Abstract
Study Design
A retrospective study.
Objective
To investigate the risk of adjacent segment disease (ASD) after L4-5 transforaminal lumbar interbody fusion (TLIF) in patients diagnosed with lumbar spinal stenosis (LSS), a prediction model for ASD is established and validated.
Methods
A retrospective study was carried out on a sample of 290 patients who underwent L4-5 TLIF at Zhongda Hospital, Southeast University, from January 2015 to January 2021. The study collected baseline data and preoperative radiographic features of L3-4 and L5-S1. The determination of the outcome variable was based on X-ray results spanning over 24 months and JOA scores. Multivariate logistic regression was used to identify the risk factors in constructing a nomogram.
Results
Independent risk factors for L3-4 degeneration after TLIF included osteoarthritis of L3-4 facet joints, L3-4 foraminal stenosis, L4 upper endplate osteochondritis, L3-4 local lordosis angle, and L3-4 spinal stenosis. Independent risk factors for L5-S1 degeneration after TLIF included osteoarthritis of L5-S1 facet joints, L5-S1 intervertebral disc degeneration, L5-S1 spinal stenosis, L5-S1 coronal imbalance, and S1 upper endplate osteochondritis. A predictive model was developed. The AUC for the prediction models at L3-4 and L5-S1 were .945 and .956. The calibration curve demonstrated good consistency between the predicted and actual probabilities. The DCA curve indicated the clinical benefit and practical value of this predictive model.
Conclusion
This study established nomograms for postoperative degeneration at L3-4 and L5-S1 based on selected preoperative radiographic features. These models provide a valuable auxiliary decision-making system for clinicians and aid in early surgical decisions.
Introductions
Transforaminal lumbar interbody fusion (TLIF), a modification of the posterior approach lumbar fusion, has been extensively used in the treatment of lumbar spinal stenosis (LSS) over the past decade. Now it has become one of the most notable procedures in spinal surgery. 1 Although TLIF generally avoids causing instability in adjacent segments, it still cannot prevent a common long-term complication of traditional lumbar fusion surgery, adjacent segment disease (ASD). ASD is a degenerative condition in which pathological changes occur in neighboring active spinal segments following lumbar or lumbosacral spinal fusion.2,3 Its imaging manifestations include a decrease in intervertebral space, disc herniation, vertebral instability, hyperplasia, and osteoarthritis of the facet joints, which are present in one to two segments adjacent to the fused segment. Previous studies have not definitively answered whether ASD results from accelerated disc degeneration due to changes in the biomechanical pattern of the lumbar spine caused by surgery or untreated underlying degenerative risk in the lumbar spine. 4 In the clinical diagnosis and treatment process, the decision regarding the segments for TLIF requires comprehensive consideration by the clinicians. This includes not only the positioning of the responsible segment but also the early intervention in potentially high-risk degenerative segments to avoid the occurrence of long-term ASD after surgery. Furthermore, most patients with LSS present with low back pain accompanied by pain and numbness in one or both lower limbs. Based on the patient’s clinical symptoms, the responsible segment for localization usually exhibits neurogenic symptoms in 1-3 segments of the lower lumbar spine. When clinicians select surgical segments, they not only consider clinical symptoms but also intervene early in potential high-risk degenerative segments revealed in imaging studies. This study conducted a retrospective analysis of clinical data from patients with LSS who underwent single-segment TLIF from January 2015 to January 2021. It primarily focused on predicting the risk of ASD in patients with L4-5 LSS requiring TLIF. The findings aim to provide a supplementary decision-making system for clinical surgeons in making early surgical decisions.
Materials and Methods
Inclusion and Exclusion Criteria
Inclusion Criteria
(1) Patients diagnosed with LSS and presenting clinical symptoms of lower back pain, accompanied by one or both lower limbs. (2) Patients who have been selected for L4-5 TLIF by the decision-makers and have been monitored for at least 24 months after surgery. (3) Availability of complete preoperative lumbar spine MRI, standard standing and dynamic lumbar X-rays, and CT scans of the lumbar spine. (4) Patients showing significant improvement in clinical symptoms postoperatively compared to preoperative conditions. (5) Patients who are awake, alert, mentally sound, have normal reading and writing abilities and can cooperate with the investigation.
Exclusion Criteria
(1) Patients with incomplete or severely missing clinical data. (2) Patients with spinal stenosis caused by various types of spinal tumors and space-occupying lesions. (3) Patients with a history of lumbar spine trauma, including old fractures, osteoporotic fractures, and ankylosing spondylitis fractures, leading to spinal stenosis. (4) Patients with a history of spinal surgeries such as spinal fractures and disc herniation. (5) Patients with systemic diseases such as rheumatoid arthritis and systemic lupus erythematosus. (6) Patients who experienced postoperative fusion device dislocation, fusion device subsidence, or non-fusion.
Patients Population
A total of 290 patients diagnosed with LSS and treated with L4-5 TLIF at Zhongda Hospital of Southeast University between January 2015 and January 2021 were included. A total of 461 patients were diagnosed with LSS and underwent L4-5 TLIF. Based on inclusion and exclusion criteria, a final cohort of 290 patients was included in the study. The specific inclusion and exclusion process is illustrated in Figure 1. Long-term postoperative follow-up is necessary to monitor the occurrence of ASD in patients who have undergone L4-5 TLIF. The outcome variables are defined using the UCLA evaluation system and lumbar JOA scores.
5
Each adjacent disc was classified as A (normal), B (general narrowing of the disc space), C (presence of osteophytes), or D (presence of endplate sclerosis). Grade A discs were defined as normal, while grade B, C, and D discs were defined as degenerative. Disc height, spinous process formation, and endplate sclerosis were recorded for adjacent discs receiving TLIF by calculating the average of the anterior, middle, and posterior disc heights. Disc space narrowing was defined as a reduction of more than 20% of normal height. The presence of osteophytes was defined as anterior endplate spurs greater than 3 mm in length on the lateral membrane. Detailed flowchart of case inclusion and exclusion criteria.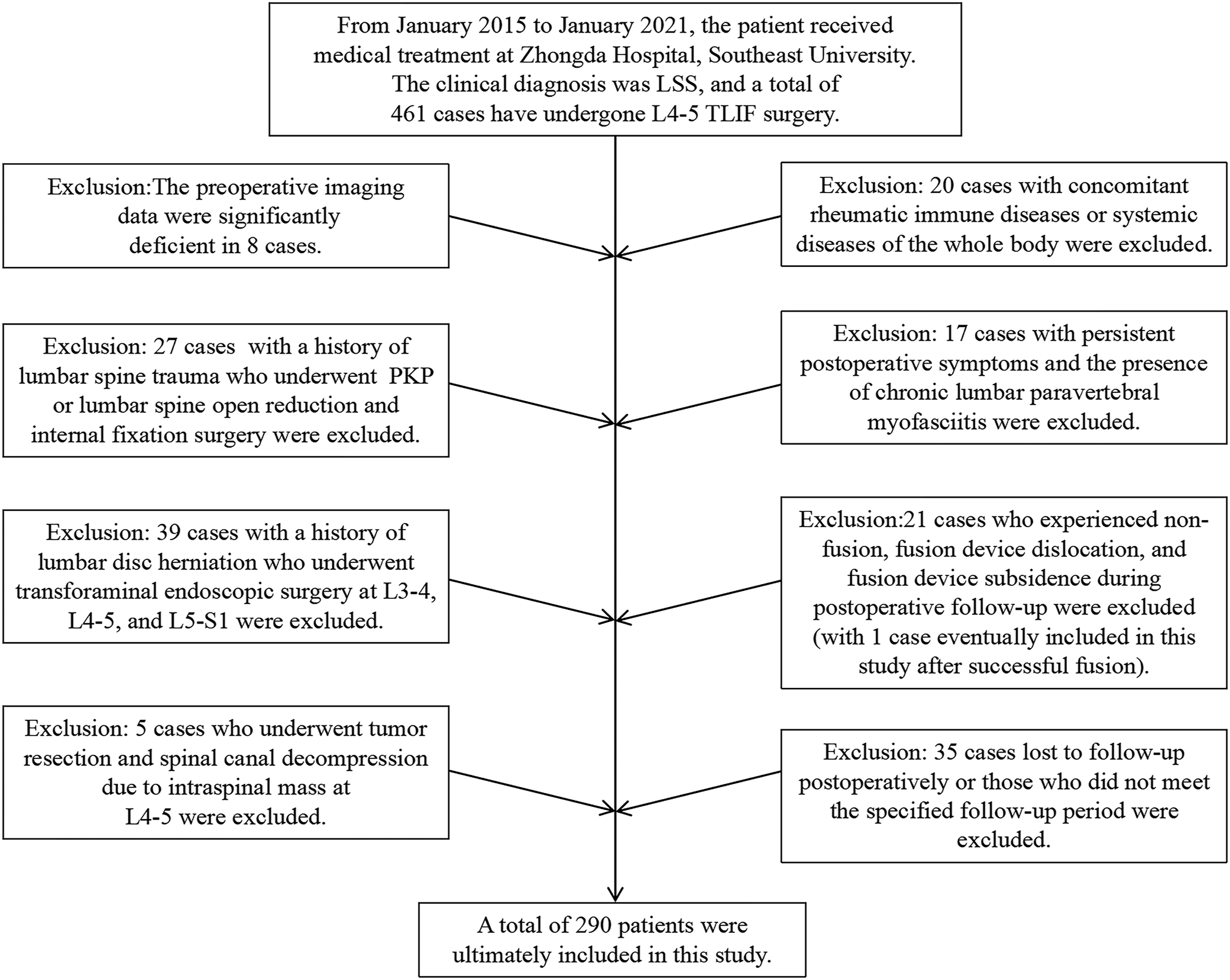
The research sample is categorized into four groups according to the existence or non-existence of degenerative alterations at L3-4 and L5-S1, specifically the L3-4 degenerative, L3-4 non-degenerative, L5-S1 degenerative, and L5-S1 non-degenerative groups. The procedures used in this study adhere to the principles of the Helsinki Declaration. Due to the observational nature of the study, informed consent was waived. Basic demographic information, radiological findings, and surgery-related data were extracted from patient records. This study has been approved by the Ethics Committee of Zhongda Hospital, Southeast University (Ethics number: 2023ZDSYLL309-P01).
