Abstract
Study Design
Retrospective case‒control study
Objectives
This study aimed to report the effects of surgical intervention on spinal stability recovery and to assess the long-term outcomes of children and adolescents with lumbar tumors.
Methods
From January 2016 to June 2021, 42 pediatric patients with lumbar tumors were selected and separated into different groups based on the surgical method used (total en bloc resection (TER) group, n = 21; piecemeal resection (PR) group, n = 21; titanium mesh (TM) group n = 23; artificial vertebrae (AV) group n = 19). The clinicopathological characteristics, treatments and related outcomes were described in detail and compared between groups, with P value ≤.05 indicating statistically significant differences.
Results
The average follow-up duration was 24.89 months, and the mean age was 14.89 ± 2.41 years. There were no significant differences in the mean operation time, average blood loss, complication rate, or length of hospital stay between the groups. The ODI, VAS and JOA scores at the final follow-up (FF) were elevated after surgery in all groups. The FF local angular drift (LOD) and lumbar angular drift (LUD) were greater in the TM group than in the AV group (P = .03, P = .001).
Conclusions
After surgery, pediatric patients with lumbar tumors can obtain satisfactory spinal stability, effective relief of pain symptoms and substantial improvements in neurological function. There was no significant difference in the invasiveness, safety or timeliness between the 2 surgical methods, so TER is recommended due to its low postoperative recurrence rate and good local control. Spinal fusion in the AV group resulted in better spinal stability.
Introduction
Spinal tumors account for approximately 4%-20% of all bone tumors, 1 and the incidence of spinal tumors in children and adolescents is relatively low, accounting for 4%-15% of all spinal lesions. According to the data from our spinal tumour centre, pediatric lumbar tumors account for approximately 22% of the total number of pediatric spinal tumors. Lumbar spinal tumors in pediatric patients cause bone destruction and neurological impairment, thus leading to pain, neurological deficits, or spinal deformities.2-4 The low incidence of spinal tumors, their nonspecific symptoms, and the strong desire for long-term survival makes it very challenging for spine surgeons to diagnose and manage patients with these tumors.
The treatments of spinal tumors in children must be individualized based on the clinical and imagological manifestations. Surgical interventions, including radical resection, segmental fusion and internal fixation, are recommended as the first-line treatment for patients with high tumour malignancy, neurological deficits, and spinal instability. Nonsurgical treatments are also considered for patients with less symptomatic vertebral destruction and focal lesions of disseminated type that are sensitive to chemoradiotherapy.5,6 Notably, incomplete and immature bone growth increases the difficulty of treating primary spinal tumors in children and adolescents. While trying to control recurrence and preserve neurological function during treatment, every effort must be made to maintain the stability of the spine and avoid affecting children’s growth and development. 7 After resection of the lesion, internal fixation and fusion may be required to maintain spinal alignment and stability. 8 Surgical resection is mainly divided into total en bloc and piecemeal resection, and spinal fusion is usually performed using artificial vertebrae or titanium mesh (TM) filled with allogeneic iliac bone. Previous studies of pediatric patients with spinal tumors mainly examined the effects of different surgical approaches on survival and local control of tumors; outcomes related to postoperative spinal stability and quality of life in children were rarely discussed.
Therefore, this study aimed to study the effects of surgical treatments in pediatric patients with lumbar spinal tumors on postoperative spinal stability and quality of life and to compare the outcomes between different surgical strategies.
Methods
Patients
The clinical features and radiological measurements were reviewed after approval by the Ethics Committee of Changzheng Hospital. The approval number was waived due to the retrospective nature of this study. Written informed consent was obtained from the patient's legal guardian, thus authorizing the treatment, imaging, and photographic documentation of the patients. The inclusion criteria were as follows: the patient was younger than 18 years old at the time of diagnosis; the tumour lesion invaded the lumbar vertebrae and was limited to 1 segment (or if 2 or more segments were involved, only 1 vertebral body needed to be treated surgically); Tomita classification: I-III/IV-VI; pathologically confirmed to have a primary spinal tumour without metastasis; and spinal instability, neurological deficits and/or complaints of unbearable pain. The exclusion criteria were as follows: patients with previous spine surgeries, congenital spinal deformity, or other medical history that might affect the postoperative evaluation; total paraplegia/neurological deficits for longer than 48 h; and patients lost to follow-up.
