Abstract
Study Design
Narrative Review.
Objective
Machine learning (ML) is one of the latest advancements in artificial intelligence used in medicine and surgery with the potential to significantly impact the way physicians diagnose, prognose, and treat spine tumors. In the realm of spine oncology, ML is utilized to analyze and interpret medical imaging and classify tumors with incredible accuracy. The authors present a narrative review that specifically addresses the use of machine learning in spine oncology.
Methods
This study was conducted in accordance with the Preferred Reporting Items of Systematic Reviews and Meta-Analysis (PRISMA) methodology. A systematic review of the literature in the PubMed, EMBASE, Web of Science, Scopus, and Cochrane Library databases since inception was performed to present all clinical studies with the search terms ‘[[Machine Learning] OR [Artificial Intelligence]] AND [[Spine Oncology] OR [Spine Cancer]]’. Data included studies that were extracted and included algorithms, training and test size, outcomes reported. Studies were separated based on the type of tumor investigated using the machine learning algorithms into primary, metastatic, both, and intradural. A minimum of 2 independent reviewers conducted the study appraisal, data abstraction, and quality assessments of the studies.
Results
Forty-five studies met inclusion criteria out of 480 references screened from the initial search results. Studies were grouped by metastatic, primary, and intradural tumors. The majority of ML studies relevant to spine oncology focused on utilizing a mixture of clinical and imaging features to risk stratify mortality and frailty. Overall, these studies showed that ML is a helpful tool in tumor detection, differentiation, segmentation, predicting survival, predicting readmission rates of patients with either primary, metastatic, or intradural spine tumors.
Conclusion
Specialized neural networks and deep learning algorithms have shown to be highly effective at predicting malignant probability and aid in diagnosis. ML algorithms can predict the risk of tumor recurrence or progression based on imaging and clinical features. Additionally, ML can optimize treatment planning, such as predicting radiotherapy dose distribution to the tumor and surrounding normal tissue or in surgical resection planning. It has the potential to significantly enhance the accuracy and efficiency of health care delivery, leading to improved patient outcomes.
Keywords
Introduction
Spine tumors, both primary and metastatic, have been steadily increasing in incidence in recent years and are a major burden of oncologic care.1,2 Primary tumors of the spine are rare, but metastases to the vertebral column are much more prevalent, and present in up to 70% of cancer patients. 3 Spine metastases can lead to epidural spinal cord compression causing substantial axial pain, vertebral body fractures, as well as myelopathy or radiculopathy. Spine tumors can be classified using their origin and location within the spine. Metastatic tumors originate outside the spine and are often indicative of disease progression. Primary bone tumors of the spine as well as primary tumors of the spinal cord or exiting nerves originate in the spine. These soft tissue tumors are commonly referred to as intradural or extradural based on their location relative to the dura mater. Being able to distinguish tumor type is critical, as they require different approaches to management and treatment. 4
The location of primary, metastatic, or intradural lesions can also impact the burden of treatment due to location-related intrinsic surgical morbidities and their obvious influence on outcomes. Additionally, location can impact radiation dose delivery given each pathology has a specific response to radiation. 5 As the field of spine oncology continues to expand, there is a need for new tools that will aid physicians in creating individualized plans for oncologic care.
Background on Machine Learning
Machine learning (ML) involves the development of algorithms that learn from data and make predictions without explicit programming. ML models have been around for almost 5 decades and can be utilized in health care given the large swaths of patient data.
6
In recent years, ML has been applied to various fields of medicine, including neurosurgery, with the goal of improving diagnosis, treatment, and patient outcomes. Artificial neural networks (ANN) are the primary ML algorithms utilized within clinical outcomes research.
7
The common types of neural networks include ANNs, Convolutional Neural Networks (CNN), and Recurrent Neural Networks (RNN) (Figure 1, Table 1). ANN is primarily used for tabular data, CNN for visual data such as radiographs, and RNN for sequence data.
8