Surgical Procedure
After successful general anesthesia induction, the patient is positioned prone with the abdomen suspended. The routine lumbar back surgical area is disinfected with iodine, drapes are placed, and a sterile film is applied. A 4 cm midline incision is made at the L4-5 level, extending through the skin, subcutaneous tissues, and lumbar fascia. The subperiosteal peeling technique is used to dissect the bilateral spinous processes, retracting the sacrospinalis muscles, and ultimately exposing the entry points for pedicle screws at L4-5. Guidewires are inserted into the pedicle screws, confirmed for accurate positioning through X-ray, and appropriate-sized pedicle screws are placed after widening the incision. The left side of the L4 lamina and lower facet joint are excised, and the upper half of the left side of the L5 facet joint and thickened ligamentum flavum are removed. The neural foramen is enlarged, and the annulus fibrosus of the disc is cut at the L4-5 left intervertebral space. Nucleus pulposus and calcified tissue are gradually removed, and the cartilage endplates of the upper and lower vertebral bodies are scraped off. Adequate autogenous crushed bone is filled into the intervertebral space. An appropriately sized interbody fusion cage is filled with bone chips and inserted into the L4-5 intervertebral space from the left side. Bilateral connecting rods are placed, and the pedicle screws are pressurized and locked. Satisfactory positions of pedicle screws and the fusion cage are confirmed through X-ray. The nerve roots are checked to ensure relaxation, with no observed compression. The spinal canal is clear above and below. The wound is rinsed repeatedly with saline solution. Negative pressure drainage tubes are placed on either side of the surgical incision in the spinous process. The incision is closed layer by layer, and sterile dressings are applied with pressure. The same primary surgeon performs all procedures.
Data Collections
Basic Demographic Information: Demographic data includes age, gender, height, weight, BMI, and duration of the condition. Radiological Features and Clinical Symptom Scores: The radiological factors include the preoperative Pfirrmann grade of L3-4 and L5-S1 discs, the foraminal stenosis, osteoarthritis of the lumbar facet joints, Modic changes in endplates, Schizas grading of lumbar spinal stenosis, the height of adjacent disc, and the sagittal and coronal imbalance. Postoperative follow-up imaging was assessed using the UCLA evaluation system from the University of California, and clinical symptoms were evaluated using the lumbar spine JOA score. All radiological data were evaluated by two associate senior spine surgeons. For the UCLA evaluation system of L3-4 degeneration, the crude agreement rate between the two observers was 96.55% (95% CI: .933, .998). Cohen’s kappa coefficient was .904. For the UCLA evaluation system of L5-S1 degeneration, the crude agreement rate between the two observers was 94.14% (95% CI: .899, .984). Cohen’s kappa coefficient was .838. Due to the high crude agreement rates between the two evaluators, Cohen’s kappa coefficient suggests strong consistency between them. The consistency test indicates that the difference between Cohen’s kappa coefficient and 0 is statistically significant. In cases where the two evaluators disagreed, an independent judgment was made by a third chief physician. Follow-up Outcome Definition: For patients undergoing single-level L4-5 TLIF, the occurrence of ASD in adjacent segments needs to be determined through long-term postoperative follow-up. The outcome variable was defined using the UCLA evaluation system and lumbar spine JOA score. JOA score was assessed during the last follow-up; scores categorized as “excellent” and “good” were defined as no clinical symptoms, while “fair” and “poor” scores were defined as having clinical symptoms. Combined with the UCLA evaluation system, “radiographic adjacent segment degeneration” and “presence of clinical symptoms” were defined as ASD occurrence, while other cases were defined as no ASD.
Statistical Analysis
SPSS version 27.0 was utilized for statistical analysis. Count data were presented as cases (%) and analyzed using the χ2 test for group comparisons. Measurement data values were expressed as χ ±s and group comparisons were performed using a t test. Ranked data between groups were compared using the Mann-Whitney rank sum test. Multivariate regression analysis was conducted using R4.3.0 software for multifactorial analysis to establish and validate the model. Results were considered statistically significant when P < .05. Patients were randomly assigned to training and validation sets in a 4:1 ratio. Independent risk factors were identified through both univariate and multivariate regression analyses. Nomograms were developed using independent risk factors selected from these analyses. The predictive accuracy of the nomograms was assessed using the area under the receiver operating characteristic (ROC) curve, and calibration charts were plotted to evaluate the calibration ability of nomograms. Decision curve analysis (DCA) was employed to assess net benefits. The prediction model mentioned above was built based on the training cohort. The accuracy of the nomograms was subsequently verified in a validation group utilizing the aforementioned methodology.
Result
Baseline Patient Data
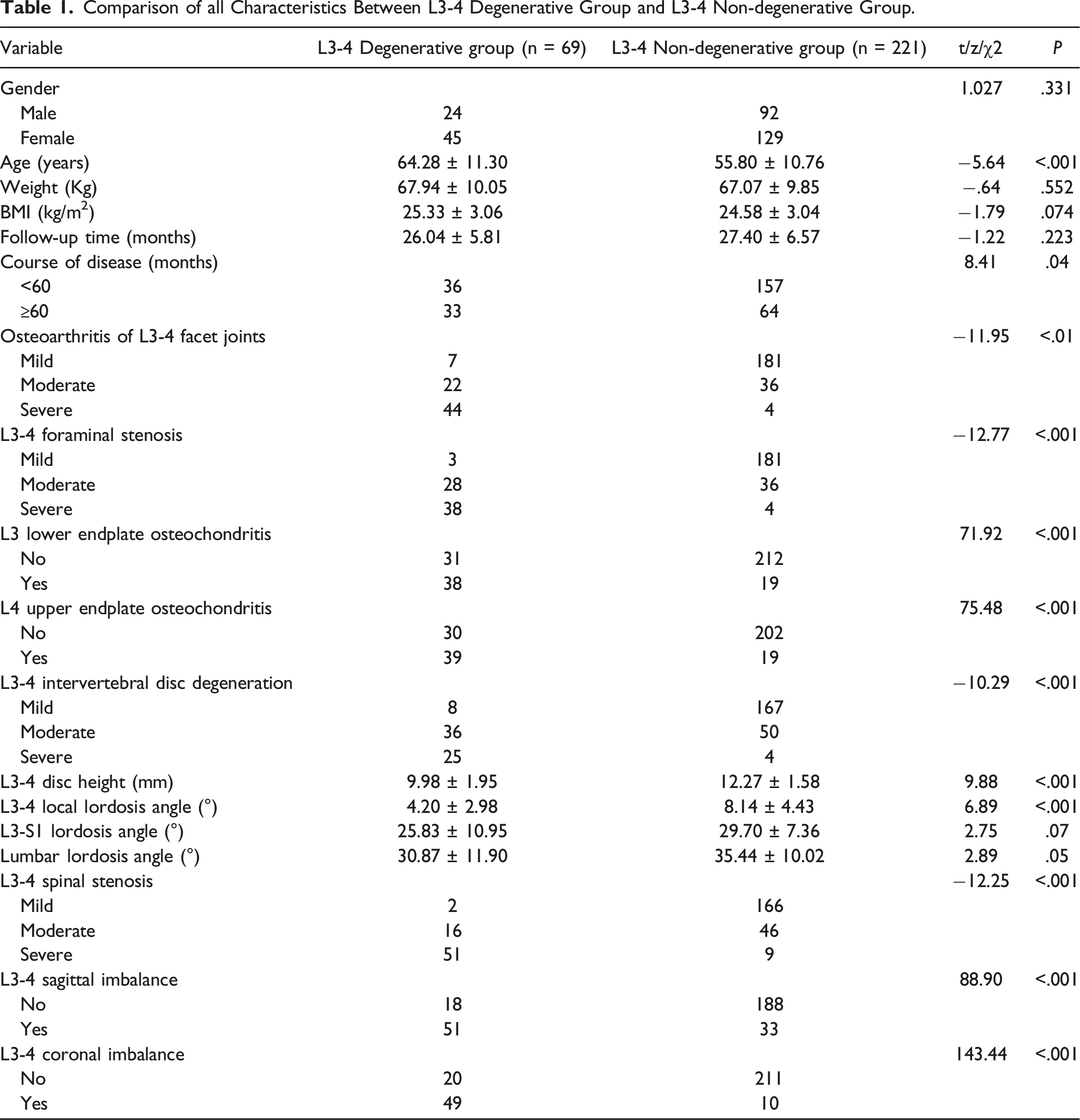
Comparison of all Characteristics Between L3-4 Degenerative Group and L3-4 Non-degenerative Group.