Patients were followed up at 3, 6, 9, and 12 months after surgery; every 6 months for the next 2 years; and once a year thereafter. Patient outcomes were based on the comprehensive outpatient radiological (e.g., postoperative radiography, CT, MRI) and functional assessments. Our study measured 4 main outcomes. Recurrence-free survival (RFS) was defined as the interval between surgery and recurrence or until June 2021 for patients without recurrence. Overall survival was not included because a majority of patients survived during the postoperative follow-up period. Stability was measured as follows. Two lines were drawn on the standing lateral X-ray to measure the local lordosis angle (1 parallel to the upper end-plate of the cephalic vertebra of the diseased segment and the other parallel to the lower end-plate of the caudal vertebra of the diseased segment), and the angle of their intersection was the local lordosis angle. Additionally, 2 lines were drawn on the standing lateral plain film to measure the lumbar lordosis angle (1 parallel to the upper endplate of T12 and 1 parallel to the upper endplate of S1), and their intersection was the lumbar lordosis angle. A convex angle value indicated that a patient was positive for lumbar lordosis and negative for local kyphosis. Neurological and QoL indicators, including JOA and VAS scores, were assessed using questionnaires. Bony fusion between contacts was evaluated by contrast-enhanced CT. Postoperative complications were defined as complications that required additional treatments, including surgery, medical treatment and others, within 30 days after surgery.
Indications for surgery were assessed by the same treatment group surgeons. Approval for this study was obtained from the ethics committee of our institution.
Surgical Procedures
Baseline Characteristics of the Enrolled Pediatric Patients.

When the P value >.05, there was no statistically significant difference between the groups.
Abbreviations: TER: total en bloc resection; PR: piecemeal resection; LCH: Langerhans’ cell histiocytosis; ABC: aneurysm bone cyst.
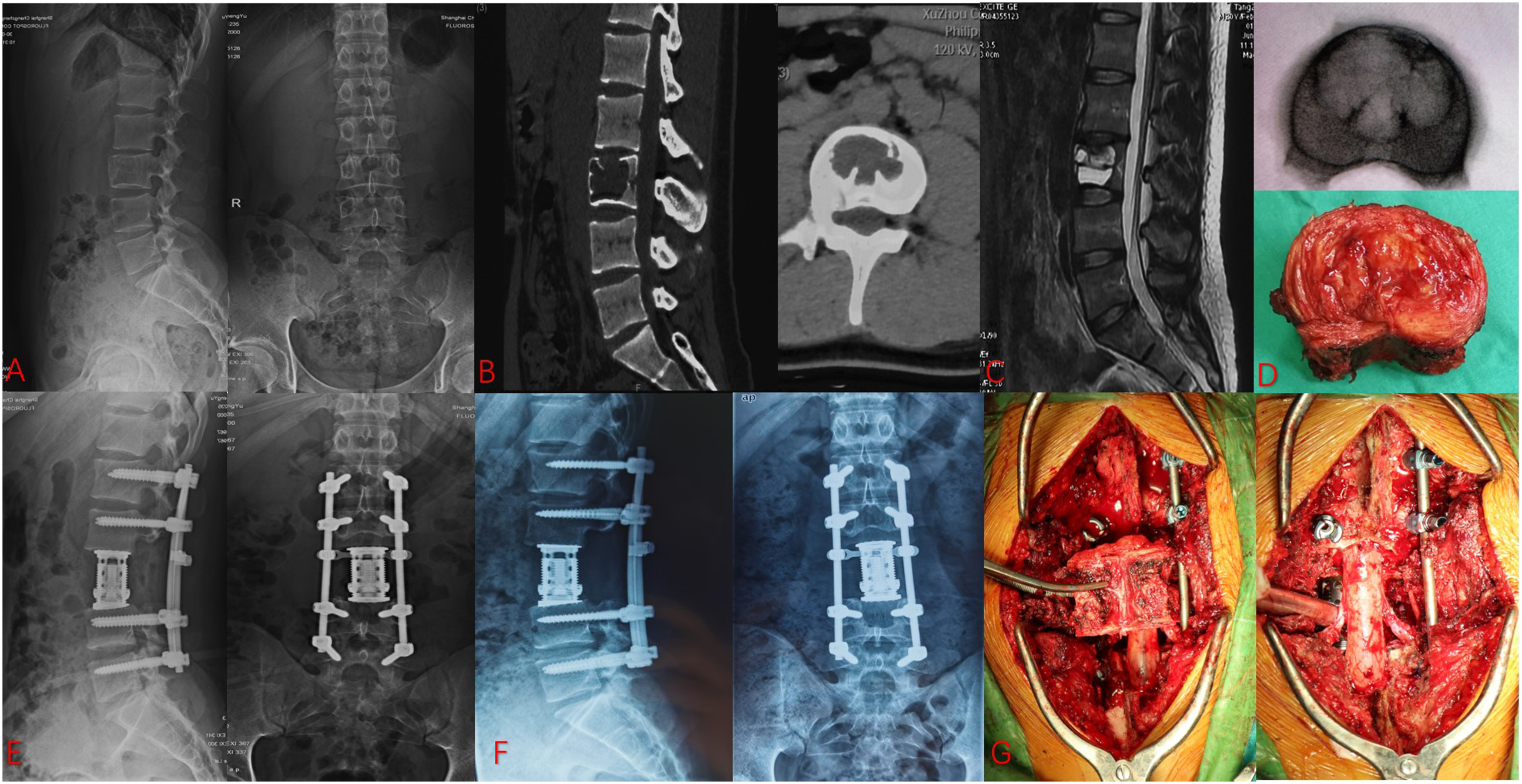
An 18-year-old boy (A) X-ray showed the lumbar curvature before surgery; (B) Preoperative CT scans showed the osteolytic fracture; (C) Preoperative transverse section of the lesion (MRI scan) (D, G) Intraoperative picture showed the total en bloc resection and instrumentation; (E) Postoperative X-ray showed instrumentation and the lumbar curvature; (F) At the final follow-up, X-ray showed good bone fusion and the lumbar curvature.
