Abstract
Study Design
A retrospective cohort study.
Objective
The goal of this study was to develop a useful clinical prediction nomogram to accurately predict the cancer-specific survival (CSS) of patients with primary spinal cord tumor (SCT), thereby formulating scientific prevention and aiding clinical decision-making.
Methods
In this study, patients with SCT diagnoses from the surveillance, epidemiology, and end results (SEER) database (2000-2018) were taken into account. Initially, a nomogram was created using the CSS-associated independent factors that were determined from both univariate and multivariable Cox regression analyses. Furthermore, the nomogram’s capacity for calibration, ability to discriminate, and actual clinical effectiveness were assessed through calibration curves, receiver operating characteristic (ROC) curves, and decision curve analysis (DCA), respectively. Finally, a strategy for categorizing SCT patients’ risk was developed.
Results
This study included 909 SCT individuals. A novel nomogram was developed to forecast SCT patients’ CSS, taking into account age, histological type, tumor grade, tumor stage, and radiotherapy. These factors were identified as independent prognostic indicators for CSS in SCT patients. Elderly SCT patients with distant metastasis, advanced tumor grade, received radiotherapy, and confirmed lymphoma have a poor prognosis. Meanwhile, the risk classification system could differentiate SCT patients and realize targeted management.
Conclusions
The developed nomogram has the ability to accurately forecast the CSS in SCT individuals, aiding in precise decision-making during clinical practice, enhancing health planning, maximizing treatment advantages, and ultimately improving patient prognosis.
Introduction
Primary spinal cord tumor (SCT) is relatively rare, accounting for 5% of central nervous system (CNS) tumors.1,2 It was reported that the total age-specific incidence rates for children (0-19 years old) and adults (≥20 years old) were .27 and 1.27 per 100 000 people, respectively. 3 They were cauda equina tumors (4.0%), spinal meninges (26.0%), and the spinal cord (70.0%), in order of the incidence of tumor occurrence. 4 In the early stage of the tumor, some patients have no apparent symptoms or even no symptoms, so it is easy to ignore and delay consultation time. As the disease progresses, the outward growth of the tumor is restricted by the surrounding bony structures. It can only compress the spinal cord or nerve roots, thus causing neurological dysfunction, such as urinary dysfunction or even paraplegia, which seriously affects the quality of patients’ daily lives. 5
To close the gaps in this research field, Engelhard et al published a description of the clinical presentation, histology, and management of 430 individuals with primary SCTs as early as 2010. 1 Then, in 2012, Duong et al described the epidemiology of SCTs in detail with a larger sample of data, which helped improve the diagnosis and treatment of SCTs and opened the way for subsequent studies. 3 In addition, with the promotion and application of magnetic resonance imaging (MRI), the optimization of intraoperative microsurgical equipment, and the refinement of operator microsurgical techniques, most SCT patients can achieve complete tumor resection and obtain a good prognosis. However, even with these conditions becoming perfect and mature, some patients still have a poor prognosis.
Therefore, it is essential to perform a distinct examination in order to identify the most pertinent prognostic factors associated with the survival rate of SCT individuals and implement personalized strategies to enhance the efficacy of the therapy. However, as far as we know, there has been no investigation into the creation of predictive models for determining the survival of SCT patients based on more comprehensive factors. In comparison to overall survival (OS), cancer-specific survival (CSS) provides a more intimate correlation with patient prognosis influenced by tumors and provides more accurate recommendations for treating such individuals. Hence, CSS served as the final objective of this investigation, with the goal of discovering independent prognostic indicators associated with the CSS of individuals with SCTs. To do this, relevant data from the surveillance, epidemiology, and end results (SEER) database were examined, and a novel nomogram and risk categorization system were developed.
Methods
The Database
The SEER dataset gathers information from 18 registries, encompassing approximately 30% of the population in the United States. This comprehensive cancer database offers valuable evidence to support medical practice and research worldwide. The SEER database was utilized to extract the pertinent information of all patients with SCT using SEER Stat 8.3.9.2. With reference number 16336-Nov2020, the data was collected from 2000 to 2018. The SEER database is accessible to the public, and the obtained data does not reveal individual personal details. As a result, there is no need for approval from the ethics committee or informed consent from the patient.
Patient Selection
Our study’s inclusion criteria were as follows: (1) patient’s primary sites were spinal meninges (C70.1), spinal cord (C72.0), and cauda equina (C72.1); 6 (2) presence of a primary tumor; (3) availability of complete follow-up data; and (4) classification of patient’s death as ‘alive’ or ‘dead due to cancer’. The exclusion criteria were as follows: (1) excluding cases that were not the primary tumor; (2) excluding cases where information about age, sex, race, marital status, tumor stage, tumor grade, surgery, radiotherapy, chemotherapy, and the ICD-O-3 histology/behave were unknown; and (3) excluding cases with a survival time of less than 1 month. Finally, a total of 909 SCT patients were included in this study and were randomized, with a 7:3 ratio, into a training group comprising 70% of the patients and a validation set of 30%. The training group was used to determine the independent prognostic predictors and establish the prognostic nomogram and risk classification system for SCT patients. Subsequently, the nomogram and the risk classification system were confirmed using the validation set.
Variable Definition
This study examined various variables, such as patients’ demographic features (including age, sex, race, and marital status), disease features (histological type of tumor according to ICD-O-3, grade of tumor, and stage of tumor), as well as treatment details (including surgery, radiotherapy, chemotherapy, and the sequence of surgery and radiation). The optimal age cut-off values, determined by the X-tile software, were 52 and 66 (Supplemental file 1). 7 There were 3 categories for races: white, black, and other. The categories of sex were male and female. The categories for marital status were married and unmarried. Yes and no categories were assigned to surgery, radiotherapy, and chemotherapy. The classification of tumor grade included grades I, II, III, IV, B-cell/pre-B/B-pre, and T cell. The classification of tumor stage included local, regional, and distant categories. The histological type was divided into ependymoma, lymphoma, glioma, embryonal tumor, and others based on the new 2021 WHO Classification of Tumors of the Central Nervous System (fifth edition). 8 The main endpoint of our study was CSS, which was defined as the duration from the day of diagnosis until death solely caused by this tumor (alive or dead due to cancer).