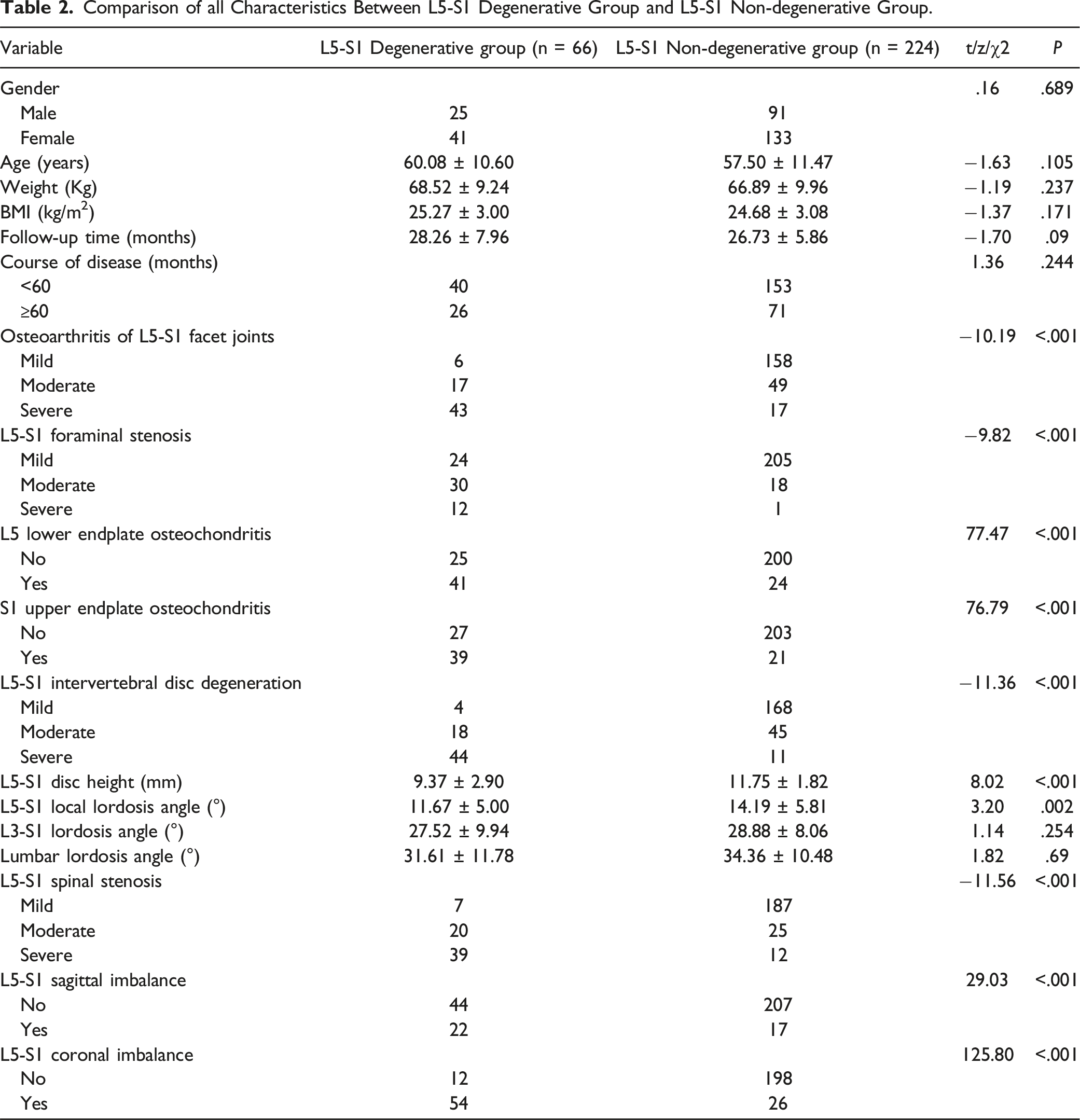
Comparison of all Characteristics Between L5-S1 Degenerative Group and L5-S1 Non-degenerative Group.
Univariate Analysis
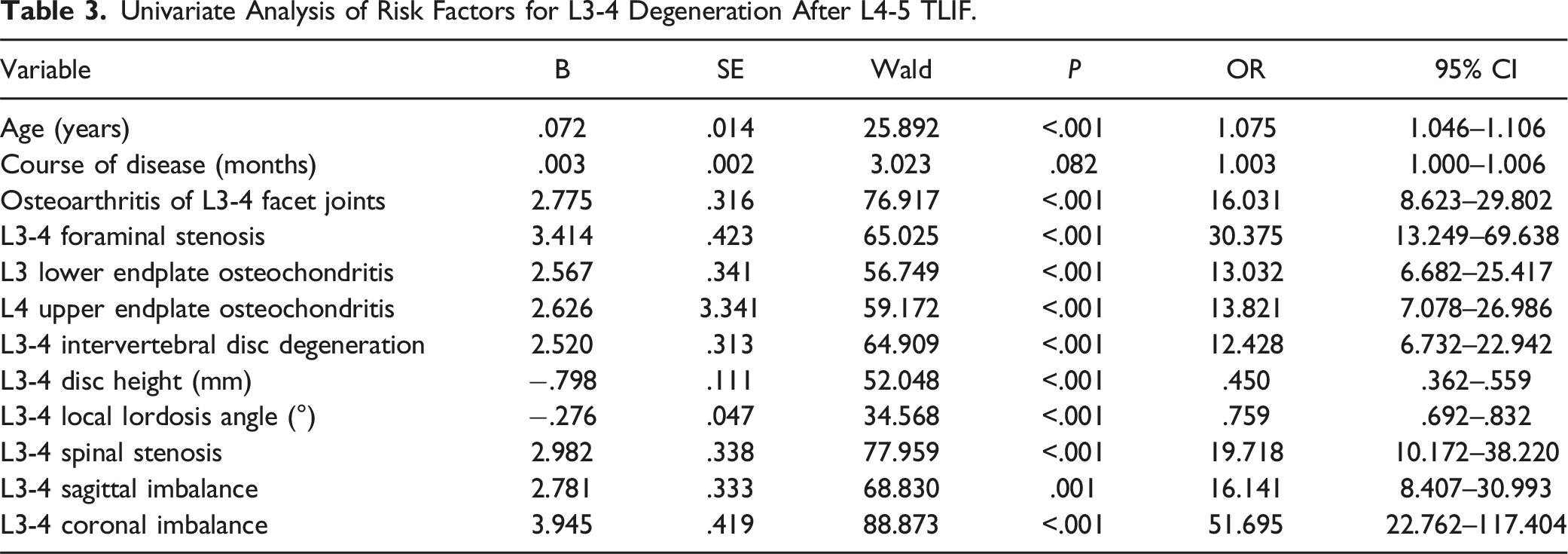
Univariate Analysis of Risk Factors for L3-4 Degeneration After L4-5 TLIF.
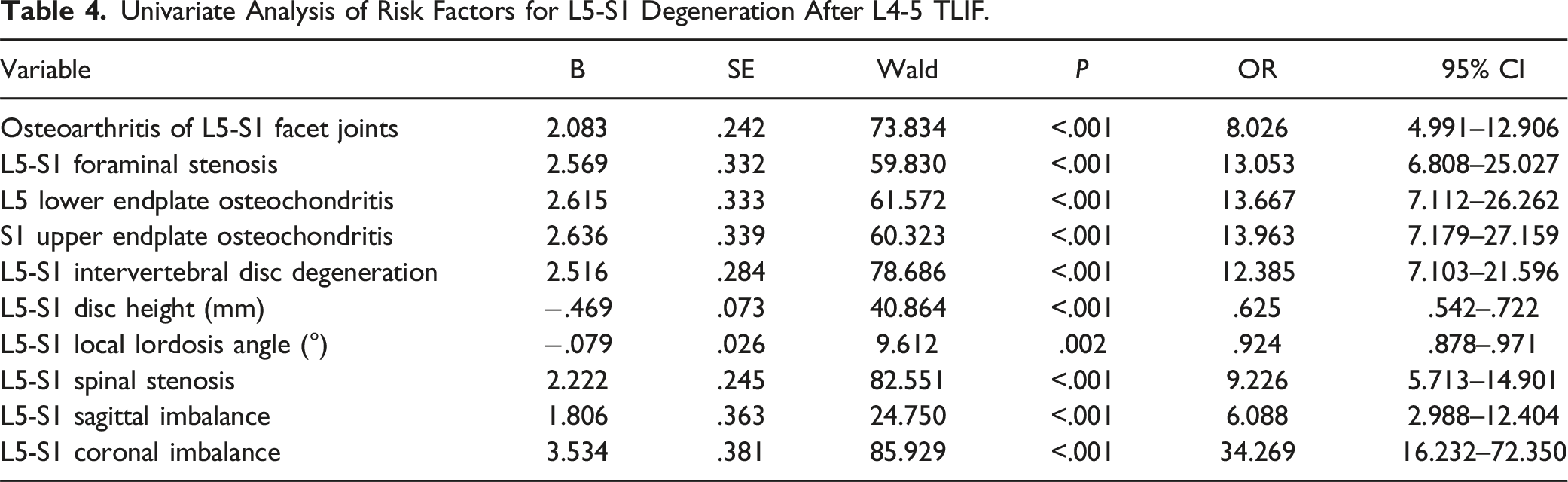
Univariate Analysis of Risk Factors for L5-S1 Degeneration After L4-5 TLIF.