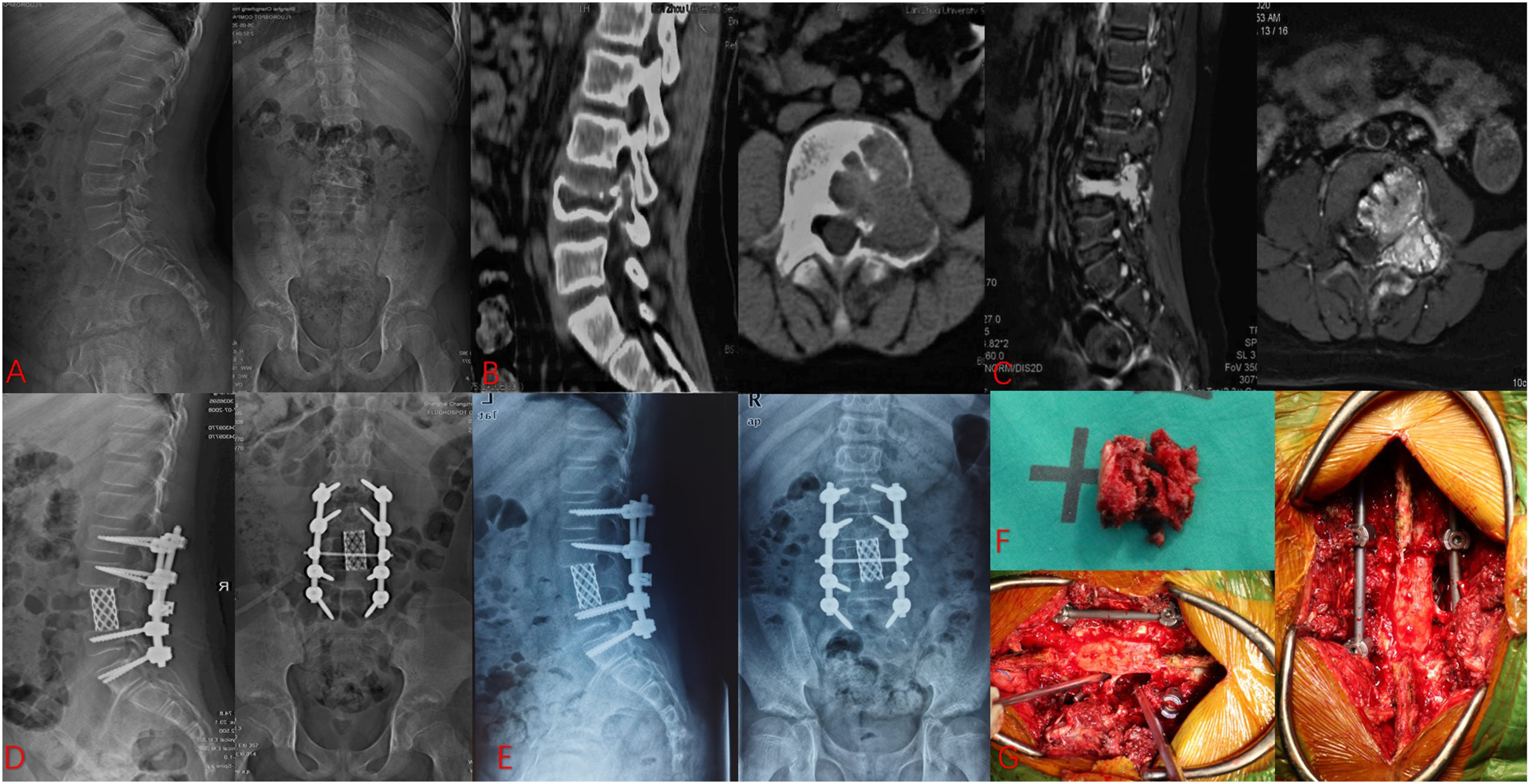
An 11-year-old boy (A) X-ray showed the lumbar curvature before surgery; (B) Preoperative CT scans showed the osteolytic fracture; (C) Preoperative transverse section of the lesion (MRI scan); (D) Postoperative X-ray showed instrumentation and the lumbar curvature; (E) At the final follow-up, X-ray showed good bone fusion and the lumbar curvature; (F,G) Intraoperative picture showed the piecemeal resection and instrumentation.