Statistical Analysis
This study utilized SPSS (22.0) and R software (4.0.3). To demonstrate the substantial differences among the included factors, initial analysis involved the utilization of Kaplan-Meier survival curves and univariate Cox regression analysis. Univariate Cox regression analysis included variables such as age, sex, race, marital status, tumor grade, histological type, tumor stage, surgical procedure, radiotherapy, chemotherapy, and the sequence of surgery and radiation. Then, the variables with P-values less than .05 were selected to perform multivariable Cox regression analysis in order get rid of any confounding influences between the factors associated to CSS mentioned above and to identify the independent prognostic predictors of CSS for SCT patients. A nomogram was created using the independent prognostic indicators listed above to forecast the 3-, 5-, and 10-year CSS for SCT patients. In the meantime, each independent prognostic predictor connected to CSS was provided with a specific point via this nomogram. One can trace a vertical line from the total score row to the bottom timeline to determine the patient’s 3-, 5-, and 10-year mortality risk. Next, calibration curves for 3, 5, and 10 years were created in order to verify the nomogram’s accuracy. The bootstrap-corrected concordance index (C-index) was used to assess the recognition ability of the nomogram. Additionally, decision curve analysis (DCA) was used to show how useful the nomogram was. Furthermore, receiver operating characteristic (ROC) curves for CSS at 3, 5, and 10 years were created utilizing the area under the curve (AUC) values of the corresponding variables in order to evaluate the nomogram’s capacity to distinguish. Furthermore, the scores of every CSS-associated individual prognostic factor were combined to determine the patient’s risk score for mortality, and the optimal threshold for the risk score was determined by X-tile software. Subsequently, a system for categorizing the mortality risk of SCT patients into subgroups of low, middle, and high risk was developed. Ultimately, distinctions among the 3 subcategories were acquired by utilizing Kaplan–Meier survival curves.
Results
Baseline Features
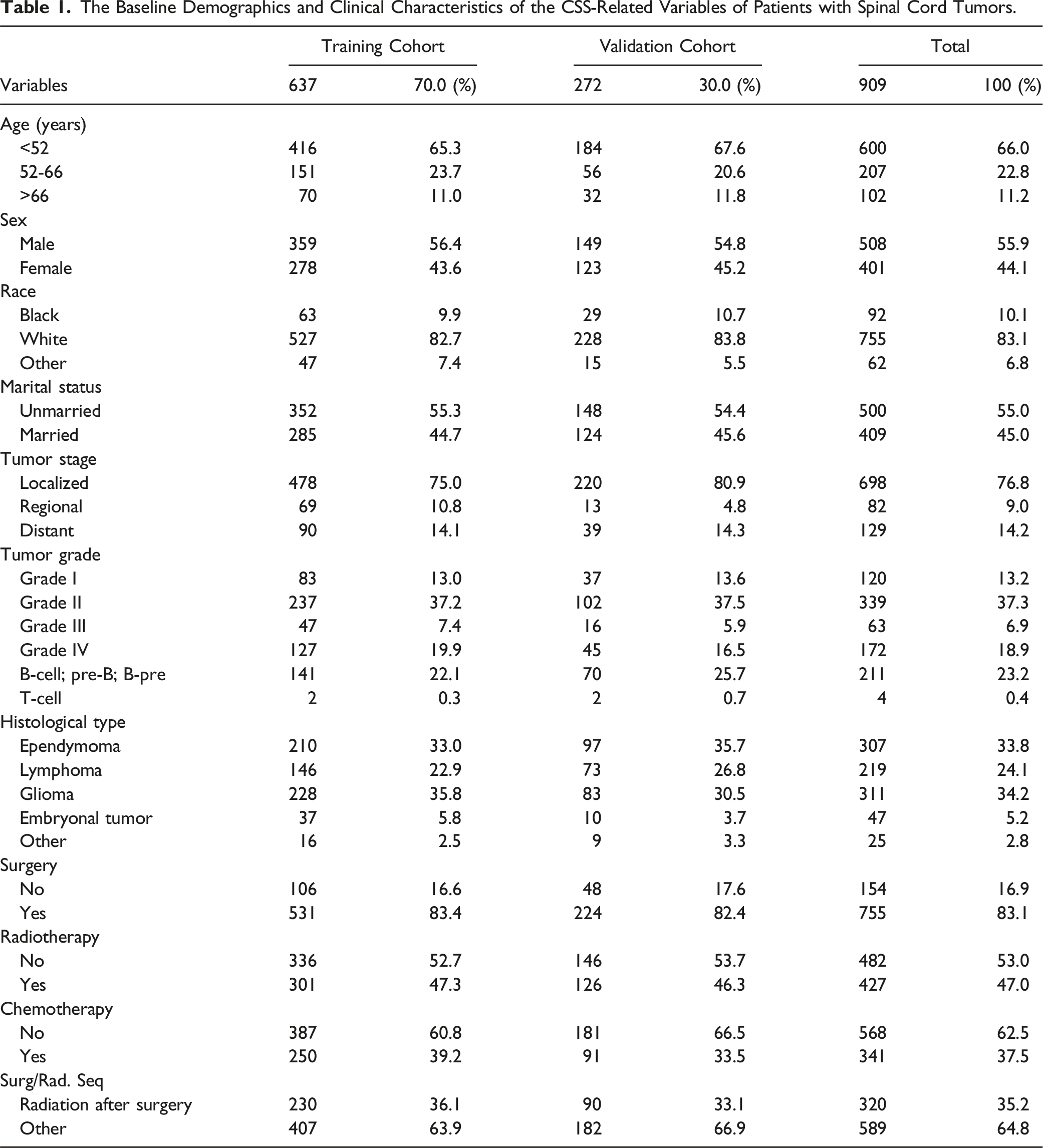
The Baseline Demographics and Clinical Characteristics of the CSS-Related Variables of Patients with Spinal Cord Tumors.