Multivariate Logistic Regression Analysis
After comparing the intergroup differences in basic patient information and conducting univariate logistic regression, the following variables were selected for the multivariate logistic regression for the L3-4 ASD: age, osteoarthritis of the L3-4 facet joints, L3-4 foraminal stenosis, L3 lower endplate osteochondritis, L4 upper endplate osteochondritis, L3-4 intervertebral disc degeneration, L3-4 disc height, L3-4 local lordosis angle, L3-4 spinal stenosis, L3-4 sagittal imbalance, and L3-4 coronal imbalance. Similarly, for the L5-S1 degenerative and non-degenerative groups, the following variables were included in the multifactor binary logistic regression: osteoarthritis of the L5-S1 facet joints, L5-S1 foraminal stenosis, L5 lower endplate osteochondritis, S1 upper endplate osteochondritis, L5-S1 intervertebral disc degeneration, L5-S1 disc height, L5-S1 local lordosis angle, L5-S1 spinal stenosis, L5-S1 sagittal imbalance, and L5-S1 coronal imbalance.
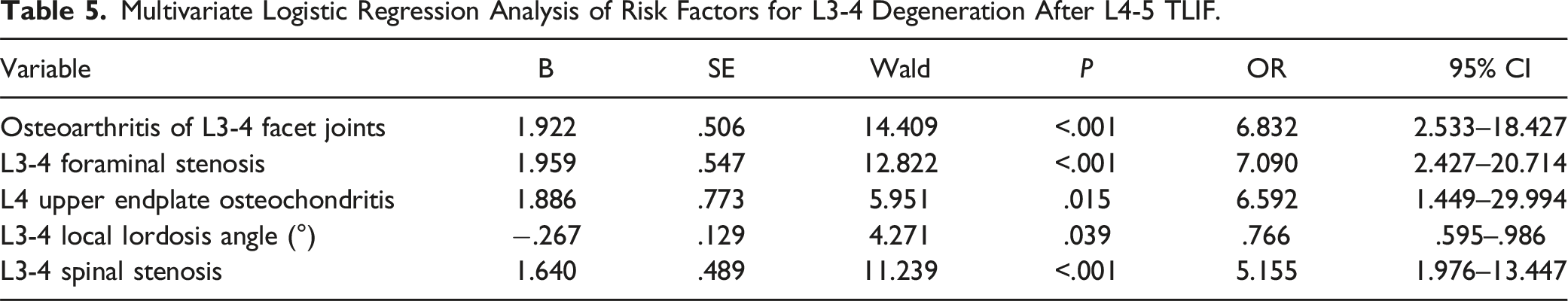
Multivariate Logistic Regression Analysis of Risk Factors for L3-4 Degeneration After L4-5 TLIF.
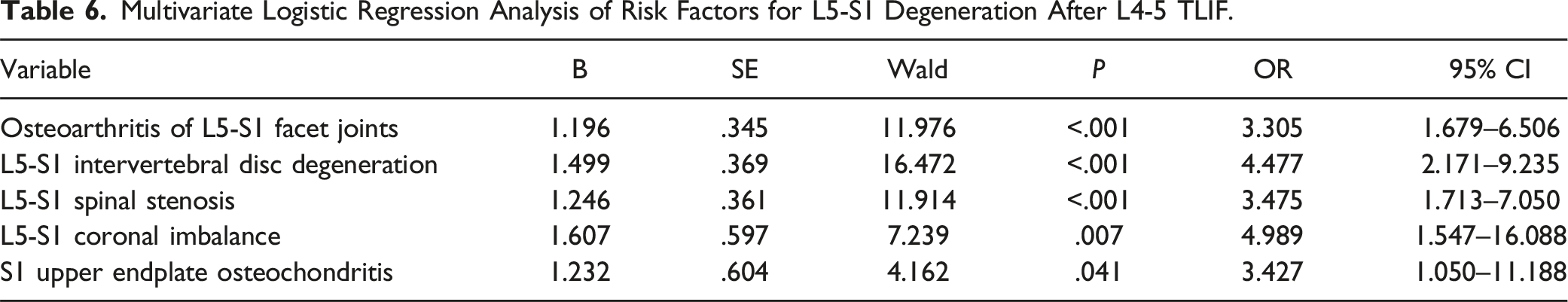
Multivariate Logistic Regression Analysis of Risk Factors for L5-S1 Degeneration After L4-5 TLIF.
Model Evaluation
Construction and validation of the model for L3-4 and L5-S1 degeneration based on identified independent risk factors. This study focuses on establishing and validating prediction models for ASD after L4-5 TLIF in patients with LSS. The