Abstract
Study Design
Narrative review of existing literature.
Objectives
Significant technological advancements in radiotherapy planning and delivery have enabled new radiotherapy techniques for the management of spine tumors. The objective of this study was to provide a comprehensive summary of these treatment techniques for practicing spine surgeons.
Methods
A narrative review of the existing literature on stereotactic body radiation therapy (SBRT) and particle beam therapy (PBT) for the treatment of spine tumors was performed. The characteristics, implementation and evidence supporting these strategies in the management of primary spinal neoplasms were summarized.
Results
The clinical effectiveness of SBRT for the control and symptom palliation of metastatic spinal tumors are well demonstrated in multiple clinical trials. Risks such as fracture, radiculopathy and plexopathy exist after spine SBRT, necessitating an individualized approach in a well experienced multidisciplinary setting. SBRT should be considered a key component of a well-rounded treatment plan for metastatic spine tumors in combination with surgery, vertebral augmentation, and drug therapy, where indicated, to achieve optimal patient outcomes. Additionally, PBT and SBRT are also leading to promising results for primary spine tumors, though comparative effectiveness studies and prospective clinical trials are required to establish these modalities more formally as alternatives to conventionally fractionated photon radiotherapy.
Conclusions
SBRT and PBT are emerging as effective and well tolerated treatment options for primary and metastatic spine tumors. Additional investigation is needed to personalize these treatment options and further strengthen these approaches as key components in a multidisciplinary approach to the management of spinal neoplasms.
Introduction
Radiotherapy plays an important role in the definitive and postoperative treatment of both primary and metastatic spine tumors. Spine tumors are located in close proximity to critical neural structures and can be associated with significant morbidity necessitating an integrated multidisciplinary approach for management. 1 Over the past decade, radiation technology has evolved to allow for the delivery of what is considered “ablative” doses of radiotherapy, with little increased risk to the surrounding normal tissues. 2 When delivered in up to 5 fractions, this technique is commonly known as stereotactic body radiotherapy (SBRT), whereas in the primary tumor setting we would more often consider doses of 70-80 Gy or more delivered over 7-9 weeks to be curative.
With respect to spinal metastases, SBRT has resulted in a paradigm shift such that sufficient doses of radiation can be delivered to achieve the goals of long-term local control and improved pain control compared to historic conventional palliative low dose practice.3,4 It is also postulated that SBRT results in the activation of novel radiobiological pathways such as direct vascular damage and improved immunological response. 5 SBRT has impacted surgical decision making as well, given that it provided an alternative for some histologies considered radioresistant that would have been previously operated on even if the patient was mechanically stable and without high grade epidural disease. Ultimately, SBRT has replaced the practice of en bloc resections and given rise to the concept of separation surgery to optimize SBRT outcomes. 6
In the management of primary tumours, SBRT is increasingly applied though with care and caution as traditional conformal daily fractionated radiation is still considered the standard of care. 7 What has changed with respect to fractionated photon radiotherapy is the ability to retain the technical attributes of SBRT, that minimize the margins of uncertainty and create highly conformal dose distributions that allow for dose escalation while keeping the spinal cord within tolerance, while still delivering treatment daily over several weeks. With further improvements in technology for those spinal tumours considered radioresistant, such as chordoma, particle beam therapy is also evolving with greater precision and conformality as compared to historic devices with the development of intensity modulated proton therapy (IMPT) and incorporation of CT-based image guidance.
In this narrative review, our goal was to outline the characteristics, rationale, indications, implementations, and clinical outcomes of advanced radiation technologies in the management of extradural spine tumors. This study was exempt from informed consent requirements and institutional review board approval due to the narrative review study design.
Stereotactic Body Radiation Therapy for Metastatic Spine Tumors
Characteristics of and Rationale for SBRT
Spine SBRT was initially developed as a treatment paralleling intracranial stereotactic radiosurgery (SRS); however, as opposed to a single high total dose-per-fraction delivered using invasive immobilization, the total dose of spine SBRT is typically delivered in 1-5 fractions given the application of non-invasive body immobilization devices, image-guidance and comparatively larger target volumes. 8 The application of what would be otherwise considered a SRS dose but over a small number of fractions is known as hypofractionation SRS.
Apart from hypofractionation, spine SBRT fundamentally must meet certain other criteria: more advanced immobilization technologies compared to conventional palliative radiotherapy, creation of highly shaped doses distributions that match closely to the shape of the lesion (‘conformal’), and on-board 3D image-guidance performed just prior to delivery to confirm the location of the lesion.2,9 Respiratory motion tracking may also be used, but this is typically not done for spine SBRT as the motion of the spinal cord is negligible. 10 With these techniques, the margin of uncertainty around a target for spine SBRT is typically 0-3 mm, compared to a margin of 7-10 mm for conventional radiotherapy.11-13 The benefits of margin reduction lie in the normal tissues exposure as, for example, the amount of normal tissue exposed to the full therapeutic dose for a small target ranging from 2-3 cm would reduce by more than 80%.
Indications of SBRT for Metastatic Spine Tumors
Spine SBRT is best applied judiciously in a multidisciplinary manner involving Radiation Oncologists, Spine Surgeons, Medical Oncologists and Radiologists. 14 Inappropriate use of spine SBRT where another treatment strategy might have been better can lead to potential adverse patient outcomes such as neurological compromise, lack of symptom relief from spinal instability as well as elevated rates of late radiation-related complications. Conversely, failing to apply SBRT where it is indicated could lead to unnecessarily prolonged recovery, delayed/ineffective symptom relief, and additional patient inconvenience.
Multiple decision-making frameworks have been previously published to help optimize decision making regarding metastatic spine tumors. With respect to assessment the grade of epidural disease, the Bilsky criteria are established and validated, 15 and degree of mechanical instability should be scored according to the Spinal Instability Neoplastic Score (SINS). 16 The highly cited Neurologic-Oncologic-Mechanical-Systemic (NOMS) framework defines an approach to the patient presenting with spinal metastases such that SBRT is indicated especially for radioresistant (melanoma, renal cell carcinoma, sarcoma, colorectal, thyroid, hepatocellular and non-small cell lung cancer) histologies, previously irradiated metastases (with or without low-grade epidural disease), and post-operatively following surgical decompression of high-grade MESCC. 17 Similarly, the framework also outlines scenarios where upfront decompressive/separation surgery is indicated. However, as we have gained greater confidence in the safety profile the indications for spine SBRT have gradually broadened. For example: (1) with the Canadian SC.24 randomized trial results reported by Sahgal et al, SBRT can be applied to painful target vertebrae to improve complete response rates to pain in patients expected to survive at least 3 months, (2) following any surgical procedure as long as the target is localizable, and (3) for those radiosensitive histologies such as breast and prostate, in particular, when oligometastatic.18,19 However, the application of SBRT to those metastases exquisitely sensitive to radiation, such as arising from small cell lung cancer, hematologic and germ cell primaries, remains controversial. Early prospective data have also suggested SBRT to be feasible even for inoperable patients with higher grade malignant epidural spinal cord compression (MESCC). 20
In addition to the considerations in the NOMS framework, the patient’s overall prognosis and goals of care should also be considered. Essentially, an expected survival prognosis of greater than 3 months is optimal for spine SBRT and there are a number of available prognostic scoring systems including the PRISM index developed by the MD Anderson Cancer Center.21-25 However, it is important to note that metastatic cancer outcomes are increasingly dictated by molecular status and access to novel targeted and immunologic therapies which is discussed within this focus issue.
