Abstract
Study Design
Retrospective Study.
Objectives
To compare subsidence and radiographic fusion rates of titanium-surface polyetheretherketone (PEEK-Ti) and 3D-Titanium (3D-Ti) cages, implanted within the same patient concurrently, during multi-level transforaminal lumbar interbody fusions (TLIF).
Methods
Forty-eight patients were treated with both PEEK-Ti and 3D-Ti cages during 2- or 3-level TLIF and instrumented posterolateral fusion (108 spinal levels in all). Equivalent bone graft material was implanted within each patient. Radiographic analysis of CT and/or X-ray imaging was performed retrospectively for each spinal level throughout 12-month follow-up period. Fusion was defined as bridging trabecular bone and subsidence was incursion into one/both vertebral bodies >20% cage height. Outcomes were analyzed with Fisher’s exact test.
Results
At 6-months post-operative follow-up, incidence of subsidence was significantly lower for PEEK-Ti cages, with 4.8% subsidence, compared to a 27.9% subsidence rate for 3D-Ti cages (P = .007). Fusion rates were comparable at 100% for PEEK-Ti and 95.5% for 3D-Ti. Results at 12-months showed similar but not statistically significant trends of less subsidence with PEEK-Ti than 3D-Ti cages (14.3% PEEK-Ti, 37.5% 3D-Ti), and similar fusion rates of 100% for PEEK-Ti and 91.7% for 3D-Ti. Thirty-nine out of 48 total patients were available for follow-up at 6 months and 20 patients at 12 months. CT availability at 6 and 12-months was 100% and 90%, respectively.
Conclusions
A significantly lower subsidence rate was associated with a PEEK-Ti cage, compared to 3D-Ti, 6 months after TLIF. Results may not be generalized across technologies due to differences in cage designs; additional research studies are warranted.
Keywords
Introduction
Transforaminal lumbar interbody fusion (TLIF) is a widely performed surgery for the treatment of degenerative spinal disorders, the goal of which is to stabilize the motion segment and facilitate bony fusion. High fusion rates and good clinical outcomes have been reported for TLIF with the combination of pedicle screws, polyetheretherketone (PEEK) cages, and recombinant human bone morphogenetic protein-2 (rhBMP-2).1,2 However, there is still a need for alternative materials for cages used in TLIF. Studies of TLIFs using PEEK cages without the use of rhBMP-2 have reported low fusion rates and sub-optimal osseointegration. 3 Although the PEEK bulk material has biomechanical benefits, its material surface is inert and hydrophobic, which can hinder osseointegration at the bone-implant interface. Other cage materials such as titanium alloys or more recent surface technologies such as titanium-surface PEEK have been developed, in an effort to offer improved osseointegration compared to PEEK alone. There is a need for more clinical evidence for fusion results in TLIF procedures comparing different cage material selections.
Interbody cages for TLIF are typically made of either metal, such as titanium and its alloys, or polymer materials such as PEEK. Titanium alloys possess osseointegration capacity and biocompatibility, but have a high elastic modulus compared to bone. Some studies have reported that the mismatch in modulus of elasticity between the implanted cage and the host bone could lead to stress shielding around the implant, which could be responsible for increased subsidence rates.4,5 The titanium cage also makes fusion assessment by radiographic imaging difficult, due to its high radiopacity. The trend toward increased use of PEEK cages in lumbar fusion, starting in the late 1990s to early 2000s, was due to 2 primary reasons. First, the radiolucency of PEEK allowed for fusion assessment by plain radiographs and CT with little interference. Second, there was a desire to match the cage material’s stiffness to that of bone. PEEK is an inert polymer material with elastic modulus similar to cortical bone. However, the hydrophobic surface of PEEK is less osteoconductive and discourages bony integration. The healing interface between the PEEK implant and the adjacent vertebral bony endplate has been reported to have poor osseointegration, as shown by gaps of peri-implant lucency and a biofilm layer associated with unsuccessful fusion. 6
Studies comparing titanium vs PEEK cages in interbody fusion report mixed clinical outcomes for fusion and subsidence. A meta-analysis performed by Massaad of titanium and/or titanium-coated cages vs PEEK cages in TLIF and PLIF found lower rates of fusion for the PEEK cages and no differences in subsidence, with wide ranges of subsidence between 0%-36% for titanium and 0%-31% for PEEK. 7 Furthermore, in a study comparing titanium and PEEK cages in TLIF, Nemoto and colleagues reported vertebral osteolysis in 60% of the non-union cases in the PEEK group, while osteolysis was not observed with titanium cages. 8 Anterior cervical fusions have increased subsidence in groups implanted with titanium as compared to PEEK, in both 1- and 2-level anterior cervical discectomy and fusions (ACDF).9-11
The advent of implants employing new surface technologies, such as titanium-surface PEEK (PEEK-Ti), have entered the spinal market to overcome reports of sub-optimal osseointegration and bony fusion with PEEK. Preclinical and clinical studies demonstrate that PEEK-Ti implants are safe and exhibit similar fusion rates and clinical outcomes compared to PEEK. 12 Preclinical studies in sheep have shown benefits of PEEK-Ti compared to plain PEEK, 13 and benefits of PEEK-Ti with 3D macroscale features relative to PEEK-Ti without the same features. 14 There is a need for more clinical studies on PEEK-Ti implants with larger cohorts and concurrent controls. Another recent technology in cage design has been provided by advances in implant manufacturing processes, such as additive manufacturing, or 3D printing. The 3D-printing process can be used to create a titanium implant with a specific porous structure to mimic cancellous bone. 15 In theory, this level of control over implant design could be used to create a 3D-titanium (3D-Ti) cage with favorable structural properties for increased bone ingrowth and/or decreased subsidence. Early studies suggest that 3D-Ti cages may be associated with accelerated fusion rates compared to traditional PEEK cages, with similar subsidence rates to PEEK.16-17 However, more clinical studies are needed to confirm these benefits.
The objective of this study was to compare subsidence and fusion rates of titanium-surface PEEK (PEEK-Ti) and 3D-Titanium (3D-Ti) cages implanted within the same patient during 2- or 3-level TLIF and instrumented posterolateral fusion.
Methods
Forty-eight consecutive patients who underwent 2- or 3-level TLIF with PEEK-Ti and 3D-Ti cages for degenerative spinal conditions were included. Surgeries were performed by one surgeon at a single center between April 2017 and January 2019. Thirty-six patients had 2-level TLIF, and 12 patients had 3-level TLIF. Every patient received a PEEK-Ti cage and a 3D-Ti cage at different disc levels. Primary surgical indications were most commonly spondylolisthesis, spondylosis with myelopathy, spondylosis without myelopathy, and stenosis. There were 19 patients with a primary diagnosis of spondylolisthesis, 17 patients with Grade I, and 2 with Grade 2 spondylolisthesis.
As a retrospective data review, this study was deemed exempt from Institutional Review Board (IRB) ethics committee review. All data obtained was collected per standard of care and the patients included in this analysis were not treated or followed per a study protocol. As such, informed consent was not required or obtained. All data analysis and reporting were performed on de-identified patient datasets.
Surgical Technique
A midline incision was performed followed by complete facetectomy and decompression of the nerve root. Transverse processes were decorticated and pedicle screws were placed. Next, a thorough discectomy and endplate preparation was performed. Local autograft bone was mixed with bone graft and packed into the disc spaces and interbody cages. For 2-level procedures, one level received the PEEK-Ti cage (VenturaTM NanoMetalene®, SeaSpine, Carlsbad, CA), and one level received the 3D-Ti cage, (Tritanium® TL, Stryker, Kalamazoo, MI). Cage type used for upper or lower spinal levels was not set by a predetermined randomization scheme, but arbitrarily selected by the surgeon. For 3-level procedures, the third level received either a PEEK-Ti or 3D-Ti cage by arbitrary selection. Cage height and lordotic angle used were determined by the surgeon intra-operatively. The technique for which cage height and lordotic angle to implant remained constant for all surgeries and levels. Height of the cage was assessed by using the largest shaver possible to open the interbody space without violating the integrity of the endplate. Cages were packed with the same bone graft material at each level within the same patient. Bone graft materials used in the cage were comprised of either demineralized bone matrix (DBM) putty mixed with morselized autograft or a synthetic bone void filler mixed with morselized autograft. No BMP was used for these patients. All patients underwent posterior fixation and posterolateral fusion. All patients recovered uneventfully with no postoperative implant-related complications and were discharged 3 days after surgery.
