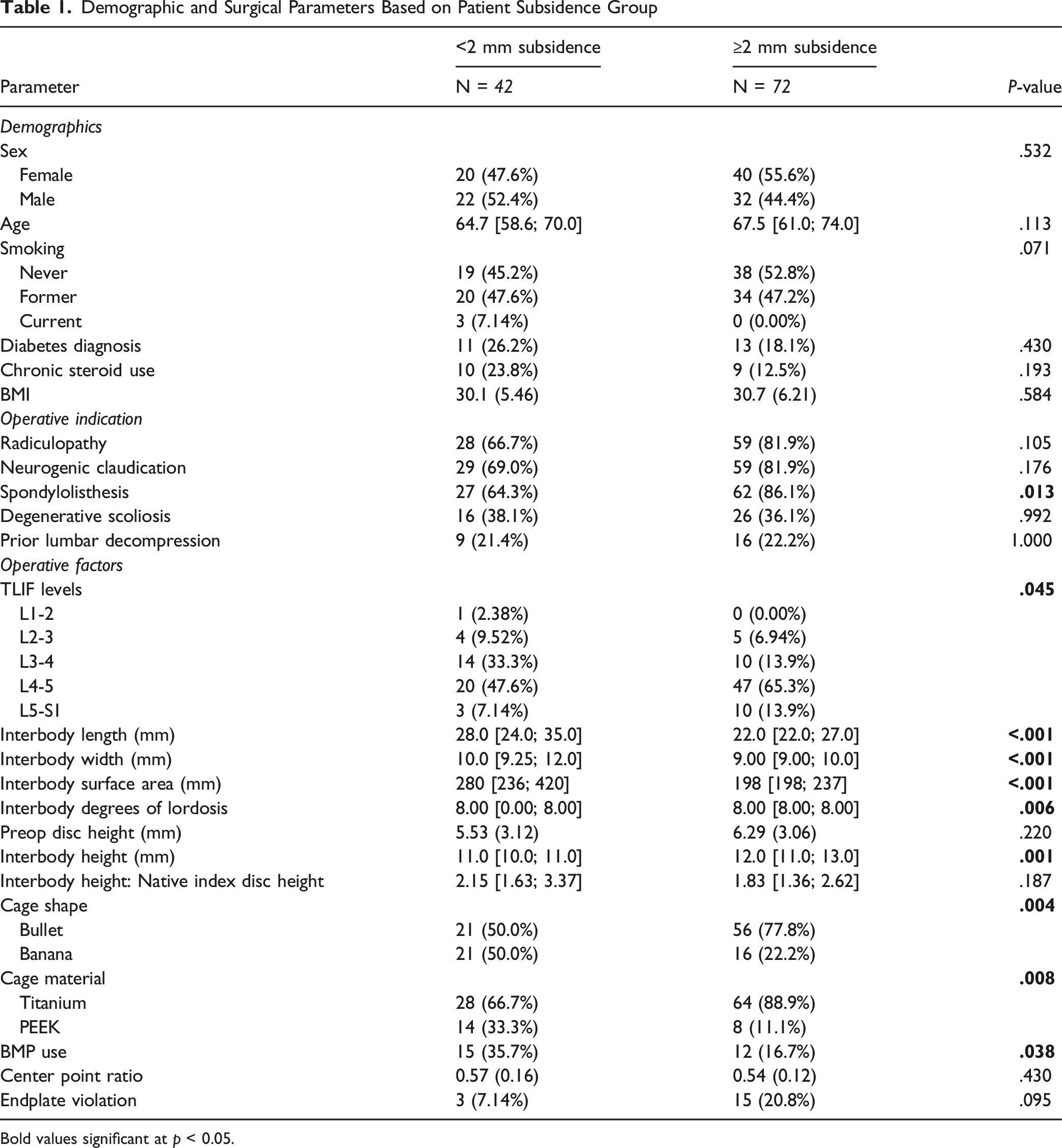
Bold values significant at p < 0.05.