Implementation of Spine SBRT
The technical and clinical aspects of establishing a spine SBRT program have become increasingly well described. We provide a brief overview of the workflow, and the reader is directed to several reviews on the subject for further details.26,27
Advanced immobilization for spine SBRT is typically performed using near-rigid body immobilization in the supine position with a vacuum cushion-based system such as BodyFIX (Elekta AB, Stockholm, Sweden). Such cushions are firm and mold to the patient’s body once made, improving setup certainty and limiting movement during treatment.28,29 A thermoplastic shell molded to the patient’s head, neck and shoulders is typically used for those tumours in the cervical and upper thoracic (T5 or above) spine. Once the patient is immobilized, computed tomography (CT) and magnetic resonance (MR) images of the treatment area are obtained to facilitate planning. In cases where SBRT is carried out post-operatively, SBRT is typically delivered 10-14 days after surgery once the wound is confirmed to be adequately healed. In most post-operative cases, using a lower strength magnet (1.5 T) with appropriate artifact-suppression sequences can minimize metallic artifacts, and the visualization of neural structures is sufficient. However, when metallic artifacts persist in obscuring the spinal cord, cauda equina or exiting nerve roots, a planning CT myelogram can be performed in the same position as treatment delivery to ensure accuracy. However, this does not obviate the need for MRI and in all circumstances, high quality MR imaging is essential for the safe delivery of spine SBRT. An alternative approach to post-operative MR imaging artifact is to use radiolucent constructs such as polyetheretherketone (PEEK).
Following fusion of the T1 and T2 sequences (with or without gadolinium contrast), the radiation oncologist then contours the target volume using established guidelines.30-32 A treatment plan is generated using multiple static beams or beam arcs to achieve highly conformal dose distributions around the target, while limiting the dose to critical organs such as the spinal cord/cauda equina, exiting nerve roots, esophagus, lungs and kidneys to safe tolerances. Typical prescription doses of SBRT ranges from 18-24 Gy in 1 fraction, 24-28 Gy in 2 fractions, 24-27 Gy in 3 fractions and 30-36 Gy in 4-5 fractions. Simultaneous integrated boost techniques may also be considered. 33 A comprehensive reference for tolerance to hypofractionated radiation has recently been reported by the HyTEC group that includes spinal cord guidelines. 34
Prior to treatment delivery, 3D image guidance with cone beam CT integrated onto the radiation linear accelerator (Linac) is typically performed with the patient in the treatment position to confirm the accuracy of the final positioning. Alternative techniques include stereoscopic X-rays (CyberKnife system or ExacTrac) and integrated onboard MRI with an MR-Linac, both of which also enables ongoing position tracking during treatment delivery.
Following treatment, patients are typically followed with serial whole-spine MRIs every 2-3 months to assess the local control of the target lesion as well as to survey for distant spine failure. The SPINO guidelines provide clinical and radiological response assessment criteria after spine SBRT, 35 and quantitative thresholds have also been recently proposed. 36
Clinical Effectiveness for Spine SBRT in Metastatic Spine Tumors
Though a significant body of non-randomized evidence has accumulated supporting spine SBRT, prospective randomized evidence is still limited to a few major trials and mixed histologies.
In terms of local control, a 1-year local control rate of approximately 85%-90% has been reported following SBRT for de novo spine metastases, 37 90% in the post-operative setting, 38 and slightly lower rates in the re-irradiation setting ranging from ∼75%-80%. 39 A subset analysis of de novo patients with long-term follow-up from the recently reported SC.24 randomized controlled trial (RCT) demonstrated a significantly improved 1-year local control rate following SBRT at 94% vs 72% following 20 Gy in 5 conventional fractions (P < .001), and this provides good comparative evidence to support superiority in local control. 4
In terms of pain control, the CCTG SC.24 Phase III clinical trial showed a significantly improved rate of complete pain response at the treated levels with SBRT compared to conventional palliative radiotherapy (35% vs 14%, P < .001) among a total of 229 patients. 3 Of note, the SC.24 study included patients with intact, not previously irradiated (de novo) spine metastases with a baseline pain score ≥2 and a maximum of 3 consecutive levels in the radiation treatment volumes. The majority (92%) of patients had ECOG status of 0 or 1. Patients were excluded if there was obvious instability (SINS ≥13), neurological compromise or had a highly radiosensitive primary (seminoma, small cell lung cancer or hematological malignancies). The SC.24 trial did not see differences in other quality of life indicators between the treatment arms during follow-up, save for financial burdens that were better with SBRT. In addition to SC.24, a phase II RCT of 55 patients showed a trend towards improved pain response (>2 point improvement on the visual analog scale) with SBRT (24 Gy in 1 fraction) compared to conventional palliative radiotherapy (30 Gy in 10 fractions) at 3 months (69.6% vs 47.8%, P = .13), which was the primary endpoint, but the difference became significantly in favor of SBRT at 6 months (73.7% vs 35%, P = .02). 40 In contrast, the RTOG 0631 Phase II/III RCT, 41 showed that the proportion of patients with a ≥3 point decrease in the Numerical Rating Pain Scale at the treated levels was not significantly different between SBRT (16 Gy in 1 fraction) and conventional palliative radiotherapy (8 Gy in 1 fraction, 41.3% vs 60.5%, one-sided P = .99, two-sided P = .01) among 339 patients. 42 No differences in quality of life indicators were also found between the treatment arms of RTOG 0631. However, the difference in dose/fractionation as well as the difference in primary outcome definitions between trials make the results difficult to juxtapose directly. The trials were also limited by their unblinded nature. At this point, it would appear that a dose of at least 24 Gy in 1 or 2 fractions are required to produce a pain response superior to conventional palliative radiotherapy.
Personalization of the Approach
There has yet to be any high-level prospective evidence that 1 SBRT fractionation scheme is better than another. Practice varies according to the program, as many began single fraction SBRT similar to brain SRS. So far, based on existing RCTs, we can derive that low dose single fraction SBRT ranging from 16 to 18 Gy may not be as efficacious as higher single fraction SBRT (20-24 Gy), or those higher biologically equivalent dose fractionated schemes such as 24-28 Gy in 2 fractions.40,41,43,44 However, the expense of dose escalation within a single fraction increases the risk of vertebral compression fracture (VCF) as the sensitivity of late adverse normal tissue effects are sensitive to dose per fraction. 45 For instance, data out of MSKCC showed a radiographic fracture rate of 39% following 24 Gy in 1 fraction. 46 However, only a subset of radiological fractures would require direct intervention, and in a follow up report, the risk of symptomatic fracture without tumor recurrence was 7.2%. 47 Nevertheless, reducing overall VCF risk would likely also reduce the risk of symptomatic VCF, and this was one of the key reasons behind why 24 Gy in 2 fractions was developed. By delivering 12 Gy over 2 successive days, there is radiobiologic protection with respect to late adverse effects and the overall risk of VCF has been consistently ∼10%. 48 This held true for a recent report of 28 Gy in 2 fractions where the rate of fracture was not increased but efficacy seemed to be superior with respect to long-term local control. 44 Interestingly, these results as well as those from the recent randomized trials, seem to support the HyTEC model on spine metastases tumour control probability (TCP) where ≥90% TCP requires ≥20 Gy in 1 fraction or ≥28 Gy in 2 fractions.43,49 It is reasonable to justify dose escalated spine SBRT in those patients oligometastatic or radioresistant disease.50,51
For high grade epidural disease, the increasing use of separation surgery serves to improve the dosimetric outcomes of SBRT.
52
The outcomes following post-operative spine SBRT have been favorable, though less so if residual epidural disease was high grade.53-55 Therefore, innovative minimally invasive techniques to address epidural disease, such as spinal laser interstitial thermotherapy (LITT), may improve outcomes.
56
For inoperable patients with MESCC, there may be a role for relaxing spinal cord constraints to optimize local control.