Postoperative Follow-up
Patients returned for clinical and radiographic follow-up at 6 months and 12 months postoperatively. Independent radiographic analysis of CT and/or X-ray imaging was performed for each spinal level to assess fusion status, implant subsidence or migration, and any evidence of screw loosening. Fusion was defined as evidence of bridging trabecular bone within the cage and/or outside the cage. Cage subsidence was defined as incursion into one or both vertebral bodies by >20% cage height. This was based on the Marchi, et al classification scheme, modified such that <20% loss of height was considered not subsided (instead of Marchi Grade 0), and subsidence was defined to encompass Marchi Grades I-III. 18 Cage migration was a composite of subsidence, migration, translation, and cage rotation.
Statistical Analysis
Subsidence was available at 1 month, 3 months, 6 months, 12 months, and 24 months, with primary analyses conducted at 6 months and 12 months.
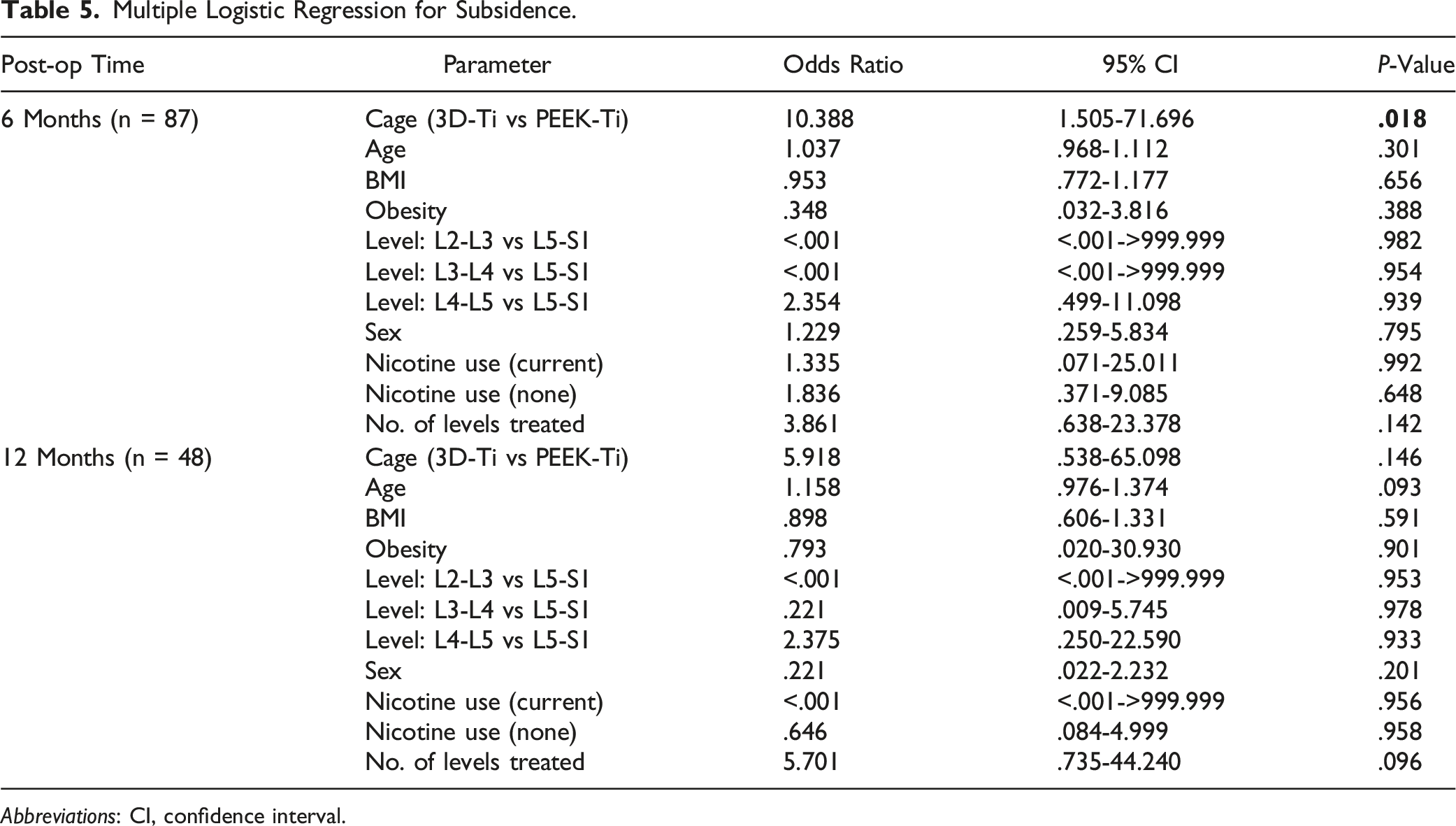
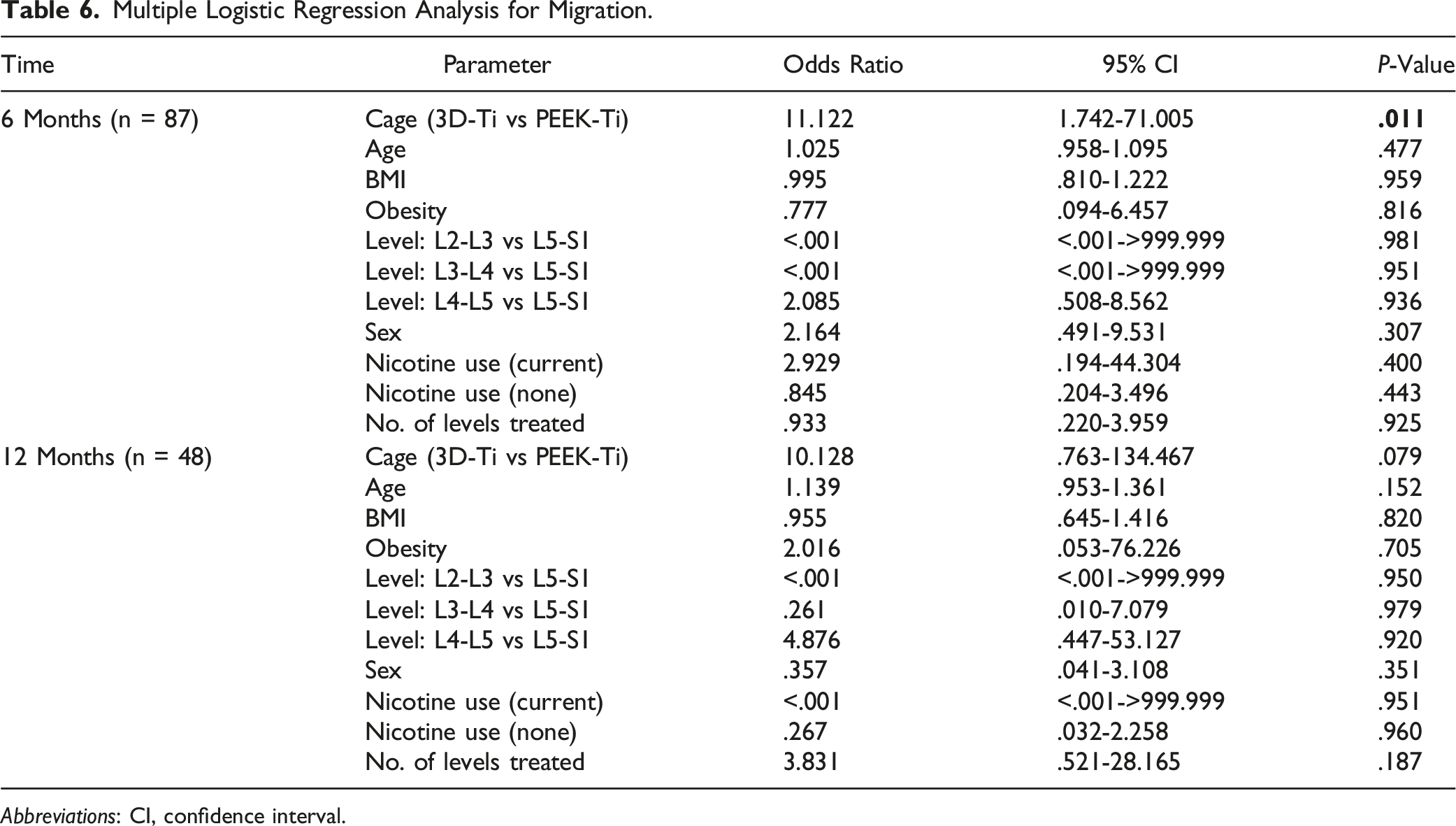
Univariate analysis was conducted to assess the association between subsidence and migration (composite of subsidence, migration, translation, cage rotation) outcomes and cage material (PEEK-Ti, 3D- Ti), age, baseline BMI, baseline obesity, sex, nicotine use, number of levels treated, level treated, graft material usage, and posterior fixation. Fisher’s exact test was used to compare categorical baseline characteristics and binary subsidence and migration outcomes at 6 and 12 months.