models were constructed based on independently identified risk factors, separately predicting degeneration at L3-4 and L5-S1 (Figures 2 and 3). Of the 290 patients, 20% were selected at random to form the validation group (comprising 58 patients), while the remaining 232 patients formed the training group. There was no significant difference found in the incidence of risk factors between the validation and training groups. A nomogram to predict the risk of L3-4 degeneration after L4-5 TLIF. A nomogram to predict the risk of L5-S1 degeneration after L4-5 TLIF.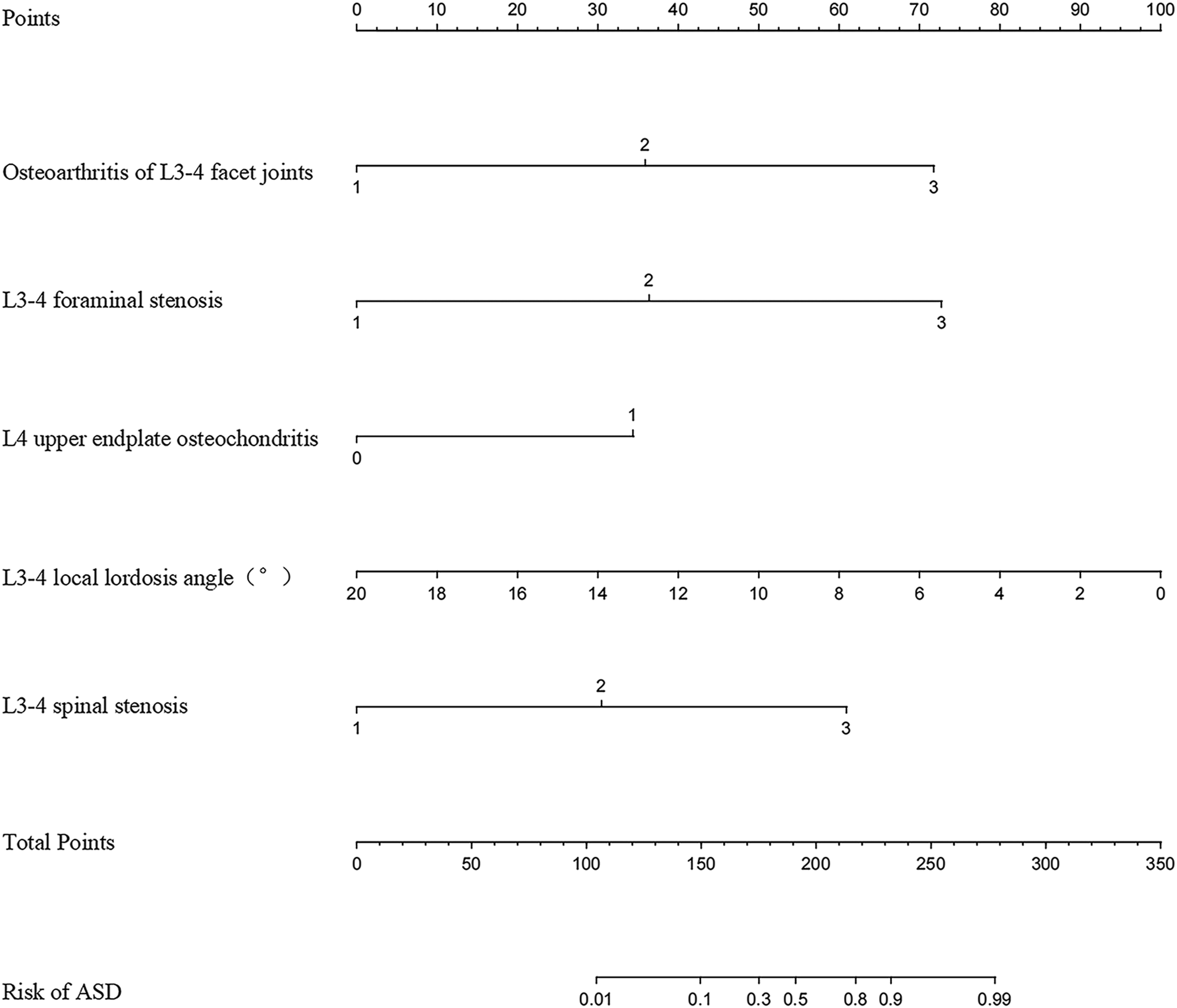
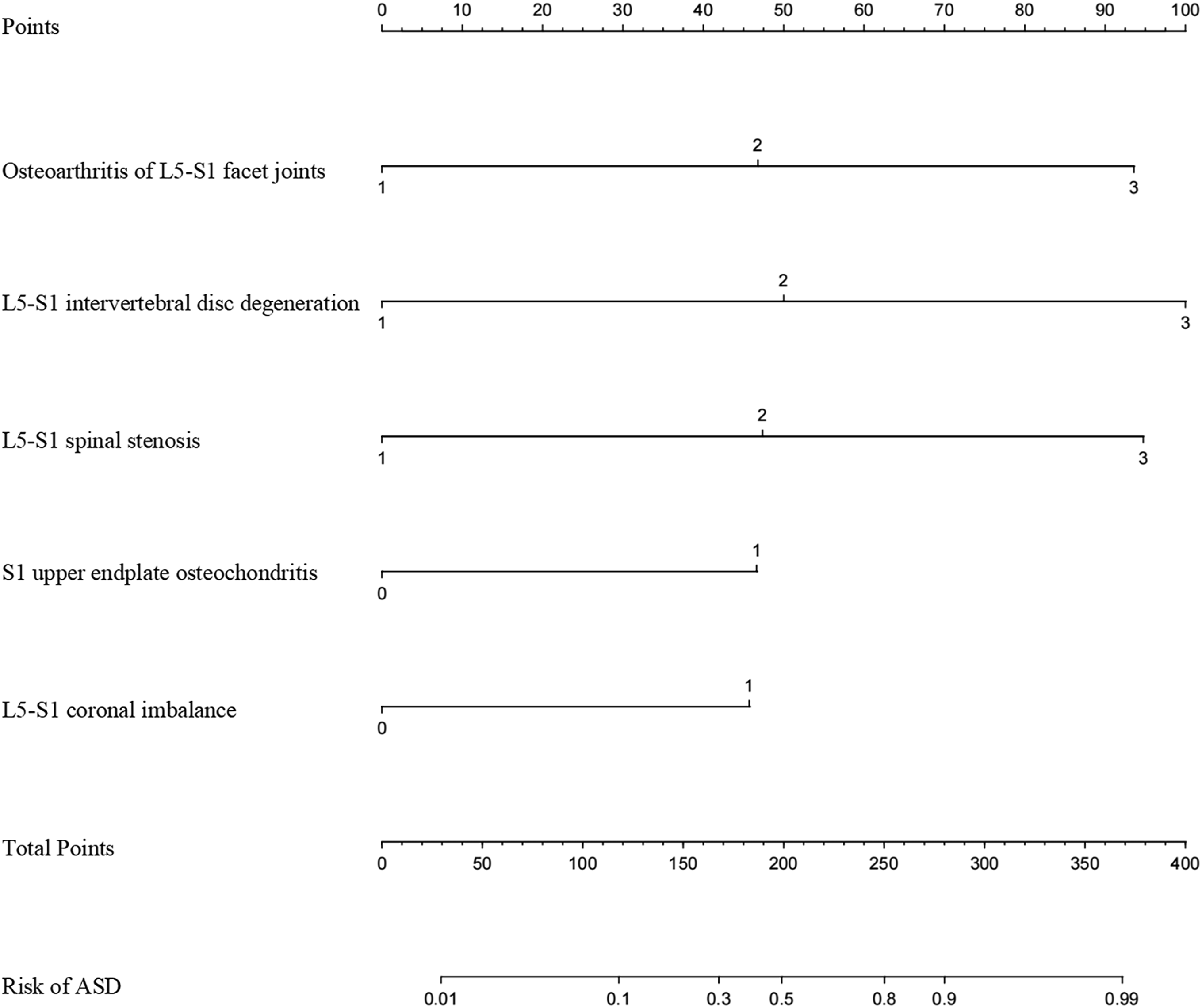
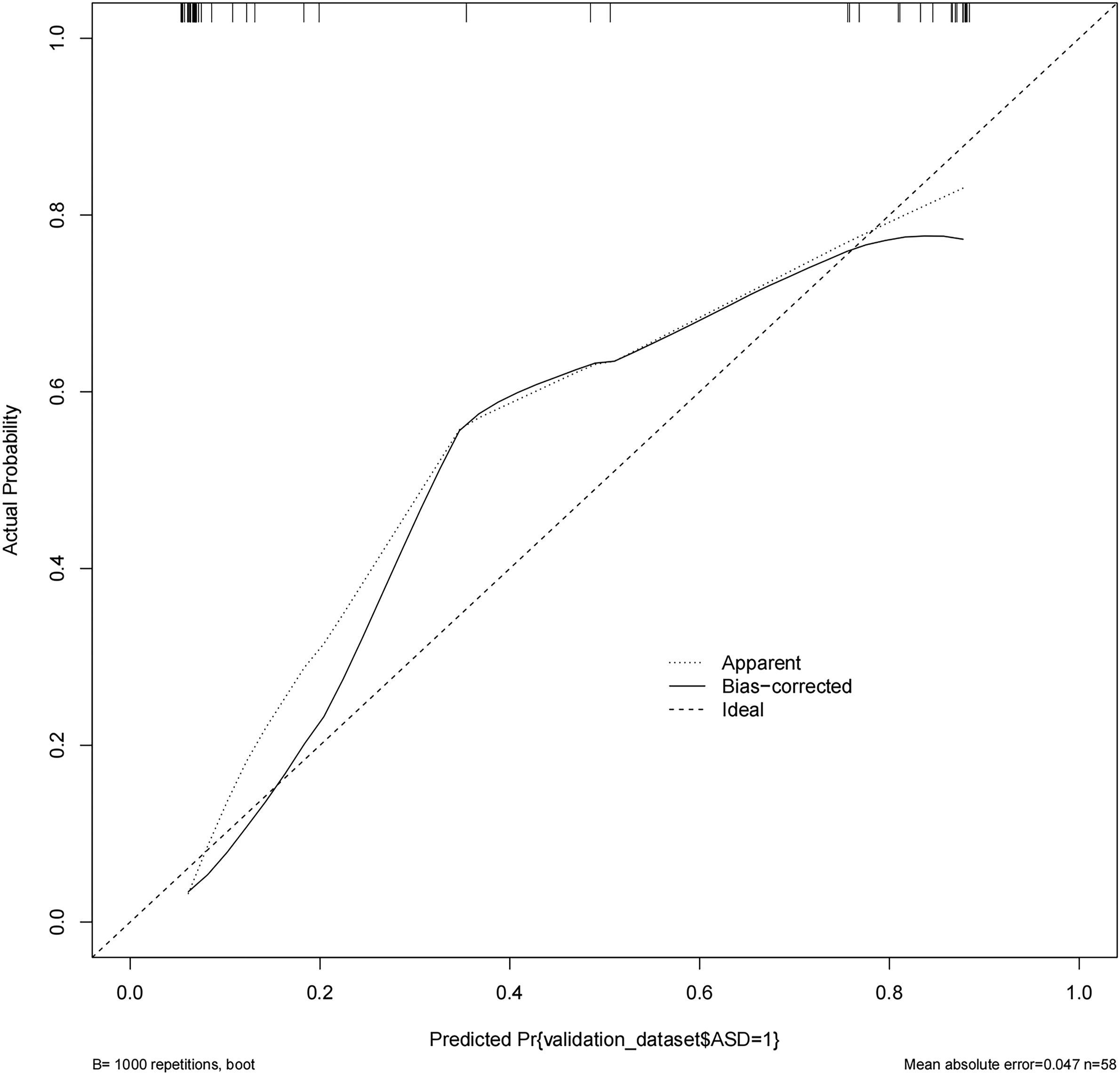
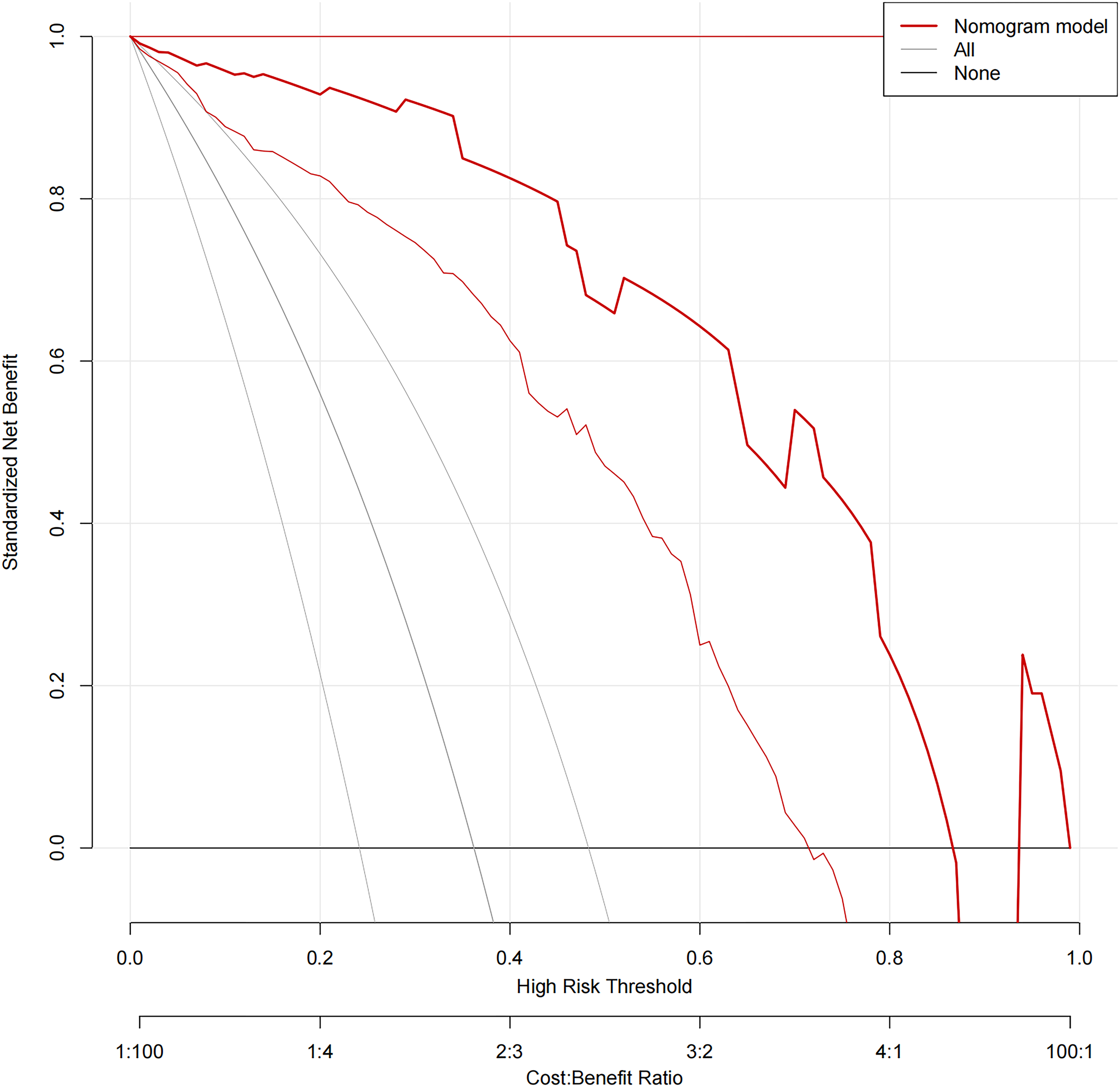
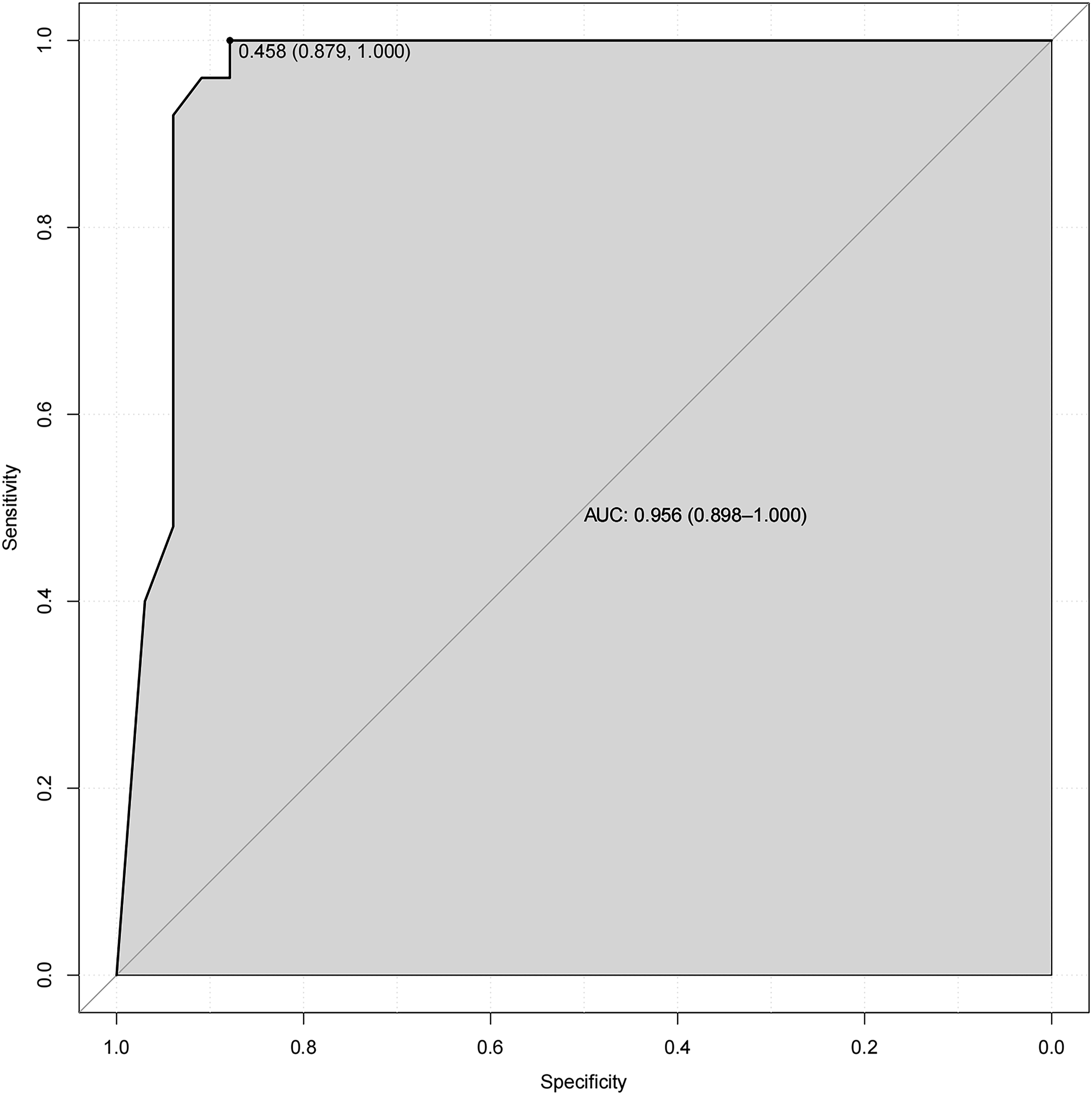
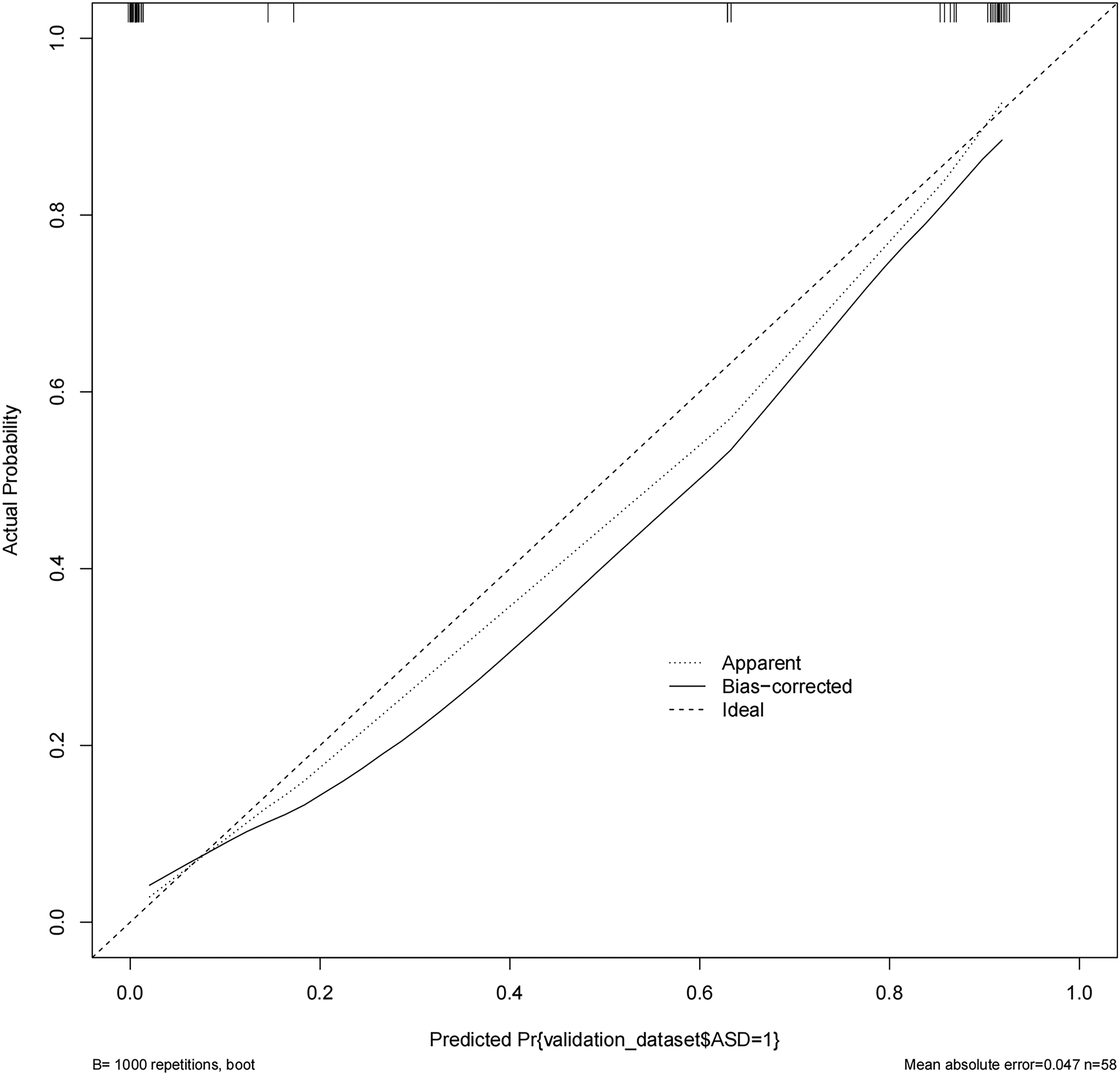
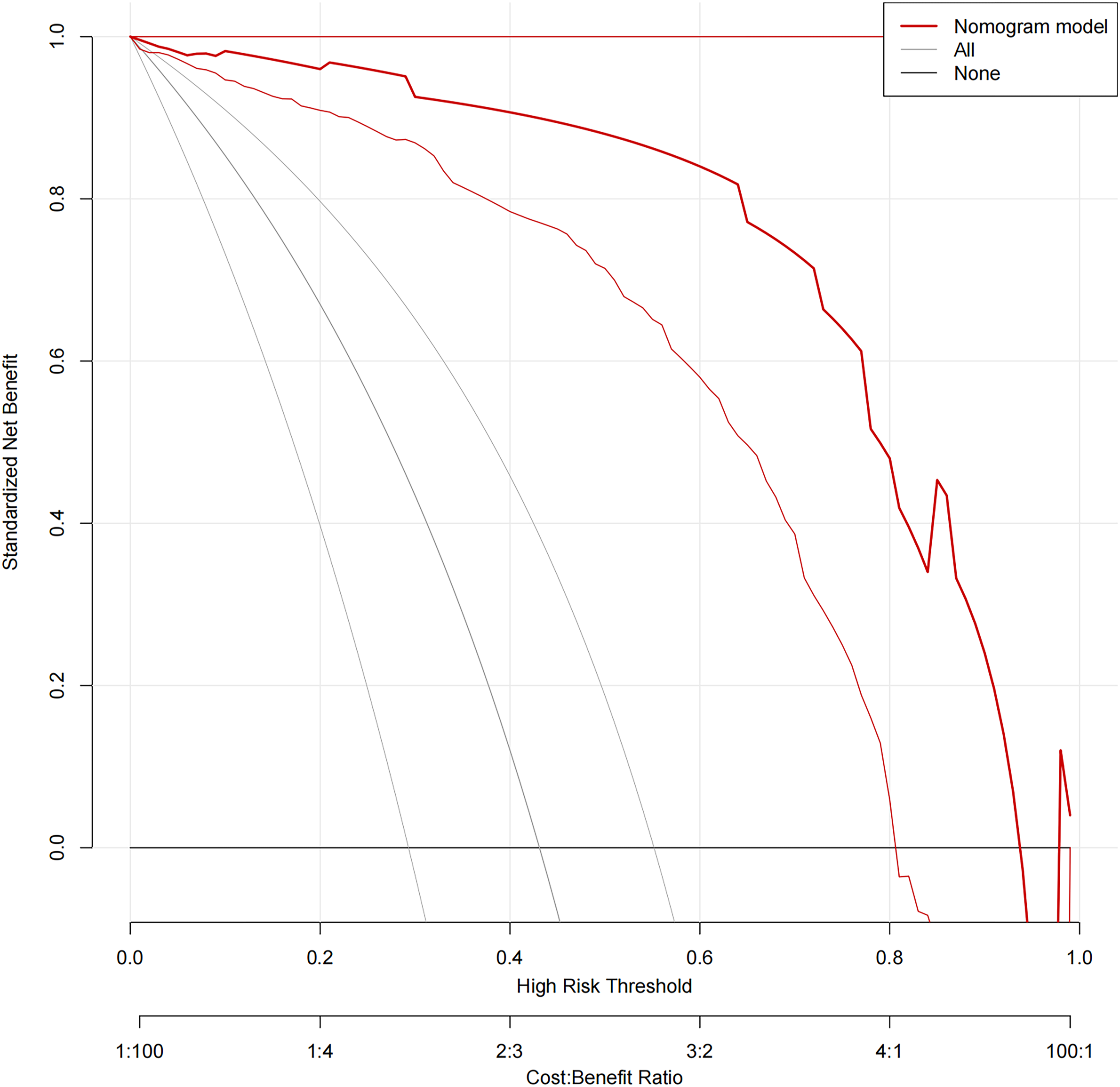
In the L3-4 degeneration nomogram, ROC curve analysis yielded an AUC of .945 in the validation set, indicating excellent predictive performance (Figure 4). Calibration curves demonstrated good consistency between predicted and actual probabilities in the validation sets (Figure 5). Decision curve analysis showed a net benefit for prediction thresholds ranging from 10% to 90% (Figure 6), indicating excellent predictive performance. In the L5-S1 degeneration nomogram, ROC curve analysis revealed an AUC of .956 in the validation set, highlighting the model’s robust predictive performance (Figure 7). Calibration curves indicated consistent alignment between predicted and actual