20
Similarly, for patients with intermediate instability pain outcomes may also be compromised with radiation alone and lends to the increasing use of minimally invasive strategies such as percutaneous instrumentation and/or cement augmentation in concert with SBRT (Figures 1 and 2).
57
An innovative study by Wardak et al examined pain outcomes in patients with a baseline pain score of at least 4 and no baseline fracture. Patients underwent SBRT followed by immediate cement augmentation and excellent 3-month pain scores.
58
Therefore, addressing the microstructural compromise of the bone integrity may be the pathomechanism behind these encouraging results as these patients did not have a baseline fracture. These results still require further validation in larger cohorts to limit over-treatment in the metastatic setting. Lastly, Steverink et al
59
performed an innovative study where 10 patients underwent pre-operative SBRT with 18 Gy in a single fraction followed by stabilization within 24 h. The tissue analyses in this study provided insight into the pathobiology of SBRT-related early tissue effects such as necrosis, desmoplasia, tumour cell apoptosis and decreased tumour vessel density. These results support the “ablative” potential of SBRT. Sagittal (A) CT and (B) MRI (T1) images of a patient with renal cell carcinoma with significant disease at L4 causing mechanical pain and instability, but minimal epidural disease, who underwent planned minimally invasive percutaneous pedicle fixation and instrumented fusion of L3-L5 prior to SBRT. Following the procedure in Figure 1, the patient’s pain was much improved, and he underwent SBRT to L3-L5, 28 Gy in 2 fractions, starting post-operative day 20. Axial 1.5 T MRI images are provided to show (A) the area of most significant disease at L4 (T1), as well as a level through the L3 titanium pedicle screws on (B) T2, and (C) T1 sequences, demonstrating generally reduced imaging artifacts on T2, which are typically of sufficient quality to allow for accurate SBRT planning without myelography. The final SBRT plan is also shown on the most representative (D) axial and (E) sagittal slices. No wound complications were observed after SBRT.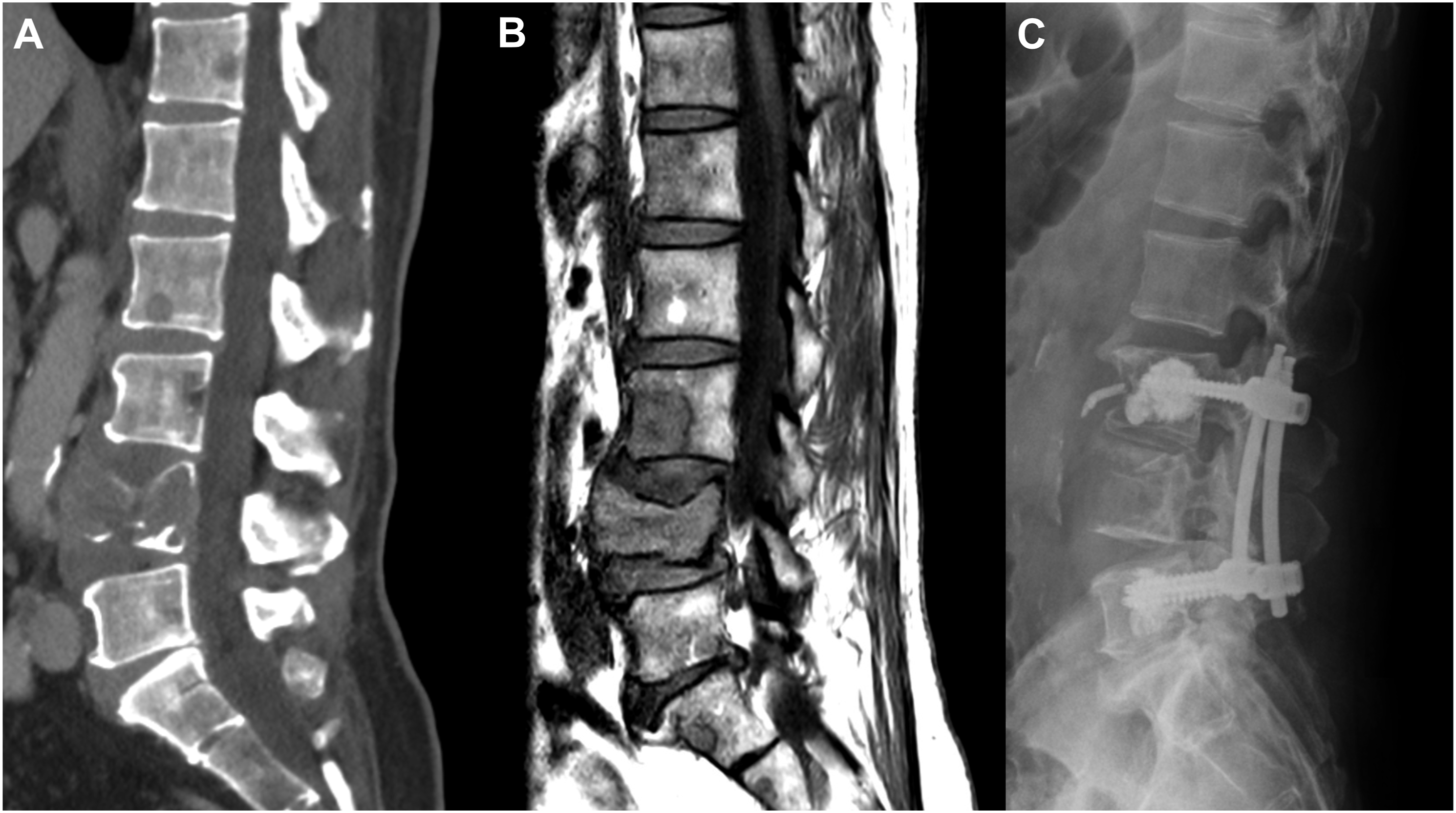
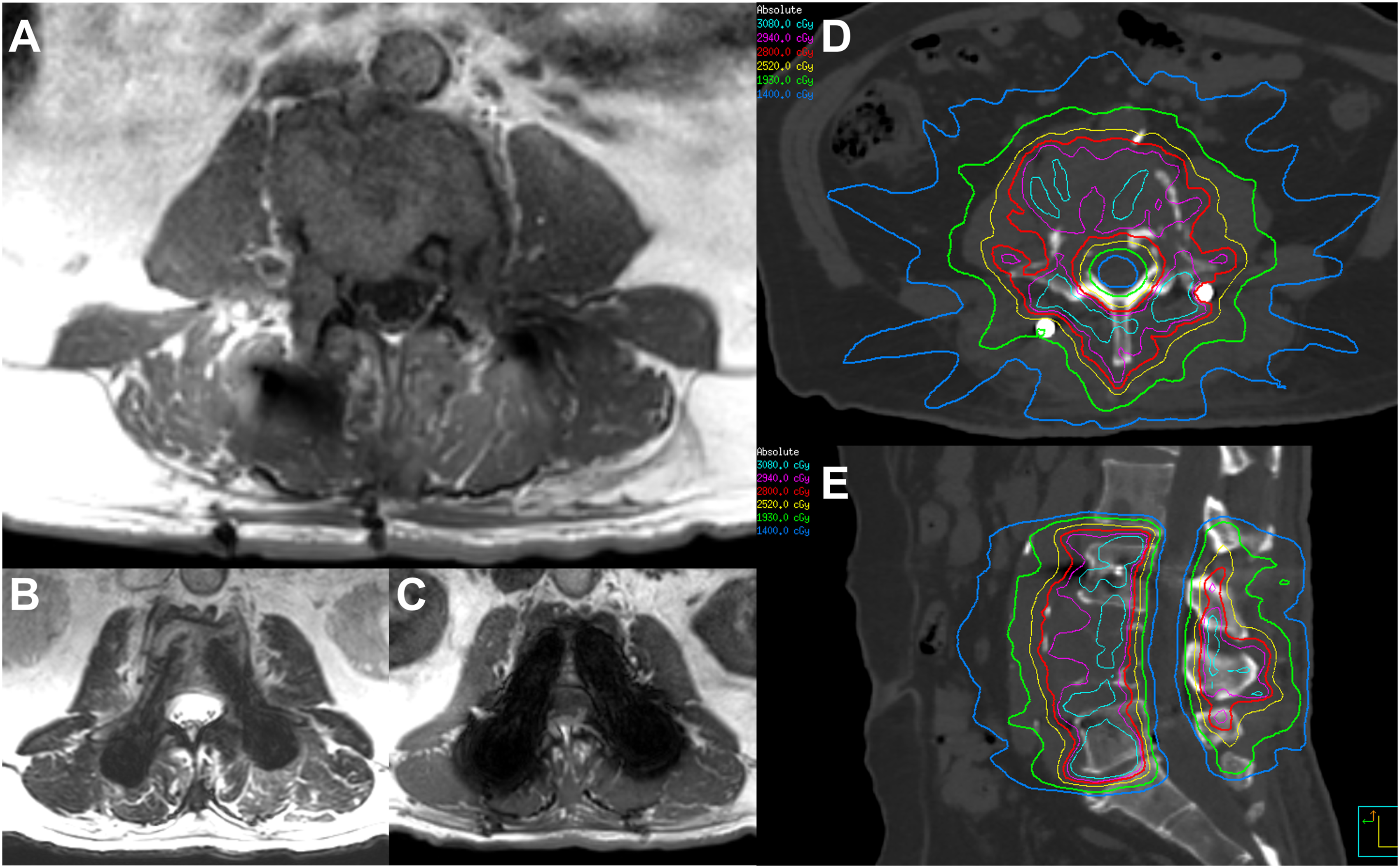
For large target volumes, high-risk re-irradiation or for physician preference to reduce the potential for adverse effects, it is common practice to increase the number of fractions of spine SBRT to 3-5. Such an approach can preserve a reasonable level of control for faster growing tumor cells while giving more breathing room to normal tissues to repair in-between fractions. An analysis of large cohort of patients with complex spine metastases treated with 30 Gy in 4 fractions of spine SBRT showed favorable local control at 84% at 2 years. 60 We are in need of dose finding spine SBRT randomized studies to determine the optimal dosing and investigations are ongoing. 61
Risks of Spine SBRT
Spine SBRT is typically well tolerated and severe adverse events are rare. In routine practice, patients may experience fatigue, pain flare reaction and site-specific acute adverse events related to radiotherapy. The pain flare reaction occurs commonly and at a similar rate as conventional palliative radiotherapy (43% vs 34% in the SC.24 trial), 3 and may be suppressed with a short course of corticosteroids.62,63 Targets close to the skin may lead to acute dermatitis, though there is a risk of causing a chronic wound if the skin dose is too high. 64 Targets close to the upper aerodigestive tract may also lead to some acute mucositis/esophagitis that is typically self-limited, though there is a risk of chronic side effects such as stenosis or fistula if a high dose is delivered to the esophagus or pharynx.3,64 However, with the application of appropriate dose constraints during radiotherapy planning, the risks of serious adverse events is expected to be very low in the de novo setting. RCTs on spine SBRT demonstrating no statistically significant differences in treatment-related side effects between SBRT and conventional palliative radiotherapy, though longer follow-up durations are likely needed for full prospective evaluations of late radiation side effects.3,40,41 Additionally, the combination of SBRT and surgery also appears to be safe, as a recent Phase I/II trial showed that spine SBRT followed by surgical stabilization within 24 hours did not result in any wound complications. 65