Postoperative Management
Patients were placed on bed rest after the operation, and the vacuum aspiration was removed within 10 days if appropriate. The internal fixation device was detected by the conventional X-ray film, and the patient was advised to wear an external brace for outdoor activity within 6 months to prevent internal fixation loosening and screw fracture caused by excessive spinal activity. All patients were followed up every month for the first 3 months and every 3 months for the next 12 months through outpatient visits. Reexaminations included evaluation of condition and neurological function, X-ray measurement of the lordosis angle and internal fixation. If necessary, MRI was performed to determine whether the tumour recurred. Patients with malignant tumors usually received chemotherapy. The chemotherapy regimen for Ewing sarcoma includes a biweekly combined treatment of vincristine/doxorubicin/cyclophosphamide or isocyclophosphamide/etoposide.12,13 The recommended drugs for adjuvant chemotherapy in osteosarcoma are methotrexate, isocyclophosphamide, doxorubicin, and cisplatin.13,14 Radiotherapy, immunotherapy, and targeted therapy were performed according to the pathological nature of the tumour. Other postoperative treatments include improved nutritional intake and specific neurotrophic and functional rehabilitation exercises. Notably, the angular change in the lordosis angle from postoperative to the final follow-up period was defined as the angular drift, which was counted as a positive value.
Statistical Analysis
The quantitative data were recorded as the mean ± standard deviation and range, and the qualitative data were described as counts and percentages. The data from preoperative, postoperative and final follow-up were analysed by paired/independent t tests or chi-square tests. Data were analysed using SPSS version 23.0. (SPSS Inc. Chicago, IL, USA). P values ≤.05 were considered statistically significant.
Results
Injury Indicators
Baseline Information
As shown in Table 1, 42 patients with the same tumour type were allocated to either group A or group B at a 1:1 ratio. The most common tumour site was L4 (n = 13), followed by L2 (n = 9), L3 (n = 8), L1 (n = 7), and L5 (n = 5). Our sample comprised 26 (61.9%) males and 16 (38.1%) females, and the mean age was 14.89 ± 2.41 years (range 10-18). The mean follow-up duration was 24.89 months (range 12-50 months). There was no significant difference in the baseline demographic characteristics of the 42 included patients between the 2 groups (P > .05). According to the Tomita classification, 20 tumors were classified as Tomita I-III, and 22 tumors were classified as Tomita IV-VI. There were no statistically significant differences in age, pathology, tumour site, tumour location or duration of the disease before surgery between the 2 groups (P > .05) (Table 1).
Peripheral Period Indicators
Comparison of Major Peripheral Period Indicators.
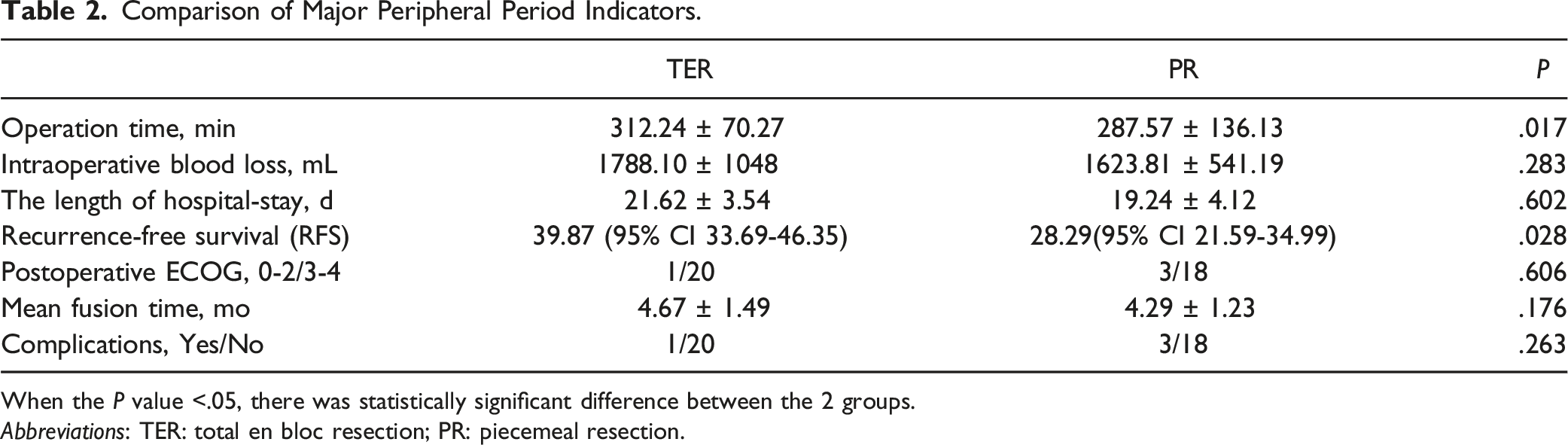
When the P value <.05, there was statistically significant difference between the 2 groups.
Abbreviations: TER: total en bloc resection; PR: piecemeal resection.
