Abstract
Study design
A randomized controlled trial.
Objective
The aim of this study is to compare the efficacy of allografts and bioactive glass-ceramic (BG) cages for anterior cervical discectomy and fusion (ACDF) in treating cervical degenerative disc disease.
Methods
We conducted a single-center, randomized controlled trial between August 2017 and August 2022. Participants were randomized into two groups, and consecutive patients requiring ACDF were randomly assigned to receive either the allograft cage or the BG cage. The surgical outcomes measured included pain levels, neck disability, surgical details, and radiological assessments.
Results
Of the 45 assessed, 40 participants were included, with 18 in the allograft cage group and 22 in the BG cage group. By the 12-month follow-up, both groups exhibited significant improvements in pain levels and disability scores, with no notable intergroup differences. Over 85% of patients in both groups were satisfied with their outcomes. Radiological assessments revealed stability in the cervical spine with both cage types post intervention. Although both materials showed a trend toward increased subsidence over time, the difference between them was not statistically significant. Fusion rates were comparable between the groups at 12 months, with BG cage showing a slightly higher early fusion rate at 6 months. No significant differences were observed between the two groups in terms of complications.
Conclusions
Both allograft and BG cages are effective in ACDF surgeries for cervical degenerative disc disease, with both contributing to substantial postoperative improvements. Differences in disc height, interspinous motion, and subsidence were not significant in the last follow-up, indicating both materials' suitability for clinical use. Future research with a larger cohort and longer follow-up is needed to confirm these preliminary findings.
Keywords
Introduction
Anterior cervical discectomy and fusion (ACDF) is a well-established surgical procedure for treating cervical spine disorders, providing significant relief for patients with cervical myelopathy or radiculopathy. 1 The success of ACDF largely depends on the effective fusion of the vertebral segments, a process that is critically influenced by the choice of graft material. Traditionally, autogenous bone grafts, often sourced from the patient's iliac crest, have been considered the gold standard for cervical interbody fusion.2,3 Despite their effectiveness, autogenous bone grafts have a disadvantage—donor site morbidity. Patients often experience significant postoperative pain, potential infection, and hematoma formation at the harvest site. These complications have led surgeons and researchers to explore alternative graft materials that can facilitate comparable fusion without the drawbacks associated with harvesting autogenous bone. 4
Over the years, graft materials have been significantly improved, evolving from autografts to allografts and, most recently, to synthetic cages. 5 Allografts have emerged as a viable alternative, minimizing complications associated with autograft harvesting. However, they introduce their own set of challenges, including the potential for graft rejection, disease transmission, and inconsistent graft properties.3,6 Recently, the ongoing search for improved materials has led to the exploration of synthetic cages, such as those made from titanium, carbon fibers, and polyetheretherketone (PEEK). Titanium cages are lauded for their robust strength and biocompatibility, but they often lead to subsidence, a notable drawback. 7 On the other hand, PEEK cages, crafted from plastic and possessing strength comparable to bone, alleviate the issue of subsidence. However, it lacks the ability to naturally bond with bone, necessitating the use of an additional bone implant. 8
Recent advancements in biomaterials have led to the introduction of bioactive glass-ceramic (BG) cages, such as the CaO-SiO2-P2O5-B2O3 BG cage.9,10 These materials not only offer mechanical robustness but also possess the unique ability to chemically bond with bone, promoting osteointegration. Its recent application in lumbar surgery has shown that the reduction in additional bone supplementation, coupled with the direct bone-binding ability of materials, can lead to a decrease in the use of proportional bone mineral protein and a potentially faster recovery. The BG cervical cage might also show particular promise in cervical spine fusion surgery.11-13 The aim of our study, a randomized controlled trial (RCT), is to compare the clinical and radiological outcomes of the traditional allograft and the innovative BG cage. The aim of this comparison is to shed light on the potential advantages and challenges of integrating BG cage in ACDF surgeries.
Methods
Trial Design and Supervision
We conducted a prospective, single-center, noncrossover RCT in the neurosurgical department to assess the efficacy of the allograft vs the BG cage in ACDF for the treatment of cervical degenerative disc disease. Our study received ethical approval from the Institutional Review Board, and ethical clearance and trial registration were obtained from our institute's Institutional Review Board (No. KC17OES10583). Prior to their inclusion, all participants granted written consent. The study's ethicality and scientific integrity were upheld by adhering to the Declaration of Helsinki and the Good Clinical Practice guidelines.
Participant Selection
Between August 2017 and August 2022, eligible patients were recruited for this trial. Participants were eligible for inclusion in the study if they were adults aged between 18 and 75 years. They must have demonstrated clinical symptoms of cervical radiculopathy or myelopathy stemming from subaxial cervical degenerative disc disease, whether due to soft disc herniation or spondylosis. Radiological validation of cervical disc degeneration, as evidenced by MRI or CT scans, was imperative. All the candidates for the study had to have endured ineffective nonsurgical management of their cervical complications for at least 3 to 6 months. A cardinal prerequisite for all participants was the voluntary signing of a written informed consent form.
The study excluded patients who had previously undergone surgery at the index level or had a history of posterior cervical spine surgery. Individuals with severe osteoporosis, identified by a T score of less than −3.5, were not considered. Moreover, those who had cervical spine degenerative disease at two or more levels or needed intervention at more than three levels were omitted from the study. Patients with less than 6 months of follow-up data were also excluded. Specific conditions, such as abscess, deformity, fracture, malignancy, and any form of allergic reaction to the implant material, rendered potential participants ineligible. Last, any known allergies to graft materials or any contraindications to surgery led to the exclusion of the candidate.
Randomization and Blinding
Given the intrinsic characteristics of the intervention, comprehensive blinding was inherently challenging. A double-blind approach was only employed in the preoperative phase. Eligible candidates were evenly randomized into either the allograft cage (Cornerstone-SR™, Medtronic) or BG cage (Novomax; CGBio Inc.) cohort. A computer-generated sequence was utilized for the allocation process, and assignments were safeguarded using sequentially numbered, sealed, and opaque envelopes. A specialized clinical research coordinator who was not involved in direct patient care or outcome evaluation, managed the allocation sequence, ensuring it was securely encased in sequentially numbered, opaque envelopes. On the day preceding the surgery, the clinical research coordinator disclosed the envelope's content, subsequently informing the operating surgeon of the patient's designated surgical group and making sure that the surgeon was apprised of the group allocation only before the surgery. To minimize potential bias in evaluating surgical outcomes, we ensured that the surgeons only saw the label and only interacted with the patients during the operation and subsequent outpatient follow-ups and were not involved in assessing outcome assessors and data analysts. While it was impractical for these assessors to remain blinded to the type of interbody cage visible in radiological images, they were completely detached from the preoperative treatment process and had no direct care responsibilities for the patients. This deliberate separation of roles considerably reduces the likelihood of bias in the assessment of patient outcomes.