Determination of the Independent Prognostic Variables
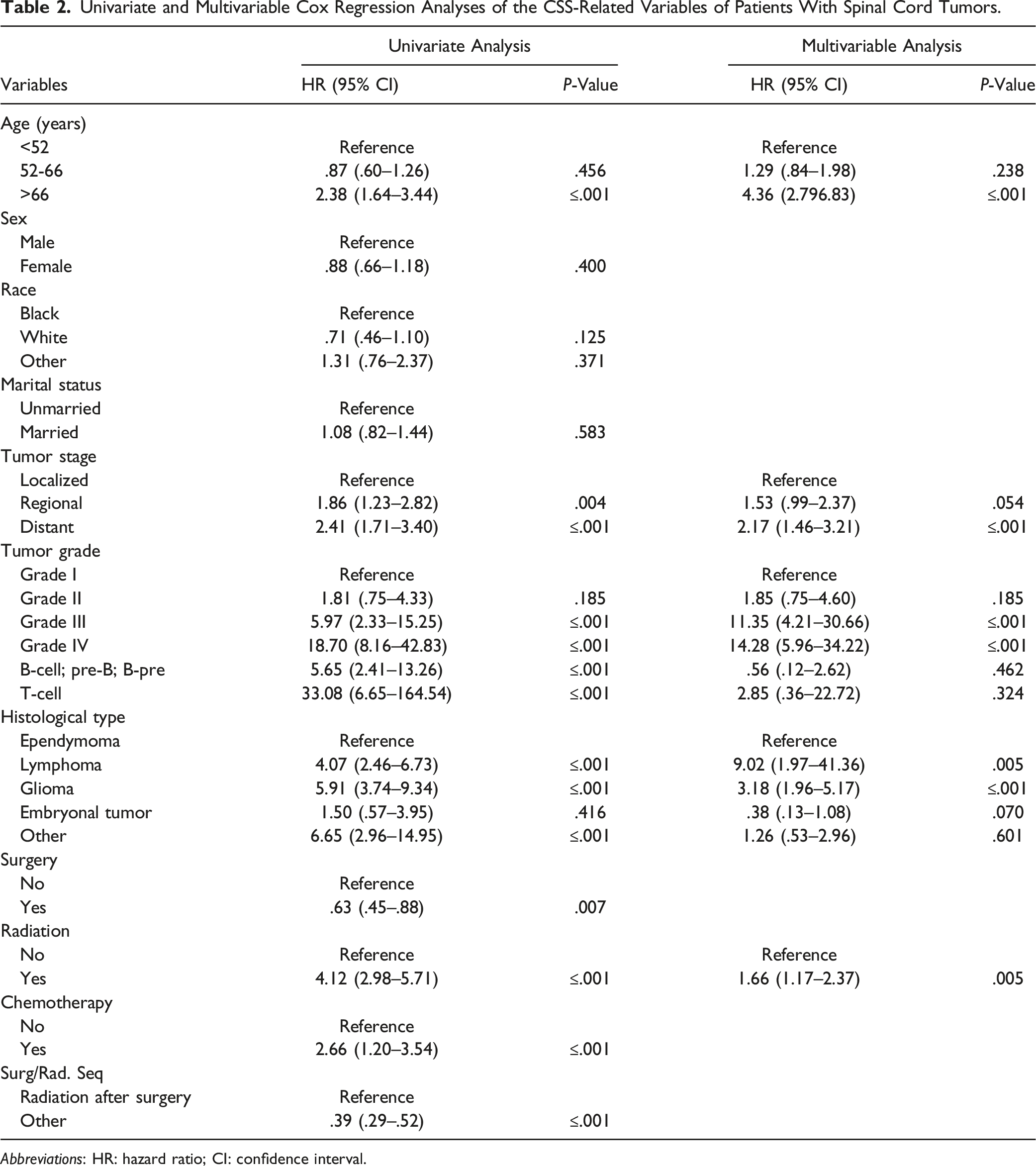
This study conducted univariate and multivariable Cox regression analysis to investigate independent prognostic factors of CSS in SCT patients. The findings of univariate Cox regression analysis indicated that variables such as age, race, tumor grade, histology, tumor stage, surgical procedure, radiotherapy, chemotherapy, and sequence of surgery/radiation were determined to be associated with CSS, whereas marital status and sex did not exhibit significant differences (Figure 1). The findings of multivariable Cox regression analysis revealed that age, tumor grade, histological type, tumor stage, and radiotherapy emerged as independent prognostic indicators for SCT patients’ CSS (Table 2). Kaplan–Meier curves were performed on the cancer-specific survival (CSS) variables of spinal cord tumor patients. (A) Age, (B) sex, (C) race, (D) marital status, (E) radiation, (F) chemotherapy, (G) surgery, (H) tumor stage, (I) tumor grade, (J) histologic type, and (K) radiation sequence with surgery. Univariate and Multivariable Cox Regression Analyses of the CSS-Related Variables of Patients With Spinal Cord Tumors. Abbreviations: HR: hazard ratio; CI: confidence interval.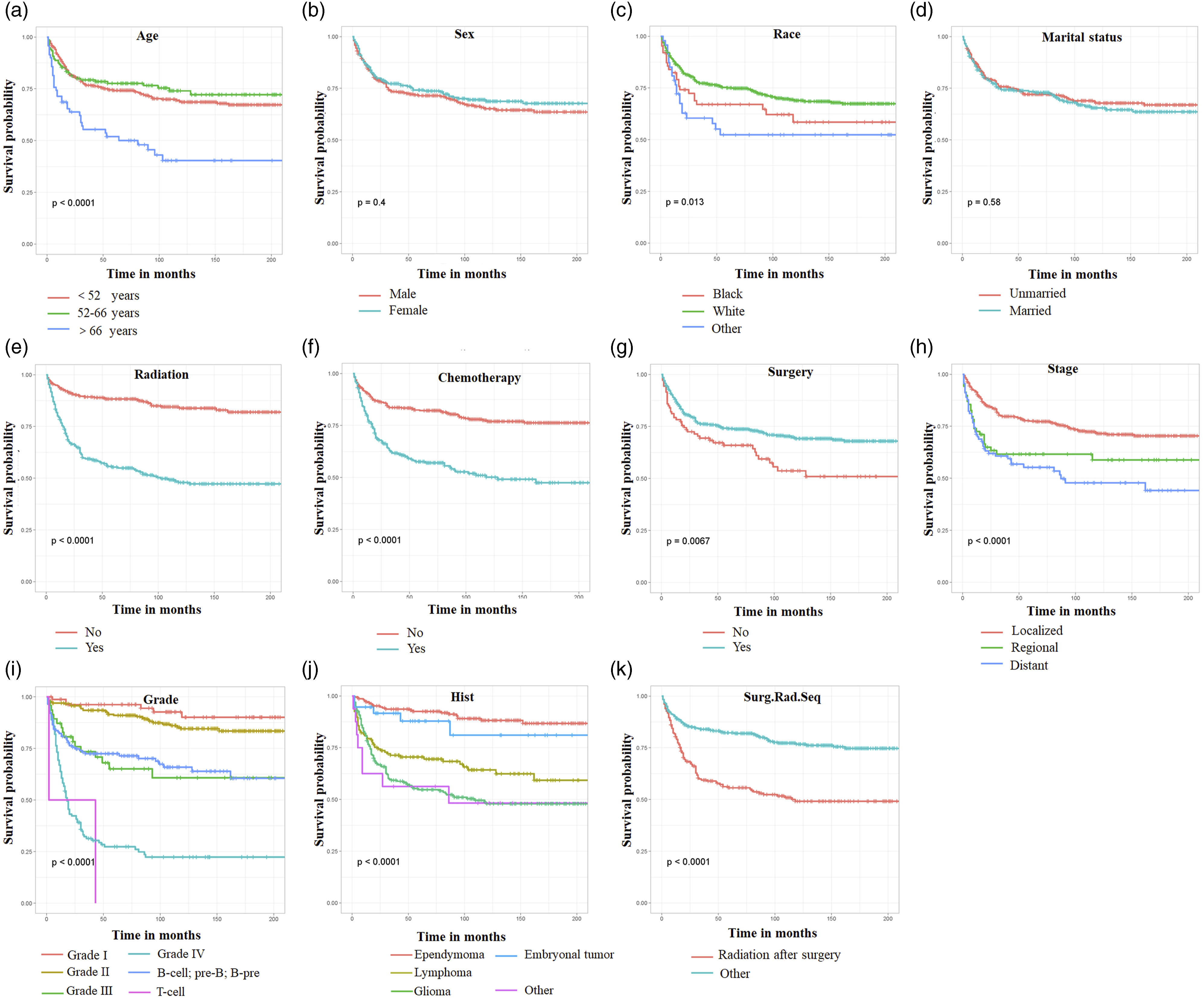
