Abstract
Study Design
Systematic review.
Objectives
The objective of this review paper was to summarize targeted molecular therapy options for spinal chordoma and chondrosarcoma, and to provide an update on the relevant clinical trials open for recruitment.
Methods
A systematic review of the current literature was performed, according to PRISMA guidelines, to summarize the latest developments in non-surgical molecular treatment options for low grade malignant primary spinal tumours. We also summarize those actively recruiting clinical trials based on clinicaltrials.gov.
Results
A total of 73 studies and completed clinical trials were reviewed. Twenty actively recruiting clinical trials (eight for chordoma and twelve for chondrosarcoma) were identified.
Conclusions
There is a strong need to find new therapeutic options to complement surgical resection and radiation therapy, which remain the cornerstone of management. Targeted therapies against molecular pathways show promise as compared to conventional chemotherapy.
Introduction
Chordoma (CH) and chondrosarcoma (CHS) are rare low-grade malignant tumours (LGMTs) affecting the spine. These diseases are characterized by slow growth and late development of distant metastasis; however, they are locally aggressive, tend to recur locally and are potentially lethal due to the biological, neurological and biomechanical consequences.
Conventional chemotherapy has limited therapeutic impact in LGMTs, and the role of radiotherapy is typically in the adjuvant post-operative setting. Ultimately, the first line of treatment is surgery with an aggressive curative procedure as the goal. The main prognostic factor for local recurrence (LR) is the quality of surgical margins (i.e., wide/marginal en bloc vs intralesional resection). 1 The localisation of the primary lesion also influences the outcome of the surgical treatment. Results from a large multicentric sacral CH cohort report an odds ratio (OR) for LR of 2.43 in the case of intralesional (Enneking inappropriate, EI) resections compared to Enneking appropriate (EA) en bloc resecetions. 2 The same study group found that the OR for LR in the case of EI surgical treatment was 7.02 in mobile spine CH patients. 3 For CHS, the LR rate in a 10 year follow-up period were 21% in EA and 48% in EI resections. However, in this large cohort, the LR risk was significantly higher in sacral CHS (45% vs 21%) which was significantly associated with disease related mortality (risk ratio = 3.6). 4
In the real world, Enneking appropriate – en bloc – resections are simply not possible for every patient due to the anatomical localization (i.e., skull base or sacrum) and the extent of disease.5,6 Moreover, radical, wide surgical resections can be usually associated with severe complications, loss of function and poor quality of life.7,8 Therefore, a significant proportion of surgical resections are Enneking inappropriate. Despite aggressive surgical resections, in either case, effective adjuvant therapy would be desirable in particular when there is residual disease.
Radiotherapy (RT) requires a high dose approach (more than 70 Gy) for both CHS and CH as conventional dosing does not yield long term local control. In particular for CH, it has been shown that a total dose less than 60 Gy given in conventional fractionation is associated with poor local control. Therefore, practice typically varies from 70 to 78 Gy, with a recommendation of at least 74 Gy with photon or protons. Dosing can vary if carbon ion therapy applied. 9 For CHS, typical radiotherapy practice is to treat adjuvantly in the range of 70 Gy in 35 fractions. The higher radiation doses required for CH are not required for CHS as it is more sensitive to the effects of radiation. 10 As a result, if the en bloc Enneking-appropriate surgical resection would result in unacceptable functional loss, or tumour-free surgical margins are not achieved, long term local control can be expected after effective radiation. The application of stereotactic body radiotherapy for LGMTs is in evolution with early encouraging results. 11
Chemotherapy has long been known to be ineffective in CH, reports of tumour response to chemotherapy (cisplatin, anthracyclines) have been anecdotal. Therefore, chemotherapy has played a limited role in the management of CH. 1 Adjuvant chemotherapy may provide some survival advantage in dedifferentiated and mesenchymal CHS, but there is little evidence supporting the effectiveness in the conventional CHS subset. 12 In a single institution study of 42 patients, conventional chemotherapy provided no benefit in disease-free survival for conventional CHS. 13 Chemotherapy resistance in CHS may be linked to the expression of multidrug resistance 1 gene (MDR-1). Moreover, the access of chemotherapeutic substances may be hindered by poor vascularity of the tumour and large amount of extracellular matrix, implying that the drugs have to diffuse relatively long distances to the tumour cells. 14
Low-grade malignant tumours are challenging to treat with conventional oncological treatment modalities (limited effectiveness of RT and chemotherapy), thus recent research and development projects aim to find new therapeutic methods targeting molecular pathways. In the previous decade, treatment of other malignancies like non-small cell lung cancer (NSCLC) has evolved to a great extent with the discovery of oncogenic driver mutations allowing for patient specific targeted treatment. For example, epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors provide a favourable treatment outcome in EGFR mutation positive lung cancers. 15 In melanoma, 5-year survival rates greatly increased compared to 10 years ago from <5% to around 30% secondary to the advancements in targeted molecular and immunotherapy. 16
This review summarizes the current state of targeted molecular and immunotherapies as well as other novel systemic therapeutic options for CH and CHS.