probabilities in the validation sets (Figure 8), and decision curve analysis demonstrated a net benefit for prediction thresholds between 15% and 90% (Figure 9), underscoring the model’s strong predictive value. Receiver operating characteristic curve analysis-model validation of L3-4 degeneration. Calibration curves of the L3-4 degeneration nomogram. The lines in the figure represent the apparent value, the bias-corrected value, and the ideal value. The apparent and the bias-corrected values are close to each other, which means the nomogram has a good predictive performance. Decision curve analysis for nomogram prediction of risk of L3-4 degeneration after L4-5 TLIF. The X and Y axes represent the threshold probability and net benefit, respectively. Receiver operating characteristic curve analysis-model validation of L5-S1 degeneration. Calibration curves of the L5-S1 degeneration nomogram. The lines in the figure represent the apparent value, the bias-corrected value, and the ideal value. The apparent and the bias-corrected values are close to each other, which means the nomogram has a good predictive performance. Decision curve analysis for nomogram prediction of risk of L5-S1 degeneration after L4-5 TLIF. The X and Y axes represent the threshold probability and net benefit, respectively.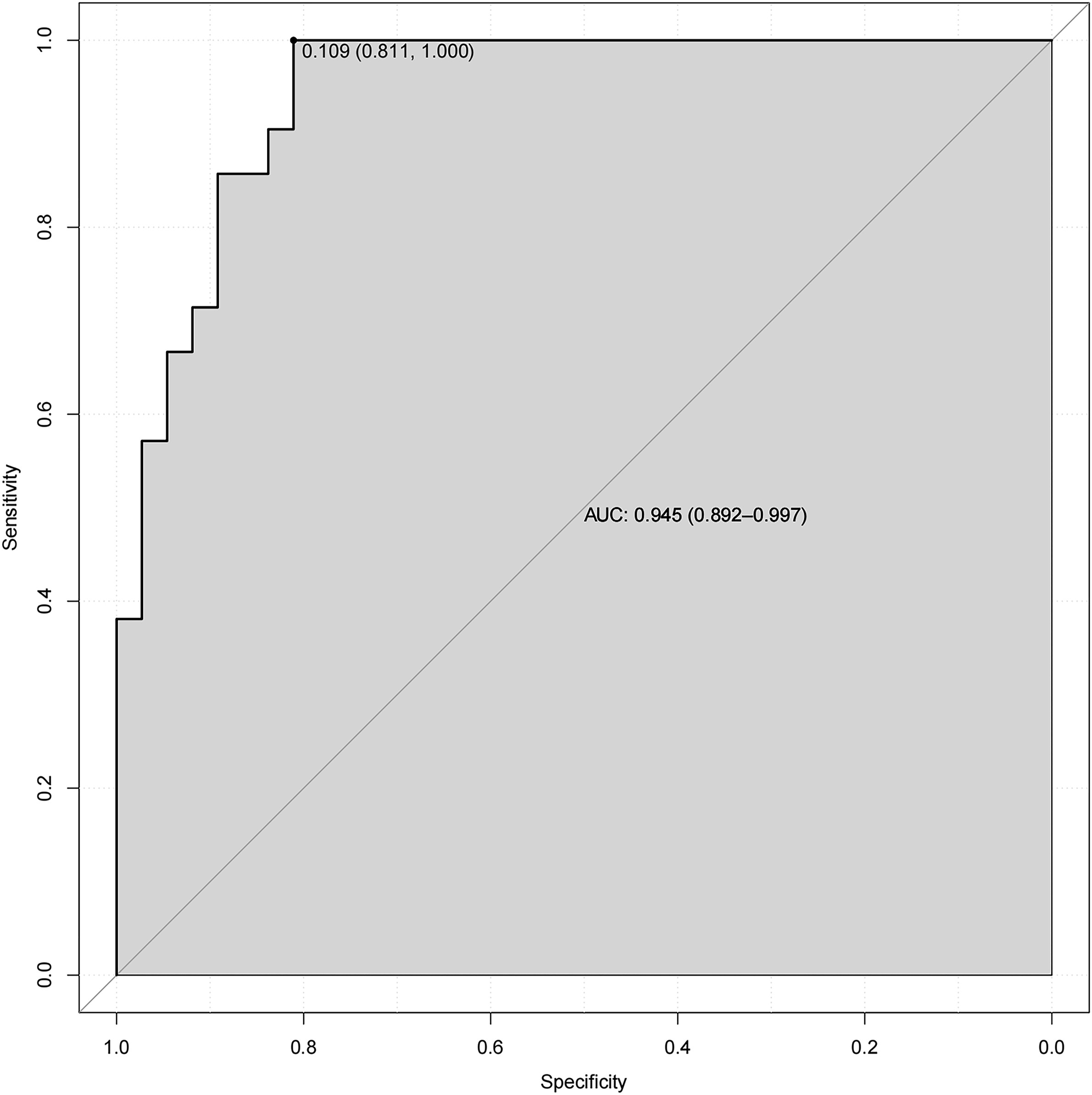
Discussion