Development and Verification of the Prognostic Nomogram
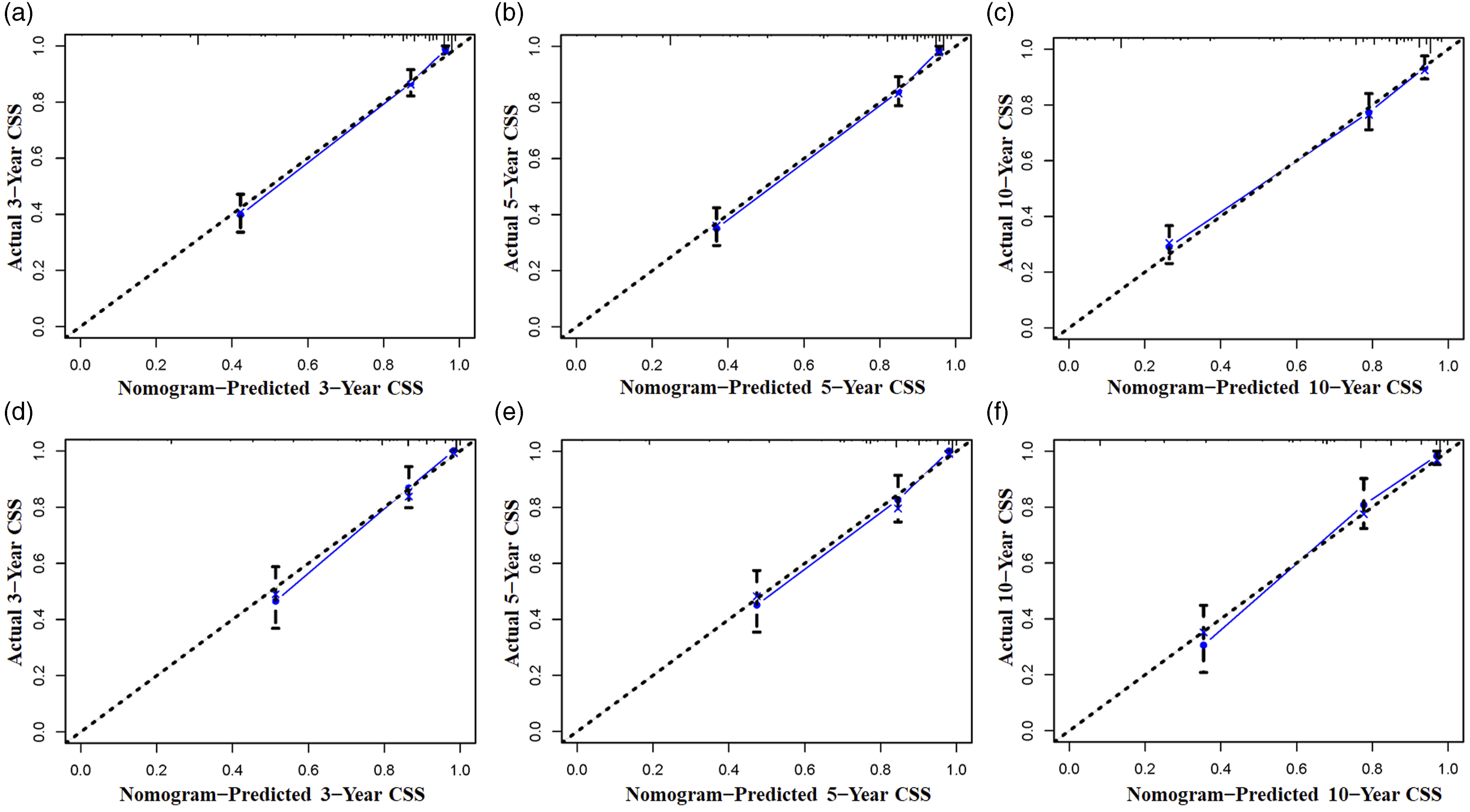
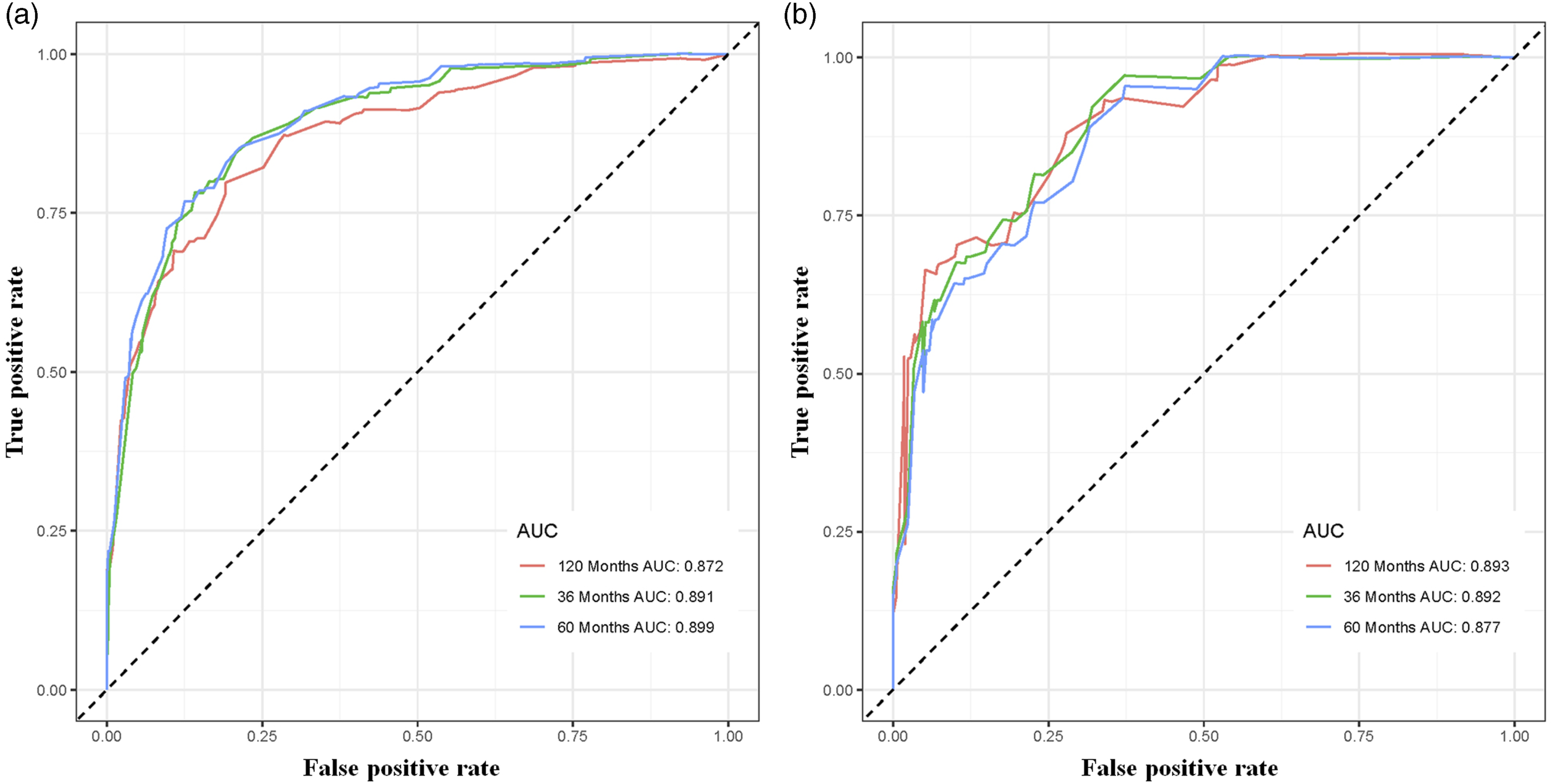
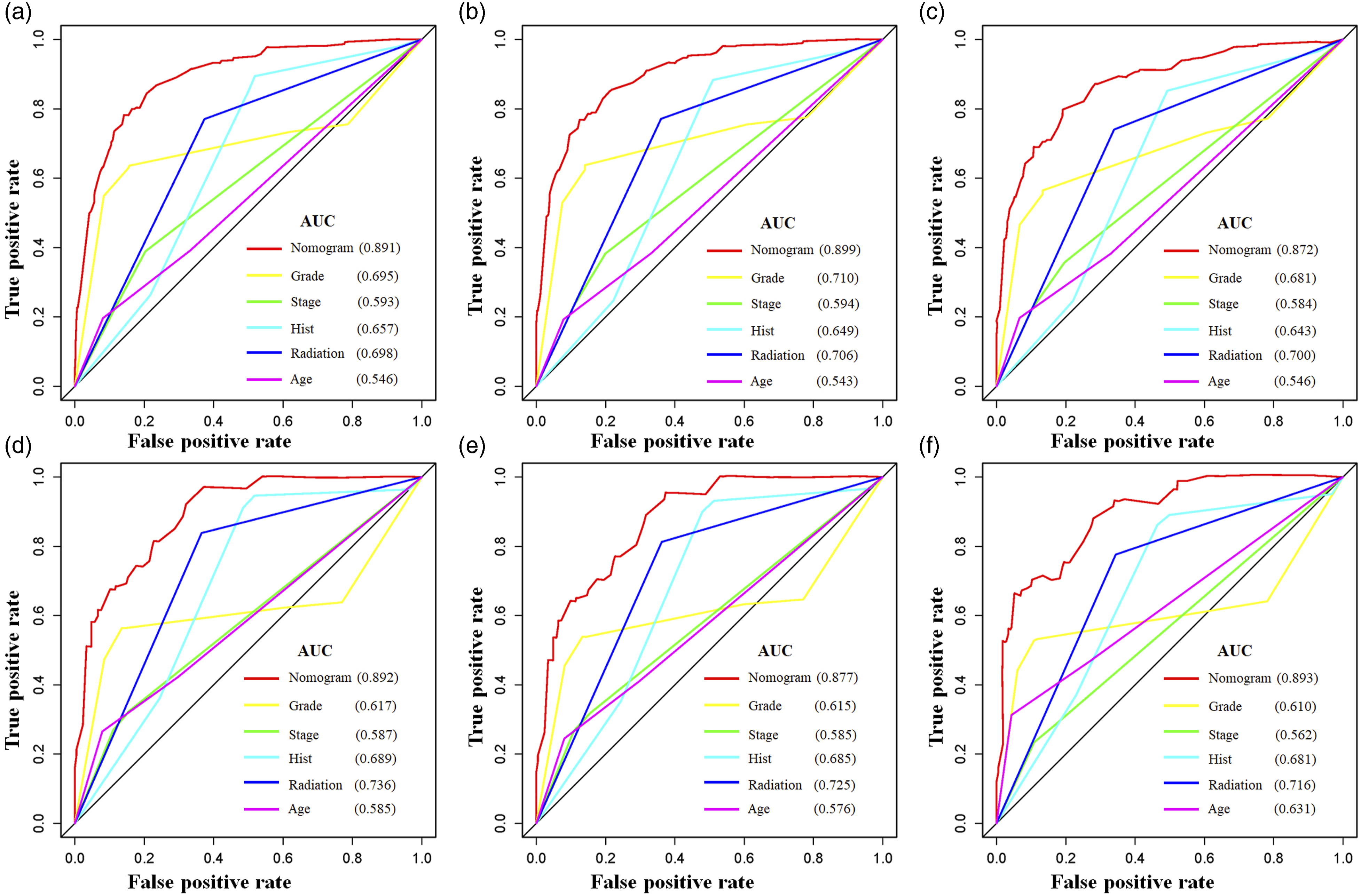
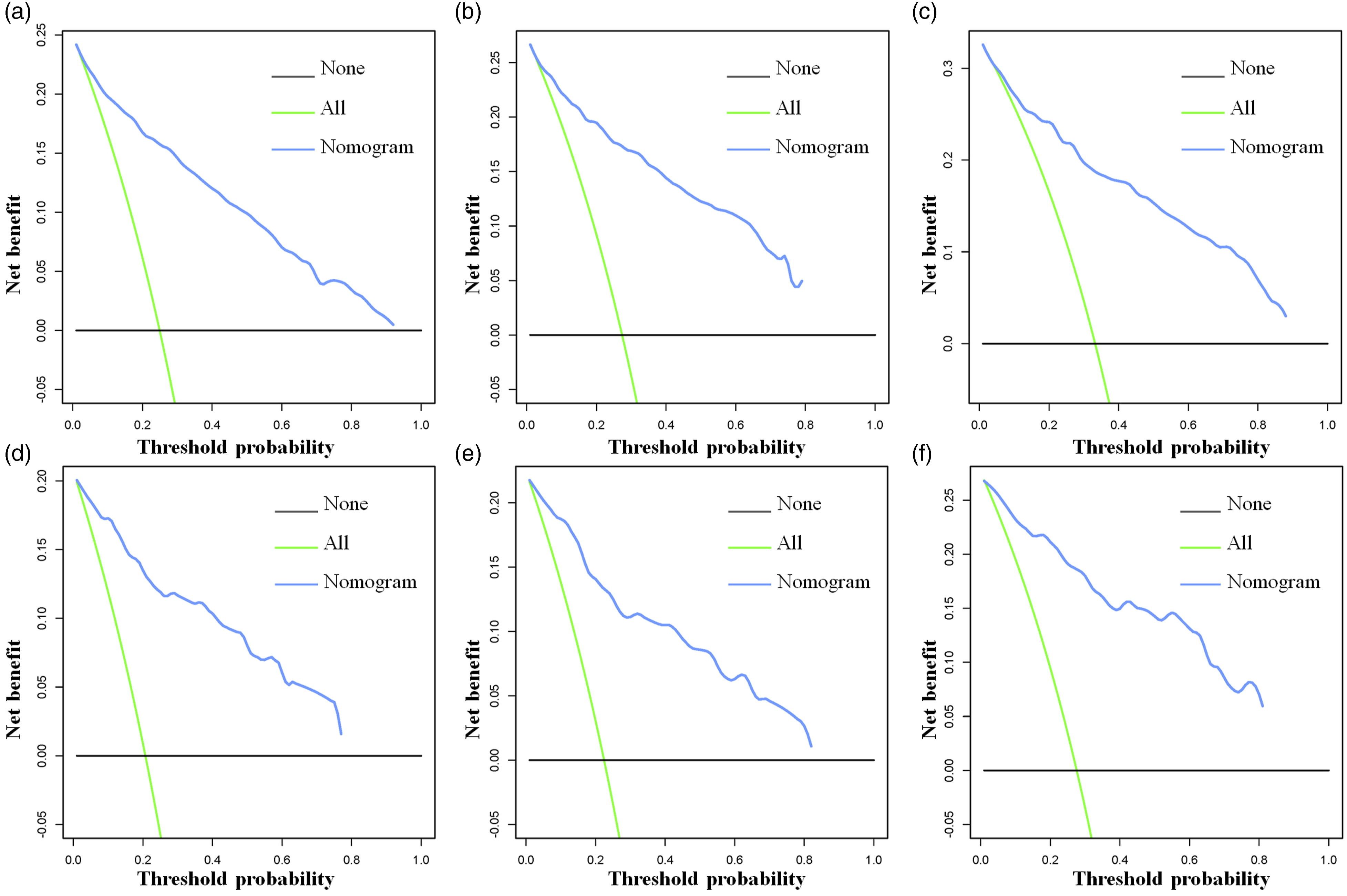
A nomogram was established using a quantitative approach, which showed a bleak outlook for older patients with advanced tumor grade, distant metastasis, confirmed lymphoma, and who underwent radiotherapy (Figure 2). At the same time, the scores of all separate prognostic predictors were obtained A prognostic nomogram was constructed to forecast spinal cord tumor patients’ 3-, 5-, and 10-year CSS. Specifically, when a patient with a primary spinal cord tumor consults about his or her survival, we can sum the points of independent prognostic factors to obtain a total score and draw a vertical line from the total points to the bottom timeline to obtain his or her 3-, 5-, and 10-year death probability. The probability of survival at the corresponding time can be obtained by subtracting the probability of death from 