Surgical Procedure
Surgery was performed by a single experienced spine neurosurgeon; the patient was positioned supine on a specialized flatbed Jackson table and underwent general endotracheal intubation. In our practice, intraoperative neuromonitoring is employed. The patient's neck was gently extended using a folded sheet placed beneath the shoulders. To ensure sterility, the patient’s neck region is prepped and draped, and an incision is made in alignment with the patient's natural skin crease. Using the Smith-Robinson approach under lateral fluoroscopy, the cervical vertebrae are accessed. The correct vertebral level is validated using fluoroscopy. The trachea and esophagus were retracted by retractors. Upon accessing the spine, comprehensive cervical discectomy is performed. Emphasis is placed on preserving the posterior longitudinal ligament first, especially during the crucial uncinectomy phase, ensuring dural sac protection. The border of the medial uncinate is meticulously discerned, and then the uncinate is methodically thinned using a high-speed diamond burr while safeguarding the sensitive nerve root beneath. Then, either the allograft or the BG cage (Figure 1), an anterior cervical plate fortified with screws, is affixed to the adjoining vertebral bodies, ensuring cage fixation. Postoperatively, patients are provided with a cervical collar, and the immobilization is undertaken for a well-defined span of four weeks. Radiographic image displaying the allograft cage and BG cage.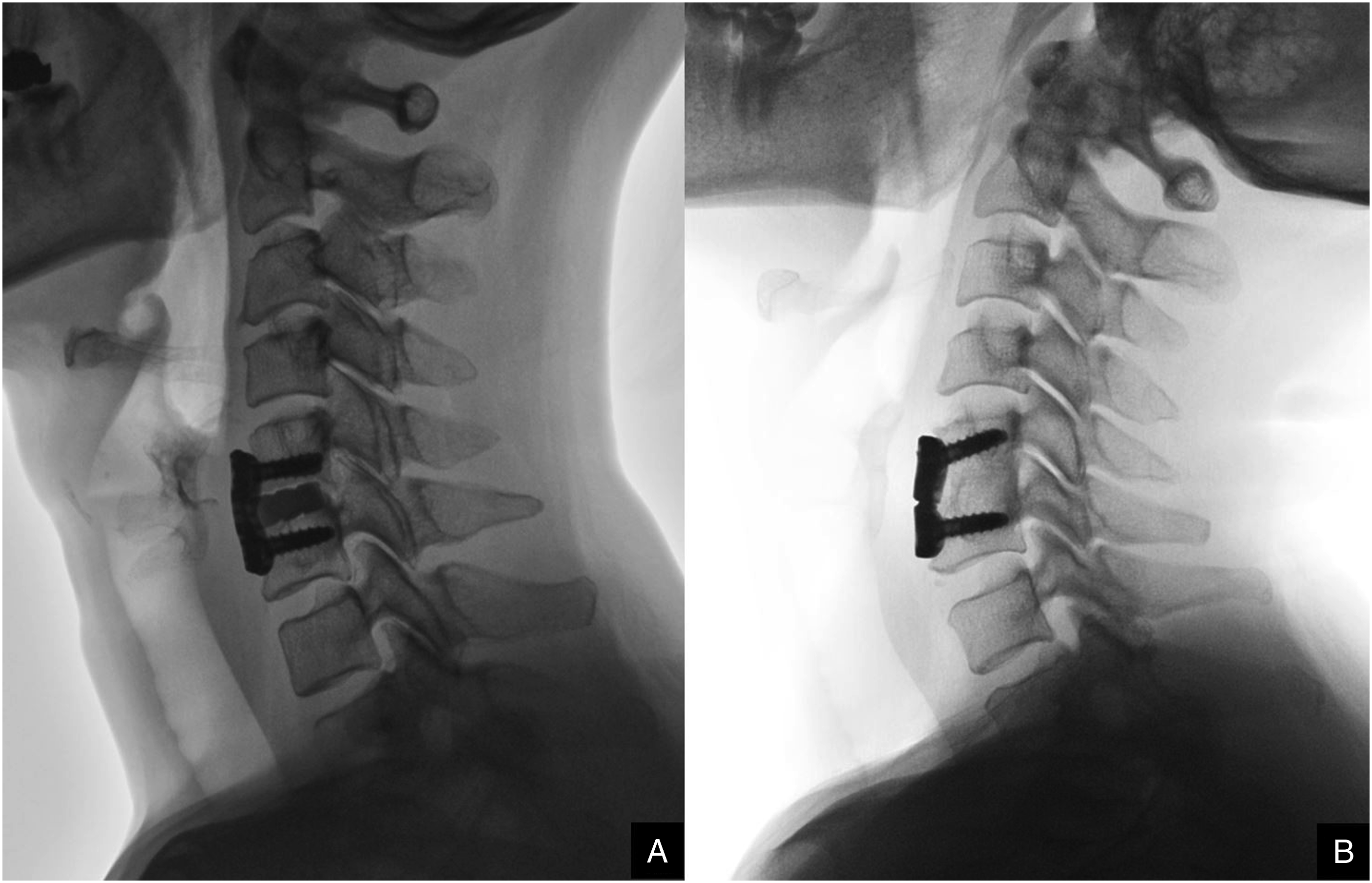
Outcome Measures
Demographic and clinical data were collected via comprehensive chart reviews. Factors under scrutiny included age, sex, bone mineral density (BMD), body mass index (BMI), surgery level, operation duration, estimated blood loss (EBL), diagnosis, prevalent symptoms, identified comorbidities, surgical approach side, Charlson Comorbidity Index (CCI), American Society of Anesthesiologists (ASA) score, and individual lifestyle elements such as cigarette use and alcohol consumption. Following their surgical procedures, patients were enrolled in a regular follow-up protocol. All participants underwent standardized pain management, physiotherapy, and meticulous wound care. Visits were scheduled for 1 month, 3 months, 6 months and 12 months postoperatively, and then on an annual basis thereafter. Subsidence and fusion status were evaluated at the 6- and 12-month follow-up visits.
The primary clinical outcomes that were measured were reflective of the patients' postoperative experiences. Pain levels in the neck and arm regions were quantified using the Visual Analog Scale. Furthermore, the Neck Disability Index was employed. The secondary clinical outcomes were the rate of return to daily life and work activities. Surgical details, encompassing the duration of the operation, blood loss volume, and length of hospital stay, were meticulously recorded. Moreover, vigilance was maintained to document any surgical complications, ensuring a comprehensive understanding of the risks such as infections, graft migrations, and neurological deficits.