ASD is a common long-term complication following lumbar fusion surgery, with an incidence rate ranging from 0 to 35.7%. This condition frequently necessitates subsequent surgeries after lumbar fusion.6,7 Despite the significant advancements achieved through TLIF, which has largely mitigated the impact on the stability of adjacent vertebral segments, it has not eliminated the occurrence of ASD. 8 Existing research indicates that preoperative ASD is considered one of the most significant factors leading to post-fusion ASD.9,10 Identifying pre-existing high-risk degenerated segments and improving the long-term quality of life of patients while effectively preventing degenerative diseases in adjacent spinal segments have become important priorities in spinal surgery. Consequently, clarifying the risk factors for ASD, developing personalized prevention and treatment strategies, and tailoring the choice of fusion segments to each patient’s specific condition have become critical to minimizing the occurrence of future ASD. One debated issue is whether asymptomatic adjacent segments showing potential degeneration on preoperative imaging should be included in the fusion construct to prevent future surgeries related to ASD in the coming years.11,12 If high-risk segments prone to degeneration after surgery can be identified through preoperative radiological features, clinicians may consider incorporating asymptomatic segments into early fusion decisions. Therefore, this study primarily focuses on predicting the risk of adjacent segment disease in patients undergoing TLIF at the L4-5 due to LSS. It aims to provide a supplementary decision-making system for clinicians in early surgical interventions.