Convolutional Neural Network (CNN) is a type of deep learning algorithm that is designed to analyze image data. It uses a series of filters to identify patterns and features in the image, allowing it to recognize objects and classify images. Artificial Neural Network (ANN) is a type of machine learning algorithm that is modeled after the structure and function of the human brain. It consists of interconnected nodes, or artificial neurons, that process information and make predictions based on the data input. Recurrent Neural Network (RNN) is a type of neural network that is designed to process sequential data, such as time series data or text. Unlike traditional neural networks, RNNs have memory and can use past information to make predictions about future events. They are often used in applications such as speech recognition and natural language processing. Definition of Terms, and ML Models.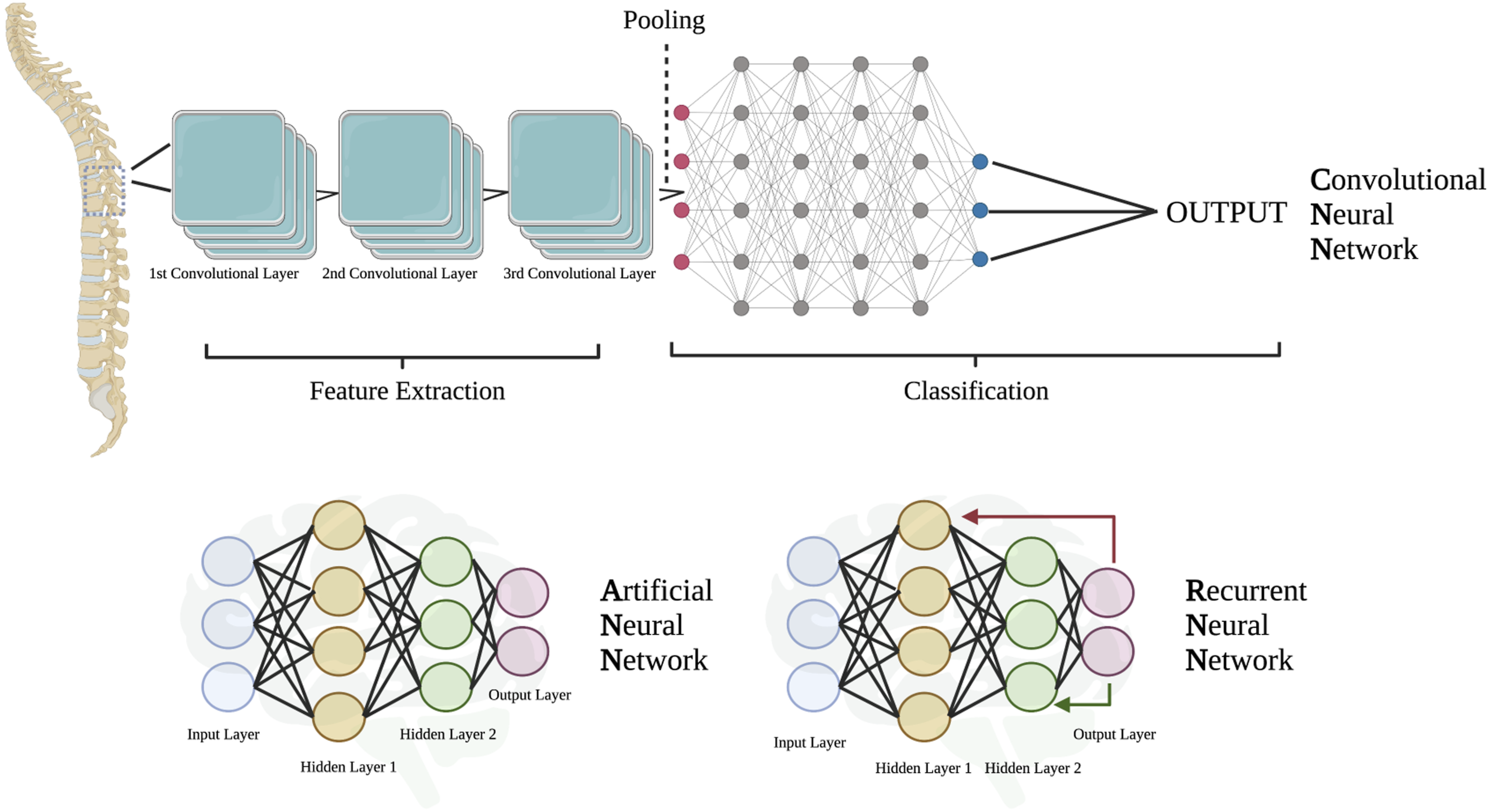

The use of ML in health care currently provides a supplementary role to physicians in developing prediction models, identifying trends and helping reduce physician workload. 9 In the field of oncology, ML is being tested to extract magnetic resonance imaging (MRI), or computed tomography (CT) data for diagnosis, 10 and predict survival rates based on individual characteristics. 11
A systematic analysis on the global burden of cancer reported 26.3% increase in global cancer cases, 20.9% increase in cancer related deaths, and 16.0% increase in disability adjusted life years from 2010 to 2019. 12 As oncologic therapy continues to improve, we expect to see an increasing incidence of patients with spinal metastases. Prediction models and trend data can assist physicians and patients to create personalized oncologic care plans based on patient and tumor characteristics. Given the increasing number of patients entering oncology clinics, ML has been put on the forefront of innovation in trying to help combat the increasing burden of oncologic care for both patients and physicians. 13
To date, several systematic reviews have examined the use of machine learning within the field of neurosurgery and its utility in neurosurgical applications; however, there are no current studies that have reviewed the use of machine learning within spine oncology. Therefore, in this study, we present a thorough and comprehensive systematic review of the literature regarding the use of ML in spine oncology.
Methods
Search Strategy
A targeted systematic review was performed and reported in accordance with the PRIMSA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. A literature search was performed using Embase (Elsevier), PubMed (National Library of Medicine), Web of Science, Scopus, and Cochrane Library databases from inception to April 14, 2023, to identify studies evaluating the use of machine learning in spinal oncology. Search terms utilized to identify relevant studies were ‘[[Machine Learning] OR [Artificial Intelligence]] AND [[Spine Oncology] OR [Spine Cancer]]’.
Inclusion and Exclusion Criteria
Studies were included if they met the following criteria: (1) reported primary data with some form of machine learning used with patients with a spinal lesion; (2) reported on where the machine learning algorithm was used, and how it can be applied. Exclusion criteria were non-English studies, abstracts, oral presentations, cadaveric studies, phantom studies, case reports, review articles, systematic reviews, and studies whose only algorithm used was radiomics as radiomics does not change the surgical strategy in metastatic epidural spinal cord compression. Abstracts were screened independently by 2 authors [SBW, VM], and conflicts were resolved through discussion and decision by a third author [JW]. Potential studies were extracted for full-text evaluation, which was completed by a two-vote system by 4 of the authors [SBW, JW, VM, CSAL], with conflicts resolved by an independent third author.
Data Extraction
Articles that passed our inclusion criteria were divided into subgroups based on tumor types, primary bone (extradural), metastatic, intradural, or a combination of the types. The primary bone tumor subgroup was classified as primary spine tumors such as Ewing sarcoma, chondrosarcoma, osteosarcoma, and sacral tumors. The intradural tumor subgroup consisted of schwannoma, astrocytoma, ependymoma, and meningioma. Articles that reported on multiple tumor types were split into a separate, mixed, subgroup. Data collected included study characteristics (publication year, journal published, sample size for testing and validation groups, type of machine learning algorithm used, inclusion and exclusion criteria), patient demographics (sex, type of cancer, area of spinal lesions), procedure characteristics (machine models input data and output groups, machine algorithms validation method, machine models prediction performance). A full-text evaluation was conducted independently by 3 authors [SBW, JW, CSAL].