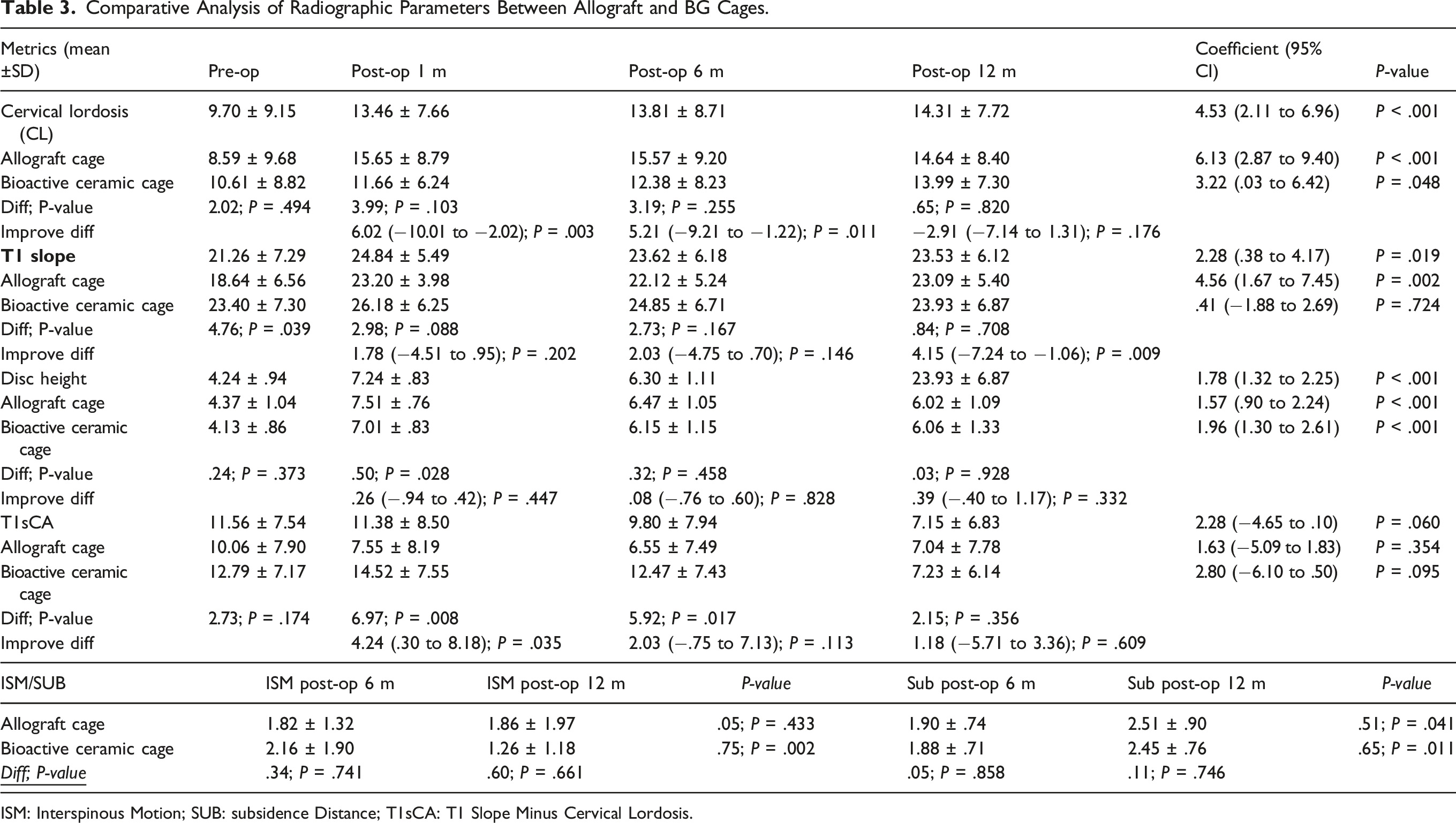
Radiological assessments, including lateral cervical radiographs, that offered insights into cervical lordosis and the T1 slope were conducted. Cage subsidence, a metric indicating a reduction of 3 mm or more in disc height postoperatively, was defined. The depth of the intervertebral space at its anterior and posterior segments determined the disc height. Another crucial radiological metric was the T1sCA or Cervical Sagittal Angle. This measurement reflects the cervical sagittal balance in that it determines the additional angle, post accounting for the T1 slope's impact, needed for the cervical sagittal angle to horizontally align (T1sCA = T1 slope−cervical lordosis; T1sCA ≤ 20°, balance; >20°, imbalance). 14 The fusion status was determined using the Bridwell interbody fusion grading system, with fusion defined as grades I and II. It is assessed by three-dimensional computed tomography and verified by separate orthopedic surgeons who are not involved in patient care. 15
Statistical Analysis
Descriptive statistics were employed to characterize the study population: continuous variables are represented as the means ± standard deviations, while categorical data are articulated in frequencies and percentages. Categorical variable comparisons between independent groups were evaluated using chi-square tests or Fisher’s exact tests. Missing data at the 12-month mark, multiple imputation to handle missing data, leveraging the chained equations approach. The imputation of missing values was conducted using the Predictive Mean Matching method in conjunction with the k-Nearest Neighbors technique, and the imputation process was adjusted using the baseline data. Concerning a limited overall number of cases, with part of the data not being normally distributed and several groups of data failing the variance chi-square test, we did not opt for variance analysis.
Therefore, to explore intergroup differences while adjusting for baseline variability between patients, we utilized a multilevel mixed-effects linear regression model. This model was designed to investigate the relationships between different groups of spinal cages used in surgery. In our model, both the intercept and slope were treated as random effects to account for individual variability. Moreover, in our effort to assess changes over varying follow-up periods within the same patient group, we employed the generalized estimating equations methodology. To aptly model the correlation structure of these repeated measurements, we assumed an exchangeable correlation matrix. If the data are a variance matrix that is nonsymmetric or highly singular, consider nonparametric tests such as the Wilcoxon signed-rank test. The primary statistical method to address the primary outcomes is intention-to-treat analysis. However, it is worth noting that such an analysis might undervalue the authentic treatment influence. Throughout our analyses, a P value threshold of less than .05 was employed to define statistical significance. Statistical analyses were meticulously executed using STATA software, while the graphical representations were visualized with GraphPad Prism.