Simple logistic regression was performed using a binary subsidence and migration outcome at 6 and 12 months with either cage material or level treated defined as the sole dependent variable. Multivariate logistic regression was performed using a binary subsidence and migration outcome at 6 and 12 months, with cage material, sex, nicotine use, number of levels treated, and level treated as factors with age, baseline BMI, and baseline obesity as covariates. A full model was fit. In addition, the forward stepwise selection method was used; a significance level of .10 was required to allow a variable into the model and a significance level of .10 was required for a variable to stay in the model. For logistic regression analyses, the odds ratio estimate for subsidence and migration based on 3D-Ti compared to PEEK-Ti, along with the 95% Wald confidence interval (CI) P-value, are reported.
Results
Baseline Patient Demographic and Surgical Characteristics.
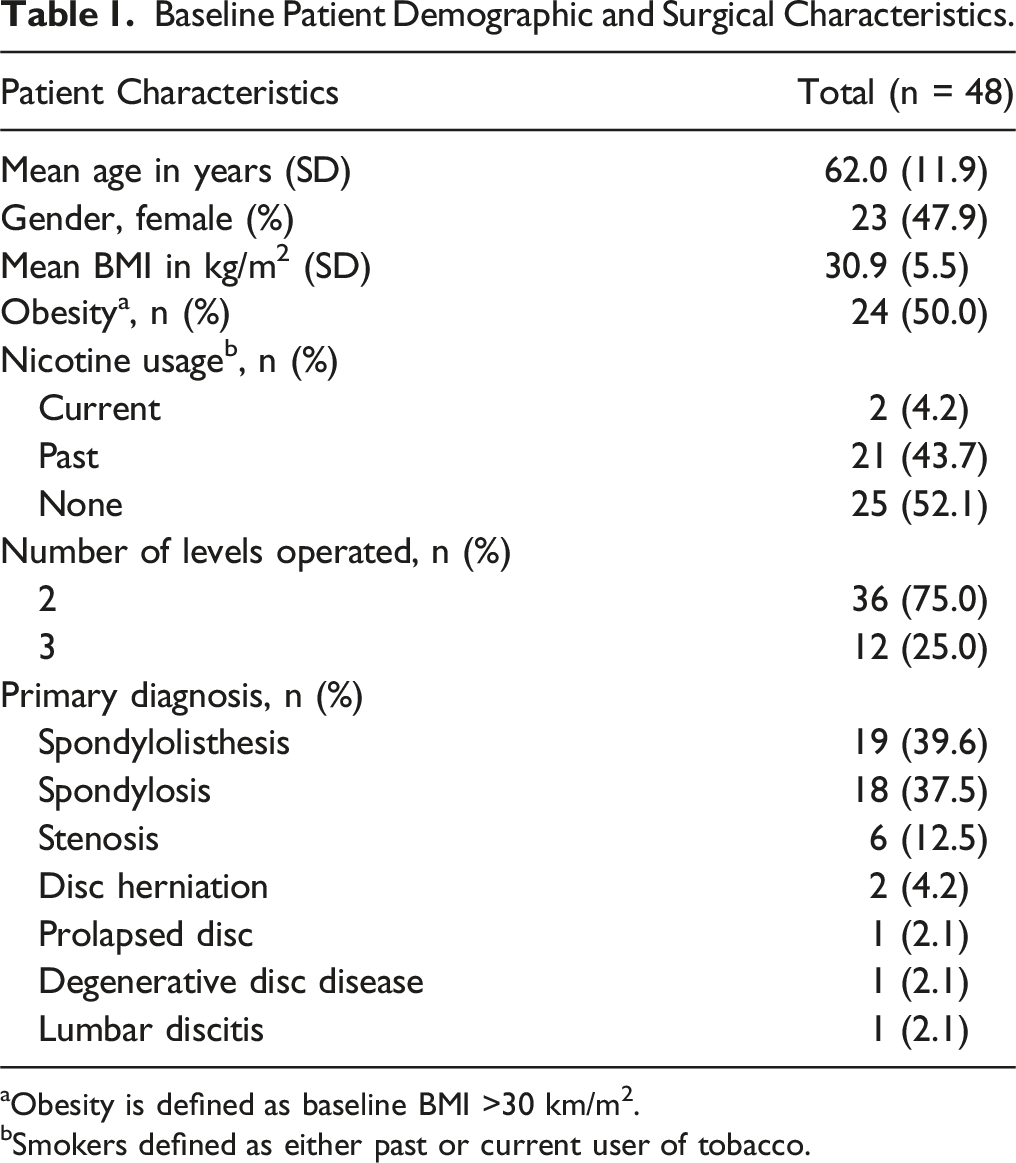
aObesity is defined as baseline BMI >30 km/m2.
bSmokers defined as either past or current user of tobacco.
Treatment Group Characteristics by Spinal Level.
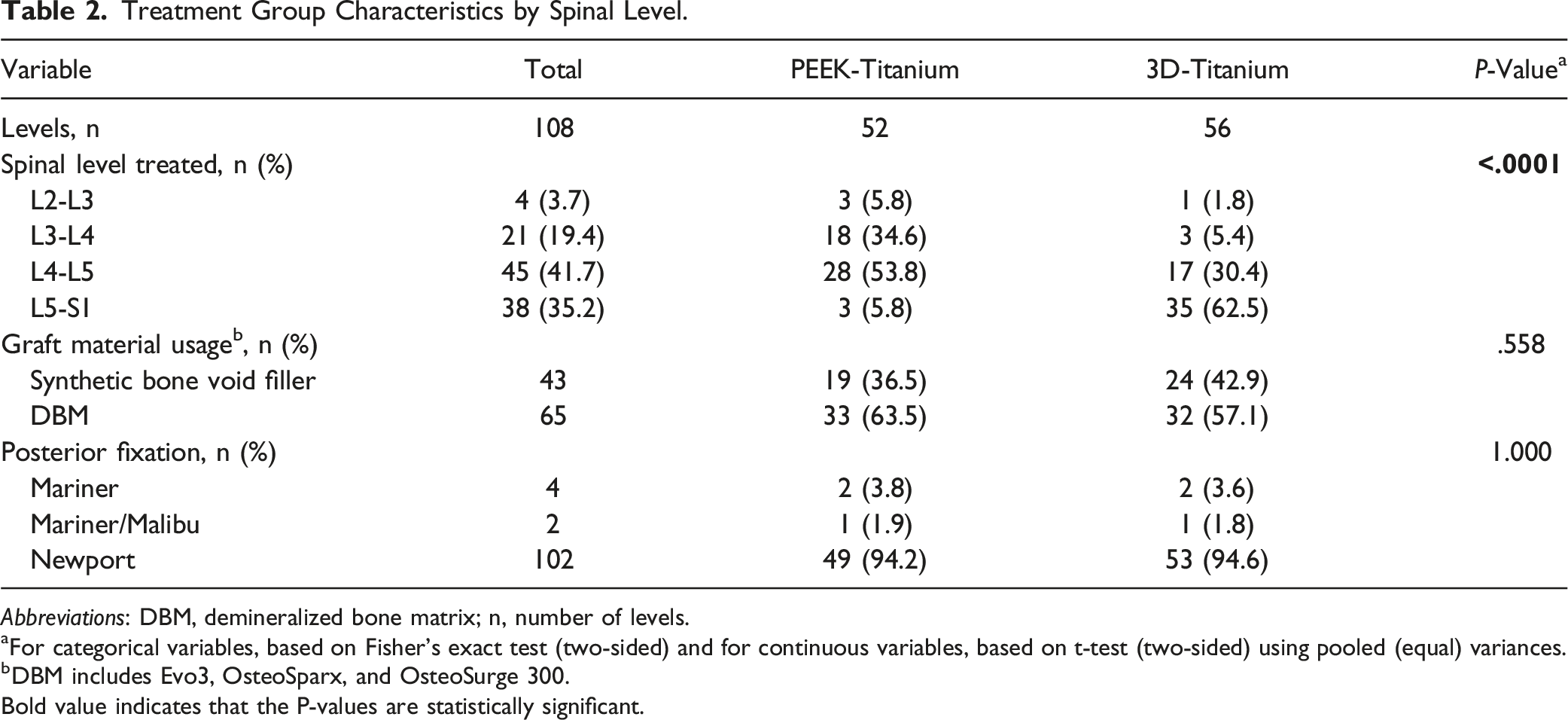
Abbreviations: DBM, demineralized bone matrix; n, number of levels.