Results
A review of the literature revealed a surge in publications on Machine Learning in Spine Oncology in the last 30 years (Figure 2). A total of 480 unique publications matched the search criteria. Four hundred and two publications were excluded during title and abstract screening due to failure to address the use of AI/ML in spine oncology. A full-text review was conducted for 77 studies. Twenty-eight of those studies were excluded due to emphasis on radiomics or were not published as a full text article. Forty-six studies met the final inclusion criteria for this systematic review (Figure 3). Bar graph of database search results of studies involving machine learning in spine surgery and spinal oncology since database inception. PRISMA flow diagram of the systematic literature search for this systematic review. Data added to the PRISMA template [from Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Reporting Items for Systematic reviews and Meta-Analyses: The PRISMA Statement. 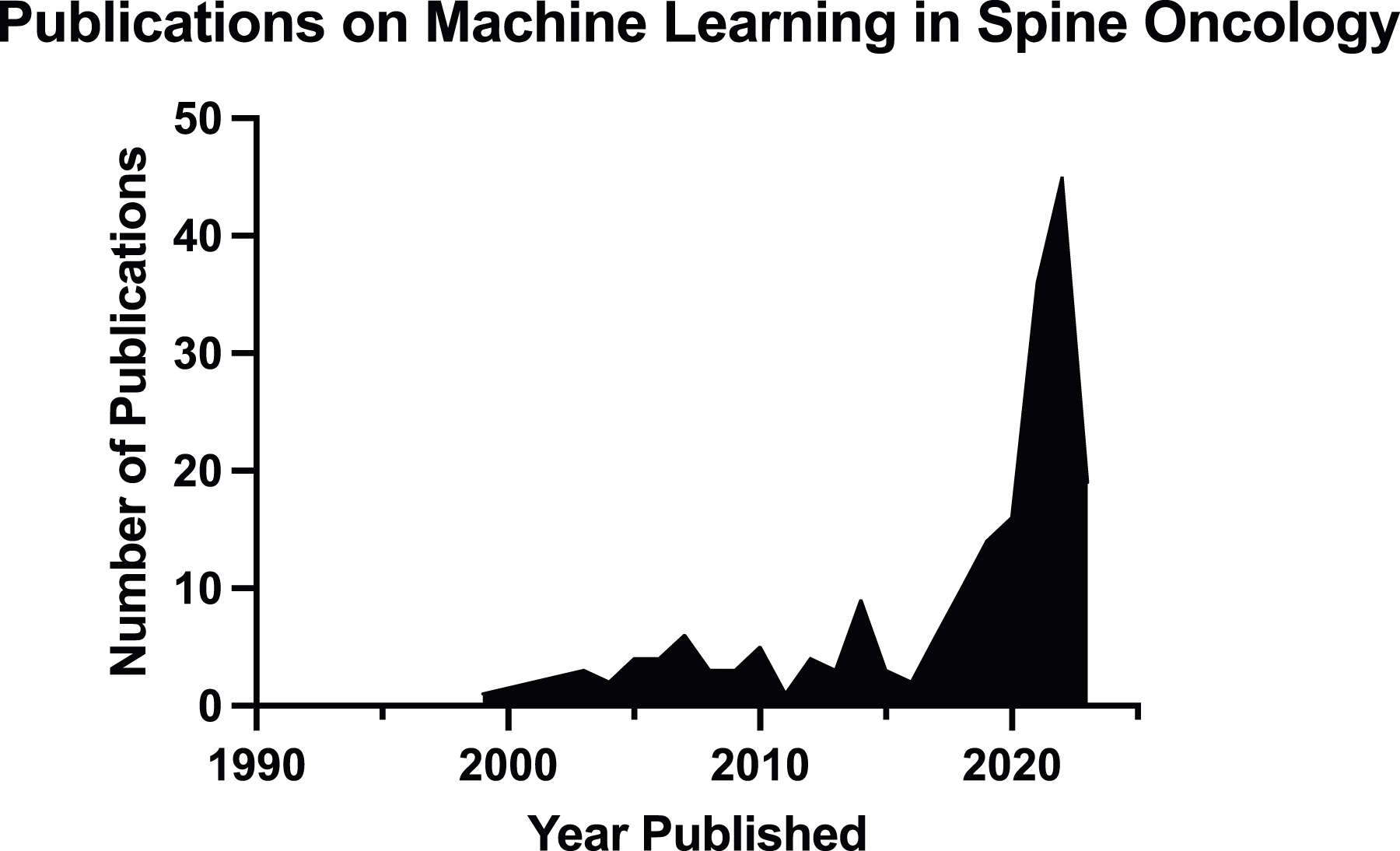
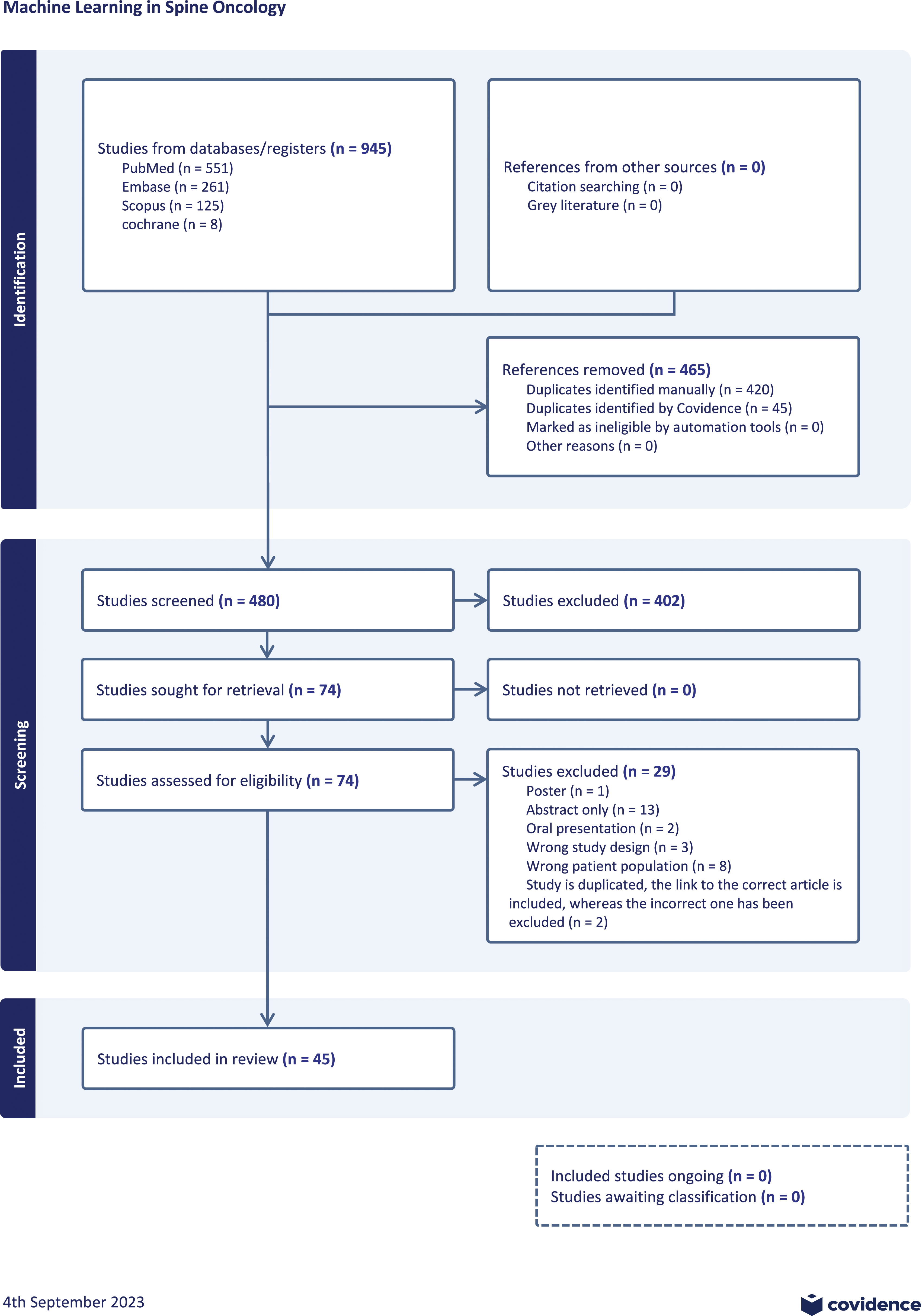
We separated the forty-six included articles based on their investigation of primary spine tumors metastatic disease, intradural tumors, or combinations of these types. Once stratified, twenty-nine publications addressed metastatic spinal tumors, 4 publications addressed primary spinal tumors, 5 publications addressed intradural tumors, and 8 publications addressed a mixture of any of the 3 tumor types.