Methods and Search Results
Search Strategies
Electronic searches were performed by March 10th, 2023 using PubMed, MEDLINE and Cochrane libraries. All searches and selection were performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines and recommendations. 17 The search strategies can be found in Supplement 1. We included studies detailing other tumour types if either CH or CHS were among them. We also performed a search on clinicaltrials.gov to find and list all ongoing clinical trials and actively recruiting clinical trials. Informed consent and institutional review board approval were not required due to the nature of the study.
Selection Criteria
Selection criteria included the following: ongoing and finished clinical trials for patients with CH and CHS at any anatomic location along the spine, we also included studies with other type of tumours as long as at least one patient with CH or CHS was included in the paper; in patients over the age of 18 undergoing treatment with at least one agent with or without other kind of therapies. Case reports were also taken in consideration as long as the above-mentioned criteria were met. Previous reviews in this subject were also included in the systematic review. Animal models and in vitro studies, studies with not specified agents, studies of non-spinal CHS or CH were excluded from the selection. All articles were screened in accordance with the selection criteria by three independent investigators. Any discrepancies were resolved through discussion until consensus was reached if no consensus was reached the senior investigator made the final decision.
Search Results
The initial search resulted 311 studies. After duplications were removed 269 article abstracts were screened. We excluded 73 articles based on their abstracts, then we thoroughly screened 196 full-text papers for potential inclusion. Animal and in vitro studies, studies with insufficient data (i.e., no drug type), studies without targeted therapy were excluded, resulting in 73 papers included in this review (3 case report, 3 case series, 21 clinical trial, 24 original article, 22 review). We also identified 20 actively recruiting ongoing clinical trials (12 for CHS, 8 for CH) from clinicaltrials.gov.
Possible Molecular Targets
Chordoma
PDGF and PDGFR Pathway
Platelet derived growth factors (PDGF) and platelet derived growth factor receptors (PDGFR) are a family of receptors and hormones that have been shown to influence the growth and invasion of numerous tumours, through modification of stem cell recruitment, angiogenesis, and modification of tumour microenvironment. 18 PDGF and PDGFR has been shown to be expressed in CH, and their overexpression is associated with worse overall outcomes and a more aggressive phenotype. 19
There are therapeutic agents such as the tyrosine kinase inhibitor imatinib mesylate target PDGF. Numerous clinical trials20-22 and case presentations23-25 have assessed the effectiveness of imatinib in CH patients. A modest clinical response was shown in 7 imatinib treated chordoma patients with long lasting stable disease and a median progression free survival (PFS) of 10 months (range: 4.2-18+ months). 21 Severe adverse events developed in a significant number of patients, therefore, the treatment was suspended in 7 cases of 26 patients. 21 Dose reduction were needed in 4 cases (28%) which also highlight the potential toxicity of this therapy in this population. 21 Stacchiotti et al reported on imatinib combined with everolimus (an mTOR inhibitor, see later) yielding a median PFS of 14 months. This result suggests that targeting more than one pathway can increase the treatment efficacy for CH. 20
VEGFR Pathway
Vascular endothelial growth factor receptor (VEGFR) physiologically plays a role in developing and sustaining angiogenesis. Tumour growth is often correlated with the level of VEGFR-induced neoangiogenesis. 26 A review concluded that VEGF inhibiting molecules modulate the tumour immune suppressive microenvironment by enhancing T-cell response. 27 Genetic profiling of CH has revealed that CH overexpresses VEGFR such that it vividly stained positive in more than 80% of CH samples.26,28
Sorafenib, another tyrosine kinase inhibitor, has profound effect on VEGFR. 29 A clinical trial reported a 12 month PFS rate of 73% in CH patients, 29 furthermore, PFS was correlated with the level of circulating VEGFR. 29 Sunitinib was also used to treat a patient with a clival CH as reported in a case report. According to the Response Evaluation Criteria in Solid Tumours (RECIST), the patient had 24 months of stable disease (SD). 23 Sunitinib also has activity against PDGFR, FLT3, KIT, CSF-1 and RET.