The Kaplan–Meier curve in Figure 3 shows that the risk of recurrence was significantly higher in the PR group (P = .028), with a mean RFS time of 28.29 months (95% CI 21.59-34.99) and 39.87 months (95% CI 33.69-46.35) in the PR and TER groups, respectively. Kaplan‒Meier curves of the effect of surgical method on recurrence-free survival. TER: total en bloc resection; PR: piecemeal resection.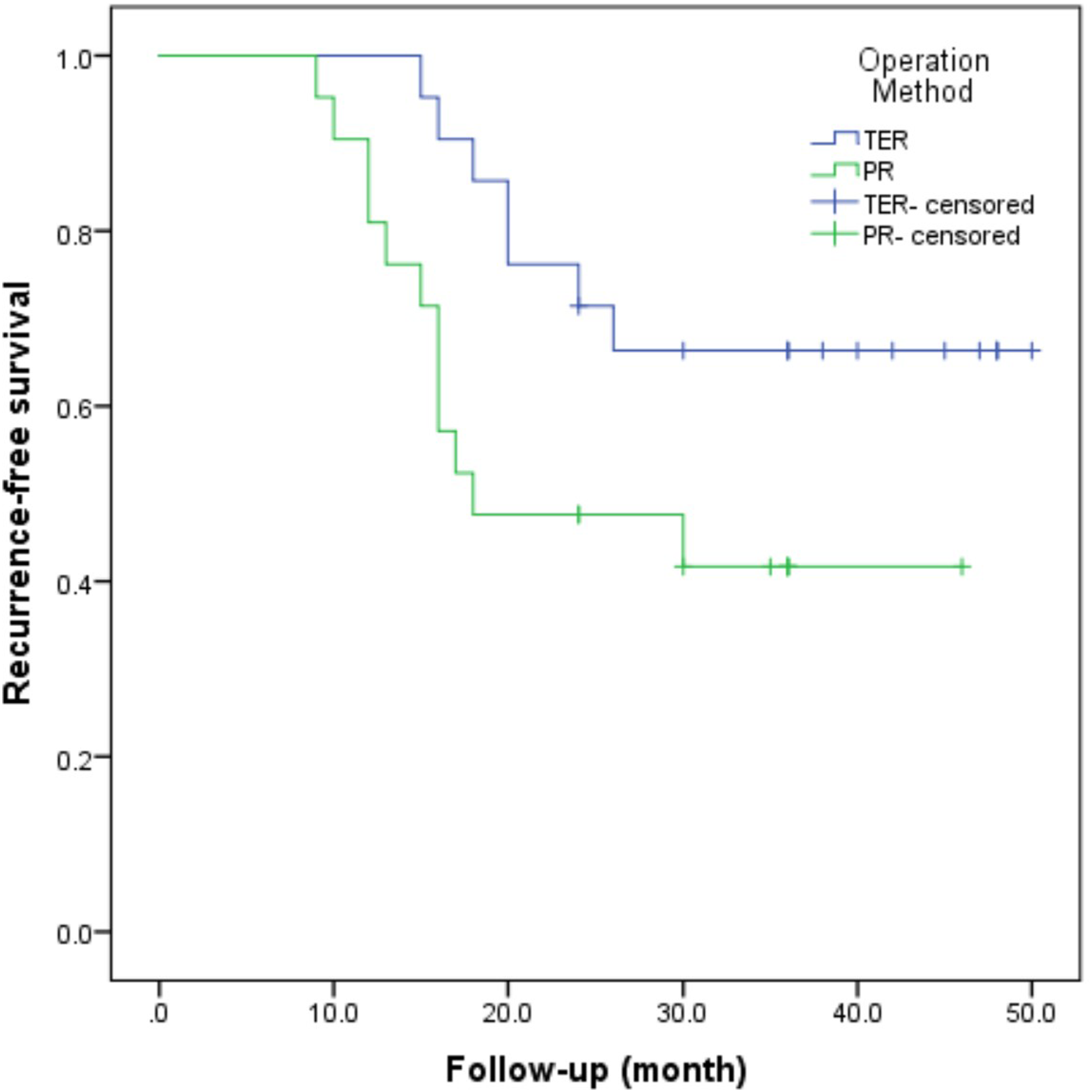
Neurological, QoL and Radiological Indicators
Comparison of AV and TM
Comparison of AV and TM in Neurological, QoL and Radiological Indicators.