VCF is perhaps the most well recognized late side effect following spine SBRT. The risk of new or worsening VCF following spine SBRT has been reported to be 14% in a systematic review. 48 High-dose single-fraction spine SBRT may be associated with a higher risk of VCF, though there is controversy and data from different studies may be difficult to juxtapose due to different techniques, timings at which toxicity was evaluated, as well as how VCF was defined. 45 Additional predictors of VCF include lytic disease, existing VCF prior to SBRT, spinal deformity, age, and percent involvement of the vertebral body with tumor. 48 Further study is required to investigate whether prophylactic vertebral augmentation or stabilization might be beneficial in patients at high risk of developing new or worsening VCF following spine SBRT.66,67
Symptomatic radiation myelopathy is very rare following spine SBRT. In the de novo setting and appropriate planning techniques, the risk of radiation myelopathy is much less than 1%. 68 The HyTEC project to better define normal tissue tolerances in hypofractionated radiotherapy reviewed existing data and provided expert guidelines on the dose limits to the spinal cord in both the de novo and reirradiation settings. 34 Additional strategies to minimize the risk of radiation myelopathy include maximizing the time between radiotherapy courses when re-irradiating and using longer fractionation schemes to reduce the biologically effective dose to the spinal cord.
Though the risk of myelopathy is low, the risk of SBRT to exiting nerve roots or adjacent nerve plexi is increasingly recognized. Radiation-induced radiculopathy and brachial/lumbosacral plexopathy is observed more commonly than myelopathy in the SBRT literature, though the data for spine SBRT specifically is sparse.69-71 However, re-irradiation with spine SBRT seems to be a risk factor for these events. In a cohort of spine SBRT patients with mature follow-up data, the risk of plexopathy is 2% at 3 years and 5% at 5 years. 70 Clinicians are encouraged to minimize the possibility of these toxicities by contouring the brachial plexus and lumbosacral nerve roots to document and limit the dose to these structures using institutional or published guidelines. 72 However, a balanced approach should be taken as loss of disease control can result similarly in neurological compromise. Additionally, radiation-induced myositis, characterized by muscle weakness/spasms/pain with radiographic changes indicative of muscular inflammation, is also a possibility. Data on this phenomenon is limited to case reports and small case series.73,74 Additional study is required to determine the optimal approach to minimize radiation-induced radiculopathy, plexopathy and myositis in the de novo and re-irradiation settings.
Challenges of Imaging-Based Response Assessment
MRI is the imaging modality of choice for the follow-up and response assessment following spine SBRT. 35 The SPINO guidelines indicates one of the criteria for disease progression is “unequivocal increase in the size of the tumor” without defining a measurement cut-off. 35 There are currently no widely accepted imaging-based guidelines for response assessment following spine SBRT. Early work has demonstrated that the minimum detectable difference in GTV on follow-up spine MRI is 10.9% with high inter- and intra-rater reliability. 75 Recently, a quantitative imaging-based response assessment model from a single-centre data has been proposed based on linear measurements of GTV in each sector of the involved vertebral bodies. The model proposed that an increase of >3 mm in any dimension of the lesion, combined with >1.67-fold increase in the product of longest orthogonal measurements on the same axial plane (largest lesion dimension on any axial plane, multiplied by the lesion dimension perpendicular to it), was a strong predictor (odds ratio: 4.6 [95% CI: 1.70-13.7)) for segment specific clinical progression. 36 This model, however, needs to be verified with larger external cohorts.
Another challenging aspect in imaging-based response assessment is pseudoprogression which has been defined by SPINO as an imaging-based, transient increase in apparent tumor size following SBRT, similar to what is observed in some cases of brain glioma following radiotherapy (Figure 3).