In a trial published in 2020, VEGFR inhibitor apatinib was used to treat CH patients with a promising PFS rate of 18 months. 30
EGFR Pathway
Epidermal growth factor receptors (EGFR) maintain and regulate the epithelium. EGFR activation by mutation and amplification has been shown to increase growth and resistance to various drugs in different tumours. 31 CH is known to be immune reactive to activated EGFR but does not harbour mutations in EGFR. Several agents have been used in CH targeting EGFR. 32 Lapatinib has shown a clinical benefit rate of 22% (complete response (CR) + partial response (PR) + SD ≥ 6 months). 33 A retrospective study assessed the responsiveness to various off label used agents in CH patients, and found a PFS of 15 months with erlotinib. 34 An attempt at trying to define the activity of cetuximab, a monoclonal antibody against EGFR is ongoing.
mTOR Pathway
The mTOR pathway is involved in cell growth, cytoskeletal reorganization and proliferation. 35 Pathological activation of mTOR promotes tumour resistance to chemotherapeutic agents. A study estimated that CH may be responsive to mTOR inhibitor rapamycin/sirolimus based on the 65% of incidence of mTOR positivity in CH. 36 mTOR inhibitors as monotherapy showed little efficacy, but combinations of everolimus plus imatinib and imatinib plus sirolimus showed promise. 37 In a single case presentation, a patient treated with imatinib combined with the mTOR inhibitor everolimus showed stable disease for 16 months. In another study imatinib combined with everolimus, amongst 9 CH patients one achieved a PR, 7 SD, and one progressive disease (PD) at 3 months. The overall clinical benefit rate (RECIST CR + PR + SD ≥ 6 months) was 89%. 38
Nuclear Export Pathway
The tumour surveillance functions of most tumour suppressor proteins are dependent on their location within the cell nucleus. In most neoplastic cells, increased nuclear export results in the inactivation of tumour suppressor proteins. Exportin 1 mediates the nuclear export of numerous mammalian cargo proteins. Exportin 1 is overexpressed in most cancerous cells and is typically correlated with poor prognosis in pancreatic, ovarian, and cervical cancers. Inhibition of exportin 1 is expected to result in activation of tumour suppressor proteins.39-41 Selinexor is a highly specific inhibitor of exportin 1, which showed promising results in solid tumours in a completed trial (NCT01607905); however, the only one recruited CH patient did not show response to the treatment. 39
Chondrosarcoma
“Classical” Molecular Targeted Therapies
Several different histologic CHS subtypes have been identified through novel signalling pathways, which encouraged investigation into the therapeutic efficacy of inhibitors of the commonly activated receptor tyrosine kinases. 42 Conventional CHS is characterized by over expression and/or activation of PDGFR, however, the tyrosine kinase inhibitor imatinib was of no clinical benefit in 26 patients with metastatic advanced CHS. 43 Moreover, dasatinib (another PDGFR inhibitor) was ineffective in 31 patients in another study. 44
A combination of sirolimus (mTOR inhibitor) and cyclophosphamide showed modest clinical activity in 10 patients. In that study, one objective response was observed, 6 had SD for at least 6 months, and 3 had PD. The median PFS was 13.4 months. 45 Selective mTOR inhibitors such as the “classical” molecular targeted therapeutic agent sirolimus can promote drug resistance through suppressing negative feedback loops that oppose their antiproliferative effect by overactivating Akt phosphorylation. 45