In the reviewed articles, most algorithms used supervised learning either for classification or regression purposes while a few used unsupervised learning models that focus on clustering outcomes. Classification models include Random Forest (RF), Support Vector Machine (SVM), Decision Tree analysis, Bayes Classifier. These models were most often used in spine oncology to classify variables such as tumor detection or differentiation,14-27 overall mortality,28-35 or adverse events related to surgery. 36 Additionally, we found that regression algorithms including LASSO, logistic and multivariate logistic were used to predict continuous variables such as overall survival and prognosis.15,33,37-45 Some studies used pretrained models such as ResNet50, Unet++, and ImageNet as a foundation, which were then further trained with their data through 50-100 epochs (number of complete passes through the training dataset). Large scale comparative statistics were absent due to high variability of study aims, algorithms, and verification metrics.
Metastatic Spine Disease
Summary of Machine Learning Studies Focused on Metastatic Disease in Spine Oncology.
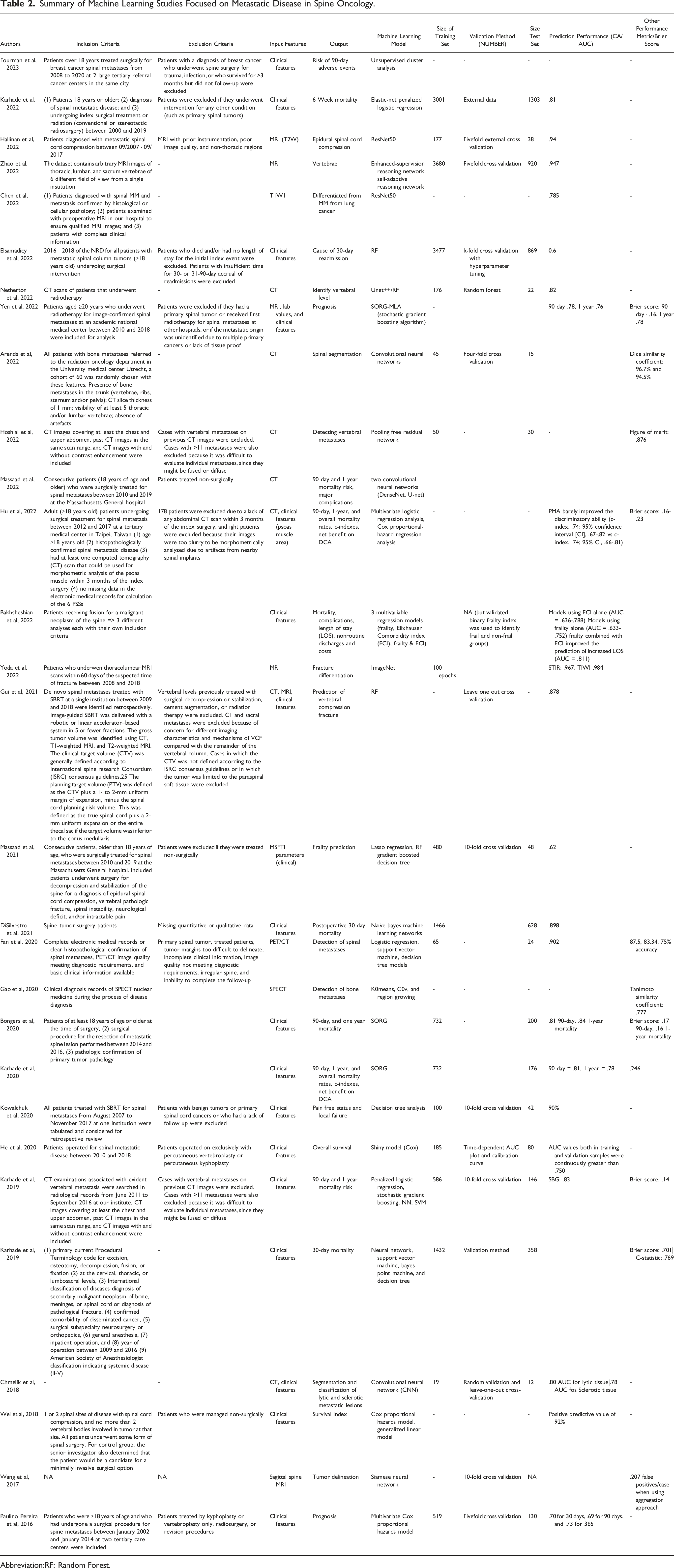
Abbreviation:RF: Random Forest.