35
In pseudoprogression, the tumor stabilizes or decreases in size on follow-up imaging without any interventions. Literature regarding pseudoprogression in spine following SBRT is limited.76-80 The incidence of pseudoprogression in the literature varies from 14% to 18% to 37%77,79,80 with case series demonstrating pseudoprogression happening 3 weeks to 3 years following SBRT.76,78 In addition, pseudoprogression is dependent on the histology of the primary tumor. In a study limited to sclerotic metastasis from prostate cancer and lytic metastasis from renal cell carcinoma,
77
it was demonstrated that in patients with prostate cancer compared to renal cell carcinoma pseudoprogression had an earlier onset (4 vs 6 months) and resolved quicker (9-12 vs 6-18 months). While we should be cognisant of pseudoprogression phenomena, further studies are required to more clearly define temporal and size cut-offs. Clinicians should keep pseudoprogression in mind when mild radiological or clinical progression is suspected soon after spine SBRT without clear neurological worsening or instability, and a multidisciplinary approach should be taken with experienced radiation oncologists and surgeons to closely monitor these patients and consider biopsy or surgery when there are uncertainties about true progression. Sagittal T1 weighted images of the thoracic spine at the level of T11 in a patient with metastatic breast cancer treated with 24 Gy in 2 fractions to T11 and T12 demonstrating pseudo-progression at the T11 vertebral target. (A) Pre-SBRT scan demonstrating the metastatic lesion as the abnormal marrow, depicted by arrowheads, (B) sagittal MRI showing the radiation isodose lines encompassing T11 and T12 and the treated volume (C) Follow-up MRI 3 months post SBRT demonstrating increasing abnormal marrow signal and (D) follow-up MRI 6 months post SBRT demonstrating marked decrease in the size and extent of marrow signal abnormality without any interval systemic therapy indicating pseudo-progression.
Currently, MRI is the standard of care by which assessments are performed. 81 However, there is increased use of PET imaging and spinal perfusion imaging that may enable more accurate response prediction, including pseudoprogression, following SBRT.82,83 Advanced imaging technologies may aid in better distinguishing pseudoprogression from true recurrence.
Emerging Radiation Therapy Techniques for Primary Spine Tumors
Primary malignancy of the spine is a relatively rare entity which presents unique challenges for the multi-disciplinary team. Common histologic subtypes including chordoma, chondrosarcoma and osteosarcoma which are locally aggressive as well as radioresistant. When a primary spinal bone tumor is suspected, a carefully planned biopsy to establish diagnosis while minimizing the risk of seeding is imperative. Given the risk of locoregional seeding, en bloc resections following Enneking principles are preferred when feasible. 84 However, these procedures are technically challenging with significant risks requiring referral to a high-volume center to optimize outcomes. 85 In a recently conducted survey of international chordoma experts from a variety of disciplines, most supported en bloc resections when feasible with acceptable morbidity for newly diagnosed chordoma of the spine. 86 There was a lack of consensus on the role of RT with en bloc resection with some omitting RT, some utilizing neo-adjuvant RT and some using adjuvant RT. If en bloc resection would result in significant morbidity, intralesional resection with adjuvant or neo-adjuvant RT was generally preferred. Intralesional resection alone for chordoma is generally non-curative. As discussed below, when patients with chordoma or chondrosarcoma undergo an intralesional resection, adjuvant radiotherapy has been implemented to reduce the likelihood of local recurrence. 87
Role of Particle Beam Therapy for Chordomas and Chondrosarcomas
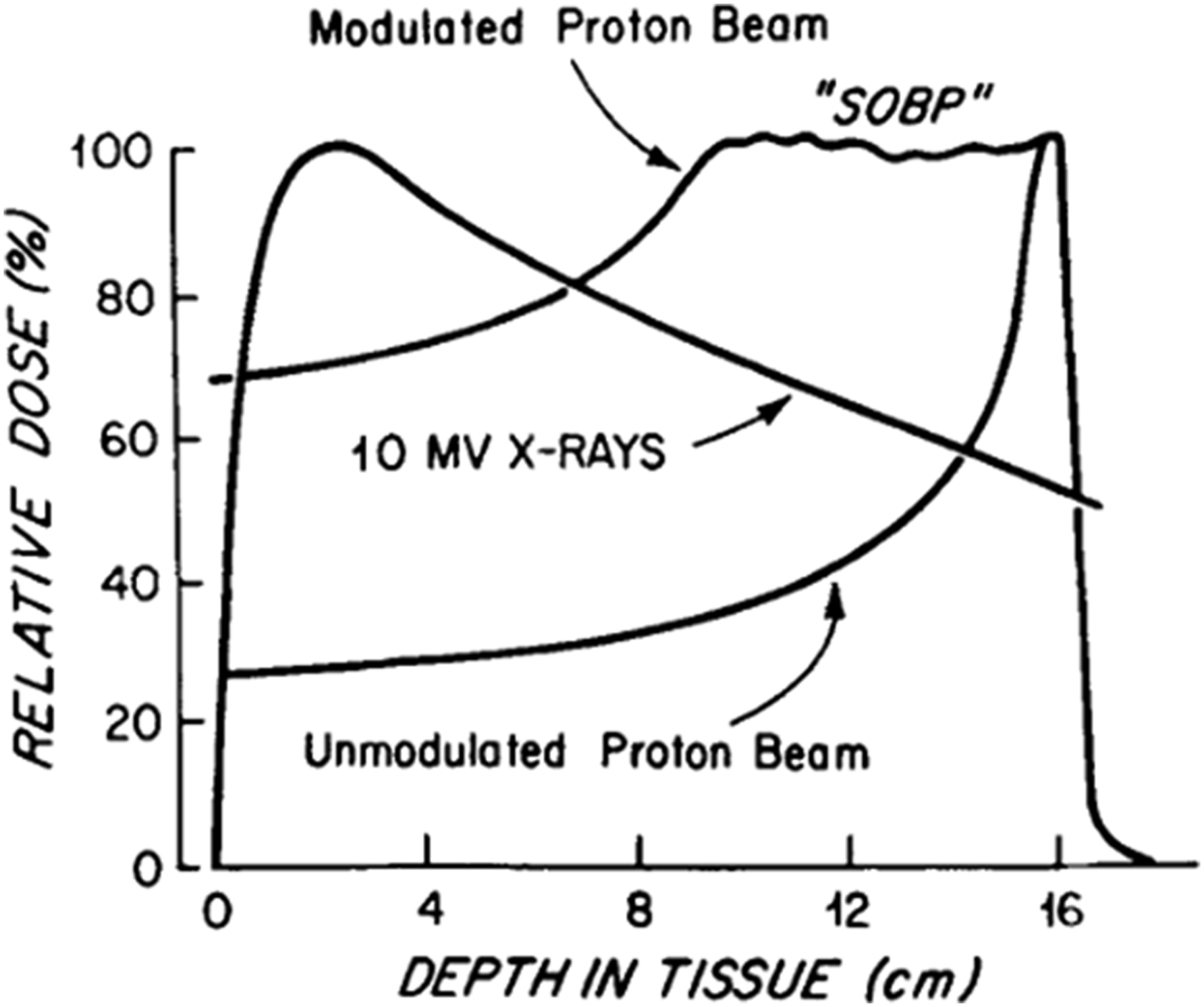
Particle therapy, such as proton or carbon ion, has been utilized historically to dose escalate beyond what can be traditionally achieved with standard 2D/3D photon-based radiotherapy. The physical characteristics of an unmodulated proton beam allow for a relatively low dose in the entrance region of the beam followed by a spike in dose deposition near the end of the particle’s range followed by a rapid dose fall off to essentially zero leading to minimum dose delivery distal to the target (the “Bragg peak”). 88 Multiple modulated proton beams can be superimposed on one another in order to create a “spread-out Bragg peak” allowing the beams to encompass a target while still largely eliminating dose beyond the target.