A phase II clinical trial with pazopanib, an anti-VEGFR tyrosine kinase inhibitor was conducted in 26 patients with extraskeletal myxoid CHS (EMC). EMC is extremely rare subtype of CHS; it is marked by a specific translocation with the gene NR4A3. 46 22 patients were evaluable for analysis at the primary endpoint. Four patients had a RECIST objective response. They concluded that pazopanib had a clinically meaningful antitumour activity in patients with advanced and progressive EMC, and could be a second line treatment option if the anthracycline-based first line chemotherapy fails (NCT02066285). 46
HDAC Inhibitors
Histone deacetylase (HDAC) inhibitors cause hyperacetylation of histones which leads to gene transcription and inhibits cell proliferation, invasion, formation of metastases and neoangiogenesis. 47 HDAC inhibitor treatment induces apoptosis, cell cycle arrest and differentiation in numerous tumour cells including CHS. 47 There was a phase II clinical trial involving romidepsin a HDAC inhibitor used on 40 CHS patients. The trial ended in 2017, only one patient finished the trial, the median PFS was 1.9 months, and the median survival was 12.2 months. There was no patient with complete response. 48
IDH Mutations
Isocitrate dehydrogenase (IDH) is an enzyme that helps in the conversion of isocitrate to α-ketoglutarate. IDH1 is localized in the cytoplasm, IDH2 and 3 act in the mitochondria. Mutation on IDH1 and 2 can lead to gain-of-function by which these enzymes can convert α-ketoglutarate to oncometabolite D-2-hydroxyglutarate leading to DNA hypomethylation. 49 IDH1 and 2 mutations are early events in different tumourigenesis. 49 Around 54% of CHS contain mutations in IDH1 or IDH2. 49 These mutations elevate oncometabolites (D-2-hydroxyglutarate, 2-HG), leading to DNA hypermethylation. 49 Ivosidenib, a mutant IDH1 inhibitor, in a phase I clinical study showed proof of clinical activity, tolerability and significant reduction of the 2-HG (by up to 98%) in plasma and in the tumour in 21 patients with advanced CHS. 50 Clinical trials evaluating response rates and PFS in patients with CHS are ongoing.
Hedgehog Signalling Pathway
The Hedgehog signalling pathway plays a crucial role in many developmental processes such as pattern formation, proliferation, differentiation, and apoptosis. 51 In a phase II clinical trial hedgehog pathway inhibitor saridegib was used on patients with metastatic or locally advanced CHS, the trial was discontinued early after the analysis of available data showed that saridegib was not superior to placebo. 42
Src Family Pathway
The Src kinase family is a family of non-receptor tyrosine kinases. It interacts with cytosolic, nuclear and membrane proteins. Dysregulation of the Src family contributes to progression of cellular transformation, and oncogenic activity. In CHS, the Src pathway seems to be active and inhibition by dasatinib has led to reduced CHS viability in in vitro studies. 44 Unfortunately, in a phase II clinical trial, dasatinib failed to achieve local control for at least 6 months in more than 50% of the patients, thus proved ineffective in CHS (NCT00464620). 44
RET Pathway
Rearranged during transfection (RET) gene is a proven proto-oncogene. Genomic alterations involving RET are oncogenic drivers of numerous tumours. 52 RXDX-105 is a VEGFR sparing multikinase inhibitor with activity against RET. In a phase I/Ib clinical trial, it was shown that adverse event profile of RXDX-105 appears manageable, and showed preliminary efficacy in patients with RET alterations in CHS (NCT01877811). 53
Other Agents
Currently there are numerous chemotherapeutic agents, herbal agents, molecular targets, pathways that are under excessive research in the treatment of chemotherapy resistant CHS. 42 Most of these substances are in the in vitro or in animal model in vivo stage and promising human results are lacking. 42
Ongoing Clinical Trials
Ongoing Clinical Trials Open for Recruitment in Chordoma (A) and Chondrosarcoma (B).