Subsidence Bone Quality Risk Factors
Bone Quality Based on Patient Subsidence Group
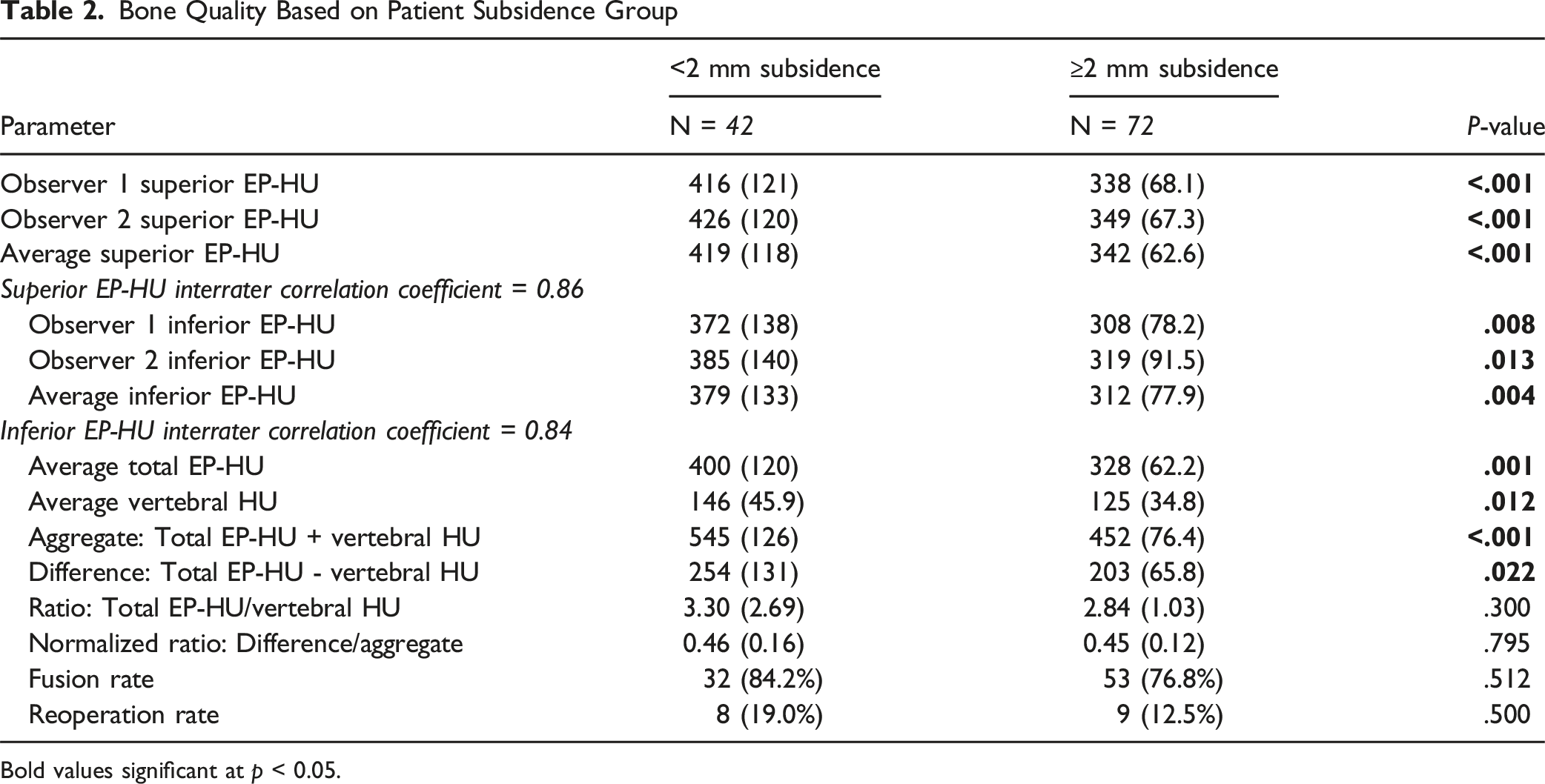
Bold values significant at p < 0.05.