In contrast, standard megavoltage X-rays will enter tissue and build-up to a peak dose deposition followed by a gradual decrease in dose as a function of depth (Figure 4). This leads to additional dose delivered beyond a target and can limit the amount of radiation which can be safely delivered over a treatment course particularly in regions with nearby critical structures such as the skull base and spine. Given the radioresistant qualities of many primary spine malignancies, this can in part explain the historically poor outcomes associated with traditional 2D/3D radiotherapy when delivered following intralesional resection. Princess Margaret Hospital published their experience treating 48 patients with chordomas of the sacrum, base of skull or mobile spine, between 1958 and 1992 with 2D/3D photon therapy to a median dose of 50 Gy delivered over 25 fractions.
89
The median time to local failure was 35 months and ultimately 38 of the 48 patients suffered local-only disease failure. Relative dose at different depths in tissue for the unmodulated proton beam, 10 MV X-rays and modulated proton beam demonstrating “spread-out Bragg peak” (SOBP). Adapted from Munzenrider et al. Strahlentherapie und Onkologie.
88
Given these suboptimal outcomes with photon therapy alone, investigators at Massachusetts General Hospital explored the addition of proton therapy to photons with the purposes of safely dose escalating beyond 65 Gy. 90 Initially, ten patients with chordomas/chondrosarcomas of the skull base or cervical spine were treated with mixed photons/protons for which none suffered a neurologic injury and 9 were disease free at last follow-up. Ultimately this positive early experience led to the development of a Phase II clinical trial evaluating high-dose photon/proton radiotherapy for non-metastatic spinal sarcomas. 91 Fifty patients with either primary or locally recurrent (after prior surgery) disease were enrolled and the most common histologic subtypes were chordomas (58%) and chondrosarcomas (28%). Depending on histology, the cumulative dose to gross disease ranged from 59.4 Gy to 77.4 Gy. Impressively, the 5-year local control was 78%, and of the 36 patients treated in the primary setting (rather than locally recurrent setting) only 2 suffered a local failure. No myelopathic late toxicities were noted though 5 patients did suffer Grade 3 toxicities including 2 patients with Grade 3 sacral neuropathies upon receiving ∼77.4 Gy. Nevertheless, there appears to be a clear local control benefit for dose escalation using combined proton/photon approaches over traditional low-dose photon therapy alone.
More recently, carbon ion radiotherapy has emerged as a treatment option for oncologic patients with radioresistant disease. 92 There are roughly a dozen carbon ion therapy centers worldwide which offers several potential advantages over proton therapy including a higher relative biological effectiveness, a lower oxygen enhancement ratio and better physical dose distribution due to a reduction in lateral scattering. 92 In a recently conducted meta-analysis, the 5 year local control following carbon ion radiotherapy for patients with skull base/sacral chordoma amongst 4 studies was 80% and late toxicities uncommon. 93 Despite promising early results, high quality data from ongoing prospective clinical trials are necessary to establish the safety and efficacy of carbon ion radiotherapy for primary spine malignancies. 94
Particle beam technology continues to evolve and pencil beam scanning (PBS) approaches further improves dose conformity around complex targets over traditional passive scattered approaches.95,96 At the end of range for a standard proton beam, the linear energy transfer (LET) and radiobiologically effective (RBE) dose spikes which can lead to toxicity if not accounted for.97,98 PBS offers the possibility of LET/RBE dose optimization which may offer local control as well as normal tissue toxicity benefits. 99 Nevertheless, PBS offers dosimetric advantages over passive scattered particle therapy, and can be potentially combined with IMRT to develop highly conformal radiation plans for patients with primary malignancies of the spine even in the presence of instrumentation as illustrated in the following example.
Particle therapy does have unique challenges. The significant upfront capital costs limit widespread adoption of particle therapy worldwide. 92 According to the Particle Therapy Co-Operative Group, there are 14 carbon ion facilities and 117 proton facilities as of January 2024. 100 As such, access is a significant barrier for patients and providers. Moreover, certain clinical scenarios may render particle therapy inferior to photon-based RT. For instance, patients with instrumentation appear to have inferior outcomes following proton-based radiotherapy for spinal chordoma relative to patients without instrumentation. In the aforementioned Phase II clinical trial conducted out of MGH, 5 of 16 patients (31%) with spine stabilization hardware suffered a local failure compared with 4 out of 34 patients (12%) without hardware suggesting the presence of spinal stabilization as a negative prognostic factor. 91 This may be due to a confounding variable such as a higher likelihood of residual gross disease or more challenging location in the spine. In a separate report from Paul Scherrer Institute, patients with chordomas of the spine or sacrum were treated with proton therapy alone (n = 31) or a mix of protons with photons (n = 9) to a median dose of 74 CGE. Of the 19 patients without stabilization hardware, 1 experienced a failure. However, of the 21 patients with stabilization hardware, 12 (57%) suffered a local failure. This highlights a potential challenge with utilizing particle therapy in patients with hardware and why centers are combining photons with particles when significant hardware exists in-field.
Advances in Photon Radiotherapy for Primary Spine Tumors
Intensity modulated radiation therapy (IMRT) is a radiation technique which utilizes inverse-planning to vary the intensity of radiation within a given field to develop complex radiation plans allowing for concave isodose lines to conform high dose regions while limited exposure to nearby organs at risk. In combination with advanced image guidance tools (IGRT) integrated with the LINAC, precise delivery of these complex plans allows for further dose escalation beyond what was safely feasible with 2D/3D photon techniques. This is particularly useful in the mobile spine/sacrum where disease and at-risk tissue lay in close proximity and safe dose escalation with IMRT-based photon radiation may offer improved local control for radioresistant disease. IMRT-based high-dose treatment courses have been associated with excellent 5-year local control for chordomas/chondrosarcomas of the skull base. 101 A recently published Cochrane review compared outcomes for chordoma patients treated with protons or photons (including IMRT) to a dose of >60 Gy. 102 They found no difference in local control or toxicity between the modalities.
IMRT combined with IGRT and rigid immobilization can allow for the precise delivery of ablative stereotactic radiation doses to spinal targets as discussed in the previous section. The biologically equivalent dose of 24 Gy delivered in a single fraction is higher than the doses achieved with protons or carbon ion therapy. 95 Single fraction SRS shows promise as an emerging treatment option for patients with chordoma of the spine and sacrum in early clinical reports. Investigators at Memorial Sloan Kettering Cancer Center published a preliminary report on 24 patients treated to a median dose of 24 Gy in 1 fraction demonstrating 95% local control with a median follow-up of 24 months. 103 In follow-up, they published the results of 35 patients with de novo chordoma of the mobile spine and sacrum with a median post-SRS follow-up of 39 months. 7 Of the 32 patients who received 24 Gy in 1 fraction, the 5-year local recurrence free survival was 90%. Hypofractionated SBRT also shows similar preliminary efficacy outcomes. 104 Longer follow-up as well as prospective investigation is necessary to validate the results and determine the optimal indication and dose/fractionation for chordoma treated with spinal SBRT.
Radiotherapy plays a key role in optimizing local control for patients with many primary spinal spine tumors particularly when intralesional resections are performed. Dose escalation is clearly associated with improved local control. A variety of techniques may be utilized to achieve adequate dose including particle therapy, IMRT and SBRT. Ongoing prospective investigations such as the randomized trial of protons vs carbon ion radiation therapy in patients with chordoma of the skull base (HIT-1) are critical to provide insight on the safety and efficacy of these emerging platforms. 105 Similarly, prospective investigations are necessary to evaluate the optimal integration of SBRT, particle therapy and IMRT, with surgery in primary spinal tumors.