1. For example, a 38 years old patient diagnosed with distant metastasis primary spinal cold lymphoma (pre-B cell) received no radiotherapy. The corresponding total points of his or her is 27 (38 years old) + 88 (lymphoma) + 48 (distant metastasis) + 10 (pre-B cell) + 27 (no radiotherapy) = 200, and the corresponding death probability at 3, 5, and 10 years are .156, .183, and .253, respectively, while the patient’s corresponding probability of CSS at 3, 5, and 10 years are .844, .817, and .747, respectively. The Detailed Point of Each Independent Prognostic Predictor in the CSS Nomogram. The calibration curves of the nomogram forecast the 3-, 5-, and 10-year CSS of patients with spinal cord tumors in the study’s training set (A-C) and validation set (D-F). The receiver operating characteristic (ROC) curves in the training (A) and validation (B) sets of patients with spinal cord tumor. Comparison of prediction accuracy between the constructed nomogram and each CSS independent predictor in this study. The ROC curves of the constructed nomogram and each CSS independent predictor at 3 (A), 5 (B) and 10 (C) years in the training set and 3 (D), 5 (E) and 10 (F) years in the validation set. Decision curve analysis (DCA) was performed on the training and the validation sets in this study. The 3-(A), 5-(B), and 10-(C) year CSS in the training set and the 3-(D), 5-(E), and 10-(F) year CSS in the validation set of patients with spinal cord tumors are predicted by the DCA of the nomogram.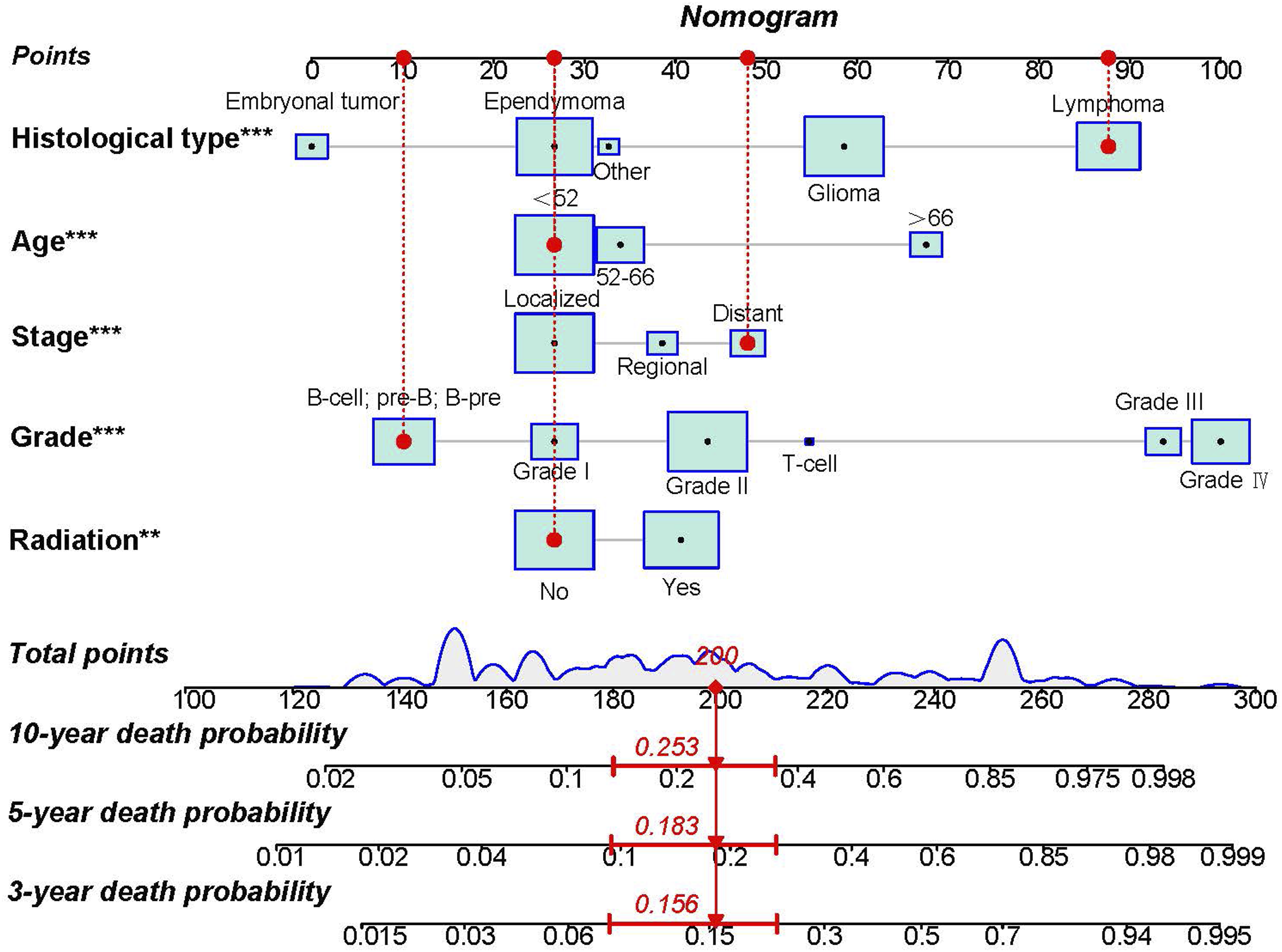

Risk Classification System for CSS
As shown in Table 3, the total score of the patient was determined by adding up the points from every individual prognostic predictor. 200 and 252 were found to be the optimal thresholds for the total score using the X-tile software (Supplemental file 1). These thresholds were then used to categorize the patients into 3 mortality risk classification subgroups: low risk (<200), middle risk (200-252), and high risk (>252). Furthermore, a plot of the Kaplan-Meier curve was made (Figure 7). Figure 7 demonstrates that the risk classification system effectively categorized SCT patients into 3 subgroups in both sets, revealing notable distinctions. This indicates that the nomogram successfully differentiated the prognosis among various subgroups of SCT patients. The training set