Results
Baseline Characteristics
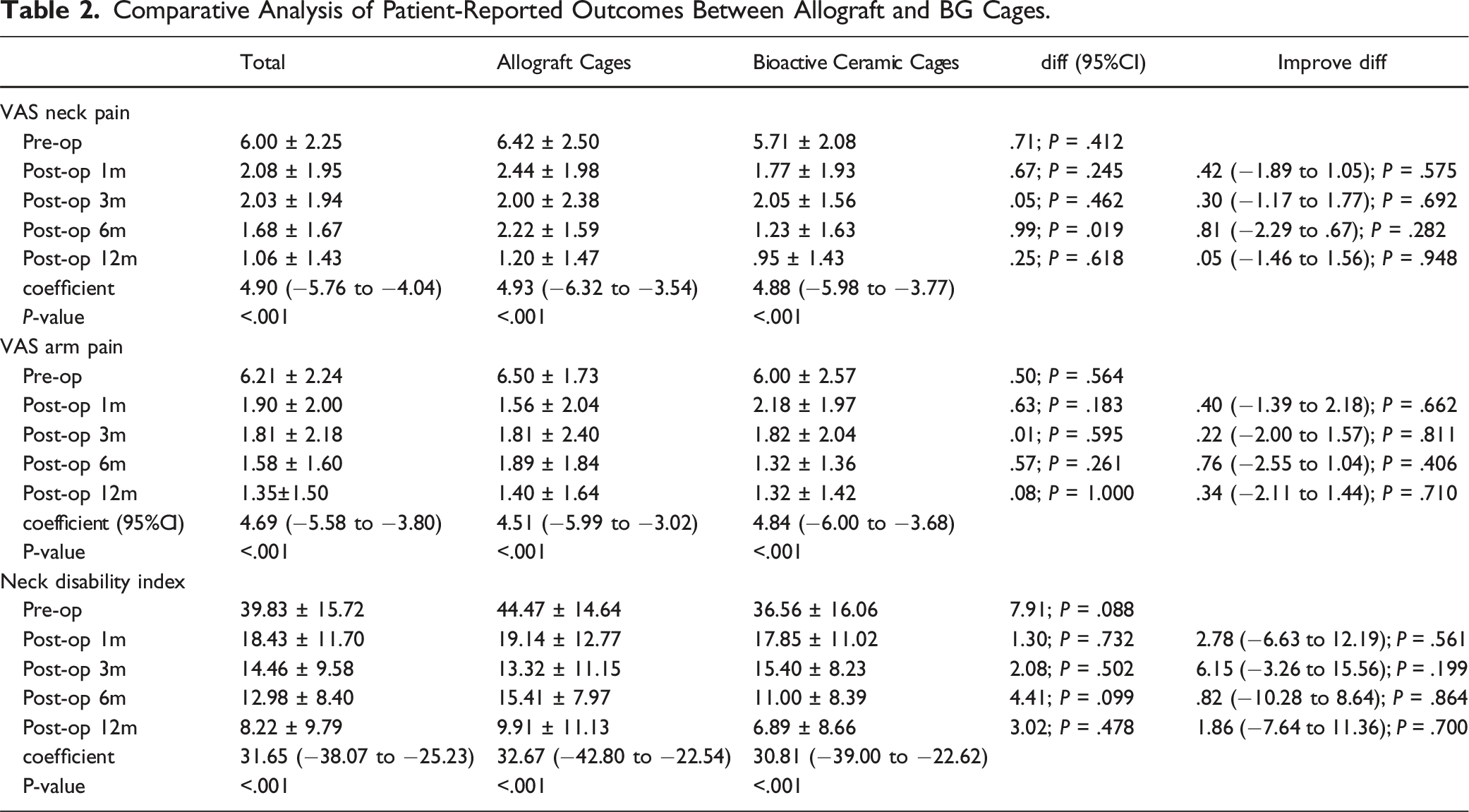
Out of the 45 individuals assessed for eligibility, three patients were excluded from the analysis due to their decline to participate in the randomization process, while the other two patients, although they agreed to the randomization process, were excluded because they were unlikely to comply with the follow-up protocol due to geographical distance and conflicting schedules. Finally, a total of 40 (52 levels) participants consented to participate in the research and were incorporated in the analysis. Patients were randomly assigned to the treatment groups: allograft cage (n = 18; 24 levels) and BG cage (n = 22; 28 levels). No treatment crossover occurred. The research's follow-up flow diagram is detailed in Figure 2. No significant disparities were detected between the groups regarding baseline characteristics (P > .05) (Table 1), as outlined in Table 1. Notably, only one patient from each group continued smoking and consuming alcohol. The C5-C6 segment was the primary operative single level, with C5-C7 representing the prevalent 2-level ACDF segments Table 2. Flowchart diagram illustrating the participant enrollment process. Demographic Characteristics of the Patients Treated With Allograft and BG Cages. Comparative Analysis of Patient-Reported Outcomes Between Allograft and BG Cages.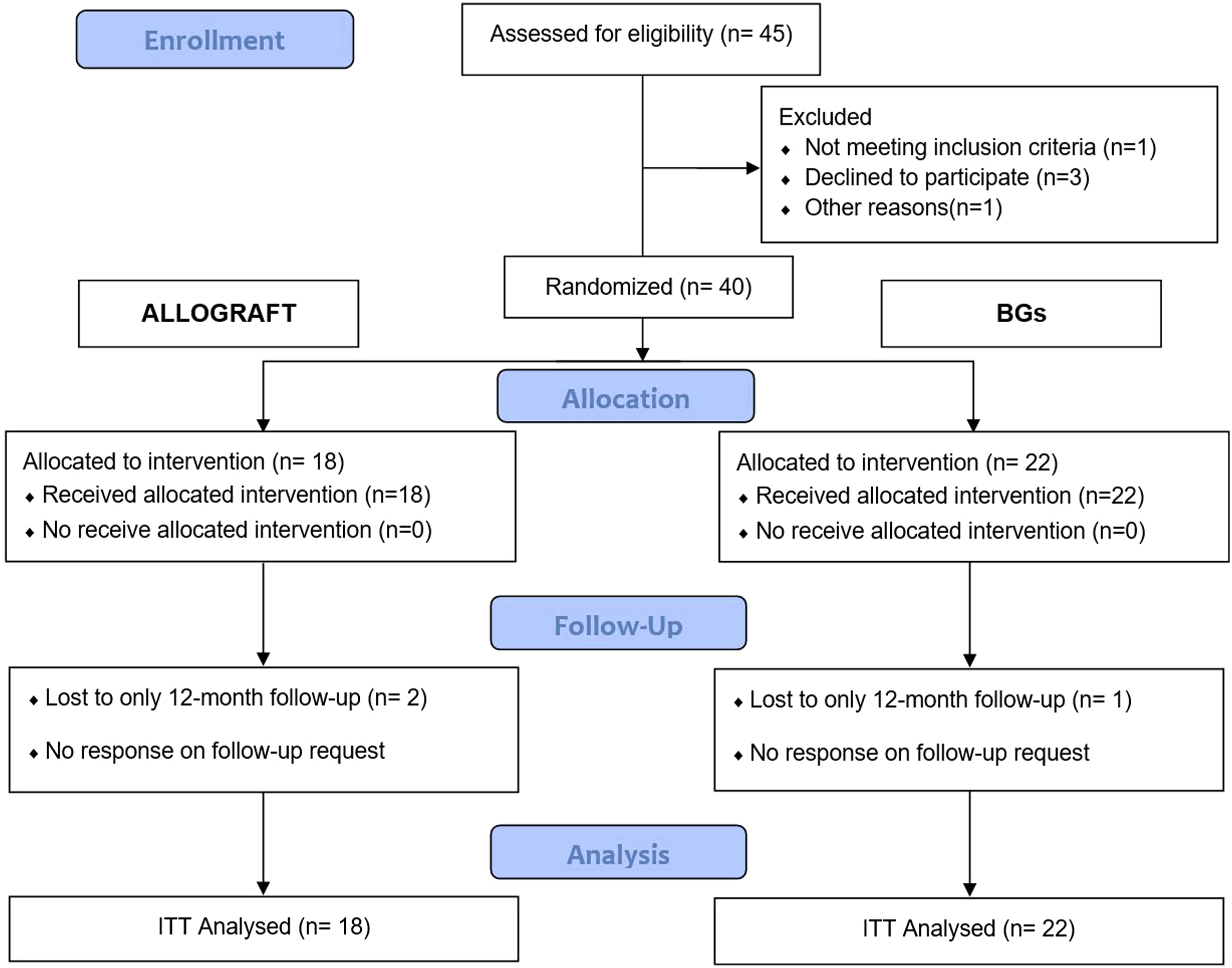
Patient-Reported Outcomes
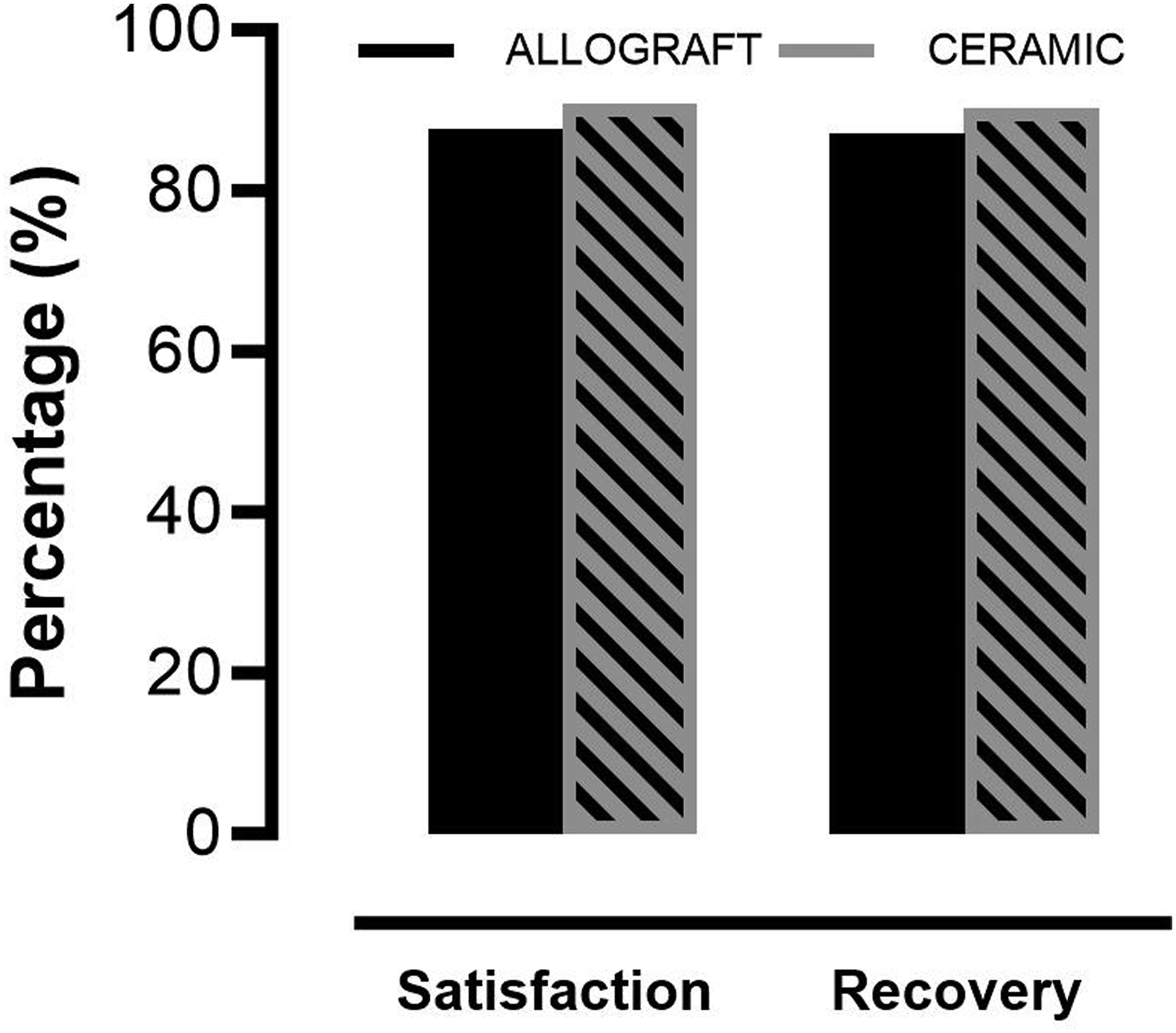
Patient-reported outcomes revealed significant improvements in both groups by the 12-month follow-up relative to their preoperative data (Figure 3). Table 3 elucidates that the postoperative comparisons between the groups showed no noteworthy differences in VAS scores for neck (1.20 ± 1.47 vs .95 ± 1.43; P = .618) or arm pain (1.40 ± 1.64 vs 1.32 ± 1.42; P = 1.000) or the Neck Disability Index values (9.91 ± 11.13 vs 6.89 ± 8.66; P = .478). A consistent pattern was confirmed by the mixed-effects linear regression model, which demonstrated parallel outcomes across groups when the preoperative and postoperative 12-month follow-up data were compared. At the 12-month evaluation, both groups were comparable in terms of minimal clinically important difference in neck pain and disability index scores. Specifically, 83.33% of patients in the allograft group and 88.24% of patients in the BG cage group showed a MCID improvement beyond 2.6 in neck VAS scores, underscoring the lack of a significant difference (P = 1.000). Reflecting this positive trend, more than 85% of patients in both groups recovered and reported that were satisfied with their outcomes at the 12-month review, and 76.92% of patients in the allograft cage group and 60.00% of patients in the BG cage group returned to work (P = .435) (Figure 4). Line graphs depicting patient-reported outcomes pre- and post-operatively. Comparative Analysis of Radiographic Parameters Between Allograft and BG Cages. ISM: Interspinous Motion; SUB: subsidence Distance; T1sCA: T1 Slope Minus Cervical Lordosis. Bar Graphs showing recovery rates and patient satisfaction at the final postoperative follow-up.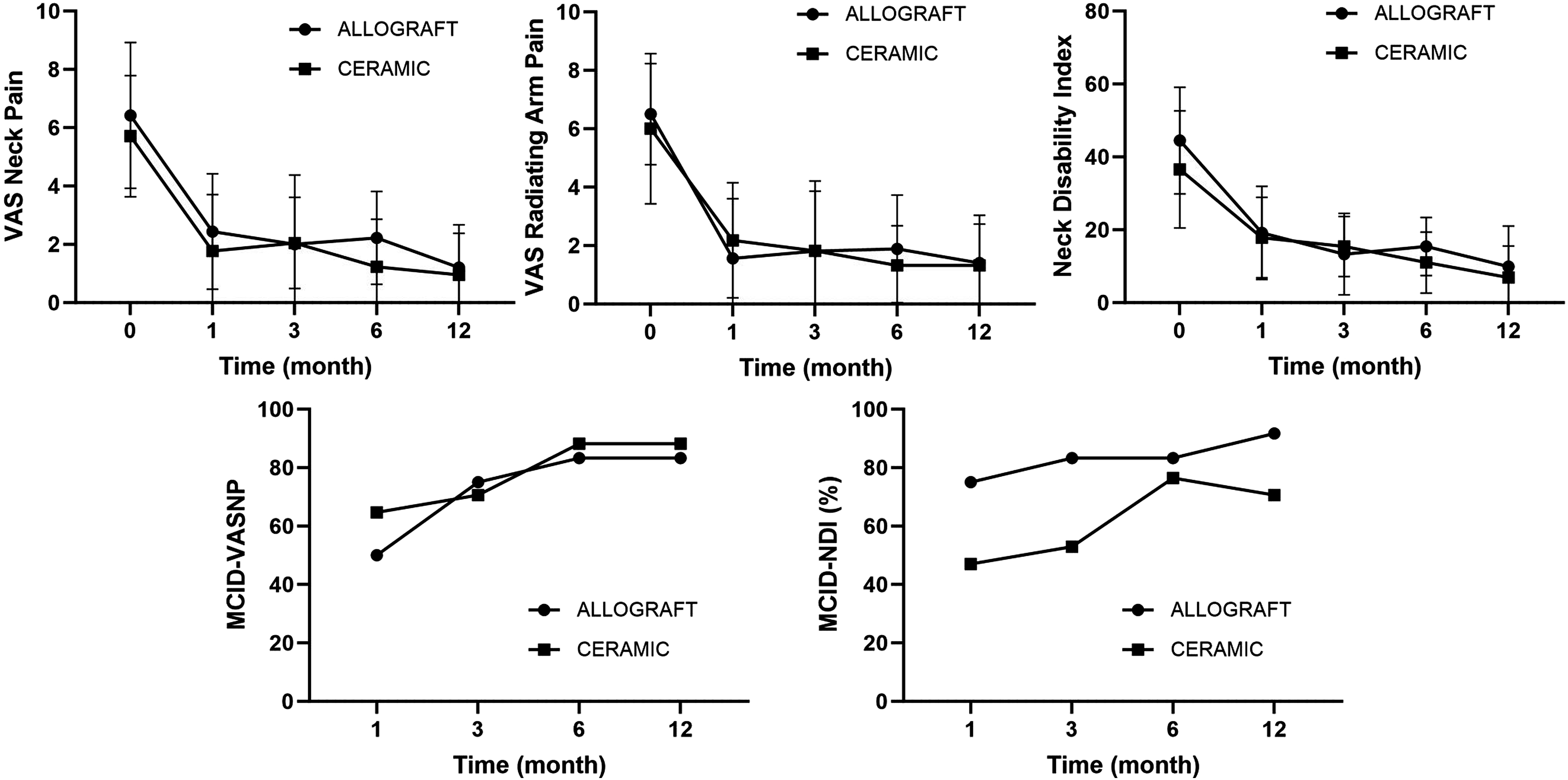
Radiological Outcomes and Assessments
Upon postoperative assessment across various time points, both the allograft cage and BG cage consistently displayed no notable significant differences. The data suggest that both cage types provided commendable stability of the cervical spine post intervention. (Figure 5) At the 12-month mark, there was a significant increase in cervical lordosis across both groups, with no discernible intergroup variability. Notably, the allograft cage group demonstrated a higher disc height than the BG cage group at the 6-month postoperative follow-up, registering a statistically significant difference of 5.21 (P = .011); however, there