Case example
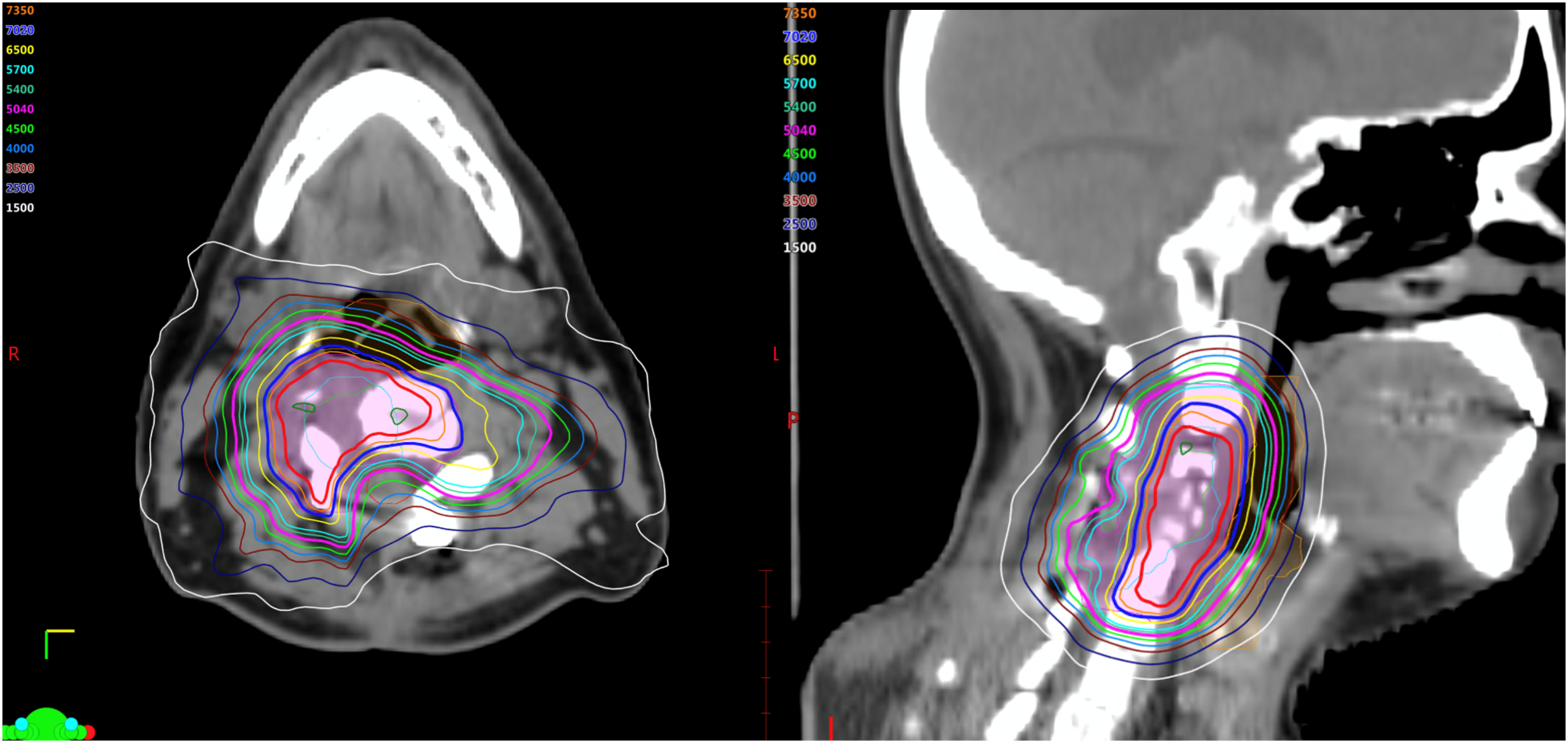
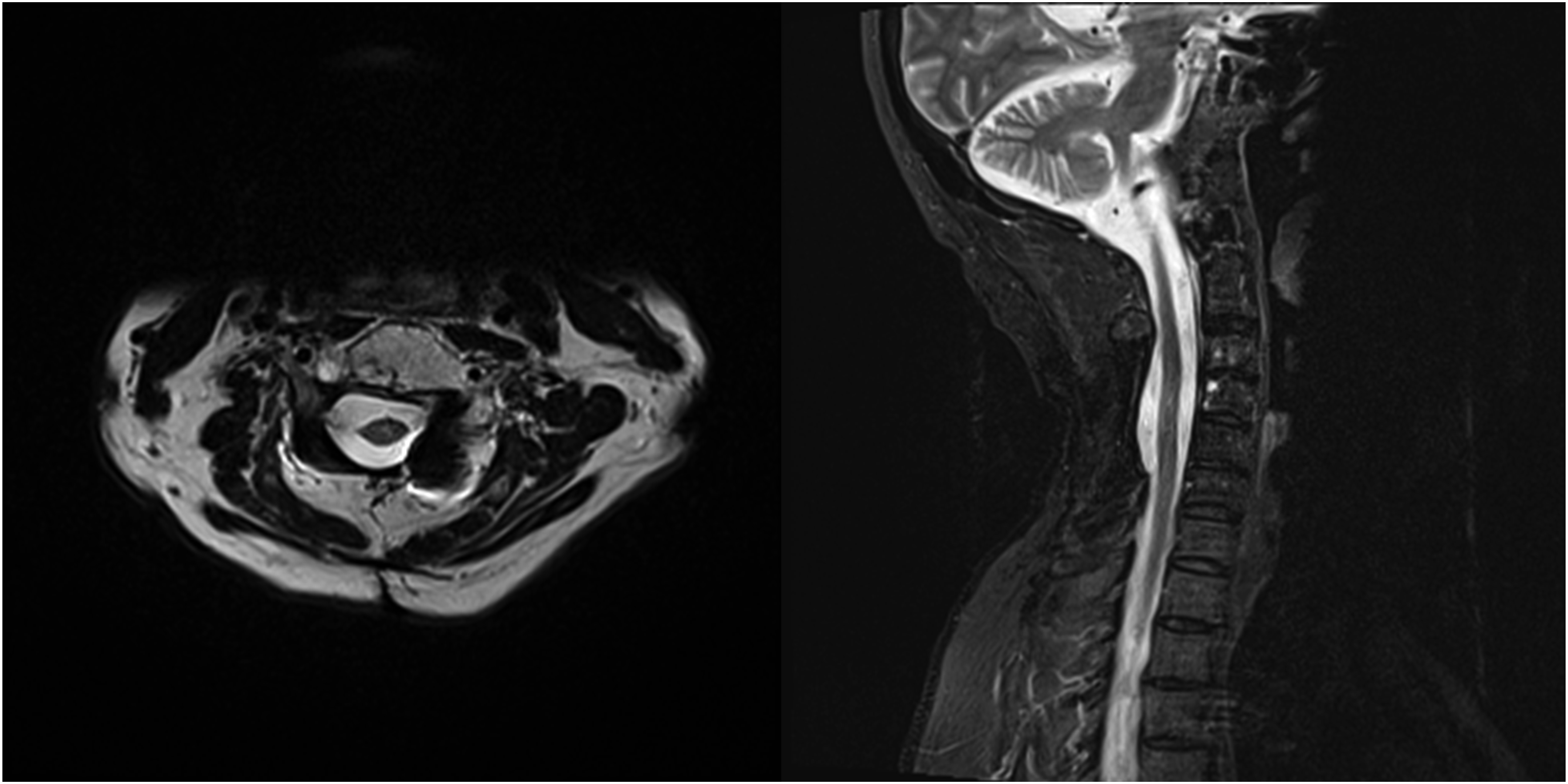
This is a 47-year-old patient who presented from an outside facility with 3 weeks of neck pain radiating to bilateral upper extremities and an MRI demonstrating a T2 hyperintense lesion at C4-C5 with epidural extension and invasion into the neural foramena on the right (Figure 5). Due to the acuity of his symptoms, he underwent decompression and stabilization with pathology revealing chordoma. Post-operative imaging confirmed gross residual disease. Given the location, presence of hardware and gross residual disease, the patient was treated with adjuvant radiation combining IMPT with IMRT. He received 50.4 CGE over 28 fractions with IMPT followed by 2 sequential IMRT based cone down boosts first to 70.2 Gy then 77.4 Gy. The composite plan is shown in Figure 6. He tolerated the treatment well without significant toxicity and remains disease free 5 years later (Figure 7). Axial and sagittal MR images of the initial chordoma at C4/5 prior to treatment. Composite dose distribution for the combined postoperative intensity-modulated photon radiation therapy (IMRT) and intensity-modulated proton therapy (IMPT) treatment courses delivered to the residual chordoma. Axial and sagittal MR images 5 years post-therapy, demonstrating durable disease control with combined IMRT/IMPT treatment for chordoma with gross residual disease postoperatively.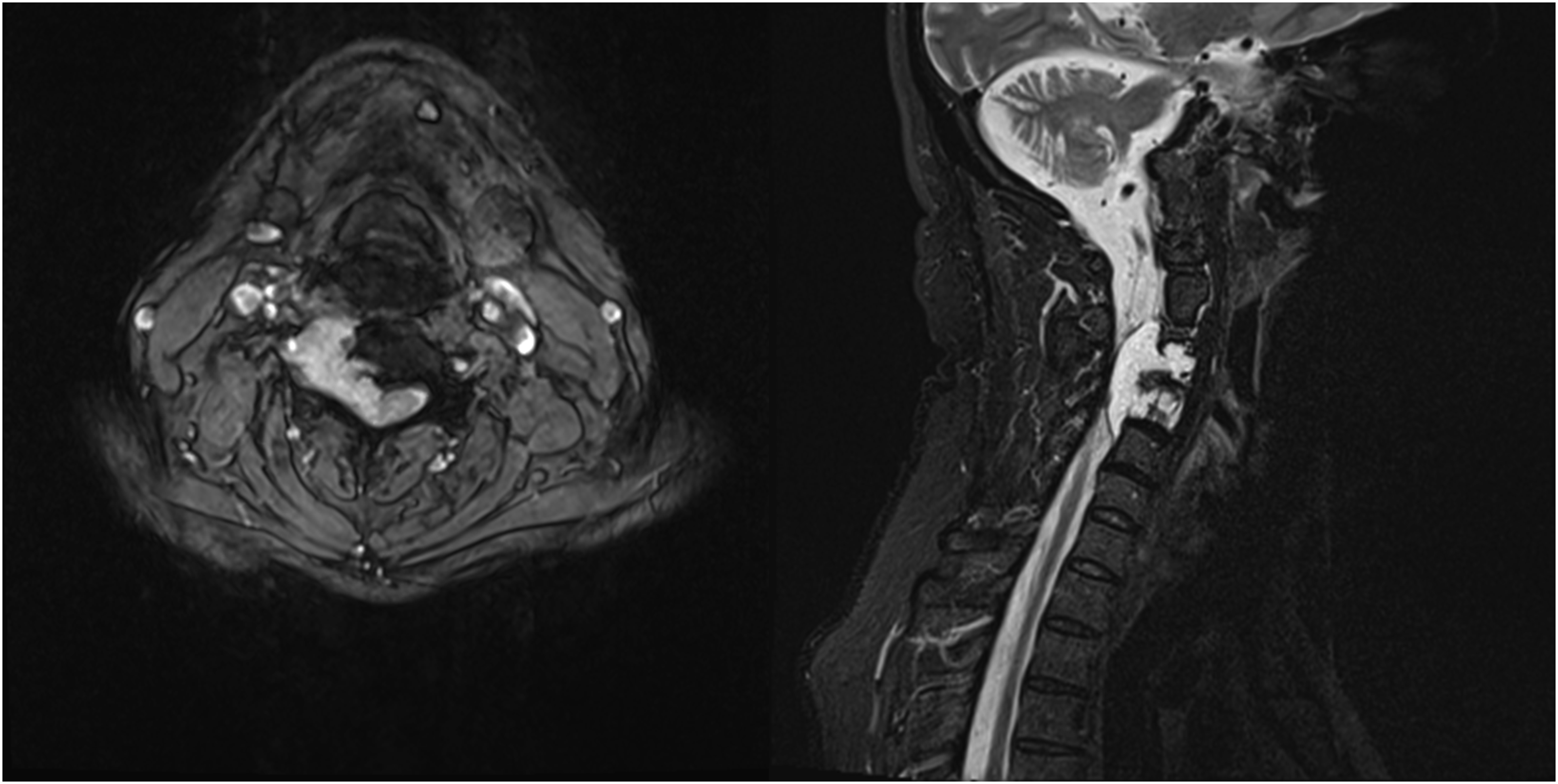
Conclusion
SBRT for metastatic spine tumors in the de novo, post-operative and re-irradiation settings are being increasingly supported by retrospective and prospective evidence as a safe and effective treatment modality. An individualized and multidisciplinary approach should be taken to incorporate SBRT as a key component of care for eligible patients with metastatic spine tumors. Additional study is required to better understand late toxicities from spine SBRT. Technological advances have also enabled additional radiotherapy options that may eventually supersede conventionally fractionate photon radiotherapy in the treatment of primary spine neoplasms.
Footnotes
Acknowledgments