aFor categorical variables, based on Fisher’s exact test (two-sided) and for continuous variables, based on t-test (two-sided) using pooled (equal) variances.
bDBM includes Evo3, OsteoSparx, and OsteoSurge 300.
Bold value indicates that the P-values are statistically significant.
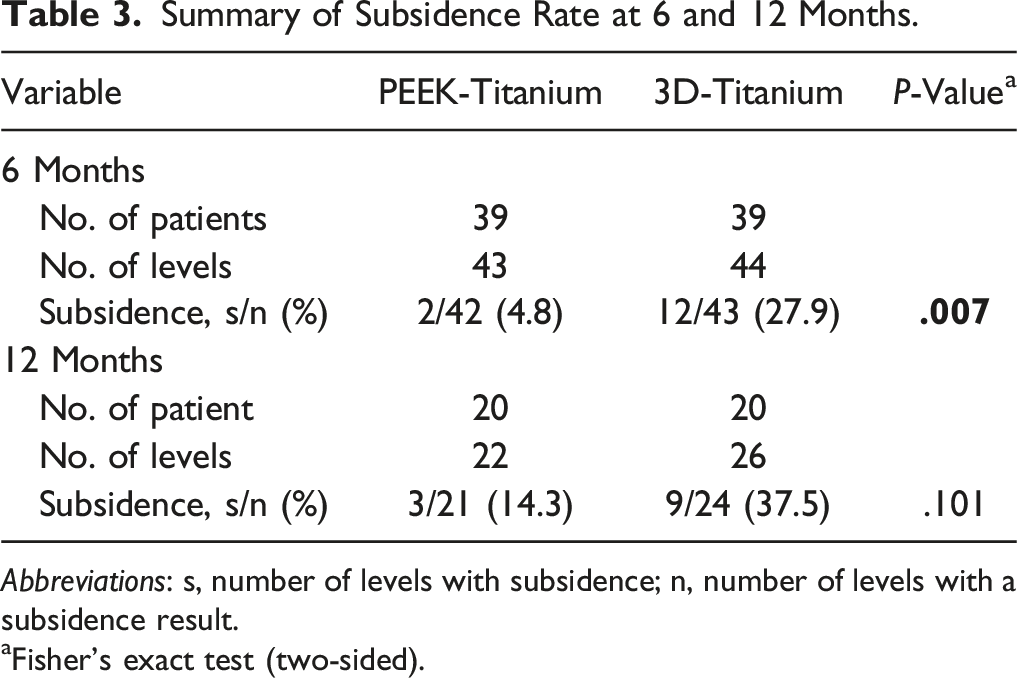
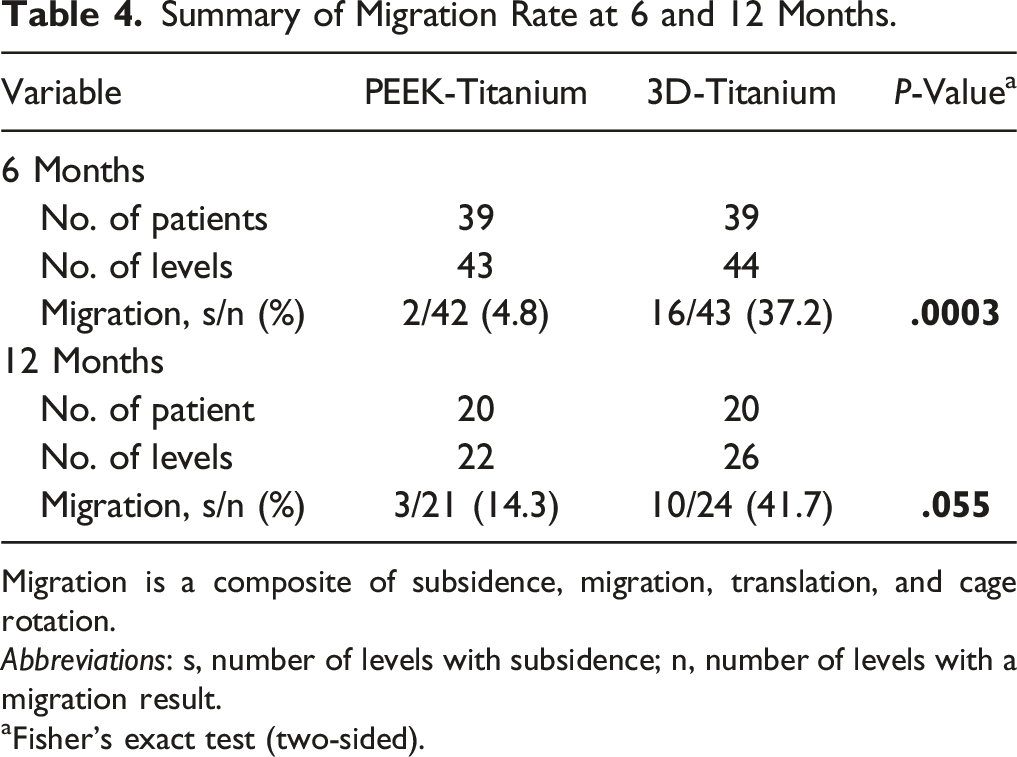
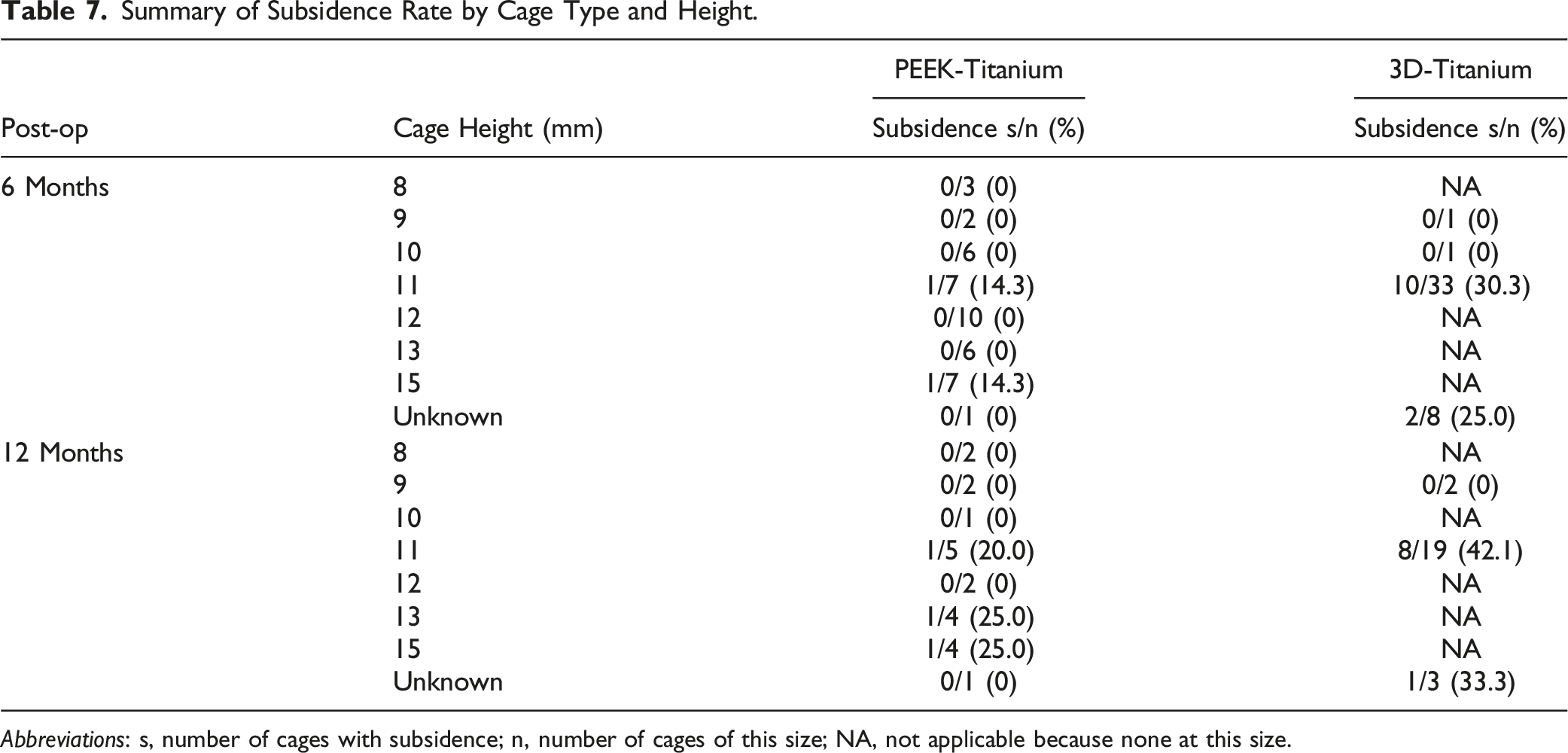
At 6-months post-op, the incidence of cage subsidence was significantly higher for 3D-Ti cages than for PEEK-Ti. Cage subsidence occurred in 27.9% (12 of 43 levels) of 3D-Ti cages, about 6X more often than in PEEK-Ti cages, which had 4.8% (2 of 42 levels) incidence, P < .01 (Figure 1, Table 3). Cage migration also occurred more for 3D-Ti cages, which had 37.2% (16 of 43 levels) migration, compared to 4.8% (2 of 42 levels) for PEEK-Ti, P < .001 (Table 4). Of the patients with cage subsidence and/or migration, there were 2 reports of low-grade residual lower back pain and 1 report of persistent radiculopathy at 6 months post-op. The majority of patients with cage subsidence had symptoms resolve. Results at 12-months post-op were similar, with higher incidence of cage subsidence and migration of 3D-Ti implants than PEEK-Ti implants. At 12-months, 3D-Ti cage migration occurred in 10 of 24 levels (41.7%) compared to only 3 PEEK-Ti levels (14.3%), though the difference was not statistically significant, P = .055. There was one patient with cage subsidence who reported reoccurrence of pain at 12 months post-op and needed reoperation. Spinal level was not a significant variable in subsidence or migration rate differences at either 6-months or 12-months post-op by multiple logistic regression analysis (Tables 5 and 6). At 6-months, subsided 3D-Ti cages were at L4-L5 (41.7%, 5 of 12 cages) and L5-S1 (58.3%, 7 of 12 cages) levels. Migrated 3D-Ti cages had a similar distribution, with 43.8% (7 of 16) at L4-L5, and 56.3% at L5-S1. For PEEK-Ti cages, only 2 cages subsided and both were at L4-L5. Similarly, the heights of the implanted cages, shown in Table 7, were not found to be a factor in cage subsidence, although the small sample size and variation in cage heights between groups did not allow for robust statistical comparison. Overall, the 3D-Ti cages ranged from 