Predictive Logistic Regression Model for Subsidence Based on EP-HU and Aggregate HU Metrics
Bold values significant at p < 0.05.
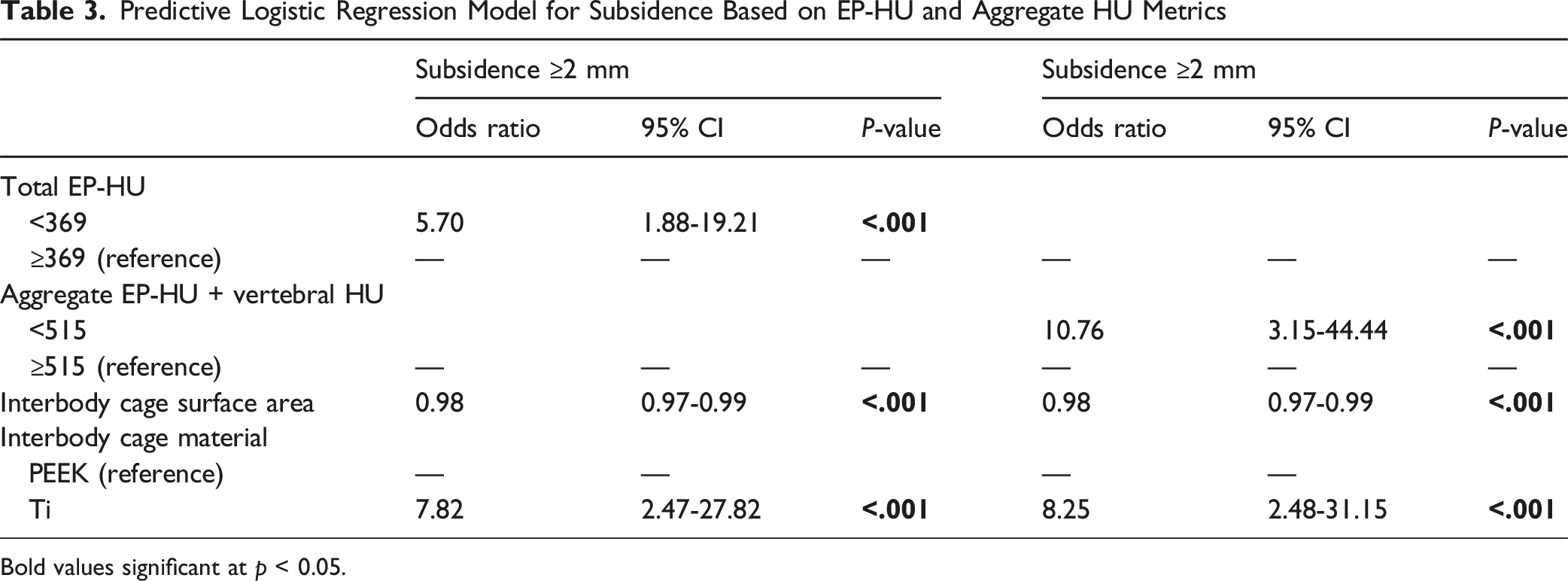
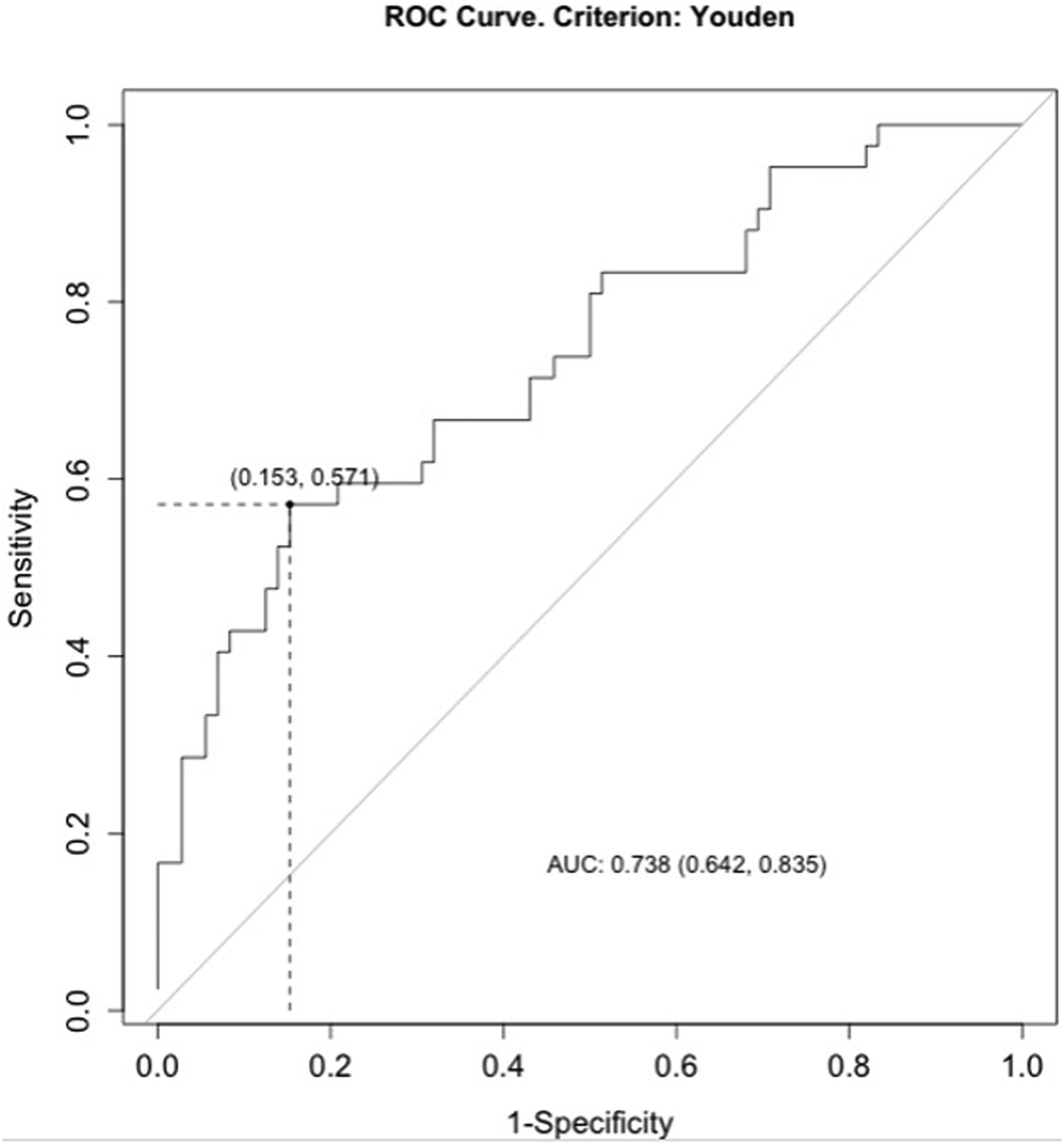
ROC identified an optimal EP-HU cutoff of 369 (area under curve [AUC] = 0.704, sensitivity: 57.1%, specificity: 79.2%) to predict subsidence (Figure 3). The AUC of the vertebral HUs with respect to subsidence was 0.640, which was below the