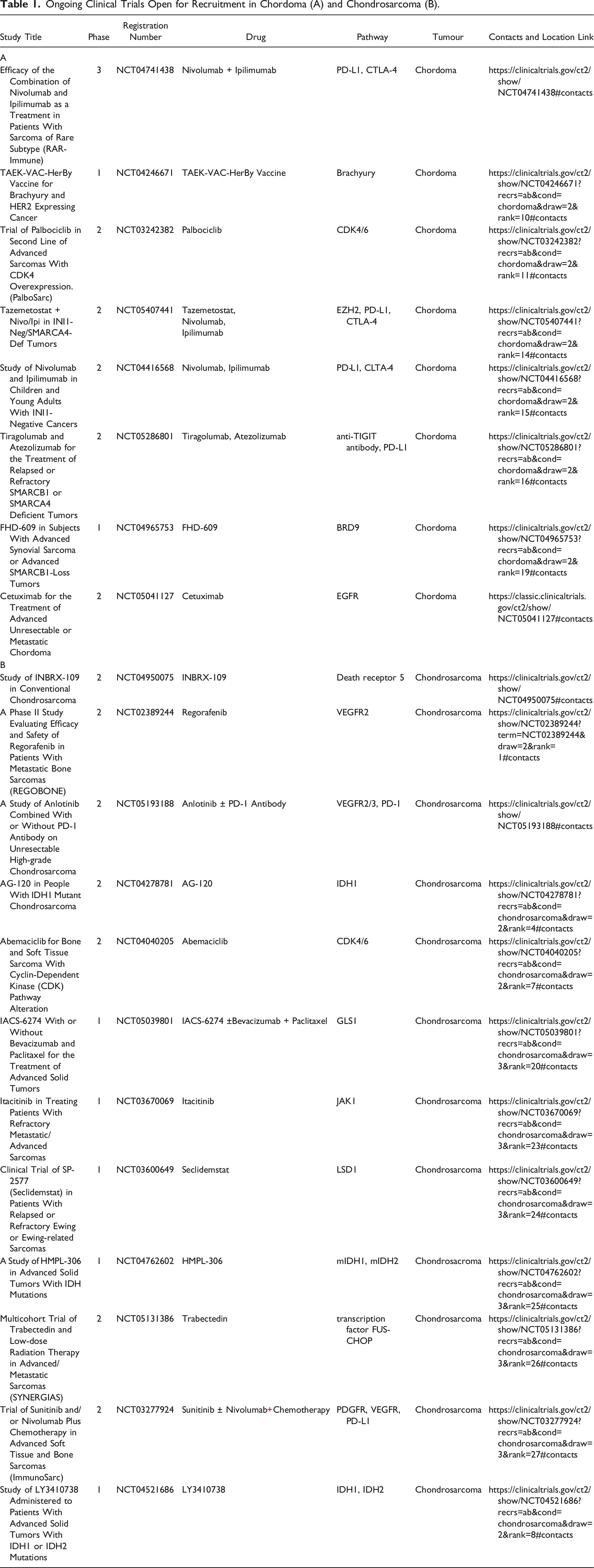
Chordoma
Brachyury
Brachyury is an oncogene transcription factor of the T-box gene family, with high expression in many malignancies (i.e.,: chordoma). It facilitates tumour resistance to chemotherapy and radiotherapy, contributes to proliferation. 54 During the embryogenesis, brachyury regulates mesodermal differentiation to notochord, and maintains stem cells during the development of neural axis. 55 CH in almost all cases overexpress brachyury, however its role of the level of overexpression in the prognosis has not been fully elucidated.56,57
There have been a few trials with brachyury expression targeting vaccination. One trial, focusing on the which was a combination of different acting agents was withdrawn before the first patient enrolment (NCT03647423, withdrawn). Clinical trial with GI-6301 yeast-brachyury vaccine was in phase II (NCT02383498, discontinued), however due to ineffectiveness the trial was discontinued early. 58
There is an ongoing clinical trial with afatinib, an EGFR inhibitor which has some effect on brachyury (NCT03083678, active not recruiting).