9-11 mm in height, with most being 11 mm, whereas the PEEK-Ti cages had a wider range from 8-15 mm, with the most common cage height being 11 or 12 mm. Incidence of subsidence at 6- and 12-month post-operative follow-up for PEEK-Titanium and for 3D-Titanium cages in 2- and 3-level TLIF. * indicates statistical difference, P < .01. Summary of Subsidence Rate at 6 and 12 Months. Abbreviations: s, number of levels with subsidence; n, number of levels with a subsidence result. aFisher’s exact test (two-sided). Summary of Migration Rate at 6 and 12 Months. Migration is a composite of subsidence, migration, translation, and cage rotation. Abbreviations: s, number of levels with subsidence; n, number of levels with a migration result. aFisher’s exact test (two-sided). Multiple Logistic Regression for Subsidence. Abbreviations: CI, confidence interval. Multiple Logistic Regression Analysis for Migration. Abbreviations: CI, confidence interval. Summary of Subsidence Rate by Cage Type and Height. Abbreviations: s, number of cages with subsidence; n, number of cages of this size; NA, not applicable because none at this size.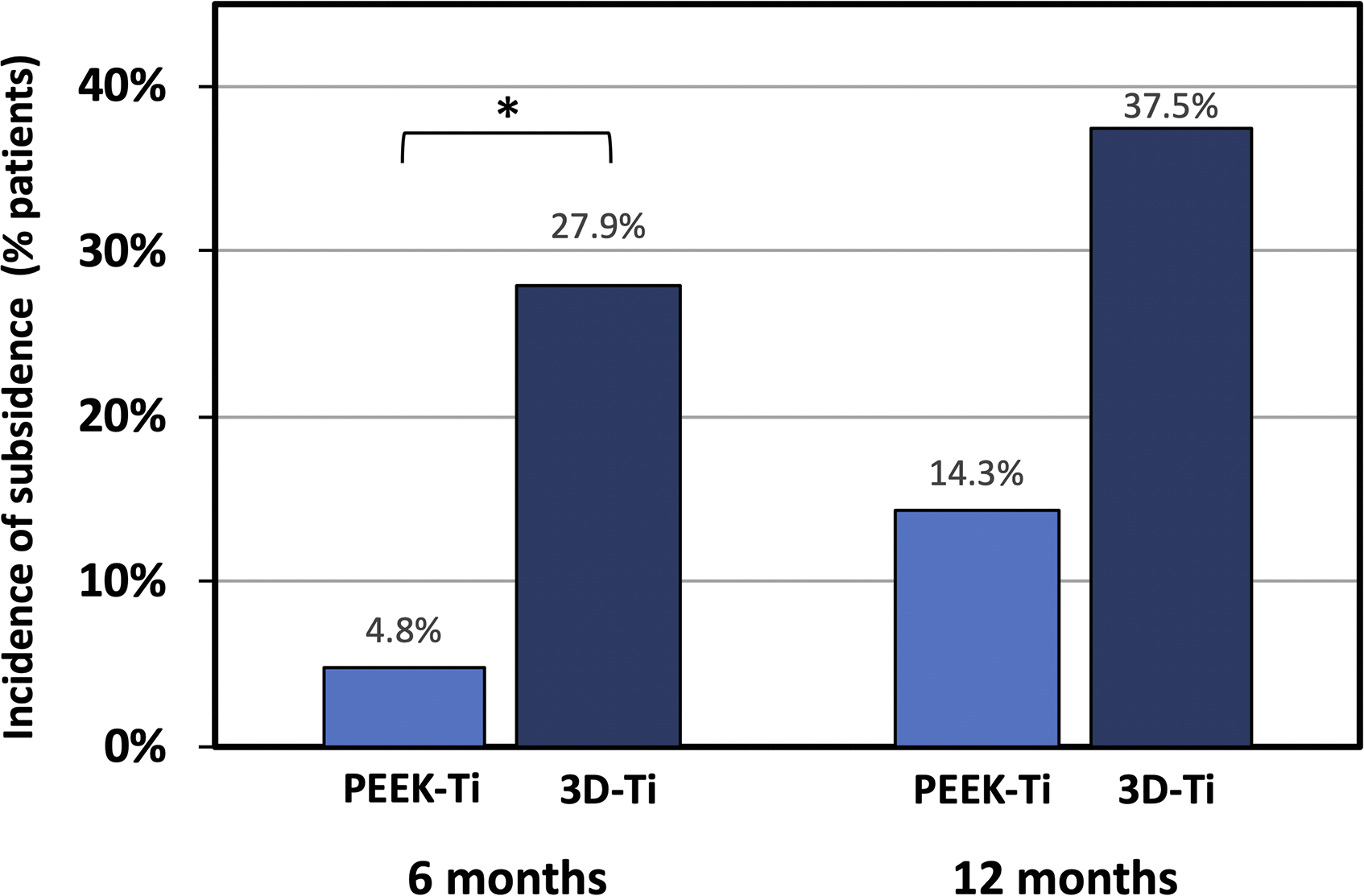
Cage subsidence was not associated with a higher rate of pseudoarthrosis in this study. Fusion rates for the PEEK-Ti and 3D-Ti implants were comparable between groups at both 6 months and 12 months (Figure 2). At 6 months, PEEK-Ti levels showed 100% fusion (42 of 42 levels fused) and 3D-Ti levels had a 95.5% fusion rate (42 of 44 levels fused) (Figure 2). At 12 months, fusion rates were 100.0% (21 of 21 levels fused) for PEEK-Ti implants and 91.7% (22 of 24 levels fused) for 3D-Ti implants. The differences in fusion rates were not statistically significant. Neither spinal level nor bone graft type was a significant factor affecting fusion rate. Figure 3 shows a case example of a 59-year-old male with 2-level TLIF demonstrating bony fusion at 6-months. Bony bridging is visible inside and outside the cages at both levels at 6 months by CT imaging (Figure 3C). At 12-months post-op, the PEEK-Ti cage level, L5-S1, is solidly fused and the 3D-Ti cage at L4-L5 is partially fused but subsided. Another case example in Figure 4 illustrates the progression of bridging bone formation at 6 months into a solid bony