Several studies evaluated a specific cancer type as it metastasized to the spine. One study attempted to predict
Of the studies that evaluated mortality or adverse events, Karhade et al 32 reported an area under the curve (AUC) of .81 for predicting 6-week mortality using an Elastic-net penalized logistic regression. Fourman et al 36 used unsupervised cluster analysis to identify 5 groups that had distinct outcomes following open spine surgery for metastatic breast cancer. High functioning patients and those who were ER/PR positive had the best outcomes, while patients with structural instability had the worst outcomes. Bongers et al 29 used clinical features to develop an algorithm that could predict 90-day and 1 year mortality with AUC of .81 and .84 respectively, following surgical resection of metastatic tumor to the spine. Karhade et al 34 reported an AUC of .83 and a Brier score of .14 when predicting mortality risk at 90-days and one year. Karhade et al 50 then described an algorithm that could similarly predict 90-day and 1-year mortality rates with AUC of .81 and .78, respectively. Massaad et al 35 used a deep learning model to risk stratify patients into high and low risk based on imaging and clinical features focusing on age, sex, frailty, muscle radiodensity, and subcutaneous fat. They reported significant differences in survival between groups and suggested use of their model in preoperative planning. He et al 37 predicted overall survival with an AUC over .75 using clinical features such as primary tumor type, site of metastasis, Frankel grade, and number of surgical segments. Hu et al 31 predicted 90-day, 1-year, and overall survival with a c-index of .74 using logistic regression analysis. Wei et al 41 developed a survival index with a positive predictive value of 92% using Cox proportional hazards model. Bakhsheshian et al 28 were able to predict mortality, complications, and length of stay with an AUCs of .788, .723, and .811 respectively using multivariate regression models. Karhade et al predicted 30-day survival with a c-statistic of .769 and a brier score of .701. 34 DiSilvestro et al described an algorithm that predicted post operative mortality using clinical features with an AUC of .898. 30
The next most common focal area was tumor detection or differentiation. Zhao et al. Developed an algorithm that assigned a metastatic lesion to a vertebral level with an AUC of .947.
16
Chen et al were able to differentiate metastatic disease of origin between MM and lung cancer with an AUC of .785.
14
Fan et al used a support vector machine and Positron emission tomography/CT scans to detect spine metastases with an AUC of .902.
15
Gao et al used single-photon emission computed tomography to detect bone metastases reporting a Tanimoto similarity coefficient of .777.
27
Netherton et al used CT data run through Unet++ and RF to determine the vertebral levels of metastatic lesions with an AUC of .82.
22
Arrends et al reported a Dice Similarity Coefficient (DSC) of 97% and 95% for internal and external validation, respectively.
17
Wang et al
24
demonstrated a 44.8% reduction in false positives for automated rates of detection of spinal metastases, from .375 to .207 using a Siamese Neural Network. Hoshai et al developed an algorithm that improved figure of merit (FOM) for physician and resident detection of vertebral metastases from .848 to .876 (
Several studies evaluated prognosis, attempting to predict post-operative outcomes. Gui et al 43 used clinical features to predict vertebral compression fracture rates, AUC = .878, using an RF model. Elsamadicy et al 42 predicted cause of 30-day readmission, AUC = .6, using clinical features, primarily frailty status, input into an RF model. Yen et al 45 evaluated overall prognosis using a stochastic gradient boosting algorithm and MRI data combined with laboratory values and other clinical features with an AUC of .78 and .76 for 90-day and 1-year prognosis, respectively. They also reported Brier scores of .16 and .78 for 90-day and 1-year prognosis, respectively. Kowalchuk et al evaluated pain free status and local failure using decision tree analysis with an AUC of .90. 52 Massaad et al used LASSO regression and an RF gradient boosted decision tree to predict frailty with an AUC of .62. 44 Paulino Pereira et al 48 predicted post operative prognosis at 30-days, 90-days, and 1-year with AUCs of .70, .69, and .73 respectively using Multivariate COX proportional hazards model. (Table 2).
ML in metastatic spine disease had the most evidence of all types of spine cancer. ML tools displayed mixed results with overall utility in determining origin of tumor, survival, and surgical outcomes using both imaging and patient demographic factors.
Primary Bone Tumors
Summary of Machine Learning Studies Focused on Primary Site Malignancy in Spine Oncology.
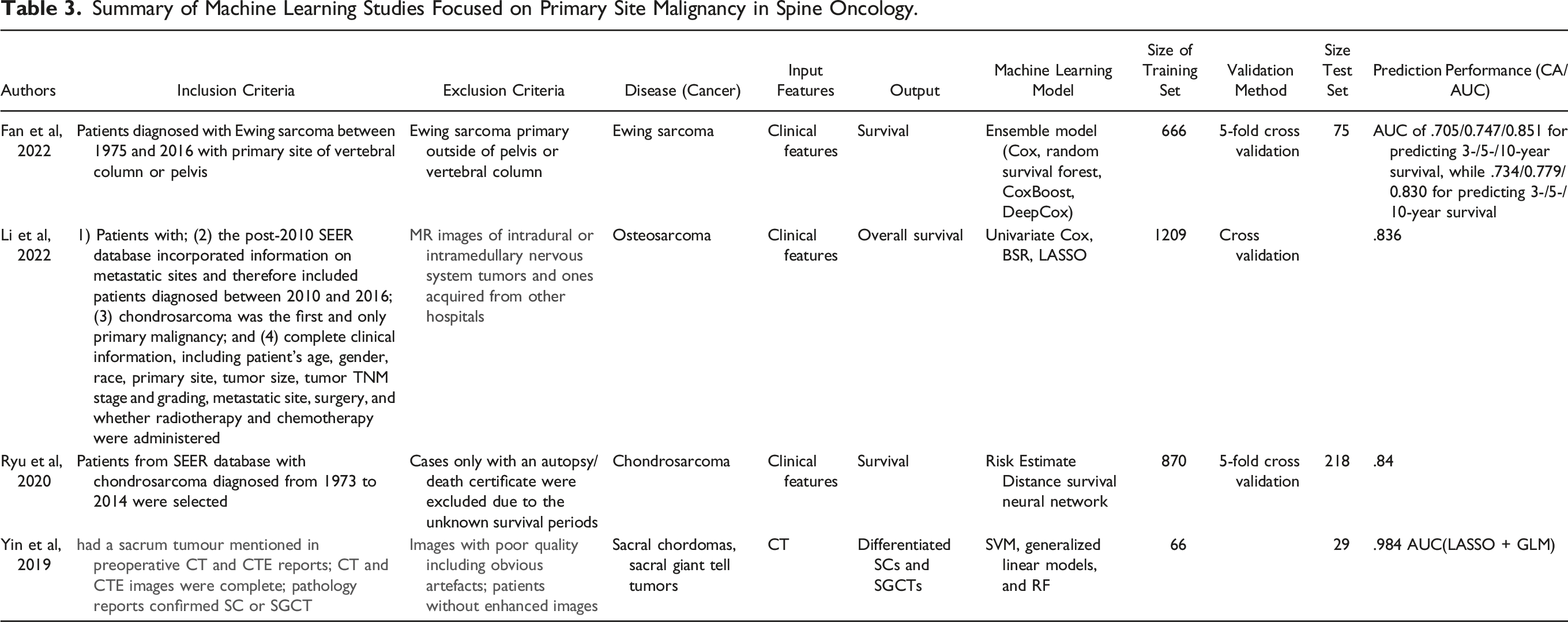
Most of the articles that discussed primary spine lesions were investigating overall survival using clinical features, with each article looking at a different primary tumor type. Fan et al 15 reported that an ensemble model combining CoxBoost and Cox with feature selection outperforming the traditional TNM system for survival rates of Ewing sarcomas with an AUC of .851 for 10-year survival. Li et al 38 developed an open access web platform using BSR, UC, and LASSO that predicts overall survival for chondrosarcoma patients, demonstrating high AUC’s > .8 for internal and external validation sets. Lastly, Ryu et al trained a neural network to predict survival of chondrosarcoma, and the average AUC for the 5 subgroups, created based on predicted percentage survival, using the test set was .84. 40
The last article in the primary section focused on tumor differentiation. Yin et al used multiple ML models, including LASSO, SVM, and GLM to determine which yielded the best results with 2 feature sets. They showed that LASSO + GLM had the highest accuracy of any combination of ML models, and that using CT-enhanced features yielded better differentiation of sacral chordomas and sacral giant cell tumors with an AUC of .984. 53 Overall, these studies show that various ML models can be used for different purposes in the field of spine oncology, with similar AUCs between models and desired output classes. (Table 3).