was no difference in the maintenance of disc height between the two groups at the last follow-up time. The T1 slope in the allograft cage cohort was significantly improved at the 12-month review, in contrast to its preoperative measurement. While the T1 slope was not improved in the BG cage group, intergroup comparisons at the 12-month follow-up revealed no significant disparities, as reflected by a mean difference of 1.18 (range: −5.71 to 3.36). Throughout the follow-up period, segmental kyphosis was absent in all patients. Trend graph illustrating radiographic measurements comparisons at each follow-up interval.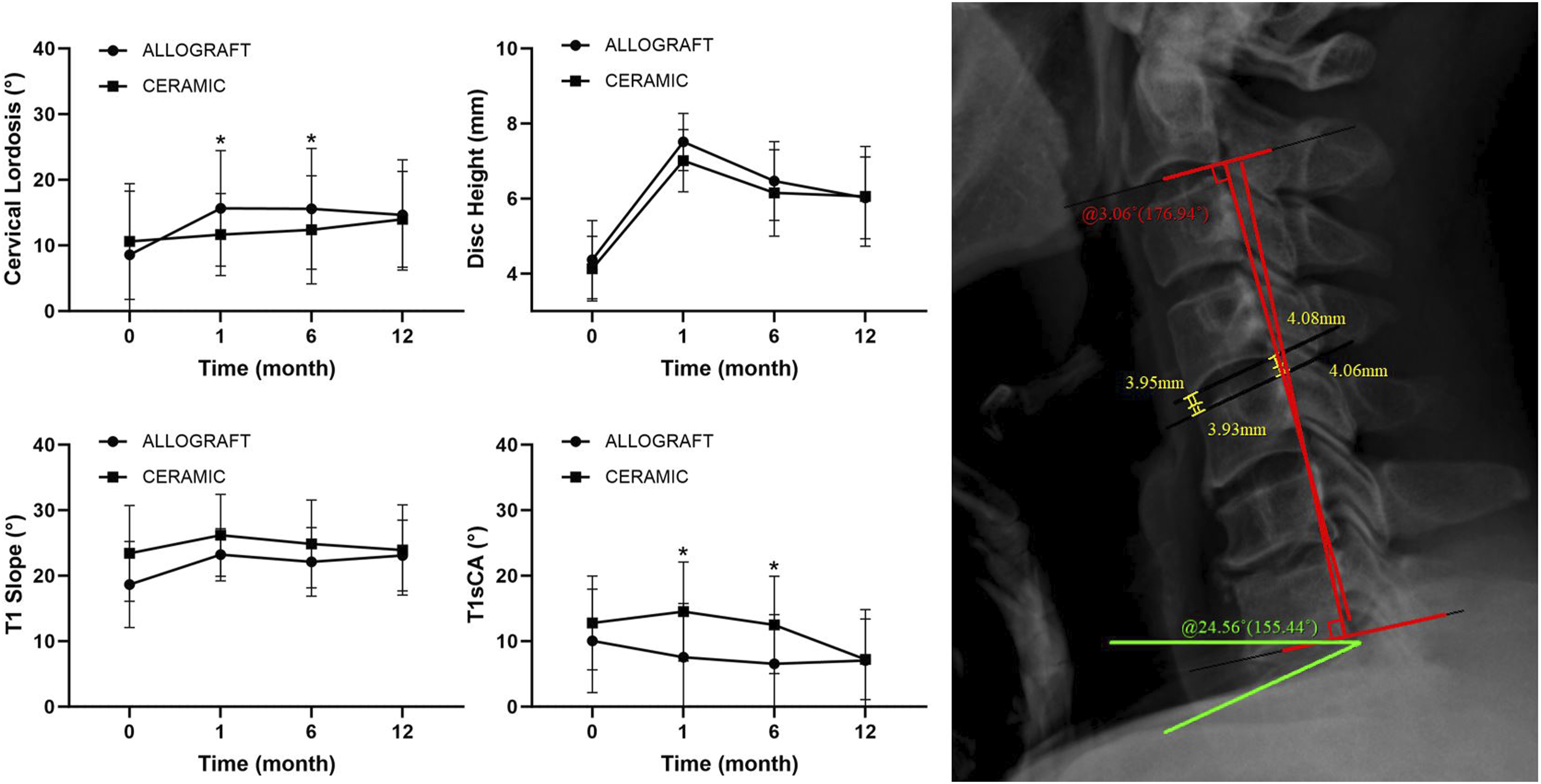
For ISM, patients who were implanted with an allograft spacer exhibited a slight increase in the mean value from 1.82 ± 1.32 at 6 months to 1.86 ± 1.97 at 12 months postoperatively. Conversely, the group with bioactive ceramic spacers showed a more pronounced fluctuation, with an initial mean of 2.16 ± 1.90 that decreased to 1.26 ± 1.18 at the 12-month mark. The difference in means between the groups at both time points did not reach statistical significance (P = .741; P = .661). Regarding subsidence, allograft spacer recipients had a mean value of 1.90 ± .74 at 6 months that increased to 2.51 ± .90 at 12 months, a change that was statistically significant (P = .041). Similarly, those with bioactive ceramic spacers began with a mean of 1.88 ± .71 and ended with 2.45 ± .76, also showing a statistically significant change over time (P = .011). Nonetheless, the difference in subsidence between the two materials at each time interval was not statistically significant, with P values of .858 at 6 months and .746 at 12 months. While both materials showed a trend toward increased values over time, the bioactive ceramic spacers significantly reduced the ISM in patients at 12 months postoperatively, which was not observed in the allograft group. Subsidence increased significantly over time for both materials, yet the comparative difference between them remained statistically nonsignificant.