fusion at the 12-month follow-up. The progression of new bone formation is occurring at both spinal levels but is particularly pronounced and evident at the PEEK-Ti level, L4-L5, due to greater visibility enabled by the cage material (Figure 4B and C). Fusion rate at 6- and 12-month post-operative follow-up for PEEK-Titanium and for 3D-Titanium cages in 2- and 3-level TLIF. Differences were not statistically significant. Case example of 59-year-old male with 2-level TLIF, with a 3D-Titanium cage implanted at L4-L5 and titanium-surface PEEK (PEEK-Ti) cage at L5-S1. Both cages were filled with a mixture of DBM and autograft. (A) Intra-operative radiograph, (B) 6-month follow-up radiograph, (C) 6-month follow-up CT demonstrating bridging bone present at both levels, (D) 12-month follow-up CT showing PEEK-Ti cage level is fused. 3D-Ti is partially fused but has subsided. Case example of 55-year-old male who underwent 2-level TLIF at L4-S1. (A) Intra-operative radiograph showing placement of PEEK-Ti cage at L4-L5 and 3D-Ti cage at L5-S1. (B) Lateral CT at 6-month follow-up shows subsidence of 3D-Ti cage into the inferior endplate of L5. Bridging bone is observed at both levels. (C) Lateral CT at 12-month follow-up shows fusion at both levels, with substantial subsidence of 3D-Ti cage into the inferior endplate of L5 and moderate subsidence of PEEK-Ti cage into the superior endplate of L5.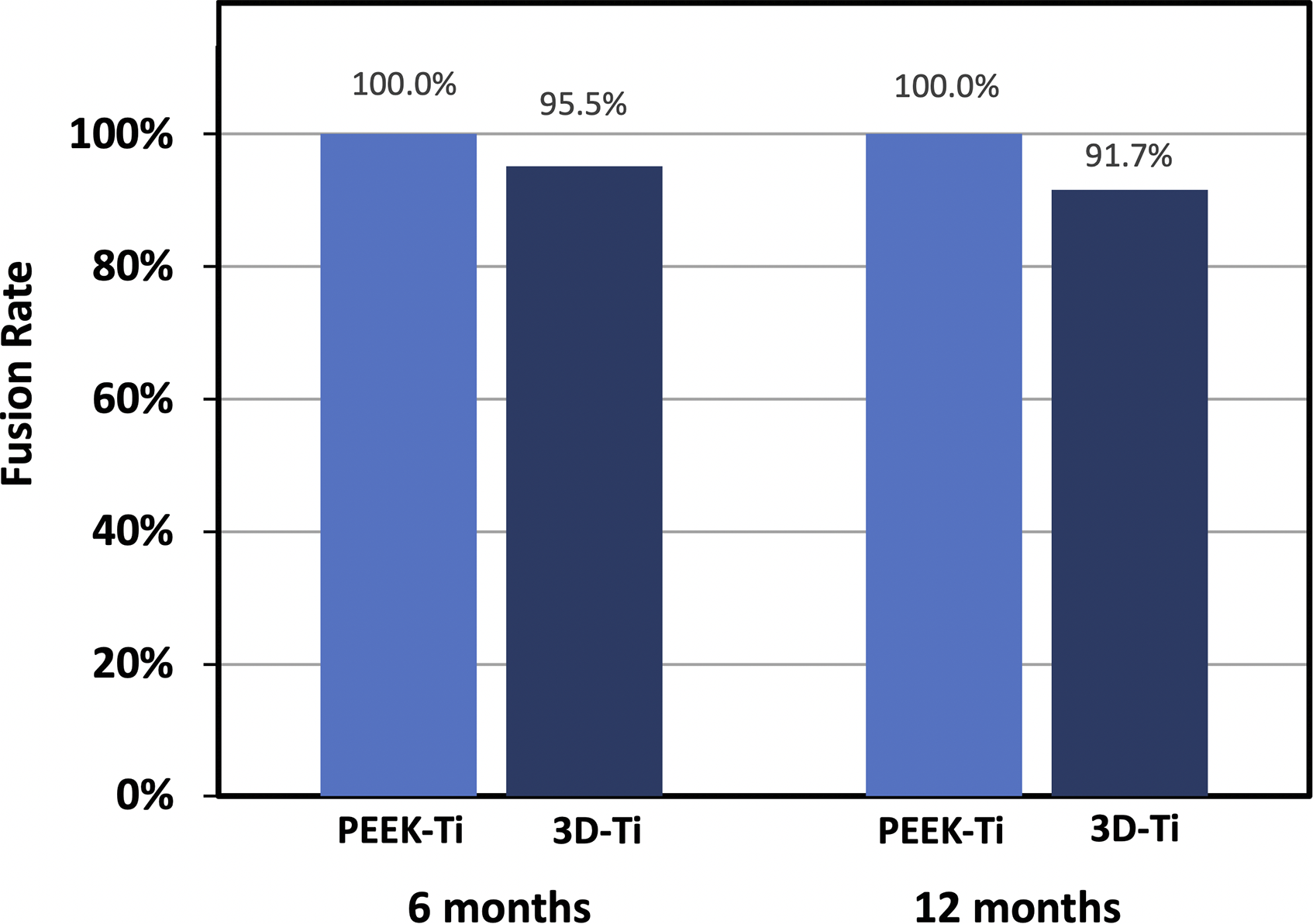
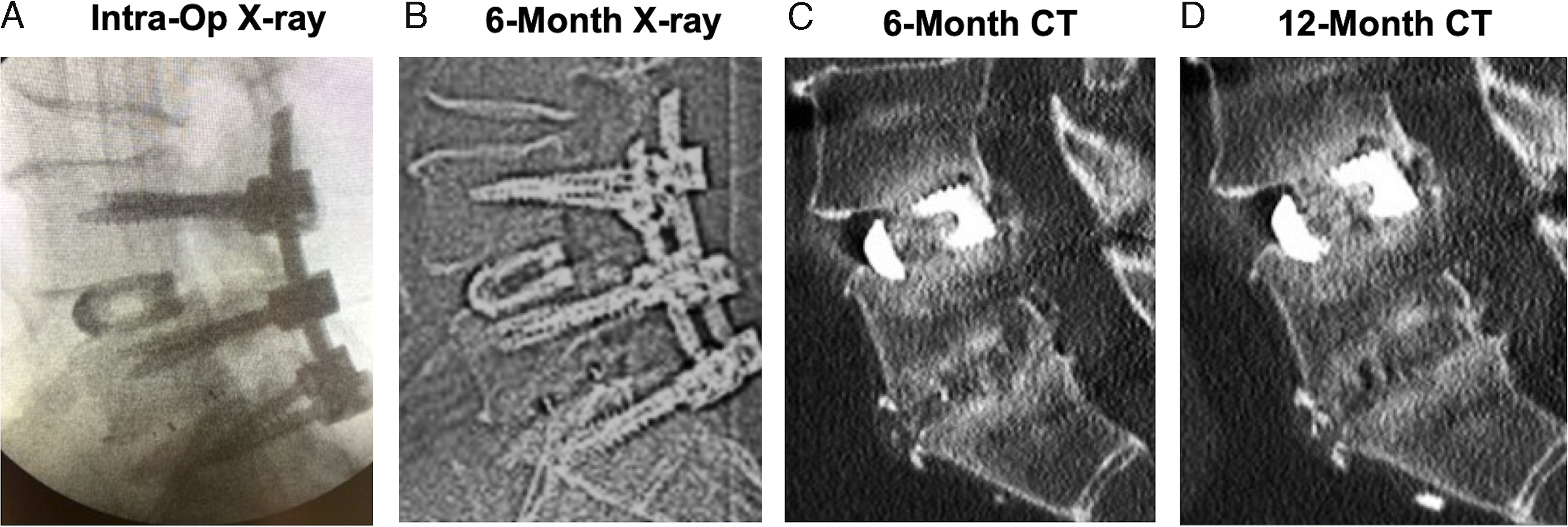
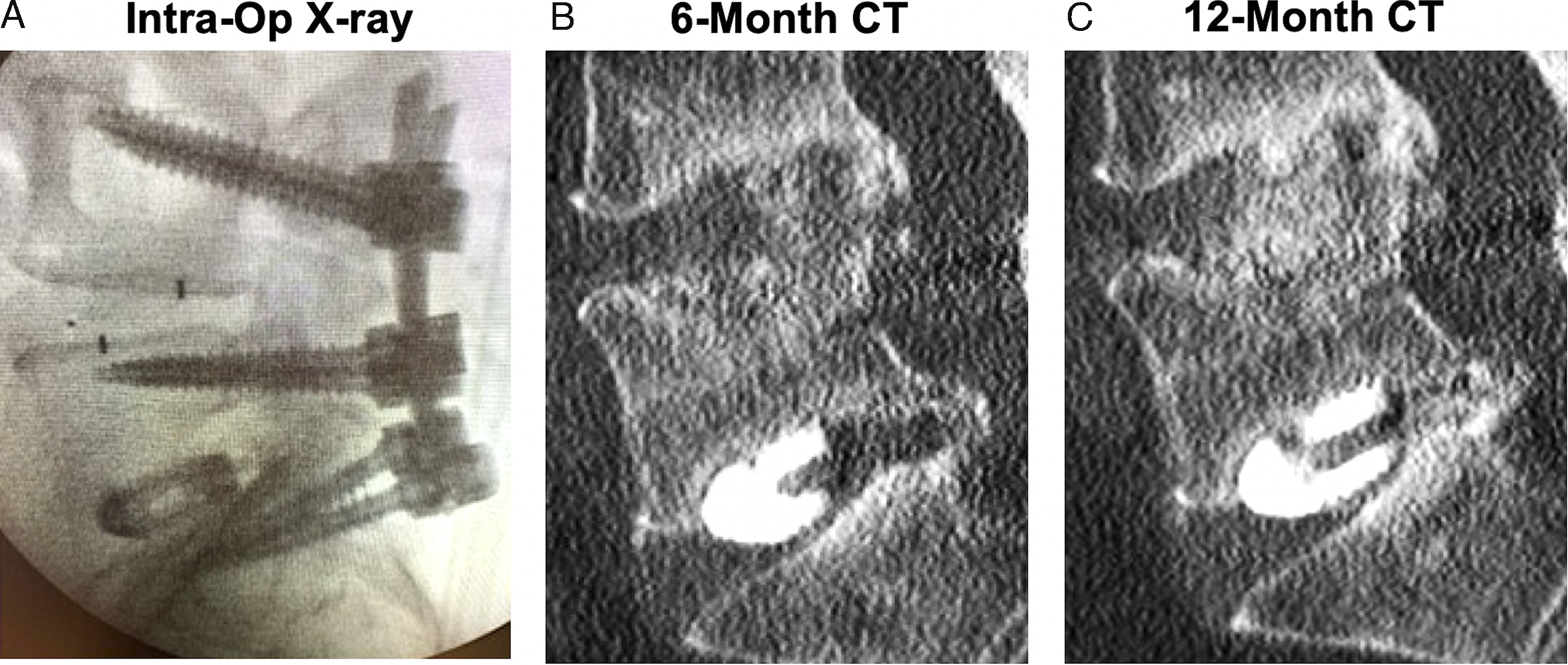
Cage subsidence occurred both superiorly and inferiorly to the implants and was often accompanied by the appearance of bony remodeling. Figure 3 demonstrates cage subsidence in a 2-level TLIF with a 3D-Ti cage implanted at L4-L5 and a PEEK-Ti cage implanted at L5-S1. Both the 6- and 12-month follow-up CTs (Figure 3C and D) demonstrate substantial subsidence of the 3D-Ti cage into the superior endplate of L5. The PEEK-Ti cage at L5-S1 shows moderate subsidence upwards into L5 but to a lesser extent than the incursion of the 3D-Ti cage into L5. Similarly, Figure 4 illustrates a different patient’s 2-level TLIF in which the PEEK-Ti cage was implanted at L4-L5 and 3D-Ti cage implanted at L5-S1. In this case, the 3D-Ti cage also exhibited greater subsidence upwards into the inferior endplate of L5, as seen in the 6-months CT (Figure 4B). The loss of disc height was maintained at the 12-months follow-up (Figure 4C).