ML in primary bone (extradural) tumors of the spine demonstrates evidence in predicting tumor origin and overall survival, providing surgeons another tool in the management of oncological spine patients.
Intradural
Summary of Machine Learning Studies Focused on Intradural Malignancy in Spine Oncology.
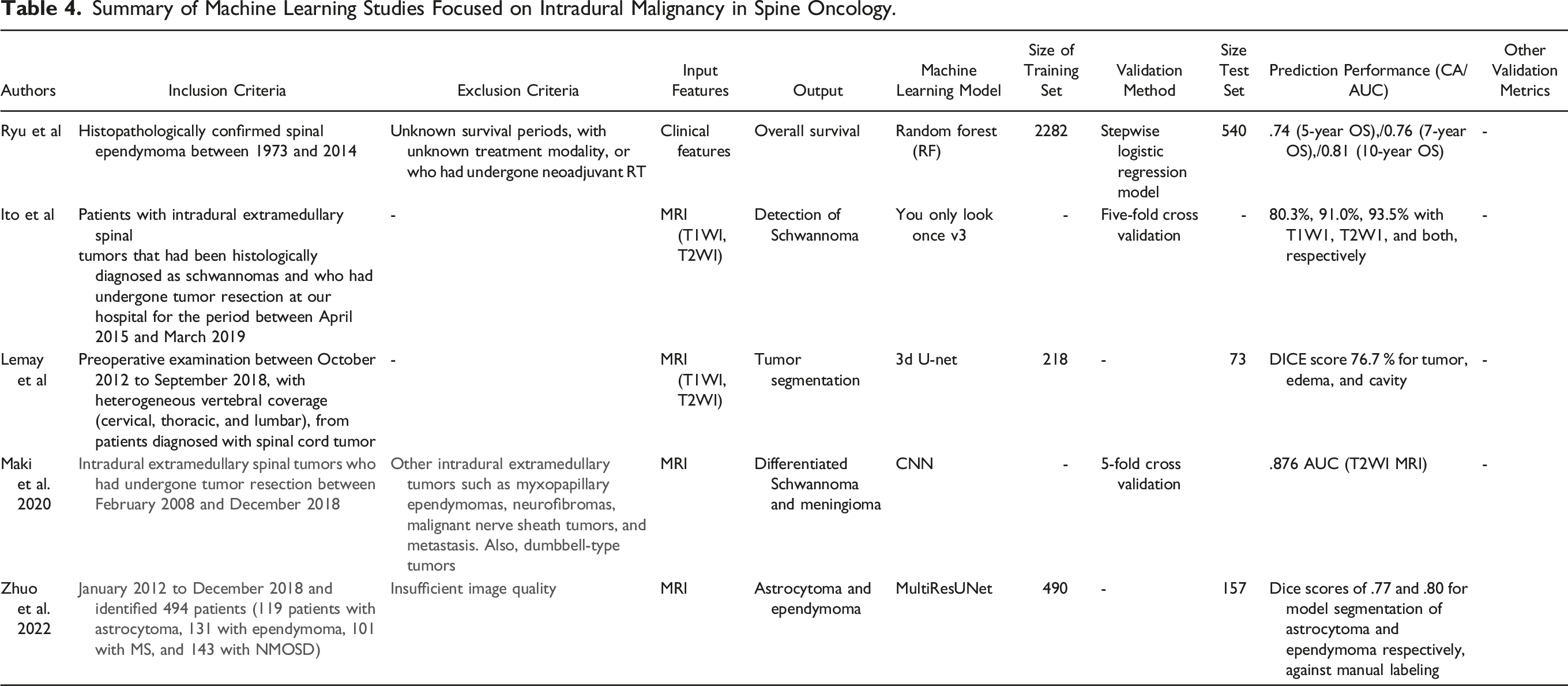
The largest subset of ML use with intradural spine tumors was investigating tumor detection, and segmentation. Of those that focused on detecting tumors, both investigators used magnetic resonance imaging (MRI) as the input for the ML models, and focused on less common tumors like schwannomas, astrocytoma, ependymomas, and hemangioblastomas. Ito et al 19 used YOLO version 3 to detect Schwannomas on MRIs, and the results using T2W images compared favorably to those of 2 physicians with the accuracy being 91%, 90.2%, and 89.3%, respectively. For segmentation, Lenmay et al showed that the 3d U-Net model was able to better segment spine lesions including astrocytoma’s, ependymomas, and hemangioblastomas when including tumor, cavity, and edema as compared to tumor only with Dice scores of 76.7% and 61.8%, respectively. 20 Zhuo et al developed a deep learning framework for segmentation and classification of spinal cord lesions included astrocytoma and ependymoma with AUC .99. 54
The other articles focused on overall survival, or tumor differentiation. Ryu et al 39 found that a RF model investigating survival patterns using patient and tumor characteristics of ependymomas performed better than a stepwise logistic regression model with 5-year and 10-year survival, with the AUC being .74, and .81, respectively. The last article for the intradural section focused on tumor differentiation. Maki et al 21 used a Convolutional NN (CNN) to differentiate schwannomas from hemangiomas, with the accuracy of the CNN being comparable to 2 trained radiologists both using T1W 81%, 82%, and 81%, respectively, and for T2W MRI being 80%, 69%, and 73%, respectively. Overall, these studies showed that ML is a helpful tool in tumor detection, differentiation, segmentation, and predicting survival of intradural spine tumors (Table 4).