acceptable (0.7) threshold to facilitate a cutoff analysis. The aggregate measure of EP-HU + vertebral HU resulted in an improved AUC of 0.738 indicating the greatest sensitivity and specificity for subsidence prediction. The cutoff value of the aggregate HU measure was 515 (sensitivity: 57.1%, specificity: 84.7%) (Figure 4). Receiver Operating Curve for the Probability of Subsidence Based on EP-HU Receiver Operating Curve for the Probability of Subsidence Based on EP-HU + Vertebral HU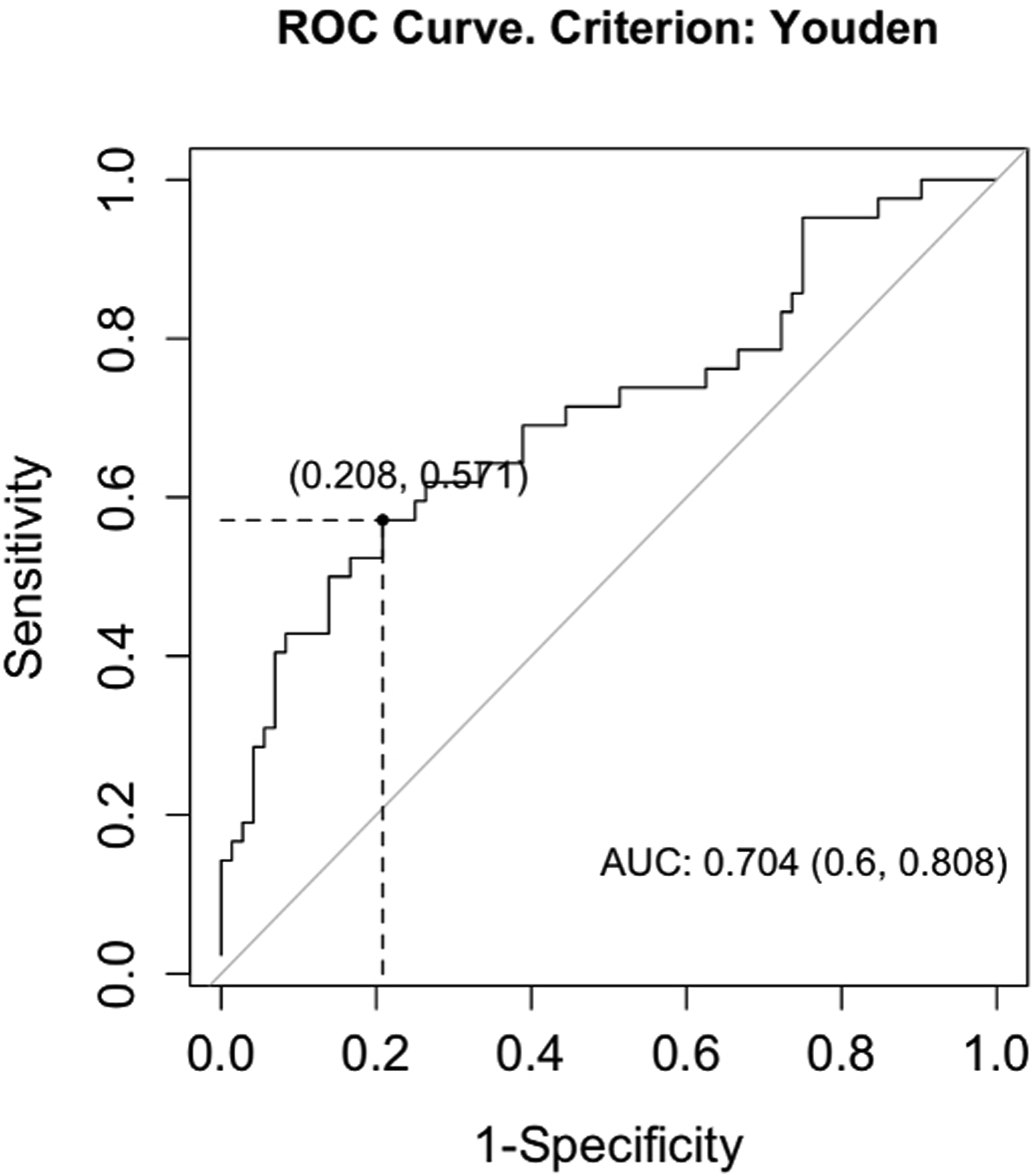
Fusion Status and Reoperation Rates
There was no significant association between subsidence and pseudoarthrosis where patients without subsidence experienced an 84.2% fusion rate compared with a fusion rate of 76.8% in patients with subsidence (Table 3). There was no significant difference in the 1 year all-cause reoperation rate between subsidence groups (Table 3).