Growth Factors Pathways Targeting Trials
Multiple kinases are under clinical trials targeting VEGFR, PDGF or PDGFR and EGFR pathways. Regorafenib a multi-kinase inhibitor proved to be safe to use in patients with CH and CHS. In a phase II clinical trial (NCT02389244, active recruiting) 46 patients were involved from 2014 to 2019. Forty patients were evaluated for efficacy, 24 on regorafenib and 16 on placebo. It showed promising result with a median PFS at 19.9 weeks vs 8 weeks in the placebo cohort. Moreover at 12 weeks, 13 patients were non-progressive vs 5 in the placebo cohort (Figure 1). The trial is scheduled to finish in 2025.
59
Regorafenib in recurrent thoracic chondrosarcoma. T2 weighted MRI of a 79 year old male patient with a 6-year long history of thoracic chondrosarcoma. A large recurrent tumour at ThVI-VII (A) was surgically treated with a partial, intralesional resection (B), but the Grade II chondrosarcoma developed a fastly growing local recurrence in 3 months (C). Regorafenib treatment was initiated and showed stable disease status at 3-month FU (D).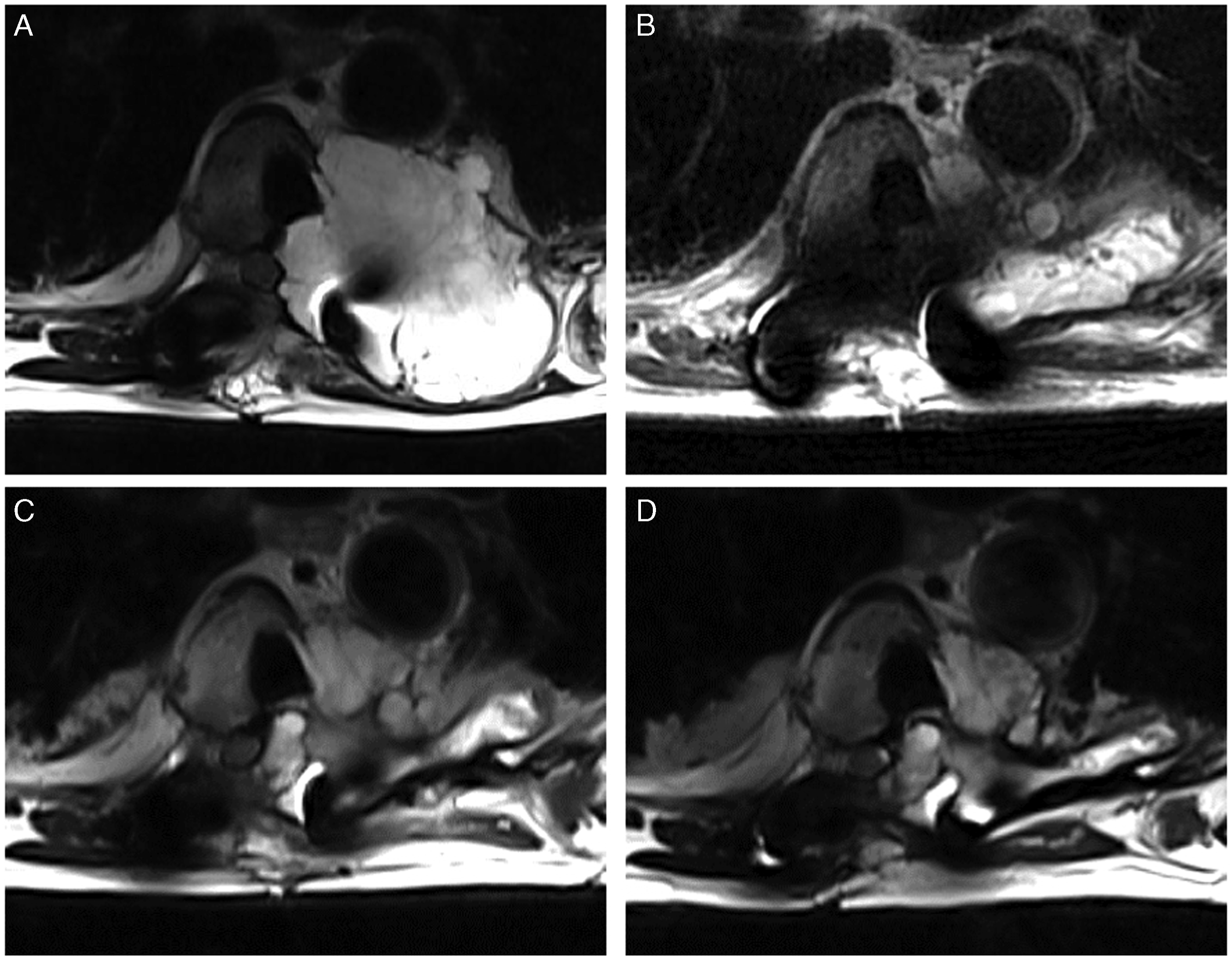
Anlotinib is another multi-kinase inhibitor with activity against VEGFR, PDGFR and c-kit. Its efficacy vs placebo was previously proven in soft tissue neoplasms such as NSCLC, renal cell carcinoma, and medullary thyroid carcinoma. 60 There is an ongoing phase II clinical trial in locally advanced, unresectable or metastatic CH with anlotinib with no reported results yet (NCT04042597, unknown status).
A phase II clinical trial (NCT 00874874, unknown status) showed promising results with a new tyrosine kinase inhibitor, sorafenib, in CH. The 9-month PFS was 91.7% with sorafenib (n = 27), however there was no association between the change in VEGFR and the PFS.
Cetuximab an EGFR inhibitor monoclonal antibody is under investigation in a phase II clinical trial in unresectable or metastatic CH patients (NCT05041127, active recruiting).
CDK4/6 Pathway
Cyclin-dependent kinases (CDK) play important role in sarcoma cell cycle. CDK4 is a key regulator of G1 phase progression and G1-S transition. CDK4 with cyclin D1 forms a complex and phosphorylates retinoblastoma (Rb) protein and inactivates its function. This causes cell cycle progression by relieving the Rb mediated inhibition of transcription factor E2F. Studies have discovered activation of CDK4/6 pathway in CH due to deletion of p16 tumour suppressor gene. P16 