With only 5 studies, ML for intradural tumors lacks robust volume of evidence however each manuscript demonstrated significant results where ML techniques assisted in tumor detection, segmentation, and survival.
Mixed Studies
Summary of Machine Learning Studies Focused on Both Primary and Metastatic Site Malignancy in Spine Oncology.
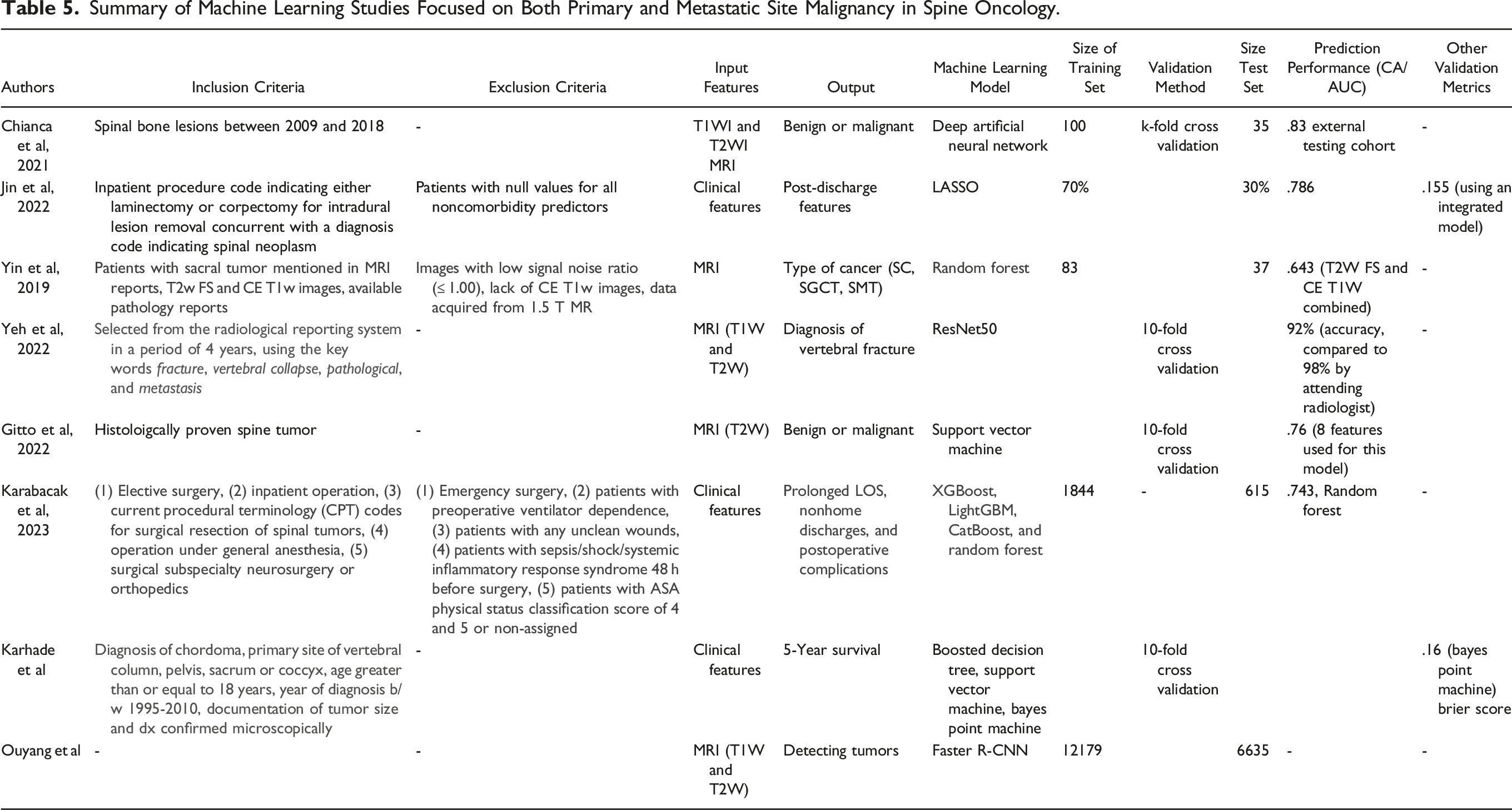
Two studies used MRIs to predict malignancy status of spine lesions. Chianca et al 55 used a deep artificial NN to predict malignancy, finding that using features extracted from 3D Slicer heterogeneity CAD and a 2-label classification, yielded the highest accuracy of 86%, compared to PyRadiomics feature extraction and 3-label classification. Gitto et al 56 used a SVM and found that the best performance using the lowest number of features had an AUC of .78, with only 8 features used for tumor classification. 2 studies used clinical features to predict nonhome discharge and 90-day post-discharge readmission. Jin et al 57 found an integrated model with LASSO that combined patient-specific and tumor-specific features had an AUC of .786 for nonhome discharge, and .693 for 90-day readmission. Meanwhile, Karabacak et al showed that the RF model performed the best of the ML models with an AUC of .743, and the most accurately predicted outcome was length of stay with an AUC of .745. 58 In terms of tumor differentiation, Yin et al 25 reported that using RF, features extracted from joint T2W fat saturated and contrast enhanced T1W images outperformed those separate imaging modalities with an AUC of .773 in differentiating SC, SGCT, and sacral metastatic tumors. Yeh et al 59 found that a ResNet50 algorithm was able to identify abnormal vertebrae with 92% accuracy, and it outperformed first-year residents reading the same images. For survival, Karhade et al 33 created an open access web application using a Bayes Point Machine that took clinical and patient features, and predicted survival after chordoma diagnosis, with a C-statistic and Brier score of .8, and .16, respectively. Lastly, Ouyang et al 23 used a version of a NN, denoted as faster R-CNN, to detect spine lesions, with the model being assessed via the Turing test. The study showed that human respondents could not determine if an image detection came from AI or from human detection, as shown by the misclassification rate being above 30% for all 6 respondents, which means that the ML model passed the Turing test. Overall, ML models have performed well on tumor differentiation, tumor detection, predicting post-operative discharge complications, 5-year survival, determining malignancy status, and performing as effective as early-stage trainees in diagnosing vertebral fracture type. (Table 5).