Regression Analysis
In predictive logistic regression models, total EP-HU <369 HU (EP-HU: odds ratio [OR] = 5.70, P < .001), interbody cage surface area (OR: 0.98, P < .001), and titanium cage material (OR: 7.82, P < .001) were independently predictive of subsidence. Vertebral HU was eliminated from this predictive model as the P-value was above the .01 threshold for inclusion. The predictive regression was however further optimized by utilizing the aggregate EP-HU + vertebral HU (OR: 10.76, P < .001) metric for subsidence prediction (Table 3).
Discussion
TLIF subsidence represents a common postoperative complication associated with compromised clinical outcomes and increased revision rates. The current study developed a CT-based, site-specific measurement of endplate bone and combined this with traditional vertebral HUs for an aggregate assessment of lumbar cortical and cancellous bone. The results demonstrate that EP-HUs was a stronger predictor of subsidence than vertebral HUs, however the aggregate EP-HU + vertebral HU metric demonstrated superior predictive capacity to either measure alone.
Over the past decade, CT HU and quantitative CT (QCT) have emerged as an opportunistic means of assessing regional bone quality with increased sensitivity and specificity for spinal instrumentation failure, including subsidence, compared to global skeletal bone assessments like DEXA.30,31 However, it remains unclear whether the enhanced predictive capacity of regional spinal bone quality assessments can be applied to site-specific measures of bone quality within the vertebrae itself. Furthermore, the relative dependence of spinal osteoporosis related complications on cortical vs cancellous bone quality is yet to be elucidated. In standard osteoporotic progression, patients experience early bone loss mostly within the trabecular space, as a reflection of the larger surface area available for osseous remodeling.32,33 With increasing age and osteoporosis severity, patients develop greater intracortical porosity and resultant increase in the endocortical surface. 33 This transition from predominately trabecular to predominately cortical bone loss is consistent with the increased risk for trabecular followed by cortical fractures in osteoporotic patients. 32 Subsidence, a consequence of supraphysiologic axial compressive loading of a vertebral body, is likely mediated by cortical microfracture or deformation and subsequent compaction of trabecular units allowing interbody settling to occur. Osteoporosis may mediate subsidence not only through decreased cancellous bone density, but also by changes in the cortical endplate surface that decrease strength and increase brittleness, which predispose to endplate microfracture propagation. Therefore, analysis of isolated cancellous bone metrics is unlikely to optimize subsidence prediction models and subsidence may be most probable in scenarios of cortical bone resorption seen in more advanced osteoporosis.
The current study developed an optimized CT method for endplate bone assessment in the lumbar spine that captured the entirety of the endplate cortical thickness despite variations in endplate surface topographies. This is in contrast to two CT-based endplate bone metrics proposed in LLIF studies which fail to account for scalloping and undulations of the vertebral cortex, commonly present in adults with degenerative spinal pathologies.23,34 Unlike the current study which utilized a 2 mm sagittal ROI in line with the endplate surface for EP-HU determination, one existing measure of endplate HU in literature was defined by a single outermost vertebral cross section on axial CT. 23 Not only did this fail to account for endplate thickness but it also introduced substantial inaccuracy by including the intra- and extra-vertebral space in the endplate HU determination. Another study defined a CT volumetric endplate metric by a 5 mm rectangular ROI, which missed the majority of the cortical surface with the rectangular constraint and circumscribed mostly cancellous bone provided two millimeters represents the upper bound of lumbar endplate thickness.34,35
Despite the aforementioned limitations in CT endplate assessments from LLIF literature, these prior studies did find a significant association between endplate bone quality measurement and LLIF subsidence.23,34,35 The present study substantiates this association in TLIF, while demonstrating that EP-HU had a greater probability of subsidence prediction than the 5 mm rectangular endplate metric. Moreover, our study also corroborates the finding from LLIF literature, that EP-HU compared to vertebral HU has increased probability of predicting and greater strength of association with TLIF subsidence, where preoperative EP-HU values <369 raised concern regarding TLIF subsidence risk. The current investigation is also the first to demonstrate that a combined assessment of endplate and trabecular bone density is superior to either metric in isolation for TLIF subsidence prediction. Specifically, relative to EP-HU alone the combined endplate and trabecular bone density metric, with a cutoff of 515 HU, increased specificity by 5.5% and doubled the odds ratio as a subsidence predictor. Taken together, the results underscore that subsidence is most reflective of the endplate thickness/density but also is dependent on cancellous osseous architecture, which may mediate resistance to trabecular compression when a cortical defect or deformation is present. Subsidence was also more likely to occur in patients where the EP-HU and vertebral HU were closer in absolute value. Mechanistically, this may suggest that subsidence is more likely to occur in patients with later stage osteoporosis where cortical porosity and cortical thinning is present in addition to the trabecular bone changes seen with earlier disease.