is one of the most frequently mutated tumour suppressor gene, it has a pivotal role in the transition of G1 to S phase of cell cycle.61,62 There is an ongoing phase II clinical trial (NCT03110744, completed) with CDK4/6 inhibitor palbociclib on locally advanced/metastatic CH. Where the median PFS of 5.75 months (n = 26) and overall survival rate of 74% at 2 years was observed. 63
PD-1, PDL-1 Pathway
Programmed death 1 (PD-1) and its ligands are expressed on the surface of tumours and lymphocytes. PD-1 and its ligands role have been studied in tumour progression. Currently the interaction between the host immune system and the tumour is poorly understood. Studies have shown that CH cells demonstrate inducible PD-1 expression which can be targeted by drugs. 64 Nivolumab binds to the PD-1 receptor and blocks its interaction with its ligands, thus prevents PD-1 pathway from inhibition the immune response. Based on this molecular interaction, a few clinical trials with nivolumab on chordoma have been launched (NCT02989636 active not recruiting, NCT03623854 active not recruiting, NCT03886311 active recruiting, NCT04416568 active recruiting),65-68 but there have not been any results reported yet.
Chondrosarcoma
CHS seems to be more resistant to conventional targeted therapies than CH. There are a lot of new therapies in development mostly in in vitro phase. Beside a few ongoing clinical trials there are some previously promising ways which have proved to be ineffective recently. 42
Immunotherapy
Nivolumab and pembrolizumab (both anti-programmed death 1 (PD-1) antibodies have recorded objective response in 2 patients with dedifferentiated CHS.69,70 These results are encouraging to patients with CHS, however severe/lethal neurologic immune-related adverse events have been associated with immune checkpoint inhibitors, including those targeting PD-1. 71
Vascular Endothelial Growth Factor Receptor
Anlotinib a novel tyrosine kinase inhibitor acting on VEGFR combined with PD-1 antibody is under a phase II clinical trial recruiting unresectable, high-grade CHS patients. The trial started in early 2023 (NCT05193188, active recruiting). 72 REGOBONE trial is a randomized, placebo-controlled, multicentric, phase II study in France about the effect of regorafenib in locally advanced or metastatic bone sarcomas including CHS and CH as well showing promising results (see above and Figure 1) (NCT02389244, active recruiting).