Discussion
In this intra-patient-controlled comparative clinical study, the use of titanium-surface PEEK (PEEK-Ti) cages in TLIF resulted in significantly lower rates of cage subsidence and migration compared to 3D-printed titanium (3D-Ti) cages at 6 months post-op, with results persisting at 12 months. PEEK-Ti cages achieved fusion rates of 100% at both 6 and 12 months, similar to 3D-Ti cage fusion rates of 95.5% and 91.7% at the same time points. High fusion rates were achieved for both PEEK-Ti and 3D-Ti cages, which is consistent with findings from previous studies.8,12 The cage subsidence rates in this study of 4.8% to 37.5% over the first 6-12 months post-op are consistent with the reported incidence of cage subsidence in TLIFs ranging from 14.8 to 34.1%, occurring within 7.2 months of surgery.19,20 Overall, the results from this study demonstrate that titanium-surface PEEK cages are safe and exhibit comparable fusion rates at 6 months and 12 months to 3-D printed titanium in TLIF, with significant benefits in lowering the risk of subsidence.
Selection between different cage materials besides titanium alloy or PEEK is a relatively new advancement in spine surgery. Newly developed interbody cage designs, such as titanium-coated PEEK and 3-D printed porous titanium, have been developed to address the limitations of PEEK cages and are most commonly compared against PEEK cages as a control cohort. As such, PEEK-Ti and 3D-Ti cages have each been shown to improve fusion performance while maintaining or lowering subsidence rates when compared to PEEK alone.21-25 In 86 patients with TLIF, PEEK-Ti resulted in higher fusion rates of 91.8% compared to 76% for PEEK at 6-months post-op, with no differences in cage subsidence. 21 Similarly for 3D-Ti, a study of 150 posterior lateral interbody fusion patients had a higher 3D-Ti fusion rate (93% compared to 82% for PEEK) and similar subsidence rate (17.9% compared to 23.4%). 25 Taken together, these results are encouraging, and the current study extends this work by directly comparing PEEK-Ti and 3D-Ti cages within the same patient in multi-level TLIF.