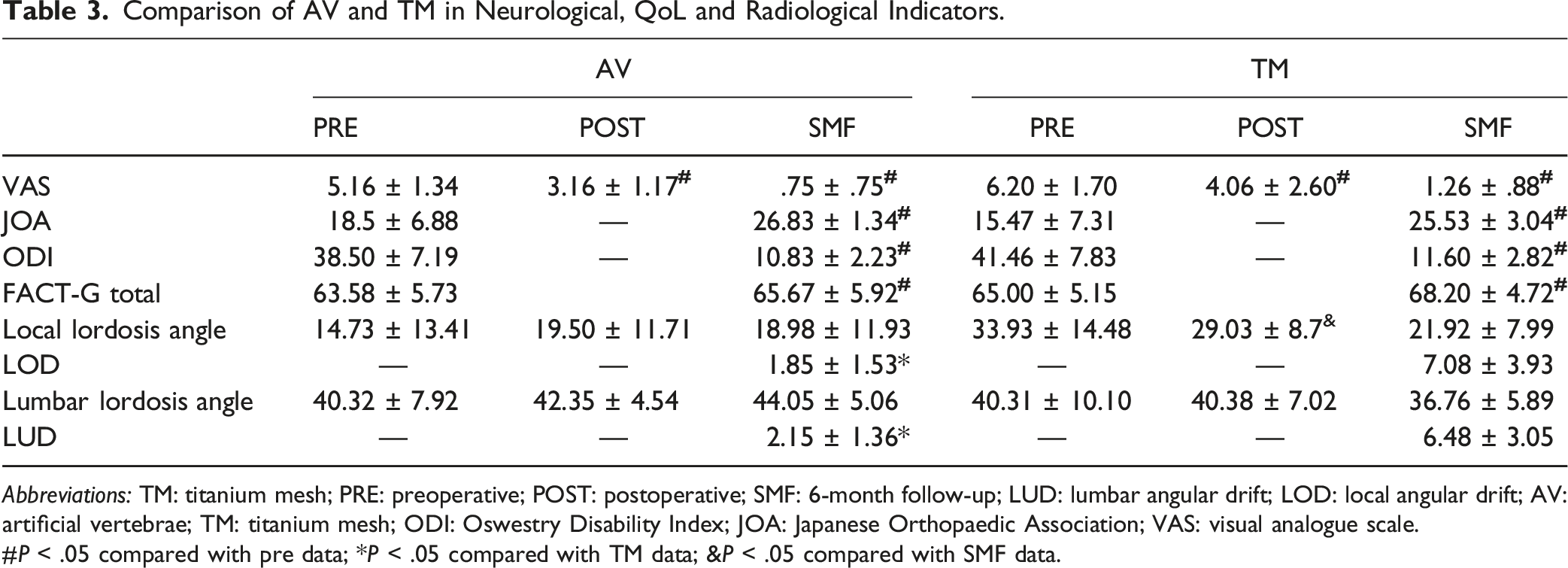
Abbreviations: TM: titanium mesh; PRE: preoperative; POST: postoperative; SMF: 6-month follow-up; LUD: lumbar angular drift; LOD: local angular drift; AV: artificial vertebrae; TM: titanium mesh; ODI: Oswestry Disability Index; JOA: Japanese Orthopaedic Association; VAS: visual analogue scale.
#P < .05 compared with pre data; *P < .05 compared with TM data; &P < .05 compared with SMF data.
At the 6-month follow-up, in the AV group, the mean lordosis angle was 44.05 ± 5.63° (range 35.0-54.7), and in the TM group, the mean lumbar lordosis angle was 36.76 ± 5.89° (range 24.4-48.8). There was a significant difference in the lumbar lordosis angle between the 2 groups (P = .002). In the TM group, the local lordosis angle at SMF was less than the local postoperative lordosis angle (P = .009). The final follow-up local angular drift (LOD) was larger in the TM group than in the AV group (P < .001). The final follow-up lumbar angular drift (LUD) was also greater in the TM group than in the TM group (P < .001) (Table 3).
Comparison of TER and PR
Comparison of TER and PR in Neurological, QoL and Radiological Indicators.
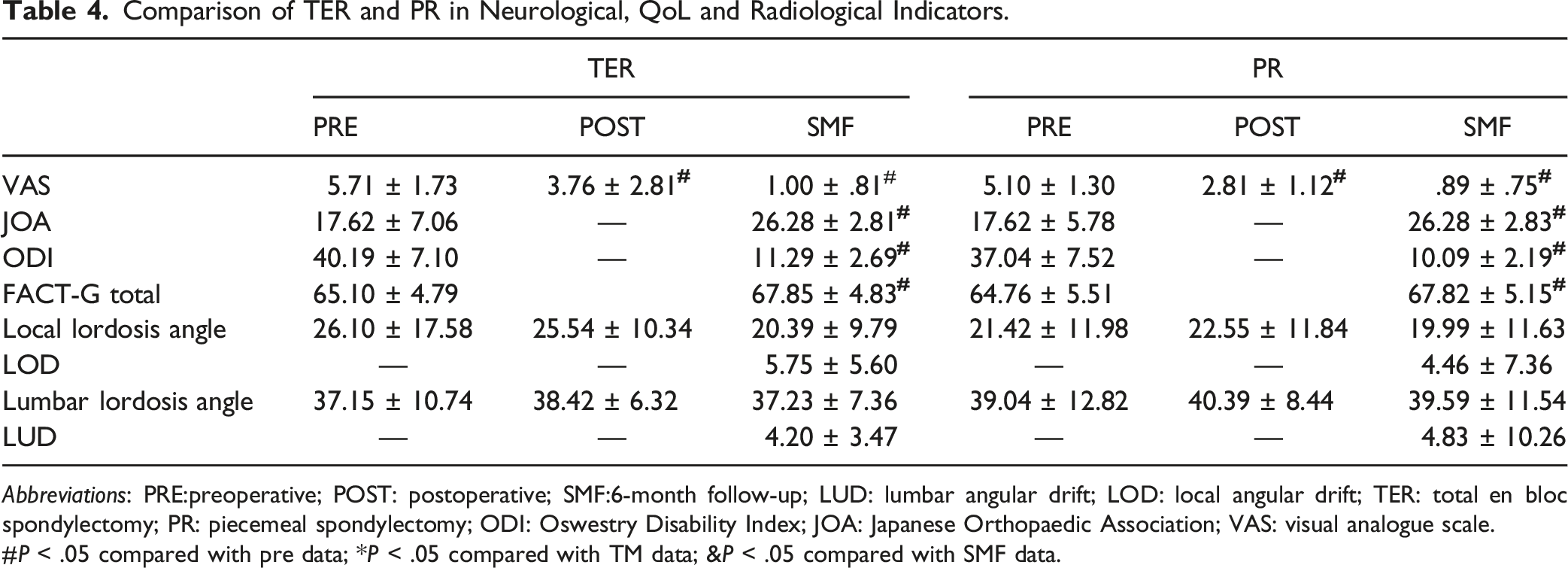
Abbreviations: PRE:preoperative; POST: postoperative; SMF:6-month follow-up; LUD: lumbar angular drift; LOD: local angular drift; TER: total en bloc spondylectomy; PR: piecemeal spondylectomy; ODI: Oswestry Disability Index; JOA: Japanese Orthopaedic Association; VAS: visual analogue scale.
#P < .05 compared with pre data; *P < .05 compared with TM data; &P < .05 compared with SMF data.
At the 6-month follow-up, in the TER group, the mean lordosis angle was 37.23 ± 7.36° (range 24.4-46.8), and in the PR group, the mean lumbar lordosis angle was 39.58 ± 11.54° (range 13.8-54.7). There was no significant difference in the lumbar lordosis angle between the 2 groups (P = .445). There was no statistically significant difference in the LOD or LUD drift between the TER and PR groups at the final follow-up (P = .795; P = .543).
Complications
In the TER group, there was 1 case of delayed wound healing due to malnutrition. In the PR group, postoperative complications were observed in 15.38% (n = 3) of patients, including surgical site infection (n = 1), dura tear and intraoperative cerebrospinal fluid leak (n = 1), and postoperative broken rod (n = 1).
Discussion