and validation set in this study underwent Kaplan-Meier survival analysis. In both the training set (A) and the validation set (B) for the CSS of patients with spinal cord tumors, patients with high-risk scores demonstrated a worse prognosis than patients with low-risk scores.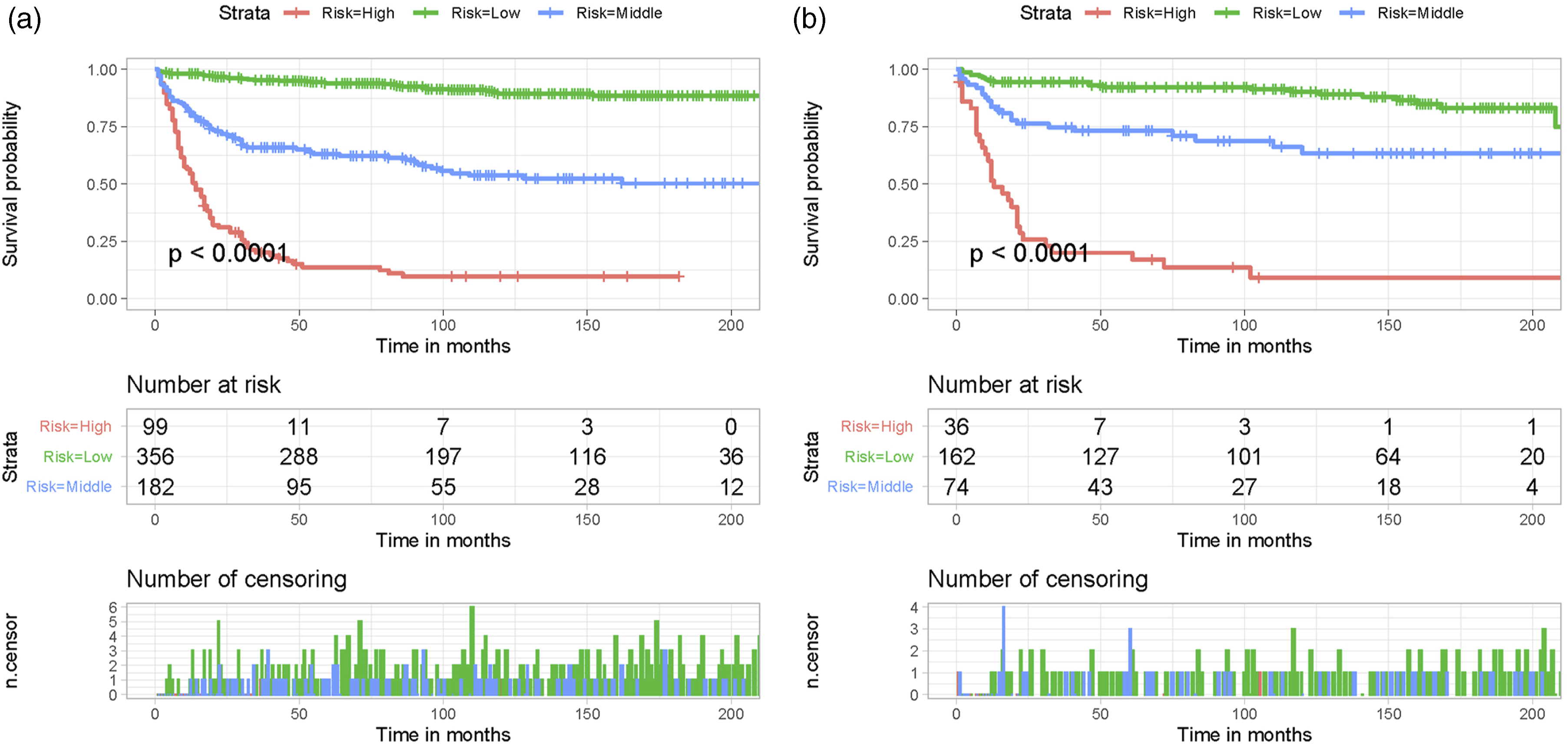
Discussion
Early research on SCTs in spinal cord, spinal meninges, and cauda equina was rare, limiting the research for developing better treatment strategies and healthcare planning for SCT patients. It was not until 2010 that researchers began to focus on this particular tumor type. 1 Although studies related to the prognosis of SCT have been reported over the past decade, they were limited to a single site or a single factor, making it impossible to analyze the overall prognosis based on information from the individual patient.1,3 Moreover, there is currently no prognostic nomogram for SCT patients. Hence, we performed a thorough study of population-based data from the SEER database to determine independent prognostic factors linked to CSS outcomes in patients with SCTs. Additionally, we created the first nomogram to forecast CSS in SCT patients.