Our study found similar fusion rates and subsidence for 3D-Ti cages at 6- and 12-months, but a significantly lower subsidence rate for PEEK-Ti cages than previously reported in 2 retrospective studies comparing PEEK-Ti and 3D-Ti cages. Makino, et al reported a case series of 63 patients in PLIF using 3D-Ti cages from the same manufacturer as the present study (Tritanium PL from Stryker) but compared to a different PEEK-Ti cage (ProSpace XP from Aesculap). 26 Similar to the current study, fusion rates were not significantly different between 3D-Ti and PEEK-Ti cages at 6- and 12-months. Likewise, their 3D-Ti subsidence rates were similar to our findings, with 20.8% at 6 months and 45% at 1 year. However, their PEEK-Ti cages had significantly higher subsidence rates of 43.7% at 6-months, accompanied by vertebral endplate cysts. 26 A different study by Segi, et al also reported a higher subsidence rate of 28% for PEEK-Ti cages compared to 15% for 3D-Ti (TM Ardis Interbody System by ZimVie) at 1 year. 27 Their 3D-Ti cages had more trabecular bone remodeling and significantly fewer vertebral endplate cysts than the PEEK-Ti cages. However, the cage geometries were vastly different, with the 3D-Ti cages having no space for bone grafting due to their uniform cancellous structure, whereas the PEEK-Ti cages were a mix of different manufacturers’ cages (MectaLIF-TiPEEK from Medacta), Concorde Pro Ti from DePuy Synthes, or Capstone PTC from Medtronic Sofamor Danek) with different geometries but all contianing graft windows. Differences in cage geometry, including microstructure of the cage surface, cage teeth shape, frame thickness, and the presence of bone graft, play a significant role in the bony remodeling response, the formation of vertebral endplate cysts, and cage subsidence. In the current study, the lower incidence of subsidence for PEEK-Ti cages compared to previous studies could be attributable to several factors, including the overall cage geometry or the method of titanium bonding to the surface of the PEEK. Depending on the method of titanium surface coating, some PEEK-Ti cages could be subject to impaction-related wear during cage insertion or wear while under continuous micromotion in an unfused segment.
There are many additional factors affecting the risk of cage subsidence which were outside the scope of the current study. Risk factors for subsidence include patient-associated factors of high body mass index, low bone mineral density, and shorter disc height. 19 In this study, pre-operative disc height was not measured, as it is not part of the surgeon’s standard of care. Surgical risk factors for subsidence include spinal level, with some reports of higher risk of subsidence at L5-S1, as well as cage position, with anterior positioning being more favorable for lowering subsidence risk. Additionally, usage of rhBMP-2 has been found to increase subsidence. Osteolysis was observed in 24.7% of TLIF levels with rhBMP-2, with subsidence occurring in 31.6% of patients with osteolysis. 28 Our study was not designed or statistically powered to assess the impact of these factors. The design of the study as an intra-patient-controlled study was aimed to eliminate variation between groups, including patient-associated risk factors such as age, obesity, and nicotine use. In addition, cage position, cage size, and bone graft were also held similar between groups by using the same surgical technique. Because this study was a retrospective data collection, assessment of cage position immediately post-operatively was not measured and is not available to compare between groups. Finally, spinal level was not able to be controlled or randomized. Retrospective analysis revealed that more PEEK-Ti cages were implanted at L4-L5 (n = 28) than at other levels, whereas more 3D-Ti cages were implanted at L5-S1 (n = 35). To assess whether this affected the results, logistic regression analysis was performed for spinal levels L2-L3, L3-L4, or L4-L5 compared to L5-S1. Odds ratios and 95% confidence intervals were calculated and spinal level was not a significant parameter. In particular, there was no difference found between spinal levels L4-L5 and L5-S1 for subsidence rate (odds ratio, 2.35; 95% confidence interval, .50-111; P = .94) or for migration rate (odds ratio, 2.09, 95% confidence interval, .51-8.56; P = .94) (Table 4 and 5). At 6 months, 3D-Ti cage subsidence occurred roughly evenly across both spinal levels (5 of 12 at L4-L5 and 7 of 12 at L5-S1), and subsidence of PEEK-Ti cages only occurred at L4-L5. If there is an effect of spinal level on cage subsidence and migration, the current study was not statistically powered to uncover it. Future prospective studies are needed to address this limitation through randomization of cage type among different spinal levels.
The current study is limited by its retrospective design, with follow-up results reported only up to 12 months, although the follow-up range was up to 26.2 months. There was a scientific interest in prospectively implanting patients with both cages; however, the data was analyzed retrospectively for fusion and migration upon seeing differences in outcomes between the cages. Finally, patient-reported clinical outcomes were not collected because each patient received both test implants (PEEK-Ti and 3D-Ti cage), such that comparisons between groups were not possible. Nonetheless, the intra-patient, single-surgeon design of the current study provides a controlled comparison of titanium-surface PEEK vs 3D-titanium cages in TLIF. This allows for a direct comparison of the cage types and eliminates the variability of using different patient cohorts or historical controls. This within-patient concurrent controlled study design has been utilized in spine surgery as an effective strategy to mitigate the methodological and logistical challenges of randomized controlled trials and to help reduce the required sample size. 29
The lower subsidence rates demonstrated by PEEK-Ti compared to 3D-Ti in this study suggests that a combined titanium-surface PEEK implant can confer the advantages of both materials – namely, the osseointegration benefits of the titanium surface, combined with the favorable biomechanical properties of PEEK for the cage bulk material. The mechanisms by which PEEK-Ti may protect against subsidence require further study. Previous studies have demonstrated that the titanium surface of PEEK-Ti promotes higher bone ongrowth and higher osteoblastic adhesion and activity, relative to PEEK. 13 The titanium surface particles may sustain stable endplate-graft contact to facilitate earlier bony bridging through the bone graft window. 30 3D-printed titanium cages would have similar initial bone-bonding properties, but may still subside long-term from a mismatch in modulus with the patient’s bone. The role of the titanium cage in the occurrence and timing of subsidence remain unclear. 3D-Ti cages from different manufacturers can vary widely in stiffness and strength due to different porosity or structural design features, as well as bone-implant contact area or surface roughness, which could all impact stress shielding over time. It is unknown if the use of 2 different cages in the same patient affects overall outcomes, fusion, subsidence or migration, and specifically if it played a role in the disparity of subsidence and migration rates observed in this study.
The results from this study may not be generalized across technologies due to differences in cage designs. For example, different methods may be used to bond the titanium coating on the PEEK implant, which can affect mechanical wear and other material properties. The commercially available PEEK-Ti implant used in this study had molecularly bonded pure commercial titanium encompassing the implant, unlike the previously studied PEEK-Ti implants which used different coating techniques. For 3D-printed titanium cages, there are various interbody geometries that may not perform the same biomechanically or biologically. These geometries include, but are not limited to, jointed structures such as a truss, lattice, diamond, or micropore design and non-jointed or wave-like structures such as a gyroid-based design. The 3D-Ti cages tested in this study had a micropore design, differing from the PEEK-Ti cages not only in material type, but also in cage geometry and cross-sectional area. With numerous geometries, 3D-Ti implants may not be easily generalized across all cages. Therefore, it is not possible to conclude with confidence that the differences in subsidence rates found in this study are attributed to the material of the implants alone.
Additional prospective, randomized studies of PEEK-Ti as compared to 3D-titanium cages of various designs are warranted, especially with larger patient numbers, patient-reported outcome measures and long-term follow-up. Ultimately, we seek to improve patient outcomes. Therefore, our future studies will concentrate on the correlation of cage material and design, and patient surgical outcome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding support was provided by SeaSpine, Inc. for retrospective data analysis and publication processing charges.