This study also corroborates known surgical risk factors for TLIF subsidence. The relationship between decreased interbody surface area and subsidence is well document in biomechanical and clinical literature, provided that subsidence is a pressure related phenomenon that may be mitigated by force distribution over a greater cross sectional area. 14 The effect of cage material on interbody subsidence, however, is a subject of ongoing debate, where the current study identified titanium relative to PEEK cages a strong subsidence predictor. It is possible that the osseoconductive properties of Ti relative to PEEK may confer earlier stability protecting against subsidence, as preliminary LLIF study results suggest. 36 While the majority of titanium interbodies in this study were porous, which reduces the overall elastic modulus to a similar range as PEEK, the edge of the porous titanium interbody cages are still solid titanium which may lead to a modulus mismatch and edge loading especially in hyper-lordotic cages, risking further subsidence progression.
This study is not without limitations including those inherent to retrospective data collection. Although patients were included in a systematic method, potentially relevant cases were identified based on availability of follow-up parameters, introducing a selection bias. Though the cohort is moderately sized, the possibility for type two errors in rare event outcomes, such as pseudoarthrosis and reoperation, cannot be excluded as this study may not be adequately powered for these secondary outcomes. Regarding the EP-HU measurement technique, a single mid-sagittal cross section may not be representative of the endplate properties in all regions. The single cross section was selected to increase reproducibility of the measurement technique and because it would be captured beneath the interbody cage regardless of variations in interbody placement and footprint. Furthermore, studies have demonstrated that the degree heterogeneity in the osseous density and architecture is much greater in cancellous relative to cortical bone. While future studies could validate the novel lumbar CT endplate assessment in quantitative CT, EP-HUs were utilized in the present study as they can be more easily applied in the clinical setting without imaging transformation and the CT image parameters and scanner type remained consistent at our institution throughout the study period. Furthermore, this study did not compare EP-HU to existing MRI-based measures of endplate and vertebral bone quality which although validated in prior studies, MRI does not provide as detailed characterization of osseous architecture and topology compared with CT assessment. Regarding subsidence assessment, a single 1 year CT timepoint did not enable determination of the clinical progression of subsidence and, while unlikely, could have underestimated subsidence that increased after the CT. Though intraoperative endplate violation is uncommon during TLIF provided that endplate preparation is less aggressive than in cervical surgeries, future investigations are required to determine the relationship between bone quality and early vs late postoperative subsidence progression. Nonethless, differences in operative technique including surgeon specific preferences for disc space and endplate preparation impart heterogeneity and their potential effect on subsidence may remain incompletely controlled for in this cohort. While postoperative CTs are regularly obtained at the institution of investigation, which enabled precise subsidence characterization, routine postoperative CT use may not be practical in many outside clinical settings.
Conclusion
Preoperative calculation of EP-HUs in addition to vertebral HUs may be considered as a more site-specific predictor of TLIF subsidence. Spine surgeons can calculate an aggregate endplate and trabecular HU score for preoperative subsidence risk stratification and consider further bone health optimization or modification of surgical technique and/or instrumentation if the score is <515 HU.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