In terms of fusion efficacy, at 6 months postoperatively, patients with allograft spacers showed a fusion rate of 45.83%, which increased to 70.83% by the 12-month follow-up. In contrast, the bioactive spacer group had a higher fusion rate at the 6-month follow-up, with 53.57% slightly improving to 75.00% at the 12-month checkpoint (Figure 6). The data illustrate a trend where bioactive spacers tend to have a marginally higher early fusion rate at 6 months regardless of single-level or double-level fusion, although by 12 months, both types of spacers achieve comparable outcomes. Composite graph showcasing the fusion rates of single-level and double-level spinal fusions. Violin plot highlights the differences in interspinous motion.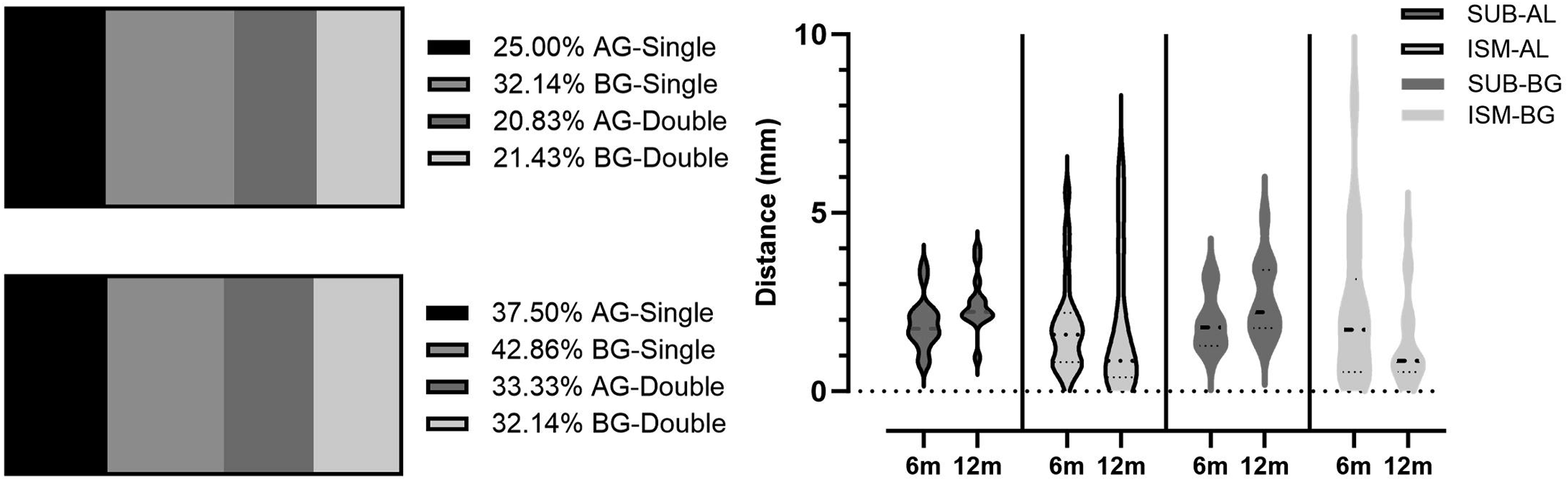
Adverse Effects
In terms of procedure durations, lengths of hospital stay, and cage implant percentages, there was a notable parity between the two cage groups. Importantly, no acute complications, such as debilitating pain, infections, or hemorrhaging, were reported. Although a minority of patients experienced postoperative discomfort or localized pain at the incision or graft sites, none of these cases warranted subsequent reoperations due to serious complications, such as postoperative hematoma. In terms of specific adverse events, 1 patient in the allograft cohort who underwent C5-6 fusion was observed to have pseudoarthrosis and absorption of the allograft at the last follow-up and another patient who was implanted with an allograft at both the C5-6 and C6-7 levels and exhibited mild swallowing difficulties that persisted up to the latest follow-up. Only one patient in the BG cage group reported postoperative transient hoarseness at the C6-7 level, which ultimately resolved completely.