Primary osseous spinal tumors are relatively rare in the pediatric and adolescent population, accounting for approximately 1% of all spinal and spinal cord tumors.15,16 The low incidence of spinal tumors in children, poor description of their symptoms by themselves, and parental neglect lead to tremendous difficulty and challenge for managing such patients. More than 50% of pediatric malignancy patients exhibit neurological symptoms, and thus, neurological symptoms are highly indicative of the diagnosis of pediatric malignancy. 15 In addition, approximately 25% of pediatric spinal tumors often cause spinal deformities, including kyphosis, scoliosis, and pronation. The deformities may be caused by direct tumour-induced bone destruction and compression fractures or muscle tension or can be the result of compensatory responses to painful symptoms due to nerve compression.17,18 Some malignancies, especially Ewing's sarcoma and lymphoma, present with nonspecific systemic symptoms (e.g., fever, night sweats) and induce a misdiagnosis of infection rather than a diagnosis of malignancy.3,19 A clear preoperative diagnosis is particularly important to guide the individualized treatment of spinal tumors in children. In addition to clinical symptoms and signs as well as imaging examination, biopsy is often required to confirm the pathology before treatment, and commonly used pathological diagnostic methods include incisional biopsy, puncture biopsy, CT-guided puncture biopsy, and intraoperative frozen pathological examination.
The current treatment strategy for pediatric spine tumors basically refers to adult spine tumors, and common treatment modalities include surgical treatments, chemotherapy, radiotherapy, and targeted therapy. Especially for the surgical treatment of spinal tumors in children, the indications and surgical procedures are basically the same as those for adults.20-22 However, unlike adults, children and adolescents have incomplete and immature growth, and there is still a lack of relevant studies on the stability of the spine and the degree of recovery of neurological function in children after surgery. The indications for surgery, surgical methods, and the selection of implant materials are controversial. Based on previous experience, patients with persistent pain, neurological symptoms, or spinal deformity usually opt for surgical treatment to remove the lesion, decompress the nerve, improve spinal deformity, and preserve spinal stability. It provides the best chance of cure and reduces the risk of recurrence.12,23,24 Surgical methods can be broadly divided into piecemeal resection and en bloc resection, and commonly used spinal fusion materials include artificial vertebral bodies and shaped TMs.