ASD is a complex postoperative complication influenced by various clinical factors. Therefore, this study focused exclusively on patients undergoing L4-5 TLIF, all operated by the same surgeon, with the same-side decompression, aiming to eliminate potential confounding surgical factors. Considering the distinct mechanical conditions of the upper and lower adjacent segments of L4-5, we separately investigated the degenerative conditions at L3-4 and L5-S1. Using the UCLA evaluation system and lumbar JOA score to define follow-up outcomes, our study identified risk factors for post-L4-5 TLIF degeneration at the L3-4 and L5-S1. In the L3-4 degeneration, risk factors for L3-4 degeneration after L4-5 TLIF included the osteoarthritis of the L3-4 facet joints, L3-4 foraminal stenosis, L4 upper endplate osteochondritis, L3-4 local lordosis angle, L3-4 spinal stenosis. In the L5-S1 degeneration, risk factors for L5-S1 degeneration after L4-5 TLIF included the osteoarthritis of the L5-S1 facet joints, L5-S1 intervertebral disc degeneration, L5-S1 spinal stenosis, L5-S1 coronal imbalance, S1 upper endplate osteochondritis. Based on these identified risk factors, predictive models for post-L3-4 and L5-S1 degeneration following L4-5 TLIF were developed, and represented as nomograms. These models exhibit good predictive performance and hold significant clinical utility.
In existing research, postoperative changes in spinal biomechanics following lumbar fusion surgery are recognized as significant factors contributing to ASD. 7 These changes include increased stress on the facet joints, increased pressure within the disc, and excessive motion in segments adjacent to the fusion site. This study suggests that pre-existing facet joint degeneration in adjacent segments is one of the risk factors for postoperative ASD. This phenomenon may be related to changes in the biomechanical patterns of adjacent segments after TLIF, resulting in compensatory motion and increased activity in adjacent segments. This compensatory activity may further stimulate proliferation, cohesion, and osteophyte formation in degenerated facet joints, resulting in the narrowing of the lateral recess and foramen. Compressed spinal nerve roots can result in symptoms in the lower limbs, potentially leading to the onset of ASD. The facet joints are surrounded by joint capsules and ligaments and contain abundant autonomic and nociceptive nerve fibers. In cases of inflammation or mechanical stimulation, these joints can become painful and may contribute to long-term back pain in patients with pre-existing facet joint degeneration. Biomechanical studies have shown that facet joints provide substantial mechanical support by resisting shear forces and tension generated during movement and loading processes. 13 Additionally, as part of the lumbar facet joint complex, they play a crucial role in maintaining lumbar stability and mobility. In this study, pre-existing facet joint degeneration was identified as one of the risk factors for postoperative ASD in adjacent segments both cranially and caudally to L4-5. 14 This finding aligns with current research results widely accepted in the academic community.
In previous studies, Modic changes in vertebral endplates were closely associated with intervertebral disc degeneration. 15 Endplates not only cushion pressure, reducing vertical stress on intervertebral discs but also serve as pathways for nutritional exchange between vertebral bodies and intervertebral discs. 16 Importantly, in this study, pre-existing L4 upper endplate osteochondritis and S1 upper endplate osteochondritis were identified as one of the risk factors for postoperative ASD. In contrast, L3 lower endplate osteochondritis and L5 lower endplate osteochondritis did not show a statistically significant association with postoperative ASD. This observation may be related to stress transmission in the lumbar spine. In particular, the cartilaginous endplate between the disc and the vertebral body may bear more vertical stress than the cartilaginous endplate between the vertebral bodies and the disc, suggesting that inflammation in the endplate of the lower vertebral body in adjacent segments may be more likely to lead to postoperative ASD.17,18 During the actual research process, a detailed classification of Modic changes was not performed. This decision was based on clinical considerations where Modic Type 2 and Type 3 changes often accompany intervertebral disc degeneration. Surgeons typically perform fusion treatment for segments affected by Modic Type 2 and 3 changes to address the chronic back pain caused by these alterations. Additionally, L3-4 and L5-S1 spinal stenosis was also identified as one of the risk factors for postoperative ASD. This suggests that the presence of asymptomatic LSS before surgery may lead to future ASD occurrences. In other words, patients with pre-existing asymptomatic LSS in non-operated segments are more likely to develop ASD after surgery.
This study demonstrates that the L3-4 local lordosis angle and the L3-4 foraminal stenosis are risk factors for postoperative degeneration at the L3-4. However, the L5-S1 local lordosis angle and the L5-S1 foraminal stenosis show no significant statistical significance concerning postoperative degeneration at the L5-S1. The sagittal plane angle of the lumbar spine has a significant impact on segmental motion and degenerative changes. The prevalent studies indicate that the sagittal plane angle of the lumbar spine serves as a predictive parameter for degenerative spinal illnesses and is deemed one of the cardinal factors in lumbar spine biomechanics. 19 In this study, the L3-4 local lordosis angle is defined as the angle between the upper endplate of L3 and the upper endplate of L4. Changes in this angle are related to the descent of the L3-4 intervertebral space and osteoporotic compressive changes in the L3 vertebral body. In clinical practice, the implantation of fusion devices within a previously fused segment results in the restoration of local segmental lordosis height. This is achieved by expanding the intervertebral space which in turn widens the previously narrowed intervertebral foramen, thereby relieving compression on the nerve root of that segment. Studies in non-operated segments suggest that lower local segmental lordosis in higher lumbar segments is more indicative of possible postoperative ASD. This observation may be related to the more prominent role of higher lumbar segments in the sagittal plane biomechanical model of the lumbar spine. It’s worth noting that the L5-S1 coronal imbalance is one of the risk factors for postoperative ASD at that level, whereas the L3-4 coronal imbalance does not show significant statistical significance concerning postoperative ASD. Spinal stability is defined as the ability of the spine to maintain its relationship and limit relative displacement during physiological positions and loading. The overall stability of the lumbar spine is maintained through the coordination of intervertebral discs, joints, ligaments, and muscles. The degenerative processes of intervertebral discs and facet joints can affect the stability of the moving segments. 20
Abei et al.'s research has proposed that alterations in spinal stability, collectively termed asymmetric degeneration, lead to increased asymmetric loading, resulting in the development of degeneration and deformities such as scoliosis or kyphosis. Asymmetric degeneration can damage facet joints, joint capsules, discs, and ligaments, potentially causing instability in one or more segments, ultimately leading to spinal stenosis. 21 In other words, changes in coronal or sagittal balance are correlated with facet joint degeneration, intervertebral disc degeneration, and intervertebral space descent. However, current research has not indicated whether sagittal and coronal balance in high and low lumbar segments have different impacts on lumbar degeneration in the lumbar spine biomechanical model. This study demonstrates that in the predictive model for postoperative ASD, the sagittal balance of higher lumbar segments is more predictive than that of lower lumbar segments, and the coronal imbalance in lower lumbar segments is more predictive than that in higher lumbar segments.
The current study has certain limitations. Firstly, the foremost challenge encountered in ASD is the extended follow-up duration and the challenge of determining the outcome of the follow-up. The retrospective study used the UCLA and lumbar JOA scores in combination to determine the outcome. However, the persistent postoperative low back pain experienced by some patients at follow-up could not be clearly distinguished from lumbar dorsal myofascitis and fatty infiltration of the paravertebral muscles. 22 X-rays still have limitations compared to MRI in determining follow-up outcomes. To address this, it is suggested to incorporate deep learning and other imaging techniques to detect risk factors in follow-up. Further research is recommended to incorporate deep learning and other methods for examining the risk factors associated with medical imaging. Additionally, the imaging data mainly used categorical variables, which resulted in the inability of multi-categorical variables to train the model more effectively than numerical variables when building the predictive model, and ultimately led to some overfitting. The study recommends increasing the sample size and introducing more precise numerical variables, such as foraminal diameter and dural sac area, to improve the predictive efficacy of the model.
Conclusion
In summary, this study investigated preoperative radiological risk factors for degeneration at L3-4 and L5-S1 after L4-5 TLIF. Five independent risk factors were identified for each segment, and predictive models were constructed using a nomogram. These models serve as a supportive decision-making tool for surgical practitioners performing lower lumbar TLIF procedures. The aim is to enable early intervention in high-risk potential degenerated segments and avoid unnecessary long-segment fusions, ultimately preventing the occurrence of postoperative ASD in the long term.
Supplemental Material
Supplemental Material - Prediction Model and Risk Factor Analysis of Adjacent Segment Disease after L4-5 Transforaminal Lumbar Interbody Fusion Through Preoperative Radiographic Features
Supplemental Material for Prediction Model and Risk Factor Analysis of Adjacent Segment Disease after L4-5 Transforaminal Lumbar Interbody Fusion Through Preoperative Radiographic Features by Zi-Jian Zhang, Lu Chen, Lei Zhu, Hang Shi, Fu-Yu Zhang, Zan-Li Jiang, and Xiao-Tao Wu in Global Spine Journal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Jiangsu Provincial Medical Key Discipline(Laboratory) Cultivation Unit (JSDW202222).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