This study selected 909 SCT patients. According to multivariable Cox regression analysis, age, histological type, tumor grade, tumor stage, and radiotherapy were identified as independent prognostic predictors for the CSS of SCT patients. Then, a nomogram to forecast SCT patients’ 3-, 5-, and 10-year CSS was developed. The nomogram’s strong discriminative power and prediction accuracy were demonstrated by the training and validation sets. The actual and expected survival rates did not differ significantly. One potential application for the nomogram is as a useful clinical prediction tool. Furthermore, the developed risk categorization system successfully segregated patients in both sets into subcategories of low-, middle-, and high-risk, exhibiting notable disparities (P < .01). This facilitated improved differentiation of patient risks and enabled effective intervention strategies.
Nowadays, surgical resection is the most effective treatment for benign, well-defined SCTs and serves the dual purpose of gross tumor excision and tissue diagnosis. 9 Obtaining a histologic diagnosis is also crucially facilitated by the surgical biopsy.10,11 Several studies have shown that a patient’s prognosis is significantly influenced by the extent of removal. 12 Patients who receive complete removal of the tumor experience more favorable results compared to those who receive partial removal of the tumor. Hence, it is crucial to extract the tumor tissue to the maximum extent feasible while preserving nerve function in order to prevent any potential relapse. However, when total tumor resection is challenging and will affect spinal cord function, the tumor should be removed to relieve compression under the premise of protecting the function of the spinal cord. Our study found that surgical treatment was a significant factor affecting patient prognosis according to univariate Cox regression analysis. However, multivariable Cox regression analysis, which excluded confounding effects among variables, did not show any correlation between surgical treatment and CSS.
Elderly SCT patients often have a poor prognosis related to decreased physical function, dysfunction, malnutrition, and comorbidities such as hypertension and diabetes. These factors hinder physicians from opting for a more vigorous treatment approach or shorten the duration of treatment, ultimately impacting the treatment outcomes.13,14 The high-grade tumor was an independent prognostic predictor for systemic metastasis and poor survival rates for SCT patients. Wright et al’s study revealed a tendency towards poorer patient survival in grade III compared to grade I and II. 15 Our findings indicate that individuals diagnosed with low-grade tumors experienced a more favorable outlook compared to those diagnosed with high-grade tumors. Additionally, compared to individuals with local or regional metastases, those with distant metastases had a poorer survival rate. The importance of enhancing early detection for individuals with SCT is further supported by these patterns. Furthermore, our research revealed that the histological type was also a separate prognostic indicator for SCTs. Patients with lymphoma as their tumor histological type had the most unfavorable prognosis. Despite being the most common kind of lymphoma, diffuse large B-cell lymphoma is characterized by its aggressive nature and has the poorest prognosis when compared to other types of tumors. 16
External radiation therapy has been utilized in adjuvant treatment for SCTs, particularly in cases of malignancy or tumors that have been partially removed. The NCCN guideline suggests that adjuvant therapy should be administered for grade III ependymoma, no matter the extent of surgical removal. 17 Nevertheless, the efficacy of radiotherapy in the management of SCTs still remains controversial. Several studies have indicated that the addition of radiotherapy does not enhance the rate of survival among patients who have undergone partial removal of the tumor, whereas radiotherapy raises the likelihood of mortality in individuals with ependymoma of low grade.18-21 Furthermore, research conducted by Saurav et al indicated that the combination of surgery and radiotherapy did not enhance the prognosis of individuals diagnosed with cauda equina tumors. 22 In our study, individuals with SCT who received radiation therapy experienced a poor prognosis, presumably related to radiation myelitis and radiotherapy-related complications, making the benefit of radiotherapy less than the side effects that came with it. Adverse effects after radiation treatment comprised of nausea and/or vomiting, fatigue, anorexia, and neurological worsening.1,23,24 Engelhard et al showed that approximately 3.5% of radiotherapy patients experienced neurological worsening. 1 Moreover, Abrey et al showed that delayed neurotoxicity was common in elderly patients with primary CNS lymphoma who received radiotherapy. 25
Although the current study has numerous benefits, such as the development of a nomogram that exhibits excellent prediction and risk classification capabilities, it does have a few limitations. First, retrospective clinical studies are bound to have selection bias. Second, certain diagnostic and treatment information, such as patient radiotherapy dose and regimen, adverse effects, etc., remains unknown. Lastly, our nomogram has temporarily not been validated in other centers or databases.
Conclusions
In conclusion, SCT individuals aged 66 years or older, with advanced grade, distant metastases, received radiotherapy, and lymphoma histological type have a poor prognosis. The 3-, 5-, and 10-year CSS of SCT patients were predicted using the nomogram and mortality risk classification system, which might be utilized as a support prediction tool in clinical practice for treatment decision-making, monitoring, and counselling.
Supplemental Material
Supplemental Material - Predicting the Probability of Tumor-Specific Survival in Patients Diagnosed With Primary Tumors in the Spinal Cord Using Nomogram Models
Supplemental Material for Predicting the Probability of Tumor-Specific Survival in Patients Diagnosed With Primary Tumors in the Spinal Cord Using Nomogram Models by Chao Huang, Tingting Tang, Zichuan Ding, Haoyang Wang, and Zongke Zhou in Global Spine Journal
Footnotes
Acknowledgments
We are thankful for the contribution of the SEER database and the 18 registries supplying cancer research information and thank all colleagues involved in the study for their contributions.
Authors’ Contributions
C H and TT T designed the study, performed the literature review, extracted the data, and analyzed the pooled data. ZC D drew the figures and organized the tables. HY W and ZK Z provided critical comments and revised the manuscript. All authors read and approved the final manuscript. C H and TT T contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82172394 and U22A20280) and West China Nursing Discipline Development Special Fund Project, Sichuan University (HXHL20003).
Provenance and Peer Review
Not commissioned, externally peer-reviewed.
Ethical Statement
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