Studies that evaluated mixed groups of spine tumors provided the ability to compare different pathologies, utilizing imaging to predict malignancy and 5-year mortality.
Discussion
Within neurosurgery, the appropriate management of spine tumors is dependent on the evaluation of various preoperative variables, including imaging studies, systemic disease burden, neurologic symptoms, and tumor biology. Patient centered care plans are critical in managing diverse patient populations with a constellation of comorbidities ML algorithms can be trained to analyze large patient datasets consisting of demographic data, clinical presentation, imaging findings, and treatment details, to identify factors associated with clinical outcomes.
Literature published on the use of ML in spine oncology has been increasing in prevalence over the last 30 years (Figure 2). Most of these studies focus on the supplementary role of ML in guiding physicians, surgeons, and other health care providers in treating patients with primary or metastatic spine tumors. Tumor detection, and differentiation using advanced imaging modalities such as MRI, CT, PET/CT, or SPECT may impact treatment pathways through detection of cancer earlier in presentation and more accurate diagnosis leading to an individualized treatment. With an aging population and an increased prevalence of metastatic cancer to the spine, these ML tools may also increase processing speed of providers that read these advanced images, helping to reduce the burden on health care systems.
Several studies compared the performance of their model to a set of experienced physicians. Yoda et al 26 developed an algorithm to differentiate between osteoporotic vertebral fractures (OVF) and pathologic vertebral fractures (PVF) that was 96.4% accuracy with TIWI. Using the same data set, 3 independent spine surgeons with 14, 10, and 7 years of experience differentiated OVF and PVF with 91.1%, 88.4%, and 85.7% accuracy. Hoshiai et al 51 found both experienced radiologists and radiology residents demonstrated increased figure of merit when reading CT scans while being assisted by a ML algorithm for lesion identification. These examples point to the efficacy of ML in tumor detection and differentiation with increasing speed and accuracy.
Current research using ML to predict mortality or adverse events focuses on 30-day, 90-day, or 1-year outcomes.29,30,32,34–36,50 A study by Li et al published results of 5-year mortality for patients with chordoma using support vector machine, bayes point machine, and neural networks reporting a Brier score of .16. Due to the high variety of pathologies, surgeons may reference this work to identify algorithms that provide data to support clinical decision making.
ML modeling and algorithmic frameworks could potentially be used for tackling other critical topics which the literature is unsure about. For instance, Gui et al demonstrated the effectiveness of a combined clinical and radiomic ML model in predicting fracture 1-year post-SBRT for metastatic spine disease, achieving high sensitivity and specificity.
Multi-institutional collaboration in developing ML application in spine oncology is an interesting strategy to increase its robustness and generalizability. By pooling diverse datasets and leveraging different sources' inputs, a collaborative effort in this regard will expedite the development of this field.
The studies cited in this review demonstrate the usefulness of ML in spine oncology. Continued research is indicated to improve ML ability to detect and differentiate tumors, predict mortality and adverse events, and aid in prognostication.
Limitations
Despite the comprehensive review, there are several limitations to our study. The articles included in this study are heterogenous in their input and output variables, machine learning algorithms, outcome measures, and differ in focuses. The prediction accuracy and AUC represent relative performances of the ML algorithms while the true reliability and generalizability is dependent upon the institution’s patient characteristics, training sets, algorithm used, input quality and size, and patient characteristics. All of these variables can affect the true accuracy and overall generalizability of ML models. Many of the studies included also use a binary outcome allowing the use of AUC measurements. If models utilized a continuous outcome, the results may have been different affecting the model’s clinical efficacy. Due to the variety in these measures, we qualitatively describe the patterns of the use of ML in spine oncology. The variety in measurements also makes quantitative analysis challenging. As with other systematic reviews, publication bias may also be a factor affecting our results. More successful models may have been more likely to be published, falsely inflating the success of ML in spine oncology. Despite this however, we provide a robust review of all the published algorithms utilized in spine oncology, and we provide valuable information regarding the application of ML and future directions of the field.
Future Directions
Current literature includes studies that have investigated the ability of machine learning to predict short- and mid-term survival after surgery for spine tumors.33,50 However, there is a paucity of literature that evaluates postoperative survival for metastatic disease using machine learning. 60 In one study, Forsberg et al investigated one- and 6-month survival in metastatic bone disease using ML, demonstrating an AUC of .76. 60 There is significant clinical utility in expanding upon Forsberg et al's work given that metastatic spine disease occurs at a higher incidence than primary bone tumors. Future studies could investigate longer term survival in patients with metastatic bone disease and stratify cohorts by demographic and clinical factors (ex: age, race, and comorbidities). A multifactorial ML-based analysis applied in a personalized approach would be valuable to neurosurgeons in counseling patients about prognosis.
Compared to metastatic disease, few studies have utilized ML for primary bone tumors.46,53,61,62 Expansion on optimizing ML models to improve accuracy, improve identification and standardization of radiomic features, and improve detection rates of spinal tumors would prove valuable in resource scarce hospitals that may lack comprehensive imaging modalities such as MRI. Thus, machine learning can support radiologists and spine surgeons in these areas as an adjunctive tool in screening for primary bone tumors based on imaging studies.
Conclusions
Machine Learning research in spine oncology aims to better understand patient outcomes, accelerate treatment planning, detect spine tumor, and differentiate between cancer types. Although these outcomes have been investigated in both primary and metastatic disease, literature trends demonstrate tumor differentiation to play a larger role in the study of metastatic disease. The utilization of machine learning in spine oncology predictive analytics is a promising translational development that has the potential to revolutionize personalized medicine and supplement the neurosurgeon’s armamentarium, also contributing to optimize the adjuvant treatment, including the refinement of radiotherapeutic isodose planning.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