We aimed to investigate changes in spinal stability and quality of life after lumbar spine surgery in pediatric patients. The results show that after surgery, pediatric patients achieved substantial improvements in pain levels, neurological function and ODI scores with no significant differences either between the 2 surgical approaches compared to each other or between the 2 spinal fusion materials. Additionally, we found that there was no significant difference in blood loss, length of hospital stay or fusion time between different surgical methods or between different spinal fusion materials.
In addition, the lumbar lordosis angle of the children did not change significantly when compared between the preoperative, postoperative, and final follow-up periods, and the difference was not statistically significant, indicating that there was no significant change in the physiological curvature of the lumbar spine after spinal fusion using pedicle screws combined with artificial vertebral body or TM implanted with allogeneic double-sided iliac bone block in pediatric lumbar tumour resection; thus, it can be inferred that the spinal stability will not be significantly damaged after pediatric lumbar tumour surgery. The TB group had a lower local lordosis angle at the final follow-up than at pre-operation and at 1 week postoperatively. Additionally, we can see that the lumbar angle drift and local angle drift at the final follow-up in the AB group were significantly lower than those in the TB group, and it can be inferred that children with lumbar fusion using artificial vertebrae will obtain better spinal stability.
Many studies have demonstrated that TER can provide excellent pain relief, provide the best chance of cure and reduce the risk of recurrence25-29; however, lumbar tumour surgery causes almost partial or even complete destruction of the anterior, middle, and posterior column structure of the vertebral body, and some people worry that it may cause iatrogenic spinal instability, which did not occur in our study. Thus, TER is recommended not only for malignant tumors but also for benign pediatric lumbar spine tumors with a high recurrence rate of potential malignancy, and spinal fusion is performed intraoperatively using artificial vertebral bodies implanted with bicortical iliac blocks. No implant fusion failure occurred in our study, and only 1 case of longitudinal rod breakage occurred. This appears to be due to several reasons: appropriately strong pedicle screws; adequate bone grafting (allograft and posterior bone grafting); and strict surgical indications (<= 2 vertebrae bodies compromised by tumour). Postoperative complications are usually associated with low compliance, malnutrition, and premature ambulation rather than the surgical procedure.
The main types of traditional spinal reconstruction are strut bone grafts and prefabricated prosthetic replacement devices. Supporting bone grafts include autologous supporting bone grafts and allogeneic supporting bone grafts.30,31 Prefabricated prosthetic replacement devices include porous TM, self-expanding TM, and polyetheretherketone fusion.11,32,33 All of the graft materials mentioned above have been used for intervertebral fusion, and the graft may fail due to postoperative slippage, fracture, resorption, or subsidence.4,34,35 Bony fusion was observed in all cases in our study, and no loosening, displacement, or dislocation of the implant was observed. The stiffness and strength-bearing quality of the artificial vertebra in combination with the pedicle screw plays a crucial supporting role. The artificial cone ensures that a sufficient contact area is achieved between the bone socket surface and the bone graft area to meet the requirements of osseointegration. The instrumentation reduces the trauma and complications caused by autogenous iliac bone and solves the problem of long-term resorption of the bone graft. More importantly, the artificial vertebra can be adjusted in length and ground slope according to the intraoperative situation, thus meeting individual treatment needs. In addition, three-dimensionally printed prostheses with the advantages of precisely matching anatomical shapes, adequate strength, and elastic modulus that is closer to that of cancellous bone are gradually being tried and applied in clinical practice. However, clinical trial data to support widespread application are still lacking.36-38
This retrospective study has several limitations, including a small sample size and relatively short follow-up period; the effect of fusion on the proximal and distal connecting segments and the effect of degenerative changes are also worth investigating. Prospective studies of a larger number of patients and longer follow-up periods (at least until skeletal maturity) may further elucidate the effect on spinal stability after pediatric lumbar tumour surgery. In addition, the type of tumour pathology and the extent of soft tissue invasion by the tumour may also have an impact on spinal stability after lumbar tumour surgery in children and requires further investigation.
Conclusion
This study showed that after posterior lumbar tumour resection and reconstruction of internal fixation, pediatric patients with lumbar tumors could obtain satisfactory spinal stability, effectively relieve pain symptoms and substantially improve neurological function. There was no significant difference in the invasiveness, safety or timeliness between the 2 surgical methods, so TER with a low postoperative recurrence rate and good local control is recommended. Spinal fusion using an artificial vertebral body implanted with bicortical bone mass results in better spinal stability.
Footnotes
Authors' Contributions
Haifeng Wei and Shaohui He designed the study and gave us several meaningful suggestions. Wenjun Chen reviewed and collected the medical records. Shuang Cao analyzed and interpreted the patient data. Yue Zhang was a contributor in writing the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: No conflict of interest exits in the submission of this manuscript, and manuscript is approved by all authors for publication. I would like to declare on behalf of my co-authors that the work described was original research that has not been published previously, and not under consideration for publication elsewhere, in whole or in part.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (82072971).