Heterogeneous Nuclear Ribonucleoprotein P2
Heterogeneous nuclear ribonucleoprotein P2 is also known as RNA-binding protein FUS/TLS (FUsed in Sarcoma/Translocated in LipoSarcoma). FUS gene rearrangement has been implicated in the pathogenesis of wide range of malignant and benign tumours. 73 Trabectedin is reported to be effective against translocation related sarcomas. In a phase II clinical trial, 5 patients with translocation related CHS (extraskeletal myxoid, and mesenchymal) were treated with trabectedin (JapicCTI-121850, no information/Japanese/completed). The sub-analysis showed a median PFS of 12.5 months and an overall survival of 26.4 months. One patient showed partial response the others showed stable disease. 74
Death Receptor 5
Death receptor 5 (DR5) is a member of the tumour necrosis factor (TNF) receptor superfamily. It binds TNF-related apoptosis-inducing ligand (TRAIL) and meditates apoptosis. INBRX-109 is a tetravalent DR5 agonist antibody that exploits the tumour specific cell death induced by DR5 activation. In a phase 1 dose escalation study, 20 patients with conventional CHS were enrolled. Two of them achieved partial response, 14 subjects had stable disease, and the disease control rate was 89%. 75 Currently the phase II study is actively enrolling (NCT04950075). 76
IDH1 Inhibitors
A phase II clinical trial investigating ivosidenib is actively recruiting CHS patient with IDH1 mutations (NCT04278781).
CDK4/6 Pathway
Patients are recruited to a single-arm, phase II study about the effect of abemaciclib (CDK4/6 inhibitor) on CHS (NCT04040205).
Conclusion
CH and CHS are different histologically, yet they share common attributes which makes them difficult to treat and achieve good local control. Although these low-grade malignant pathologies are slow growing, they are locally aggressive and ultimately cause death if not controlled. They tend to recur after initial treatment, even in the case of Enneking appropriate surgical resection, and appropriate consultation with radiation and medical oncology should be performed up-front in all patients as multidisciplinary discussions are critical to proper care.
In the past decades, several molecular pathways have emerged as potential targets to achieve better clinical outcomes. CH has more potential molecular therapeutic targets, and various tyrosine kinase inhibitors (e.g.,: imatinib-PDGFR, sunitinib-VEGFR, lapatinib-EGFR) have shown moderate to good PFS rates. One has to be cautious in interpreting the data as sample sizes are limited and toxicity profiles can be disadvantageous. Unfortunately, these drugs have yet to demonstrate significant clinical activity in CHS. 42 More molecular pathway targets are under intense research in the treatment of CHS; however, most are in in vitro or in animal model stages.
At present, in the patient presenting with a low grade malignant spinal tumour, there is no pharmaceutical treatment alternative for the Enneking appropriate surgical resection. Some recent and ongoing clinical trials with targeted molecular therapy show better oncological response compared to conventional options, and can be used to treat patients with recurrent or metastatic disease.
Supplemental Material
Supplemental Material - Latest Developments in Targeted Biological Therapies in the Management of Chordoma and Chondrosarcoma
Supplemental Material for Latest Developments in Targeted Biological Therapies in the Management of Chordoma and Chondrosarcoma by Adam Biczo, Arjun Sahgal, Jorrit-Jan Verlaan, Patel Shreyaskumar, Zsolt Szoverfi, Markus Schultheiss, Laurence Rhines, Jeremy Reynolds, Ilya Laufer, Alessandro Gasbarrini, Nicolas Dea, Ziya Gokaslan, Charles Fisher, Chetan Bettegowda, Stefano Boriani, Francis Hornicek, Rory Goodwin, and Aron Lazary in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was organized and funded by AO Spine through the AO Spine Knowledge Forum Tumor, a focused group of international Tumor experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