Discussion
The randomized controlled trial compared the preliminary clinical and radiological outcomes of allograft cages and the innovative BG cage in ACDF surgeries. The study's rigorous methodology, including ethical clearance, comprehensive participant selection criteria, and robust statistical analysis, lends credibility to the findings. In our results, both the allograft and BG cages significantly improved patient-reported outcomes, including visual analog scale scores for neck and arm pain and the neck disability index. Importantly, no significant adverse effects were reported in either group, indicating that both materials are safe for use in ACDF surgeries. Moreover, radiographic parameters such as cervical lordosis, T1 slope, and disc height changes pre- and postoperatively showed no significant difference between the two groups. The study found no significant differences in fusion rates or subsidence distance between the two groups. Generally, sinking of a body with a higher elasticity modulus into a body with a lower elasticity modulus leads to 3-dimensional changes in spinal geometry. However, in our study, the BG cage elasticity was higher than that of the allograft, and there was no difference in subsidence distance compared to the allograft cage at the 12-month postoperative follow-up. This finding aligns with previous studies that have highlighted the safety and efficacy of allografts and BG cages in spinal surgeries.16-19
A recent study adds to the growing body of evidence supporting the use of bioactive materials such as BG cages in ACDF.20,21 The comparable outcomes with traditional allografts, combined with the added advantages of superior bioactivity, eliminating the need for additional bone supplementation, set it apart. BG cage share some properties with ceramics, such as being brittle, inorganic, and nonmetallic and composed of metal-oxygen ionic bonds. The CaO-SiO2-P2O5-B2O3 spacer establishes a chemical bond with bone tissues through the formation of a silica gel layer. This layer stimulates the proliferation and differentiation of osteoblast cells, initiating the synthesis and deposition of the organic bone matrix. Bioactive glasses dissolve when exposed to body fluids, leading to the formation of apatite crystals on their surface, which allows them to chemically bond with the bone tissues. 22 This is also consistent with our finding that the BG cage has a rapid early fusion rate. Early fusion between the cage and the vertebral body in spinal surgeries can mitigate the phenomenon of stress shielding. When fusion is achieved promptly, the cage and vertebral bodies collaboratively bear loads, allowing the vertebral bone to remain engaged in stress distribution and reducing the shielding effect. This integrated load-bearing facilitates biomechanical harmony, ensuring a seamless transition of forces between the cage and the bone. Moreover, early fusion eradicates potential micromotions at the cage-bone interface, which might otherwise impede proper load transfer and induce stress shielding. 23 Furthermore, our results are different from previous studies related to the subsidence distance that showed that the BG cage group has been reported to have significantly lower rates of subsidence than the allograft bone group. 24 According to our results, both the allograft spacer and the BG cage showed similar subsidence rates at 6 months and 12 months postoperatively.
Before our research, we were concerned that BG cages can present certain challenges during implantation. These challenges include potential complications such as spacer displacement, fragmentation, and subsidence. If not addressed, these complications can escalate to more severe issues, such as fusion failure and compression of the spinal canal capsule. Several studies have highlighted intraoperative displacement in both posterior interbody fusion and extreme lateral interbody fusion procedures when using BG cages in lumbar spine surgery, identifying this as a predominant complication.13,25 It appears that the primary concern arises from the smooth surfaces interfacing with the BG cages, rather than their intrinsic properties. However, our findings from a 1-year study offer a different perspective. None of our patients experienced these complications. One plausible explanation for this discrepancy is the inherent structure of the lumbar spine that exists as a dynamic force weight-bearing issue to consider related to dynamic loading conditions that the lumbar spine experiences during daily activities, unlike cervical fusion. Additionally, an anterior plate is used in cervical fusion, and this factor further restricts the potential distance of cage migration. Actually, the spacers used in the BG cages come in two different designs, both perforated and nonperforated at the center. In our research, we used spacers without holes because we believe their bioactivity is sufficient that no additional fusion material is needed. More importantly, they support a more extensive footprint on the surface of the cage.
In our RCT, a number of pertinent limitations merit attention. At the forefront, the study's confinement to a lone institute and its reliance on a single surgeon's expertise may circumscribe the application of its findings to some surgical practices and patient demographics. Additionally, due to the relatively small sample size, the risk of a Type II error is increased, potentially leading us to miss a genuine effect or difference. Such a sample size restricts our ability to detect all but the largest of effect sizes, and it narrows the confidence with which we can generalize our findings to a larger population. 26 Coupled with this, the limited duration of our follow-up might overshadow nuanced long-term outcomes or latent postsurgical complications. Moreover, because this was not a completely double-blind protocol postoperatively, there is a possibility of unintentional biases, possibly skewing the integrity of the results. Moreover, our singular focus on the cage type devoid of holes in the BG cage group curtails the extrapolation of our findings to diverse cage configurations. Furthermore, our study did not find significant differences in radiological outcomes between the two groups, possibly because the 1-year postoperative radiological outcomes might not fully capture the long-term issues of using BG cages. Finally, the lack of a comprehensive cost-effectiveness analysis leaves a void in understanding the economic ramifications of our chosen interventions.
Conclusion
The study provides that both materials, allograft cages and BG cages, were found to be safe in ACDF surgeries, leading to significant improvements in patient-reported outcomes and radiographic alignment. Notably, the BG cage accelerated fusion at the earlier postoperative follow-up time point, representing the potential to improve patient outcomes further. Therefore, larger cohorts with longer-term follow-up research are needed to confirm these findings and explore other relevant factors.
Footnotes
Acknowledgments
Our team like to express gratitude to Dr JYC and Dr SYB for their assistance in this research.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jin-Sung Kim is a consultant for RIWOSpine, GmbH, Germany, Stöckli Medical AG, Switzerland and Elliquence, LLC, USA.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