This study was organized and funded by AO Spine through the AO Spine Knowledge Forum Tumor, a focused group of international Tumor experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AJG reports honoraria from BrainLab; CB reports consulting fees from Depuy-Synthes, Bionaut Labs, Haystack Oncology, Privo Technologies and Galectin Therapeutics, travel support from AO Spine and co-founding and membership to the Board of Advisors for OrisDx and Belay Diagnostics; SB reports consulting fees Nuvasive, travel support from Nuvasive and Stryker and participation in the AO Spine Knowledge Forum; ND reports consulting fees from Stryker, Cerapedics, Baxter and Medtronic, travel support from AO Spine, and stocks/options in Medtronic; CGF reports royalties from Medtronic, consulting fees from Medtronic and Nuvasive, and fellowship support from AO Spine, Depuy and Medtronic; ZLG reports honoraria from AO Foundation and AO Spine Knowledge Forum Tumor, payments for expert testimony from various sources, travel support from AO Spine, participation on the Advisory Board for Proprio, and stocks/options in Lenoss Medical; IL reports royalties from Globus, consulting fees from Depuy Synthes, honoraria from AO Foundation, travel support from AO Foundation, and participation on the Advisory Board for Chiefy; AL reports grants from the European Commission Horizon Europe, Hungarian Academy of Scient, and the National Research, Development and Innovation Fund, royalties from Sanatmetal Ltd., and consulting fees from Inossia Ltd. and Metronic; JR reports consulting fees from Cerapedics, honoraria from Gobus, Depuy Synthes, Cerapedics, travel support from AO Spine, chairing the Advisory Board of Cerapedics, and membership in the Steering Committee of the AO Spine Knowledge Forum Tumor; JJV reports founding and stock ownership of SentryX; LDR reports consulting fees from Icotec and Stryker, honoraria from Icotec and Stryker, and travel support from Icotec and Stryker; AS reports grants from Elekta AB, Varian, Seagen Inc., and BrainLab, consulting fees from Varian, Elekta AB, BrainLab, Merck, AbbVie and Roche, honoraria from AstraZeneca, Elekta AB, Varian, BrainLab, Accuray, and Seagen Inc., Vice Presidency of the International Stereotactic Radiosurgery Society (ISRS), co-chairing the AO Spine Knowledge Forum Tumor, membership and clinical steering committee member of the Elekta MR Linac Research Consortium, chairing the Elekta Oligometastases Group and the Elekta Gamma Knife Icon Group. All other authors declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
